Molecular Characterization of Heat-Induced HSP11.0 and Master-Regulator HSF from Cotesia chilonis and Their Consistent Response to Heat Stress
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Insects
2.2. Sample Treatments
2.2.1. Different Temperature Treatments
2.2.2. Thermal Treatment at Different Times
2.3. Total RNA Isolation and Synthesis of First Strand cDNA
2.4. Cloning and Genome Amplification
2.5. Sequence Analysis of Genes
2.6. Real-Time qPCR Analysis
2.7. Statistical Analysis
3. Results
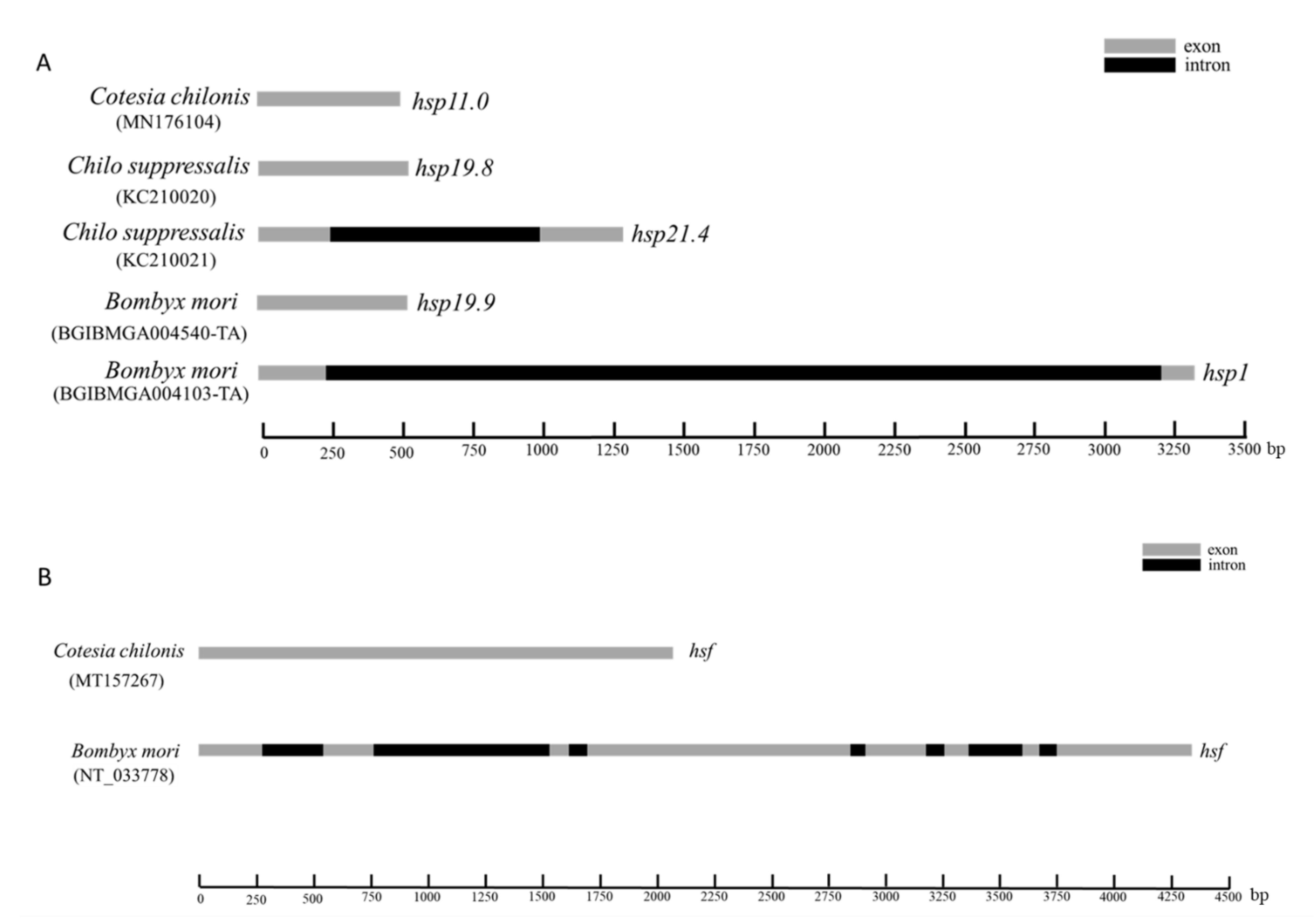
3.1. Characteristics of Sequenced Genes
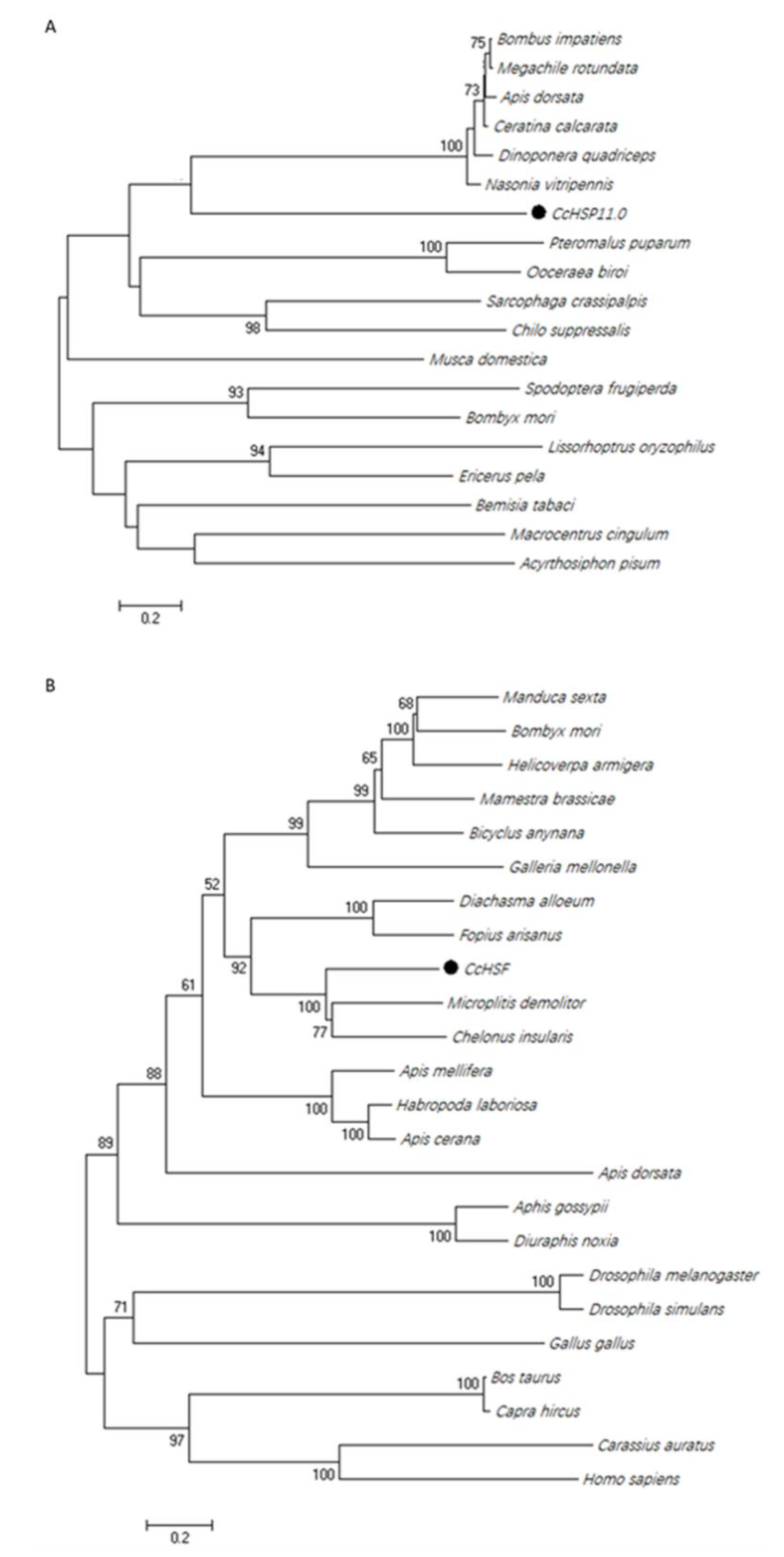
3.2. Phylogenetic Analysis of Genes
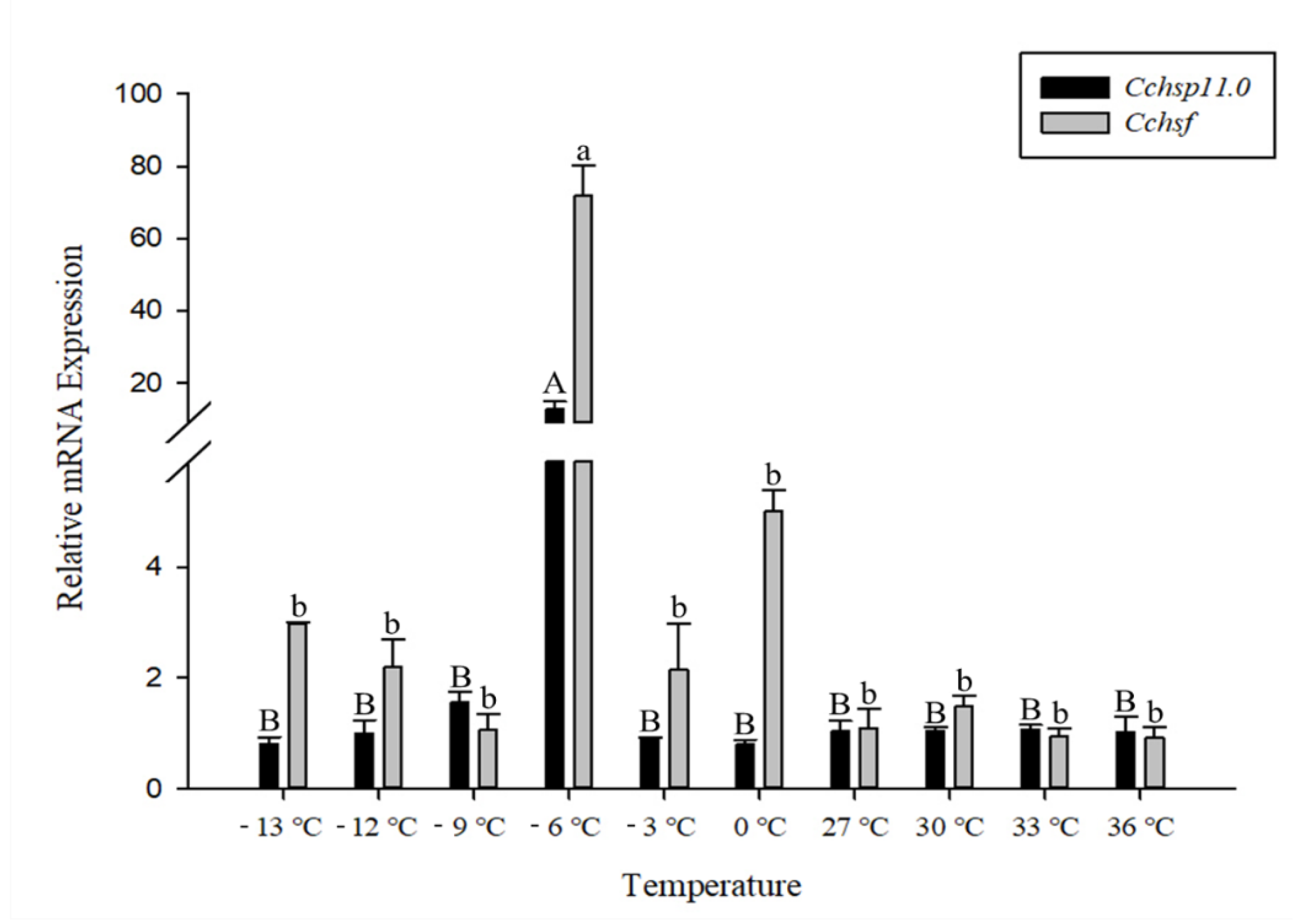
3.3. Gene Expression in Response to Different Temperatures
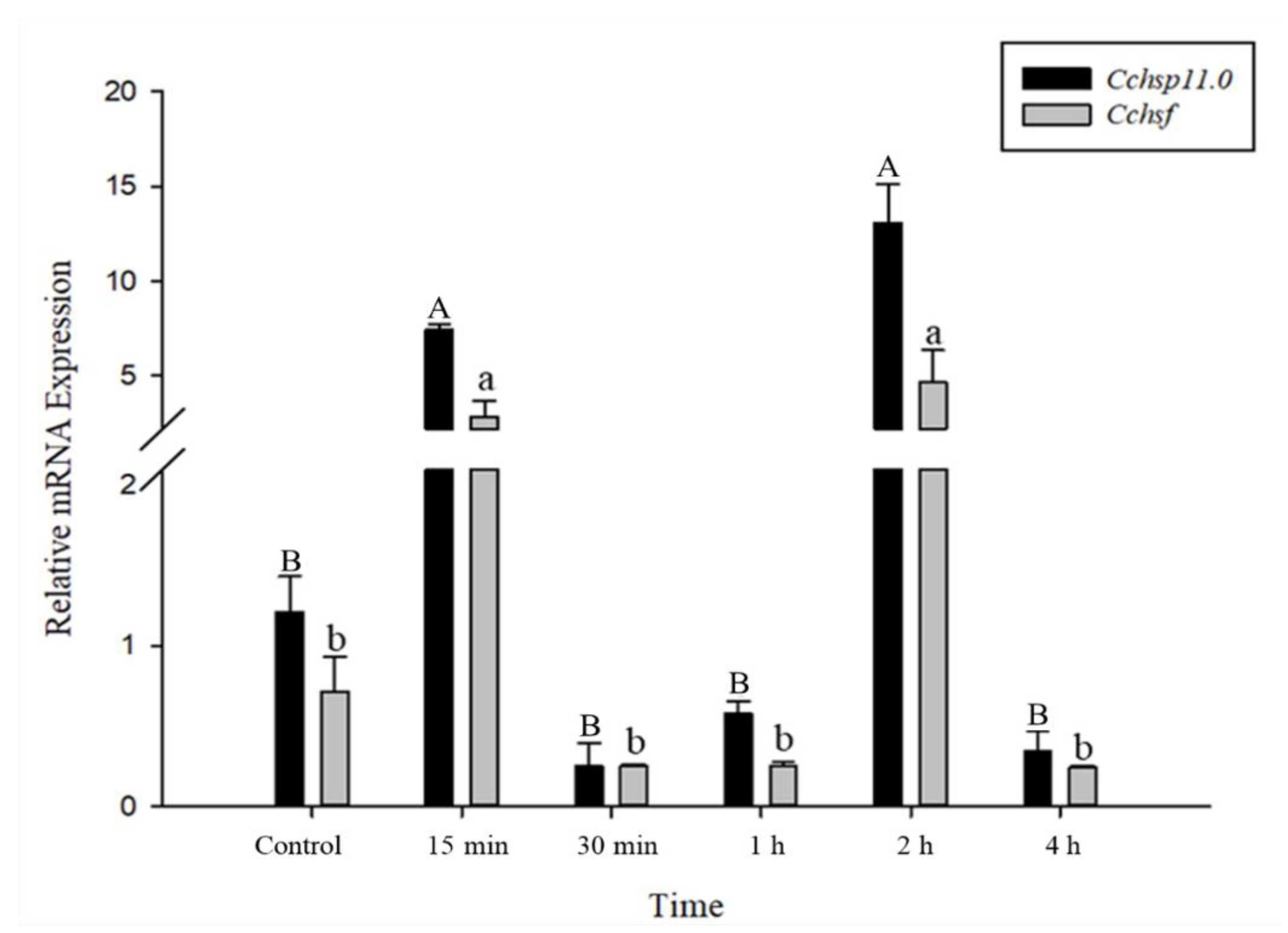
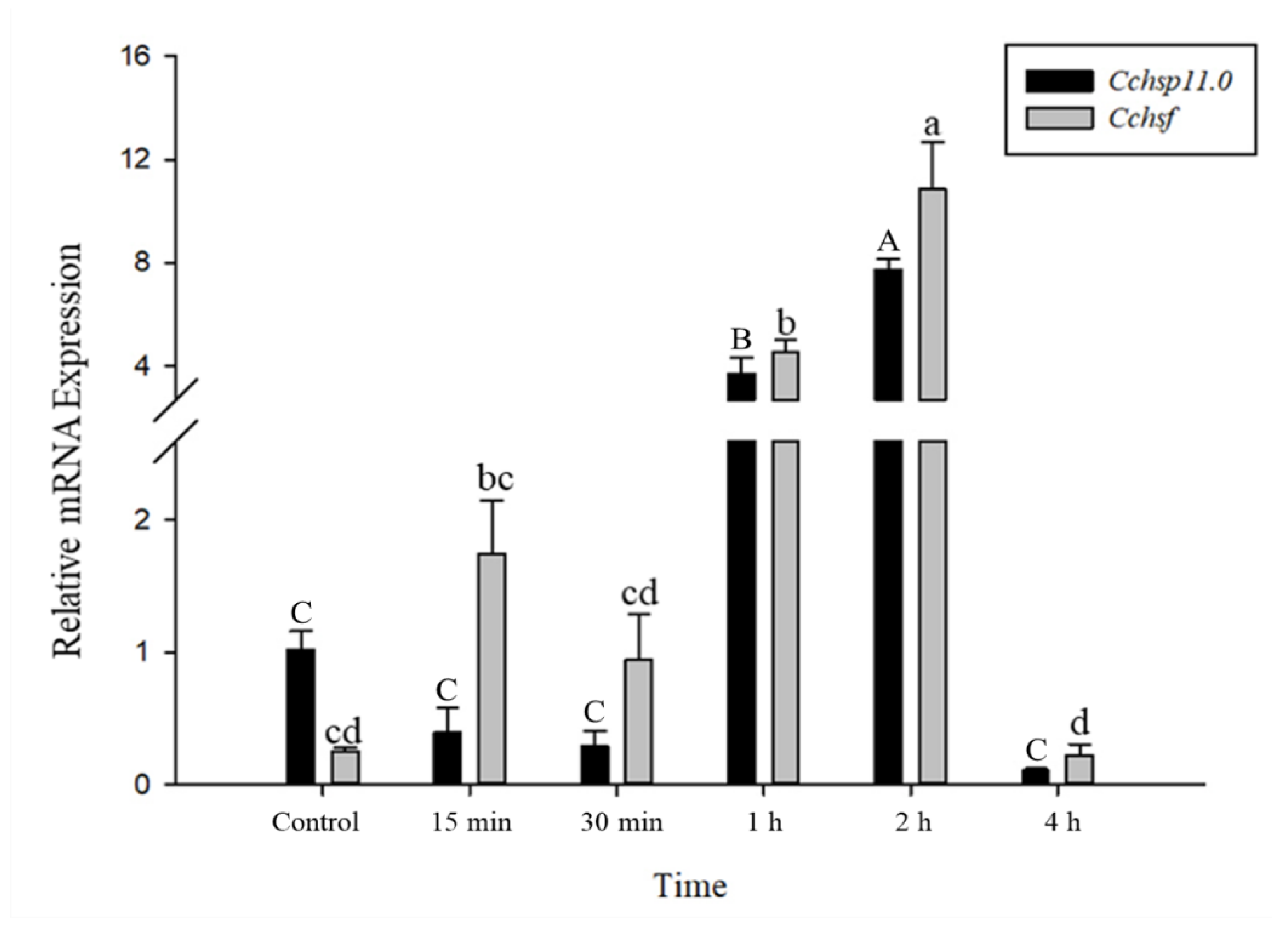
3.4. Time Course of Gene Expression After Temperature Treatments
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gao, P.; Lu, M.X.; Pan, D.D.; Du, Y.Z. Characterization of an inducible HSP70 gene in Chilo suppressalis and expression in response to environmental and biological stress. Cell Stress Chaperones 2019, 25, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.D.; Cao, S.S.; Lu, M.X.; Hang, S.B.; Du, Y.Z. Genes encoding heat shock proteins in the endoparasitoid wasp, Cotesia chilonis, and their expression in response to temperatures. J. Integr. Agric. 2018, 17, 1012–1022. [Google Scholar] [CrossRef]
- Chen, H.C.; Lou, Y.G.; Cheng, J.A. The research and application of Cotesia chilonis. Chin. J. Biol. Control 2002, 18, 90–93. [Google Scholar]
- Pan, D.D.; Liu, Z.X.; Lu, M.X.; Cao, S.S.; Yan, W.F.; Du, Y.Z. Species and occurrence dynamics of parasitic wasps of the rice stem borer, Chilo suppressalis (Walker) (Lepidoptera: Pyralidae) in Yangzhou. J. Environ. Entomol. 2016, 38, 1106–1113. [Google Scholar]
- Colinet, H.; Brent, J.; Sinclair, P.V.; David, R. Insects in fluctuating thermal environments. Annu. Rev. Entomol. 2015, 60, 123–140. [Google Scholar] [CrossRef]
- Willmer, P.C.; Stone, G.; Johnston, I.A. Environmental physiology of animals. Blackwell 2000, 71, e1333. [Google Scholar]
- Kerr, J.T.; Pindar, A.; Galpern, P.; Packer, L.; Potts, S.G.; Roberts, S.M.; Rasmont, P.; Schweigerm, O.; Colla, S.R.; Richardson, L.L.; et al. Climate change impacts on bumblebees converge across continents. Science 2015, 349, 177–180. [Google Scholar] [CrossRef]
- Garcia, R.C.; Kuprewicz, E.K.; Staines, C.L.; Erwin, T.L. Limited tolerance by insects to high temperatures across tropical elevational gradients and the implications of global warming for extinction. Proc. Natl. Acad. Sci. USA 2016, 113, 680–685. [Google Scholar] [CrossRef]
- Sinclair, B.J.; Alvarado, L.E.; Ferguson, L.V. An invitation to measure insect cold tolerance: Methods, approaches, and workflow. J. Therm. Biol. 2015, 53, 180–197. [Google Scholar] [CrossRef]
- Martin-Folgar, R.; Aquilino, M.; Ozaez, I.; Martinez-Guitarte, J.L. Ultraviolet filters and heat shock proteins: Effects in Chironomus riparius by benzophenone-3 and 4-methylbenzylidene camphor. Environ. Sci. Pollut. R 2018, 25, 333–344. [Google Scholar] [CrossRef]
- Tian, L.; Wang, X.Y.; Wang, X.P.; Lei, C.L.; Zhu, F. Starvation-, thermal- and heavy metal-associated expression of four small heat shock protein genes in Musca domestica. Gene 2018, 642, 268–276. [Google Scholar] [CrossRef]
- Feder, M.E.; Hofmann, G.E. Heat-shock proteins, molecular chaperones, and the stress response: Evolutionary and ecological physiology. Annu. Rev. Physiol. 1999, 61, 243–282. [Google Scholar] [CrossRef]
- Sørensen, J.G.; Kristensen, G.T.N.; Loeschcke, V. The evolutionary and ecological role of heat shock proteins. Ecol. Lett. 2003, 6, 1025–1037. [Google Scholar] [CrossRef]
- Horwitz, J. Alpha-crystallin can function as a molecular chaperone. Proc. Natl. Acad. Sci. USA 1992, 89, 10449–10453. [Google Scholar] [CrossRef]
- Akerfelt, M.; Morimoto, R.I.; Sistonen, L. Heat shock factors: Integrators of cell stress, development and lifespan. Nat. Rev. Mol. Cell Biol. 2010, 11, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Wiederrecht, G.; Seto, D.; Parker, C.S. Isolation of the gene encoding the S. cerevisiae heat shock transcription factor. Cell 1988, 6, 841–853. [Google Scholar] [CrossRef]
- Triandafillou, C.G.; Drummond, D.A. Heat shock factor 1: From fire chief to crowd-control specialist. Mol. Cell 2016, 63, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Kihara, F.; Niimi, T.; Yamashita, O.; Yaginuma, T. Heat shock factor binds to heat shock elements upstream of heat shock protein 70a and Samui genes to confer transcriptional activity in Bombyx mori diapause eggs exposed to 5 °C. Insect Biochem. Mol. 2011, 41, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Anckar, J.; Sistonen, L. Heat shock factor 1 as a coordinator of stress and developmental pathways. Adv. Exp. Med. Biol. 2017, 594, 78–88. [Google Scholar]
- Fujikake, N.; Nagai, Y.; Popiel, H.A.; Kano, H.; Yamaguchi, M.; Toda, T. Alternative splicing regulates the transcriptional activity of Drosophila heat shock transcription factor in response to heat/cold stress. FEBS Lett. 2005, 17, 3842–3848. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.W.; Li, X.; Yu, Q.Y.; Xiang, Z.H.; Kishino, H.; Zhang, Z. The small heat shock protein (sHSP) genes in the silkworm, Bombyx mori, and comparative analysis with other insect sHSP genes. BMC Evol. Biol. 2009, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Gu, J.; Huang, L.H.; Zheng, S.C.; Liu, L.; Xu, W.H.; Feng, Q.L.; Kang, L. Cloning and expression analysis of six small heat shock protein genes in the common cutworm, Spodoptera litura. J. Insect Physiol. 2011, 57, 908–914. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.X.; Hua, J.; Cui, Y.D.; Du, Y.Z. Five small heat shock protein genes from Chilo suppressalis: Characteristics of gene, genomic organization, structural analysis, and transcription profiles. Cell Stress Chaperones 2014, 19, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.N.; Liu, Y.; Xin, Z.Z.; Zhu, X.Y.; Ge, B.M.; Li, C.F.; Wang, D.; Bian, X.G.; Yang, L.; Chen, L.; et al. A small heat shock protein 21 (sHSP21) mediates immune responses in Chinese oak silkworm Antheraea pernyi. Int. J. Biol. Macromol. 2018, 111, 1027–1031. [Google Scholar] [CrossRef]
- Huang, L.H.; Wang, C.Z.; Kang, L. Cloning and expression of five heat shock protein genes in relation to cold hardening and development in the leaf miner, Liriomyza sativa. J. Insect Physiol. 2009, 55, 279–285. [Google Scholar] [CrossRef]
- Sun, M.; Lu, M.X.; Tang, X.T.; Du, Y.Z. Characterization and expression of genes encoding three small heat shock proteins in Sesamia inferens (Lepidoptera: Noctuidae). Int. J. Mol. Sci. 2014, 15, 23196–23211. [Google Scholar] [CrossRef]
- Song, J.; Cao, S.S.; Lu, M.X.; Du, Y.Z. Mortality and HSP genes expression in the endoparasitoid Cotesia chilonis (Hymenoptera: Braconidae) after cold acclimationat different temperatures. Ann. Entomol. Soc. Am. 2020, 113, 171–175. [Google Scholar] [CrossRef]
- Chen, X.E.; Zhang, Y.L. Identification of multiple small heat-shock protein genes in Plutella xylostella (L.) and their expression profiles in response to abiotic stresses. Cell Stress Chaperones 2015, 20, 23–35. [Google Scholar] [CrossRef]
- Chen, W.; Geng, S.L.; Song, Z.; Li, Y.J.; Wang, H.; Cao, J.Y. Alternative splicing and expression analysis of HSF1 in diapause pupal brains in the cotton bollworm, Helicoverpa armigera. Pest. Manag. Sci. 2019, 75, 1258–1269. [Google Scholar] [CrossRef]
- Sonoda, S.; Tsumuki, H. Characterization of alternatively spliced transcripts encoding heat shock transcription factor in cultured cells of the cabbage armyworm, Mamestra brassicae. Arch. Insect Biochem. 2010, 1, 49–60. [Google Scholar] [CrossRef]
- Li, Q.Y.; Li, Z.L.; Lu, M.X.; Cao, S.S.; Du, Y.Z. Selection of valid reference genes for quantitative real-time PCR in Cotesia chilonis (Hymenoptera: Braconidae) exposed to different temperatures. PLoS ONE 2019, 14, e002679. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The Clustal-X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.; Pareek, A.; Verma, J.K. The small heat shock proteins, chaperonin 10, in plants: An evolutionary view and emerging functional diversity. Environ. Exp. Bot. 2020, 4, 323. [Google Scholar]
- Wang, H.J.; Shi, Z.K.; Shen, Q.D.; Xu, C.D.; Wang, B.; Meng, Z.J.; Wang, S.G.; Tang, B.; Wang, S. Molecular cloning and induced expression of six small heat shock proteins mediating cold-hardiness in Harmonia axyridis (Coleoptera: CoCcinellidae). Front. Physiol. 2017, 8, 60. [Google Scholar] [CrossRef]
- Yang, J.; Gu, J.; Hu, Y.; Wang, N.; Wang, P. Molecular cloning and characterization of HSP60 gene in domestic pigeons (Columba livia) and differential expression patterns under temperature stress. Cell Stress Chaperones 2020, 4, 1–13. [Google Scholar] [CrossRef]
- Bale, J.S.; Masters, G.J.; Hodkinson, I.D.; Awmak, C.; Bezemer, T.M.; Brown, V.; Butterfield, J.; Buse, A.; Coulson, J.C.; Farrar, J.; et al. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Sorensen, J.G.; Loeschcke, V. Adaptation of Drosophila to temperature extremes: Bringing together quantitative and molecular approaches. J. Therm. Biol. 2003, 28, 175–216. [Google Scholar] [CrossRef]
- Song, J.; Lu, M.X.; Du, Y.Z. Molecular cloning and expression patterns of two small heat shock proteins from Chilo suppressalis (Walker). J. Integr. Agric. 2020, 19, 1522–1529. [Google Scholar] [CrossRef]
- Wang, L.H.; Zhang, Y.L.; Pan, L.; Wang, Q.; Han, Y.C.; Niu, H.T.; Shan, D.; Hoffmann, A.; Fang, J.C. Induced expression of small heat shock protein is associated with thermotolerance in female Laodelphax striatellus planthoppers. Cell Stress Chaperones 2018, 24, 115–123. [Google Scholar] [CrossRef]
- Guo, L.L. Heat shock regulatory factor Hsf’s influence on Hsp60’s expression in Invasion Whitefies. Henan Inst. Sci. Technol. 2013, 56, 145–167. (In Chinese) [Google Scholar]
- Zheng, J.B.; Mao, Y.; Su, Y.Q.; Wang, J. Cross talk between heat shock protein 10 and a heat shock factor identified from Marsupenaeus japonicus. Int. J. Biol. Macromol. 2020, 147, 1041–1052. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.M.; Overgaard, J.; Sørensen, J.G.; Holmstrup, M.; Justesen, J.; Loeschcke, V. Role of HSF activation for resistance to heat, cold and high-temperature knock-down. J. Insect Physiol. 2005, 51, 1320–1329. [Google Scholar] [CrossRef] [PubMed]
- Wu, C. Heat shock transcription factors: Structure and regulation. Annu. Rev. Cell Dev. Biol. 1995, 11, 441–469. [Google Scholar] [CrossRef] [PubMed]

| Primer Name | Primer Sequences (5′-3′) | Tm (°C) | ε b (%) | R2 c |
|---|---|---|---|---|
| Fragment verification | ||||
| hsp11.0-F | CGGGAACAAATCAACAT | 46.3 | ||
| hsp11.0-R | ACTCGGTCCATCAAAGG | 51.3 | ||
| hsf-F | AGAACGCAACAACCAAG | 50.0 | ||
| hsf-R | CAACTACAGAACCATCAGAG | 45.0 | ||
| Rapid-amplification of cDNA ends (RACE) | ||||
| hsp11.0-5′ | GAGCCAGGTCCAACAGCAACTACAG | 62.4 | ||
| hsp11.0-3′ | CGTTCAAAGAGCCGATGCTATAA | 54.8 | ||
| hsf-F | CTTGCTGCTGGAGCCTGGATCAC | 63.5 | ||
| hsf-R | ATTCCAGACATCCTACTCACCTC | 55.7 | ||
| Verification of full-length cDNA | ||||
| hsp11.0-F | AGTTATTCACCAGCAACGT | 51.1 | ||
| hsp11.0-R | GTTTGATAATTTCATAGAGC | 42.4 | ||
| hsf-F | ATCACTAATACGACTCACTATAGGG | 52.5 | ||
| hsf-R | TTTGTTTATAGTACGCAAGTCG | 51.8 | ||
| Verification of genome | ||||
| hsp11.0-F | CTCAGATCTTATTCTTTCAT | 42.6 | ||
| hsp11.0-R | GTTTGATAATTTCATAGAGC | 42.4 | ||
| hsf-F | ATCACTAATACGACTCACTATAGGG | 52.5 | ||
| hsf-R | GAGCTGAATAAATACACTCACCA | 51.8 | ||
| Real-time quantitative PCR a | ||||
| hsp11.0-F | ACAAAGTTCTCCTCCCCG | 59.4 | 90.0 | 0.988 |
| hsp11.0-R | GCAACAATGTCTGATTCACG | |||
| hsf-F | TTAGGTGCTGAAAGTGCCGA | 60.0 | 117.3 | 0.904 |
| hsf-R | AGTACGCAAGTCGAGCTGAA | |||
| Reference gene in qRT-PCR a | ||||
| H3-F | CGTCGCTCTTCGTGAAATCA | 58.1 | 97.4 | 0.978 |
| H3-R | TCTGGAAACGCAAGTCGGTC | |||
| GAPDH-F | GAAGGTGGTGCCAAGAAAG | 54.0 | 106.7 | 0.978 |
| GAPDH-R | GCATGGACAGTGGTCATAAGA | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, F.-J.; Zhu, F.; Lu, M.-X.; Du, Y.-Z. Molecular Characterization of Heat-Induced HSP11.0 and Master-Regulator HSF from Cotesia chilonis and Their Consistent Response to Heat Stress. Insects 2021, 12, 322. https://doi.org/10.3390/insects12040322
He F-J, Zhu F, Lu M-X, Du Y-Z. Molecular Characterization of Heat-Induced HSP11.0 and Master-Regulator HSF from Cotesia chilonis and Their Consistent Response to Heat Stress. Insects. 2021; 12(4):322. https://doi.org/10.3390/insects12040322
Chicago/Turabian StyleHe, Fu-Jing, Feng Zhu, Ming-Xing Lu, and Yu-Zhou Du. 2021. "Molecular Characterization of Heat-Induced HSP11.0 and Master-Regulator HSF from Cotesia chilonis and Their Consistent Response to Heat Stress" Insects 12, no. 4: 322. https://doi.org/10.3390/insects12040322
APA StyleHe, F.-J., Zhu, F., Lu, M.-X., & Du, Y.-Z. (2021). Molecular Characterization of Heat-Induced HSP11.0 and Master-Regulator HSF from Cotesia chilonis and Their Consistent Response to Heat Stress. Insects, 12(4), 322. https://doi.org/10.3390/insects12040322






