Area-Wide Elimination of Subterranean Termite Colonies Using a Novaluron Bait
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Site and Collections
2.2. DNA Extraction, Genotyping & Sequencing
2.3. Colony Breeding Structure and Colony Differentiation
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vreysen, M.; Robinson, A.; Hendrichs, J.; Kenmore, P. Area-wide integrated pest management (AW-IPM): Principles, practice and prospects. In Area-Wide Control of Insect Pests; Springer: New York, NY, USA, 2007; pp. 3–33. [Google Scholar]
- Getty, G.M.; Solek, C.W.; Sbragia, R.J.; Haverty, M.I.; Lewis, V.R. Large-scale Suppression of a Subterranean Termite Community Using the Sentricon® Termite Colony Elimination System: A Case Study in Chats-worth, California, USA. Sociobiology 2007, 50, 1041–1050. [Google Scholar]
- Guillot, F.S.; Ring, D.R.; Lax, A.R.; Morgan, A.; Brown, K.; Riegel, C.; Boykin, D. Area-wide management of the Formosan subterranean termite, Coptotermes formosanus Shiraki (Isoptera: Rhinotermitidae), in the New Orleans French Quarter. Sociobiology 2010, 55, 311–338. [Google Scholar]
- Smith, J.; Su, N.-Y.; Escobar, R.N. An areawide population management project for the invasive eastern subterranean termite (Isoptera: Rhinotermitidae) in a low-income community in Santiago, Chile. Am. Entomol. 2006, 52, 253–260. [Google Scholar] [CrossRef]
- Osbrink, W.L.A.; Cornelius, M.L.; Lax, A.R. Areawide field study on effect of three chitin synthesis inhibitor baits on populations of Coptotermes formosanus and Reticulitermes flavipes (Isoptera: Rhinotermitidae). J. Econ. Entomol. 2011, 104, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Rust, M.K.; Su, N.-Y. Managing social insects of urban importance. Annu. Rev. Entomol. 2012, 57, 355–375. [Google Scholar] [CrossRef] [PubMed]
- Su, N.-Y. Novel technologies for subterranean termite control. Sociobiology 2002, 40, 95–102. [Google Scholar]
- Blumenfeld, A.J.; Vargo, E.L. Geography, opportunity and bridgeheads facilitate termite invasions to the United States. Biol. Invasions 2020, 22, 3269–3282. [Google Scholar] [CrossRef]
- Eyer, P.-A.; Blumenfeld, A.; Johnson, L.; Perdereau, E.; Shults, P.; Wang, S.; Dedeine, F.; Dupont, S.; Bagnères, A.-G.; Vargo, E. Extensive human-mediated jump dispersal within and across the native and introduced ranges of the invasive termite Reticulitermes flavipes. Authorea Prepr. 2020. [Google Scholar] [CrossRef]
- Perdereau, E.; Baudouin, G.; Bankhead-Dronnet, S.; Chevalier, Z.; Zimmermann, M.; Dupont, S.; Dedeine, F.; Bagnères, A.-G. Invasion Dynamics of A Termite, Reticulitermes flavipes, at Different Spatial Scales in France. Insects 2019, 10, 30. [Google Scholar] [CrossRef]
- Su, N.-Y.; Chouvenc, T.; Li, H.-F. Potential hybridization between two invasive termite species, Coptotermes formosanus and C. gestroi (Isoptera: Rhinotermitidae), and its biological and economic implications. Insects 2017, 8, 14. [Google Scholar] [CrossRef]
- Jenkins, T.M.; Jones, S.C.; Lee, C.-Y.; Forschler, B.T.; Chen, Z.; Lopez-Martinez, G.; Gallagher, N.T.; Brown, G.; Neal, M.; Thistleton, B. Phylogeography illuminates maternal origins of exotic Coptotermes gestroi (Isoptera: Rhinotermitidae). Mol. Phylogenet. Evol. 2007, 42, 612–621. [Google Scholar] [CrossRef]
- Blumenfeld, A.J.; Eyer, P.-A.; Husseneder, C.; Mo, J.; Johnson, L.N.L.; Wang, C.; Grace, J.K.; Chouvenc, T.; Wang, S.; Vargo, E.L. Bridgehead effect and multiple introductions shape the global invasion history of a termite. Commun. Biol. 2021, 4, 1–12. [Google Scholar] [CrossRef]
- Thorne, B.L. Biology of subterranean termites of the genus Reticulitermes. In National Pest Control Association Research Report on Subterranean Termites; National Pest Management Association: Fairfax, VA, USA, 1998; pp. 1–30. [Google Scholar]
- Su, N.Y.; Ban, P.M.; Scheffrahn, R.H. Foraging Populations and Territories of the Eastern Subterranean Termite (Isoptera, Rhinotermitidae) in Southeastern Florida. Environ. Entomol. 1993, 22, 1113–1117. [Google Scholar] [CrossRef]
- Deheer, C.J.; Vargo, E.L. Colony genetic organization and colony fusion in the termite Reticulitermes flavipes as revealed by foraging patterns over time and space. Mol. Ecol. 2004, 13, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Howard, R.W.; Jones, S.C.; Mauldin, J.K.; Beal, R.H. Abundance, distribution, and colony size estimates for Reticulitermes spp.(Isoptera: Rhinotermitidae) in southern Mississippi. Environ. Entomol. 1982, 11, 1290–1293. [Google Scholar] [CrossRef]
- Chouvenc, T.; Su, N.-Y. When subterranean termites challenge the rules of fungal epizootics. PLoS ONE 2012, 7, e34484. [Google Scholar] [CrossRef]
- Bulmer, M.; Traniello, J. Foraging range expansion and colony genetic organization in the subterranean termite Reticulitermes flavipes (Isoptera: Rhinotermitidae). Environ. Entomol. 2002, 31, 293–298. [Google Scholar] [CrossRef]
- Calleri, D.V.; McGrail Reid, E.; Rosengaus, R.B.; Vargo, E.L.; Traniello, J.F. Inbreeding and disease resistance in a social insect: Effects of heterozygosity on immunocompetence in the termite Zootermopsis angusticollis. Proc. R Soc. Lond B Biol. Sci. 2006, 273, 2633–2640. [Google Scholar] [CrossRef] [PubMed]
- Aguero, C.M.; Eyer, P.-A.; Vargo, E.L. Increased genetic diversity from colony merging in termites does not improve survival against a fungal pathogen. Sci. Rep. 2020, 10, 1–9. [Google Scholar]
- Vargo, E.L.; Husseneder, C. Biology of subterranean termites: Insights from molecular studies of Reticulitermes and Coptotermes. Annu. Rev. Entomol. 2009, 54, 379–403. [Google Scholar] [CrossRef] [PubMed]
- Perdereau, E.; Bagnères, A.G.; Vargo, E.; Baudouin, G.; Xu, Y.; Labadie, P.; Dupont, S.; Dedeine, F. Relationship between invasion success and colony breeding structure in a subterranean termite. Mol. Ecol. 2015, 24, 2125–2142. [Google Scholar] [CrossRef]
- Hernández-Teixidor, D.; Suárez, D.; García, J.; Mora, D. First report of the invasive Reticulitermes flavipes (Kollar, 1837)(Blattodea, Rhinotermitidae) in the Canary Islands. J. Appl. Entomol. 2019, 143, 478–482. [Google Scholar] [CrossRef]
- Perdereau, E.; Bagneres, A.G.; Bankhead-Dronnet, S.; Dupont, S.; Zimmermann, M.; Vargo, E.; Dedeine, F. Global genetic analysis reveals the putative native source of the invasive termite, Reticulitermes flavipes, in France. Mol. Ecol. 2013, 22, 1105–1119. [Google Scholar] [CrossRef] [PubMed]
- Suppo, C.; Robinet, C.; Perdereau, E.; Andrieu, D.; Bagnères, A.-G. Potential spread of the invasive North American termite, Reticulitermes flavipes, and the impact of climate warming. Biol. Invasions 2018, 20, 905–922. [Google Scholar] [CrossRef]
- Evans, T.A.; Forschler, B.T.; Grace, J.K. Biology of invasive termites: A worldwide review. Annu. Rev. Entomol. 2013, 58, 455–474. [Google Scholar] [CrossRef]
- Perdereau, E.; Bagnères, A.-G.; Dupont, S.; Dedeine, F. High occurrence of colony fusion in a European population of the American termite Reticulitermes flavipes. Insectes Soc. 2010, 57, 393–402. [Google Scholar] [CrossRef]
- Perdereau, E.; Dedeine, F.; Christidès, J.-P.; Bagnères, A.-G. Variations in worker cuticular hydrocarbons and soldier isoprenoid defensive secretions within and among introduced and native populations of the subterranean termite, Reticulitermes flavipes. J. Chem. Ecol. 2010, 36, 1189–1198. [Google Scholar] [CrossRef] [PubMed]
- Eger, J.E., Jr.; Lees, M.D.; Neese, P.A.; Atkinson, T.H.; Thoms, E.M.; Messenger, M.T.; Demark, J.J.; Lee, L.-C.; Vargo, E.L.; Tolley, M.P. Elimination of Subterranean Termite (Isoptera: Rhinotermitidae) Colonies Using a Refined Cellulose Bait Matrix Containing Noviflumuron When Monitored and Replenished Quarterly. J. Econ. Entomol. 2012, 105, 533–539. [Google Scholar] [CrossRef]
- Hu, X.P. Liquid termiticides: Their role in subterranean termite management. In Urban Pest Management: An Environmental Perspective; CABI: Wallingford, UK, 2011; pp. 114–132. [Google Scholar]
- Su, N.-Y. Field evaluation of a hexaflumuron bait for population suppression of subterranean termites (Isoptera: Rhinotermitidae). J. Econ. Entomol. 1994, 87, 389–397. [Google Scholar] [CrossRef]
- Vargo, E.L. Genetic structure of Reticulitermes flavipes and R. virginicus (Isoptera: Rhinotermitidae) colonies in an urban habitat and tracking of colonies following treatment with hexaflumuron bait. Environ. Entomol. 2003, 32, 1271–1282. [Google Scholar] [CrossRef]
- Getty, G.M.; Haverty, M.I.; Copren, K.A.; Lewis, V.R. Response of Reticulitermes spp. (Isoptera: Rhinotermitidae) in northern California to baiting with hexaflumuron with sentricon termite colony elimination system. J. Econ. Entomol. 2000, 93, 1498–1507. [Google Scholar] [CrossRef]
- Keefer, T.C.; Puckett, R.T.; Brown, K.S.; Gold, R.E. Field Trials With 0.5% Novaluron Insecticide Applied as a Bait to Control Subterranean Termites (Reticulitermes sp. and Coptotermes formosanus [Isoptera: Rhinotermitidae]) on Structures. J. Econ. Entomol. 2015, 108, 2407–2413. [Google Scholar] [CrossRef]
- Kakkar, G.; Osbrink, W.; Su, N.-Y. Molting site fidelity accounts for colony elimination of the Formosan subterranean termites (Isoptera: Rhinotermitidae) by chitin synthesis inhibitor baits. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Su, N.-Y. Development of Baits for Population Management of Subterranean Termites. Annu. Rev. Entomol. 2019, 64, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.A.; Iqbal, N. Termite (order Blattodea, infraorder Isoptera) baiting 20 years after commercial release. Pest Manag. Sci. 2015, 71, 897–906. [Google Scholar] [CrossRef]
- Su, N.-Y.; Ban, P.; Scheffrahn, R.H. Use of a bait impact index to assess effects of bait application against populations of Formosan subterranean termite (Isoptera: Rhinotermitidae) in a large area. J. Econ. Entomol. 2004, 97, 2029–2034. [Google Scholar] [CrossRef]
- Messenger, M.T.; Su, N.Y.; Husseneder, C.; Grace, J.K. Elimination and reinvasion studies with Coptotermes formosanus (Isoptera: Rhinotermitidae) in Louisiana. J. Econ. Entomol. 2005, 98, 916–929. [Google Scholar] [CrossRef] [PubMed]
- Vargo, E. Polymorphism at trinucleotide microsatellite loci in the subterranean termite Reticulitermes flavipes. Mol. Ecol. 2000, 9, 817–820. [Google Scholar] [CrossRef]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef]
- Rousset, F. Genepop Version 4.7.0. Available online: https://kimura.univ-montp2.frrousset/Genepop4.7.pdf (accessed on 20 February 2021).
- Vargo, E.L.; Juba, T.R.; Deheer, C.J. Relative abundance and comparative breeding structure of subterranean termite colonies (Reticulitermes flavipes, Reticulitermes hageni, Reticulitermes virginicus, and Coptotermes formosanus) in a South Carolina lowcountry site as revealed by molecular markers. Ann. Entomol. Soc. Am. 2006, 99, 1101–1109. [Google Scholar]
- DeHeer, C.J.; Vargo, E.L. An indirect test of inbreeding depression in the termites Reticulitermes flavipes and Reticulitermes virginicus. Behav. Ecol. Sociobiol. 2006, 59, 753–761. [Google Scholar] [CrossRef]
- Bulmer, M.S.; Adams, E.S.; Traniello, J.F. Variation in colony structure in the subterranean termite Reticulitermes flavipes. Behav. Ecol. Sociobiol. 2001, 49, 236–243. [Google Scholar] [CrossRef]
- Lee, S.-H.; Yang, R.-L.; Su, N.-Y. Tunneling response of termites to a pre-formed tunnel. Behav. Process. 2008, 79, 192–194. [Google Scholar] [CrossRef] [PubMed]
- Pitts-Singer, T.L.; Forschler, B.T. Influence of guidelines and passageways on tunneling behavior of Reticulitermes flavipes (Kollar) and R. virginicus (Banks)(Isoptera: Rhinotermitidae). J. Insect Behav. 2000, 13, 273–290. [Google Scholar] [CrossRef]
- Vargo, E.L.; Leniaud, L.; Swoboda, L.E.; Diamond, S.E.; Weiser, M.D.; Miller, D.M.; Bagnères, A.G. Clinal variation in colony breeding structure and level of inbreeding in the subterranean termites Reticulitermes flavipes and R. grassei. Mol. Ecol. 2013, 22, 1447–1462. [Google Scholar] [CrossRef] [PubMed]
- Eyer, P.-A.; Vargo, E.L. Breeding structure and invasiveness in social insects. Curr. Opin. Insect Sci. 2021, 46, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, K.; Fujimoto, M.; Goka, K. Sexual and asexual colony foundation and the mechanism of facultative parthenogenesis in the termite Reticulitermes speratus (Isoptera, Rhinotermitidae). Insectes Soc. 2004, 51, 325–332. [Google Scholar] [CrossRef]
- Vargo, E.L.; Labadie, P.E.; Matsuura, K. Asexual queen succession in the subterranean termite Reticulitermes virginicus. Proc. R Soc. Lond B Biol. Sci. 2012, 279, 813–819. [Google Scholar] [CrossRef]
- Matsuura, K.; Vargo, E.L.; Kawatsu, K.; Labadie, P.E.; Nakano, H.; Yashiro, T.; Tsuji, K. Queen Succession Through Asexual Reproduction in Termites. Science 2009, 323, 1687. [Google Scholar] [CrossRef]
- Grube, S.; Forschler, B.T. Census of Monogyne and Polygyne Laboratory Colonies Illuminates Dynamics of Population Growth in Reticulitermes flavipes (Isoptera: Rhinotermitidae). Ann. Entomol. Soc. Am. 2004, 97, 466–475. [Google Scholar] [CrossRef]
- Hellemans, S.; Roisin, Y. Asexual Queen Succession in Termites. eLS 2020, 1, 13–20. [Google Scholar]
- Vargo, E.L. Diversity of termite breeding systems. Insects 2019, 10, 52. [Google Scholar] [CrossRef] [PubMed]
- Chouvenc, T.; Su, N.-Y. Subterranean termites feeding on CSI baits for a short duration still results in colony elimination. J. Econ. Entomol. 2017, 110, 2534–2538. [Google Scholar] [CrossRef] [PubMed]
- Su, N.-Y.; Scheffrahn, R.H. Laboratory evaluation of two chitin synthesis inhibitors, hexaflumuron and diflubenzuron, as bait toxicants against Formosan and eastern subterranean termites (Isoptera: Rhinotermitidae). J. Econ. Entomol. 1993, 86, 1453–1457. [Google Scholar] [CrossRef]
- Vahabzadeh, R.D.; Gold, R.E.; Austin, J.W. Effects of four chitin synthesis inhibitors on feeding and mortality of the eastern subterranean termite, Reticulitermes flavipes (Isoptera: Rhinotermitidae). Sociobiology 2007, 50, 833–860. [Google Scholar]
- Su, N.-Y.; Ban, P.M.; Scheffrahn, R.H. Remedial baiting with hexaflumuron in above-ground stations to control structure-infesting populations of the Formosan subterranean termite (Isoptera: Rhinotermitidae). J. Econ. Entomol. 1997, 90, 809–817. [Google Scholar] [CrossRef]
- Su, N.-Y. A Fluid Bait for Remedial Control of Subterranean Termites. J. Econ. Entomol. 2015, 108, 274–276. [Google Scholar] [CrossRef]
- Aguero, C.M.; Eyer, P.-A.; Crippen, T.L.; Vargo, E.L. Reduced Environmental Microbial Diversity on the Cuticle and in the Galleries of a Subterranean Termite Compared to Surrounding Soil. Microb. Ecol. 2021, 1–10. [Google Scholar]
- Chouvenc, T.; Elliott, M.L.; Šobotník, J.; Efstathion, C.A.; Su, N.-Y. The termite fecal nest: A framework for the opportunistic acquisition of beneficial soil Streptomyces (Actinomycetales: Streptomycetaceae). Environ. Entomol. 2018, 47, 1431–1439. [Google Scholar] [CrossRef]
- Aguero, C.M.; Eyer, P.-A.; Martin, J.; Bulmer, M.S.; Vargo, E.L. Natural variation in a subterranean termite’s individual immunity is not related to colony inbreeding. Ecol. Evol. 2021. In press. [Google Scholar]
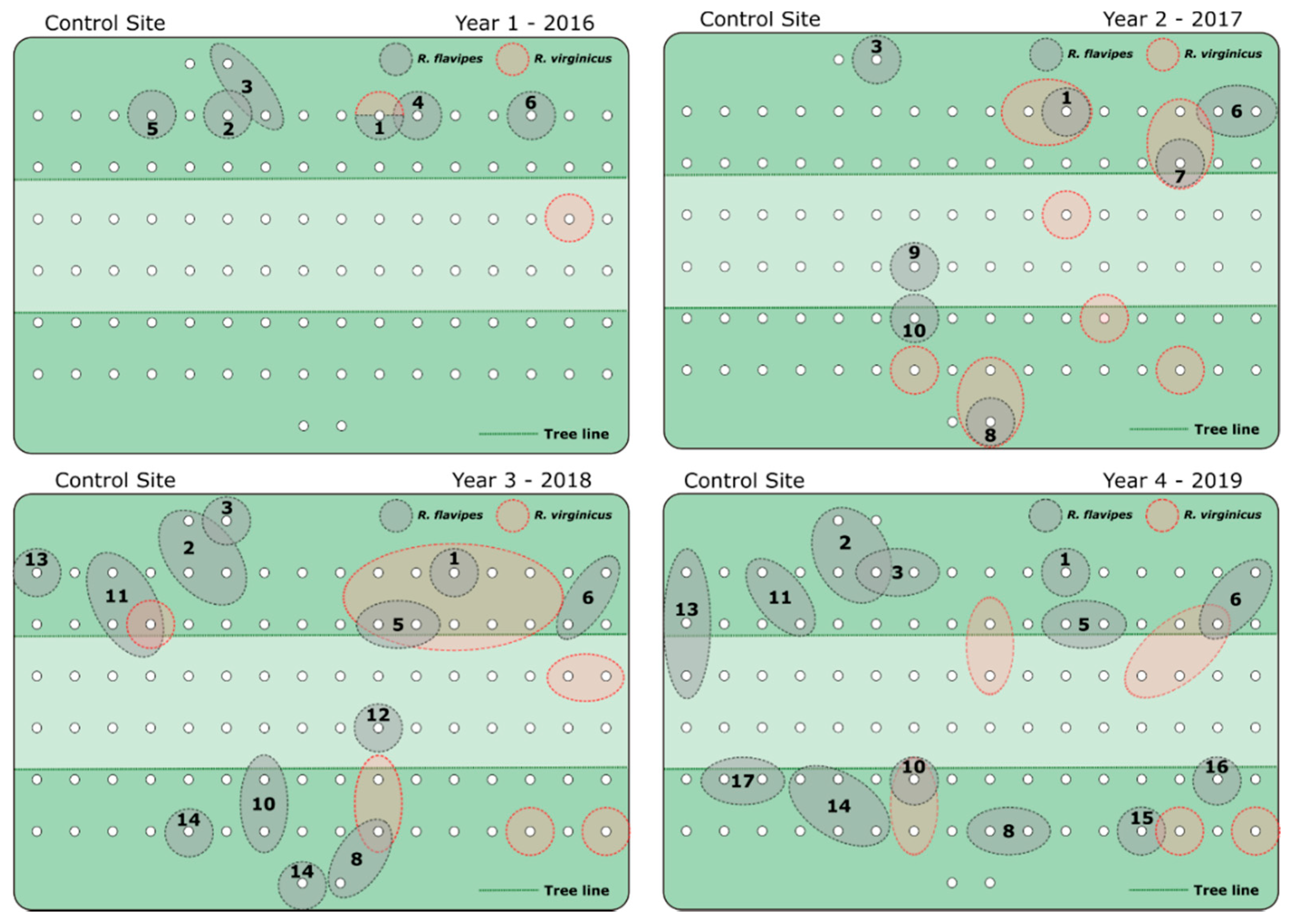
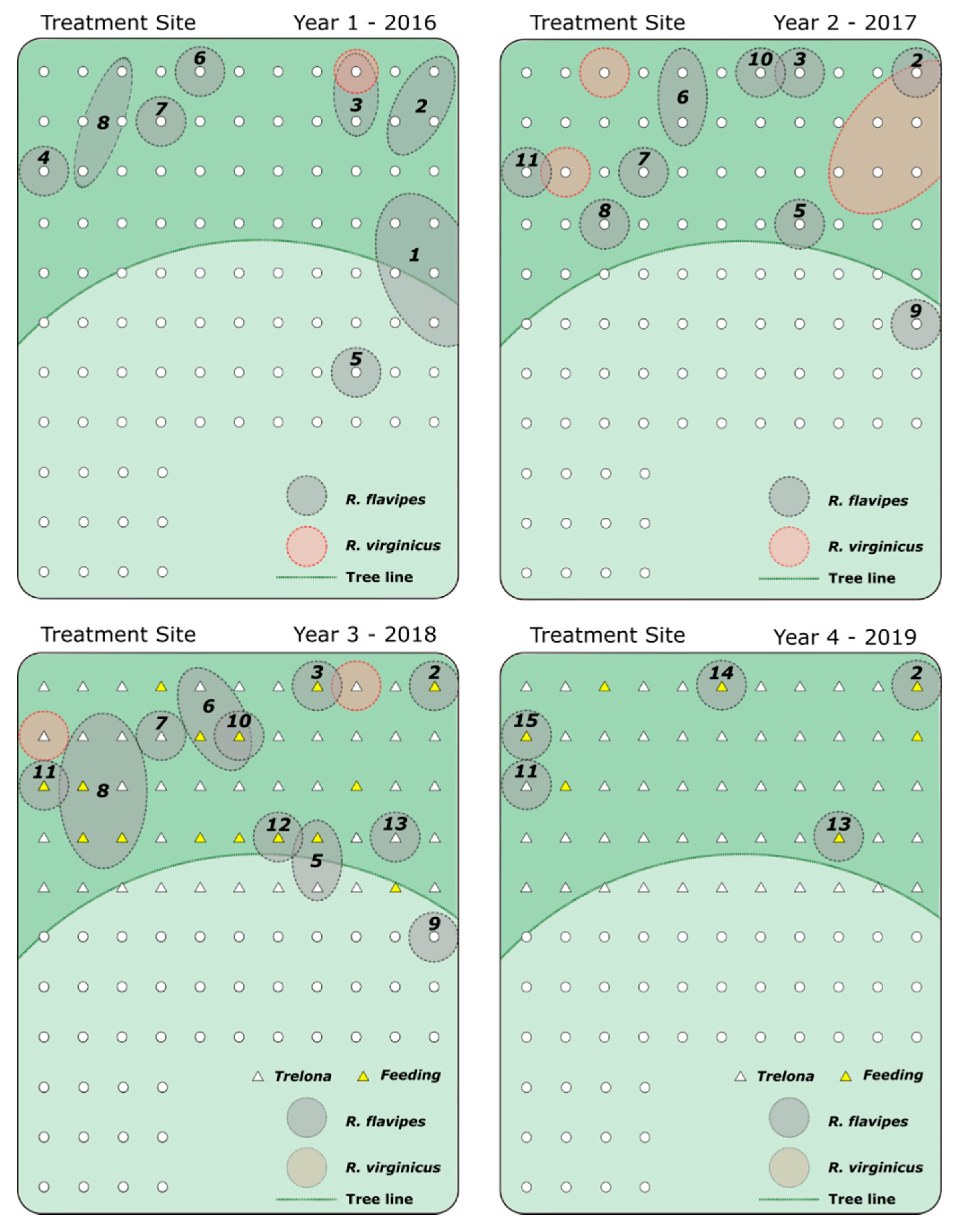
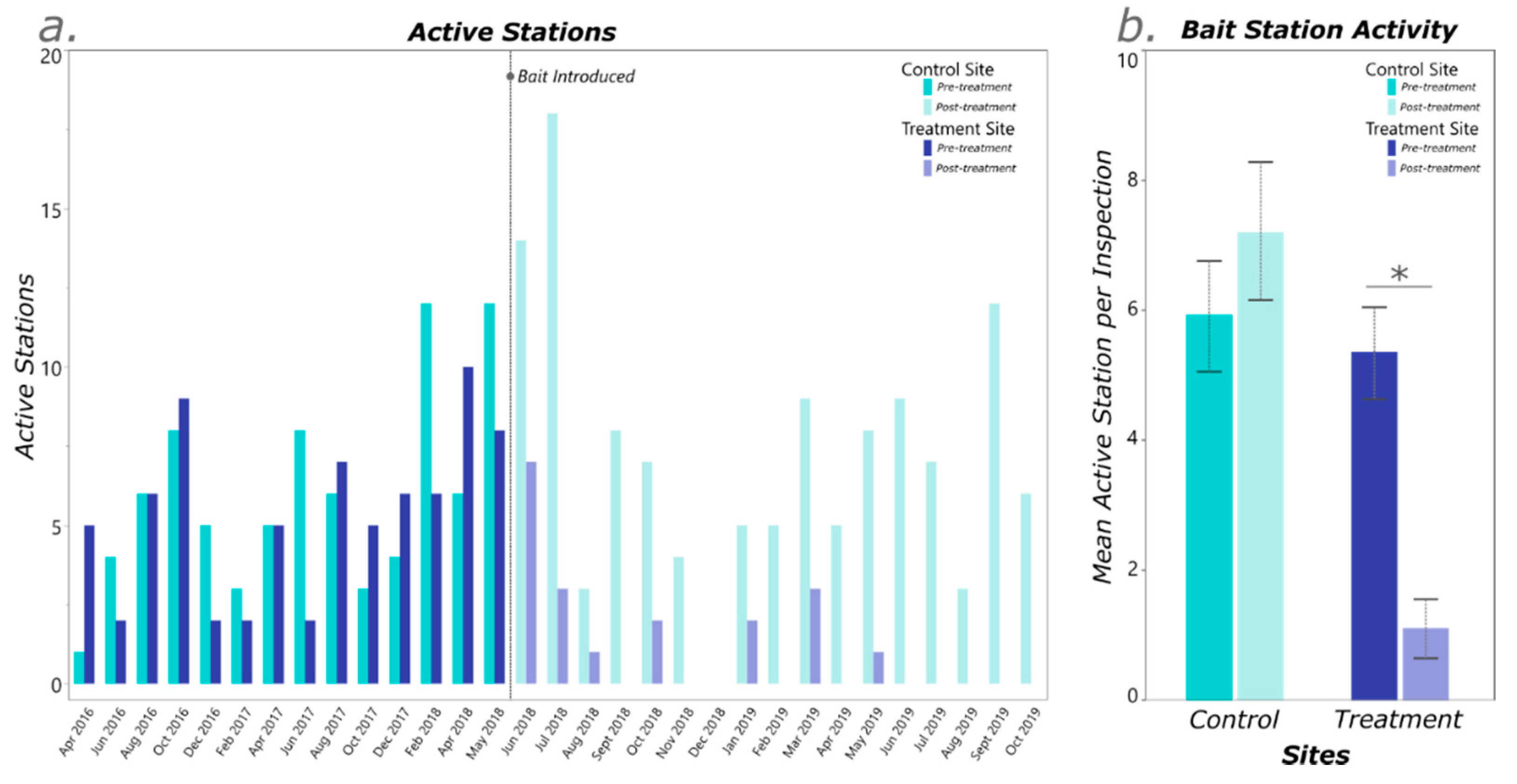

| Treatment Site | Control Site | ||||||
|---|---|---|---|---|---|---|---|
| Colony | Workers Genotyped | FIC | Family Type | Colony | Workers Genotyped | FIC | Family Type |
| 1 | 87 | −0.3338 | Simple | 1 | 57 | 0.3031 | Extended |
| 2 | 95 | −0.2628 | Simple | 2 | 123 | −0.4620 | Simple |
| 3 | 70 | −0.3608 | Simple | 3 | 84 | −0.3088 | Simple |
| 4 | 10 | 0.5610 | Extended | 4 | 9 | −0.2948 | Simple |
| 5 | 86 | −0.3590 | Simple | 5 | 87 | −0.3536 | Simple |
| 6 | 89 | 0.2324 | Extended | 6 | 177 | 0.4706 | Extended |
| 7 | 30 | −0.3126 | Simple | 7 | 7 | −0.5385 | Simple |
| 8 | 78 | 0.2104 | Extended | 8 | 116 | −0.2485 | Simple |
| 9 | 29 | −0.3764 | Simple | 9 | 10 | 0.2070 | Extended |
| 10 | 22 | −0.3057 | Simple | 10 | 107 | −0.2718 | Simple |
| 11 | 47 | 0.0439 | Extended | 11 | 89 | −0.4527 | Simple |
| 12 | 10 | −0.2289 | Simple | 12 | 7 | −0.0366 | Extended |
| 13 | 29 | −0.0950 | Simple | 13 | 56 | −0.2535 | Simple |
| 14 | 19 | −0.1060 | Simple | 14 | 101 | 0.4979 | Extended |
| 15 | 8 | 0.3058 | Extended | 15 | 30 | −0.2353 | Simple |
| v | 155 | −0.1764 | Simple | 16 | 9 | 0.1818 | Extended |
| 17 | 40 | −0.2966 | Simple | ||||
| v | 700 | −0.2772 | Simple | ||||
| Treatment Site | ||
|---|---|---|
| Colony ID | Amount of Bait Consumed (g) | Last Seen in Site |
| 1 | - | Oct-16 |
| 2 | 60 | May-19 |
| 3 | 90 | May-18 |
| 4 | - | May-16 |
| 5 | 250 | May-18 |
| 6 | 60 | Jun-18 |
| 7 | 30 | May-18 |
| 8 | 210 | Jun-18 |
| 9 | 30 | Jul-18 |
| 10 | 30 | May-18 |
| 11 | 10 | Jan-19 |
| 12 | 60 | Jul-18 |
| 13 | 120 | Jan-19 |
| 14 | 60 | May-19 |
| 15 | 30 | Mar-19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shults, P.; Richardson, S.; Eyer, P.-A.; Chura, M.; Barreda, H.; Davis, R.W.; Vargo, E.L. Area-Wide Elimination of Subterranean Termite Colonies Using a Novaluron Bait. Insects 2021, 12, 192. https://doi.org/10.3390/insects12030192
Shults P, Richardson S, Eyer P-A, Chura M, Barreda H, Davis RW, Vargo EL. Area-Wide Elimination of Subterranean Termite Colonies Using a Novaluron Bait. Insects. 2021; 12(3):192. https://doi.org/10.3390/insects12030192
Chicago/Turabian StyleShults, Phillip, Steven Richardson, Pierre-Andre Eyer, Madeleine Chura, Heather Barreda, Robert W. Davis, and Edward L. Vargo. 2021. "Area-Wide Elimination of Subterranean Termite Colonies Using a Novaluron Bait" Insects 12, no. 3: 192. https://doi.org/10.3390/insects12030192
APA StyleShults, P., Richardson, S., Eyer, P.-A., Chura, M., Barreda, H., Davis, R. W., & Vargo, E. L. (2021). Area-Wide Elimination of Subterranean Termite Colonies Using a Novaluron Bait. Insects, 12(3), 192. https://doi.org/10.3390/insects12030192






