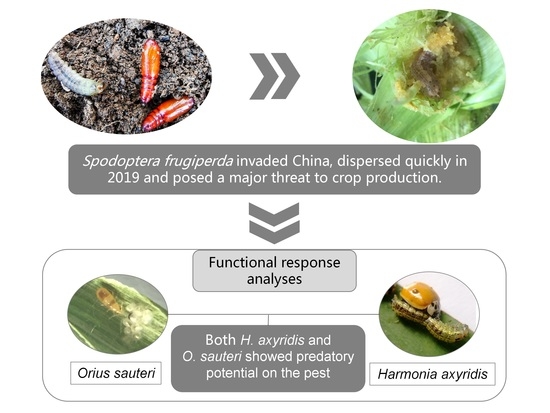
Predatory Ability of Harmonia axyridis (Coleoptera: Coccinellidae) and Orius sauteri (Hemiptera: Anthocoridae) for Suppression of Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Organisms
2.1.1. Spodoptera frugiperda
2.1.2. Orius sauteri and Harmonia axyridis
2.2. Experimental Methodology
2.2.1. Predatory Capacity of Orius sauteri and Harmonia axyridis on Spodoptera frugiperda Larvae
2.2.2. Predatory Capacity of Orius sauteri and Harmonia axyridis on Spodoptera frugiperda Eggs
2.2.3. Effects of Natural Enemy Density on Predatory Response
2.3. Statistics and Analysis
3. Results
3.1. Predation by Orius sauteri on Larvae and Eggs of Spodoptera frugiperda
3.2. Effect of Predator Density on Consumption of Spodoptera frugiperda by Orius sauteri and Harmonia axyridis
3.3. Predation by Harmonia axyridis on Larvae and Eggs of Spodoptera frugiperda
3.4. Functional Response of Predators Consuming Spodoptera frugiperda
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Boaventura, D.; Bolzan, A.; Padovez, F.E.; Okuma, D.M.; Omoto, C.; Nauen, R. Detection of a ryanodine receptor target-site mutation in diamide insecticide resistant fall armyworm, Spodoptera frugiperda. Pest Manag. Sci. 2020, 76, 47–54. [Google Scholar] [CrossRef]
- Brévault, T.; Ndiaye, A.; Badiane, D.; Bal, A.B.; Sembène, M.; Silvie, P.; Haran, J. First records of the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), in Senegal. Entomol. Gen. 2018, 3, 129–142. [Google Scholar] [CrossRef]
- Zhang, L.; Jin, M.H.; Zhang, D.D.; Jiang, Y.Y.; Liu, J.; Wu, K.M.; Xiao, Y.T. Molecular identification of invasive fall armyworm Spodoptera frugiperda in Yunnan Province. Plant Prot. 2019, 45, 19–24. [Google Scholar] [CrossRef]
- Sun, X.X.; Hu, C.X.; Jia, H.R.; Wu, Q.L.; Shen, X.J.; Zhao, S.Y.; Jiang, Y.Y.; Wu, K.M. Case study on the first immigration of fall armyworm, Spodoptera frugiperda invading into China. J. Integr. Agr. 2021, 20, 664–672. [Google Scholar] [CrossRef]
- Liao, Y.L.; Yang, B.; Xu, M.F.; Wei, L.; Wang, D.S.; Chen, K.W.; Chen, H.Y. First report of Telenomus remus parasitizing Spodoptera frugiperda and its field parasitism in southern China. J. Hymenopt. Res. 2019, 73, 95. [Google Scholar] [CrossRef]
- Wang, R.; Jiang, C.; Guo, X.; Chen, D.D.; You, C.; Zhang, Y.; Wang, M.T.; Li, Q. Potential distribution of Spodoptera frugiperda (JE Smith) in China and the major factors influencing distribution. Glob. Ecol. Consev. 2020, 21, e00865. [Google Scholar] [CrossRef]
- Sisay, B.; Tefera, T.; Wakgari, M.; Ayalew, G.; Mendesil, E. The efficacy of selected synthetic insecticides and botanicals against fall armyworm, Spodoptera frugiperda, in maize. Insects 2019, 10, 45. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.X.; Huang, J.M.; Ni, H.; Guo, D.; Yang, F.X.; Wang, X.; Wu, S.F.; Gao, C.F. Susceptibility of fall armyworm, Spodoptera frugiperda (JE Smmith), to eight insecticides in China, with special reference to lambda-cyhalothrin. Pestic. Biochem. Phys. 2020, 168, 104623. [Google Scholar] [CrossRef] [PubMed]
- Weisenburger, D.D. Human health-effects of agrichemicals use. Hum. Pathol. 1993, 24, 571–576. [Google Scholar] [CrossRef]
- Desneux, N.; Decourtye, A.; Delpuech, J.M. The sublethal effects of pesticides on beneficial arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.X.; Jaworski, C.C.; Desneux, N.; Zhang, F.; Yang, P.Y.; Wang, S. Long-term and large-scale releases of Trichogramma promote pesticide decrease in maize in northeastern China. Entomol. Gen. 2020, 40, 331–335. [Google Scholar] [CrossRef]
- Tepa-Yotto, G.T.; Tonnang, H.E.; Goergen, G.; Subramanian, S.; Kimathi, E.; Abdel-Rahman, E.M.; Flø, D.; Thunes, K.H.; Fiaboe, K.K.M.; Niassy, S.; et al. Global habitat suitability of Spodoptera frugiperda (JE Smith) (Lepidoptera, Noctuidae): Key parasitoids considered for its biological control. Insects 2021, 12, 273. [Google Scholar] [CrossRef] [PubMed]
- Shylesha, A.N.; Jalali, S.K.; Gupta, A.; Varshney, R.; Venkatesan, T.; Shetty, P.; Ojha, R.; Ganiger, P.C.; Navik, O.; Subaharan, S.; et al. Studies on new invasive pest Spodoptera frugiperda (JE Smith)(Lepidoptera: Noctuidae) and its natural enemies. J. Biol. Control 2018, 32, 145–151. [Google Scholar] [CrossRef] [Green Version]
- Sisay, B.; Simiyu, J.; Malusi, P.; Likhayo, P.; Mendesil, E.; Elibariki, N.; Wakgari, M.; Ayalew, G.; Tefera, T. First report of the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), natural enemies from Africa. J. Appl. Entomol. 2018, 142, 800–804. [Google Scholar] [CrossRef]
- Desneux, N.; O’neil, R.J.; Yoo, H.J.S. Suppression of population growth of the soybean aphid, Aphis glycines Matsumura, by predators: The identification of a key predator and the effects of prey dispersion, predator abundance, and temperature. Environ. Entomol. 2006, 35, 1342–1349. [Google Scholar] [CrossRef] [Green Version]
- Desneux, N.; Kaplan, I.; Yoo, H.J.S.; Wann, S.; O’Neil, R.J. Temporal synchrony mediates the outcome of indirect effects between prey via a shared predator. Entomol. Gen. 2019, 39, 127–136. [Google Scholar] [CrossRef]
- Desneux, N.; O’neil, R.J. Potential of an alternative prey to disrupt predation of the generalist predator, Orius insidiosus, on the pest aphid, Aphis glycines, via short-term indirect interactions. Bull. Entomol. Res. 2008, 98, 631–639. [Google Scholar] [CrossRef]
- Zhao, J.; Guo, X.; Tan, X.; Desneux, N.; Zappala, L.; Zhang, F.; Wang, S. Using Calendula officinalis as a floral resource to enhance aphid and thrips suppression by the flower bug Orius sauteri (Hemiptera: Anthocoridae). Pest. Manag. Sci. 2017, 73, 515–520. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Xiang, M.; Hou, Y.Y.; Yang, X.B.; Dai, H.J.; Li, J.T.; Zang, L.S. Impact of egg deposition period on the timing of adult emergence in Trichogramma parasit. Entomol. Gen. 2019, 39, 339–346. [Google Scholar] [CrossRef]
- Liu, P.; Jia, W.; Zheng, X.; Zhang, L.; Sangbaramou, R.; Tan, S.Q.; Liu, Y.Q.; Shi, W.P. Predation functional response and life table parameters of Orius sauteri (Hemiptera: Anthocoridae) feeding on Megalurothrips usitatus (Thysanoptera: Thripidae). Fla. Entomol. 2018, 101, 254–259. [Google Scholar] [CrossRef]
- Wang, S.; Tan, X.L.; Michaud, J.P.; Zhang, F.; Guo, X.J. Light intensity and wavelength influence development, reproduction and locomotor activity in the predatory flower bug Orius sauteri (Poppius) (Hemiptera: Anthocoridae). BioControl 2013, 58, 667–674. [Google Scholar] [CrossRef] [Green Version]
- Holling, C.S. Some characteristics of simple types of predation and parasitism1. Can. Entomol. 1959, 91, 385–398. [Google Scholar] [CrossRef]
- Miller, D.A.; Grand, J.B.; Fondell, T.F.; Anthony, M. Predator functional response and prey survival: Direct and indirect interactions affecting a marked prey population. J. Anim. Ecol. 2006, 75, 101–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassell, M.P.; Lawton, J.H.; Beddington, J.R. Sigmoid functional responses by invertebrate predators and parasitoids. J. Anim. Ecol. 1977, 249–262. [Google Scholar] [CrossRef] [Green Version]
- Zhang, A.S.; Yu, Y.; Li, L.L.; Zhang, S.C.; Men, X.Y. Functional response and searching rate of Orius sauteri adults on Frankliniella occidentalis nymphs. Chin. J. Ecol. 2007, 26, 1233–1237. [Google Scholar]
- Ge, Y.; Camara, I.; Wang, Y.; Liu, P.P.; Zhang, L.; Xing, Y.J.; Li, A.M.; Shi, W.P. Predation of Aphis craccivora (Hemiptera: Aphididae) by Orius sauteri (Hemiptera: Anthocoridae) under different temperatures. J. Econ. Entomol. 2018, 111, 2599–2604. [Google Scholar] [CrossRef]
- Gitonga, L.M.; Overholt, W.A.; Löhr, B.; Magamboa, J.K.; Muekec, J.M. Functional response of Orius albidipennis (Hemiptera: Anthocoridae) to Megalurothrips sjostedti (Thysanoptera: Thripidae). Biol. Control 2002, 24, 1–6. [Google Scholar] [CrossRef]
- Lu, Y.; Wu, K.; Jiang, Y.; Guo, Y.Y.; Desneux, N. Widespread adoption of Bt cotton and insecticide decrease promotes biocontrol services. Nature 2012, 487, 362–365. [Google Scholar] [CrossRef]
- Ragsdale, D.W.; Landis, D.A.; Brodeur, J.; Heimpel, G.E.; Desneux, N. Ecology and management of the soybean aphid in North America. Annu. Rev. Entomol. 2011, 56, 375–399. [Google Scholar] [CrossRef] [Green Version]
- Hulle, M.; Chaubet, B.; Turpeau, E.; Simon, J.C. Encyclop’Aphid: A website on aphids and their natural enemies. Entomol. Gen. 2020, 40, 97–101. [Google Scholar] [CrossRef]
- Thomine, E.; Rusch, A.; Supplisson, C.; Monticelli, L.; Desneux, E.; Lavoir, A.; Desneux, N. Highly diversified crop systems can promote the dispersal and foraging activity of the generalist predator Harmonia axyridis. Entomol. Gen. 2020, 40, 133–145. [Google Scholar] [CrossRef] [Green Version]
- Koch, R.L.; Hutchison, W.D.; Venette, R.C.; Heimpel, G.E. Susceptibility of immature monarch butterfly, Danaus plexippus (Lepidoptera: Nymphalidae: Danainae), to predation by Harmonia axyridis (Coleoptera: Coccinellidae). Biol. Control 2003, 28, 265–270. [Google Scholar] [CrossRef]
- Feng, Y.; Zhou, Z.X.; An, M.R.; Yu, X.L.; Liu, T.X. The effects of prey distribution and digestion on functional response of Harmonia axyridis (Coleoptera: Coccinellidae). Biol. Control 2018, 124, 74–81. [Google Scholar] [CrossRef]
- Huang, Z.; Qureshi, J.; Zhou, X.; Pu, Z.; Chen, G.; Yu, J.; Zhang, H. Predation and functional response of the multi-coloured Asian ladybeetle Harmonia axyridis on the adult Asian citrus psyllid Diaphorina citri. Biocontrol Sci. Technol. 2019, 29, 293–307. [Google Scholar] [CrossRef]
- Chen, X.; Xiao, D.; Du, X.; Zhang, F.; Zang, L.; Wang, S. Impact of polymorphism and abiotic conditions on prey consumption by Harmonia axyridis. Entomol. Gen. 2019, 39, 251–258. [Google Scholar] [CrossRef]
- Wang, Y.S.; Yao, F.L.; Soares, M.A.; Basiri, S.E.; Edwige, D.; Campos, M.; Lavoir, A.; Desneux, N. Effects of four non-crop plants on life history traits of the lady beetle Harmonia axyridis. Entomol. Gen. 2020, 40, 243–252. [Google Scholar] [CrossRef]
- Jaworski, C.C.; Xiao, D.; Xu, Q.; Ramirez-Romero, G.; Guo, X.J.; Wang, S.; Desneux, N. Varying the spatial arrangement of synthetic herbivore-induced plant volatiles and companion plants to improve conservation biological control. J. Appl. Ecol. 2019, 56, 1176–1188. [Google Scholar] [CrossRef]
- Wang, S.; Di, N.; Chen, X.; Zhang, F.; Biondi, A.; Desneux, N.; Wang, S. Life history and functional response to prey density of the flower bug Orius sauteri attacking the fungivorous sciarid fly Lycoriella pleuroti. J. Pest. Sci. 2019, 92, 715–722. [Google Scholar] [CrossRef]
- Van Lenteren, J.C.; Manzaroli, G. Evaluation and use of predators and parasitoids for biological control of pests in greenhouses. In Integrated Pest and Disease Management in Greenhouse Crops; Springer: Dordrecht, The Netherlands, 1999; pp. 183–201. [Google Scholar] [CrossRef]
- Pehlivan, S.; Alinc, T.; Achiri, T.D.; Atakan, E. Functional responses of two predatory bugs (Hemiptera: Anthocoridae) to changes in the abundance of Tetranychus urticae (Acari: Tetranychidae) and Bemisia tabaci (Hemiptera: Aleyrodidae). Eur. J. Entomol. 2020, 117, 49–55. [Google Scholar] [CrossRef] [Green Version]
- Zeng, G.; Zhi, J.R.; Zhang, C.R.; Zhang, T.; Ye, J.; Zhou, L.; Hu, C.; Ye, M. Orius similis (Hemiptera: Anthocoridae): A promising candidate predator of Spodoptera frugiperda (Lepidoptera: Noctuidae). J. Econ. Entomol. 2021, 114, 582–589. [Google Scholar] [CrossRef]
- Aragón-Sánchez, M.; Román-Fernández, L.R.; Martínez-García, H.; Aragón-García, A.; Pérez-Moreno, I.; Marco-Mancebón, V. Rate of consumption, biological parameters, and population growth capacity of Orius laevigatus fed on Spodoptera exigua. BioControl 2018, 63, 785–794. [Google Scholar] [CrossRef]
- Isenhour, D.J.; Layton, R.C.; Wiseman, B.R. Potential of adult Orius insidiosus [Hemiptera: Anthocoridae] as a predator of the fall armyworm, Spodoptera frugiperda [Lepidoptera: Noctuidae]. Entomophaga 1990, 35, 269–275. [Google Scholar] [CrossRef]
- Yang, S.; Yang, S.Y.; Zhang, C.P.; Wei, J.N.; Kuang, R.P. Population dynamics of Myzus persicae on tobacco in Yunnan Province, China, before and after augmentative releases of Aphidius gifuensis. Biocontrol Sci. Tech. 2009, 19, 219–228. [Google Scholar] [CrossRef]
- Lee, J.H.; Kang, T.J. Functional response of Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae) to Aphis gossypii Glover (Homoptera: Aphididae) in the Laboratory. Biol. Control 2004, 31, 306–310. [Google Scholar] [CrossRef]
- Dutra, C.C.; Koch, R.L.; Burkness, E.C.; Meissle, M.; Romeis, J.; Hutchison, W.D.; Fernandes, M.G. Harmonia axyridis (Coleoptera: Coccinellidae) exhibits no preference between Bt and non-Bt maize fed Spodoptera frugiperda (Lepidoptera: Noctuidae). PLoS ONE 2012, e44867. [Google Scholar] [CrossRef] [PubMed]
- Islam, Y.; Shah, F.M.; Shah, M.A.; Khan, M.; Rasheed, M.; Rehman, S.; Ali, S.; Zhou, X. Temperature-dependent functional response of Harmonia axyridis (Coleoptera: Coccinellidae) on the eggs of Spodoptera litura (Lepidoptera: Noctuidae) in laboratory. Insects 2020, 11, 583. [Google Scholar] [CrossRef] [PubMed]
- Symondson, W.O.C.; Sunderland, K.D.; Greenstone, M.H. Can generalist predators be effective biocontrol agents? Ann. Rev. Entomol. 2002, 47, 561–594. [Google Scholar] [CrossRef] [Green Version]
- Hou, Y.Y.; Yang, X.; Zang, L.S.; Zhang, C.; Monticelli, l.; Desneu, N. Effect of oriental armyworm Mythimna separata egg age on the parasitism and host suitability for five Trichogramma species. J. Pest. Sci. 2018, 91, 1181–1189. [Google Scholar] [CrossRef]
- Kenis, M.; Du Plessis, H.; Van den Berg, J.; Ba, M.N.; Goergen, G.; Kwadjo, K.C.; Baoua, I.; Tefera, T.; Buddie, A.; Cafà, G.; et al. Telenomus remus, a candidate parasitoid for the biological control of Spodoptera frugiperda in Africa, is already Present on the Continent. Insects 2019, 10, 92. [Google Scholar] [CrossRef] [Green Version]
- Karp, D.S.; Chaplin-Kramer, R.; Meehan, T.D.; Martind, E.A.; DeClerck, F.; Grab, H.; Gratton, C.; Hunt, L.; Larsen, A.; Martínez-Salinas, A.; et al. Crop pests and predators exhibit inconsistent responses to surrounding landscape composition. Proc. Nat. Acad. Sci. USA 2018, 11, E7863–E7870. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zang, L.S.; Wang, S.; Zhang, F.; Desneux, N. Biological control with Trichogramma in China: History, present status, and perspectives. Annu. Rev. Entomol. 2021, 66, 463–484. [Google Scholar] [CrossRef] [PubMed]



| Orius sauteri | Prey | Fitted Formula | R2 | a′ | Th | Na-max | X2, p |
|---|---|---|---|---|---|---|---|
| Nymph | larvae | Na = 0.5626N/(1 − 0.0008N) | 0.8591 | 0.5602 | 0.0008 | 700.24 | 3.333, 1.000 |
| eggs | Na = 0.7444N/(1 + 0.049N) | 0.9947 | 0.7444 | 0.049 | 15.19 | 8.000, 0.924 |
| Harmonia axyridis | Prey | Fitted Formula | R2 | a′ | Th | Na-max | X2, p |
|---|---|---|---|---|---|---|---|
| Adult | larvae | Na = 0.8322N/(1 + 0.0006N) | 0.9322 | 0.8407 | 0.0006 | 1401.1 | 2.400, 1.000 |
| eggs | Na = 1.1112N/(1 + 0.0085N) | 0.9654 | 1.1112 | 0.0085 | 130.73 | 18.250, 0.108 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di, N.; Zhang, K.; Xu, Q.; Zhang, F.; Harwood, J.D.; Wang, S.; Desneux, N. Predatory Ability of Harmonia axyridis (Coleoptera: Coccinellidae) and Orius sauteri (Hemiptera: Anthocoridae) for Suppression of Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). Insects 2021, 12, 1063. https://doi.org/10.3390/insects12121063
Di N, Zhang K, Xu Q, Zhang F, Harwood JD, Wang S, Desneux N. Predatory Ability of Harmonia axyridis (Coleoptera: Coccinellidae) and Orius sauteri (Hemiptera: Anthocoridae) for Suppression of Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). Insects. 2021; 12(12):1063. https://doi.org/10.3390/insects12121063
Chicago/Turabian StyleDi, Ning, Kai Zhang, Qingxuan Xu, Fan Zhang, James D. Harwood, Su Wang, and Nicolas Desneux. 2021. "Predatory Ability of Harmonia axyridis (Coleoptera: Coccinellidae) and Orius sauteri (Hemiptera: Anthocoridae) for Suppression of Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae)" Insects 12, no. 12: 1063. https://doi.org/10.3390/insects12121063
APA StyleDi, N., Zhang, K., Xu, Q., Zhang, F., Harwood, J. D., Wang, S., & Desneux, N. (2021). Predatory Ability of Harmonia axyridis (Coleoptera: Coccinellidae) and Orius sauteri (Hemiptera: Anthocoridae) for Suppression of Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). Insects, 12(12), 1063. https://doi.org/10.3390/insects12121063