Pathogenicity Detection and Genome Analysis of Two Different Geographic Strains of BmNPV
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Virus Collection and Purification
2.2.2. Determination of Pathogenicity of Different BmNPV to B. mori
2.2.3. Negative-Staining Electron Microscopy to Observe BmNPV Particles
2.2.4. Sequencing of BmNPV ZJ and BmNPV YN Genomes
2.2.5. Analysis of BmNPV ZJ and BmNPV YN Genome Sequences
2.2.6. Detection of Gene-Relative Expression of BmNPV by qPCR
2.2.7. BmNPV YN and BmNPV ZJ Differential Genes Analysis
3. Results
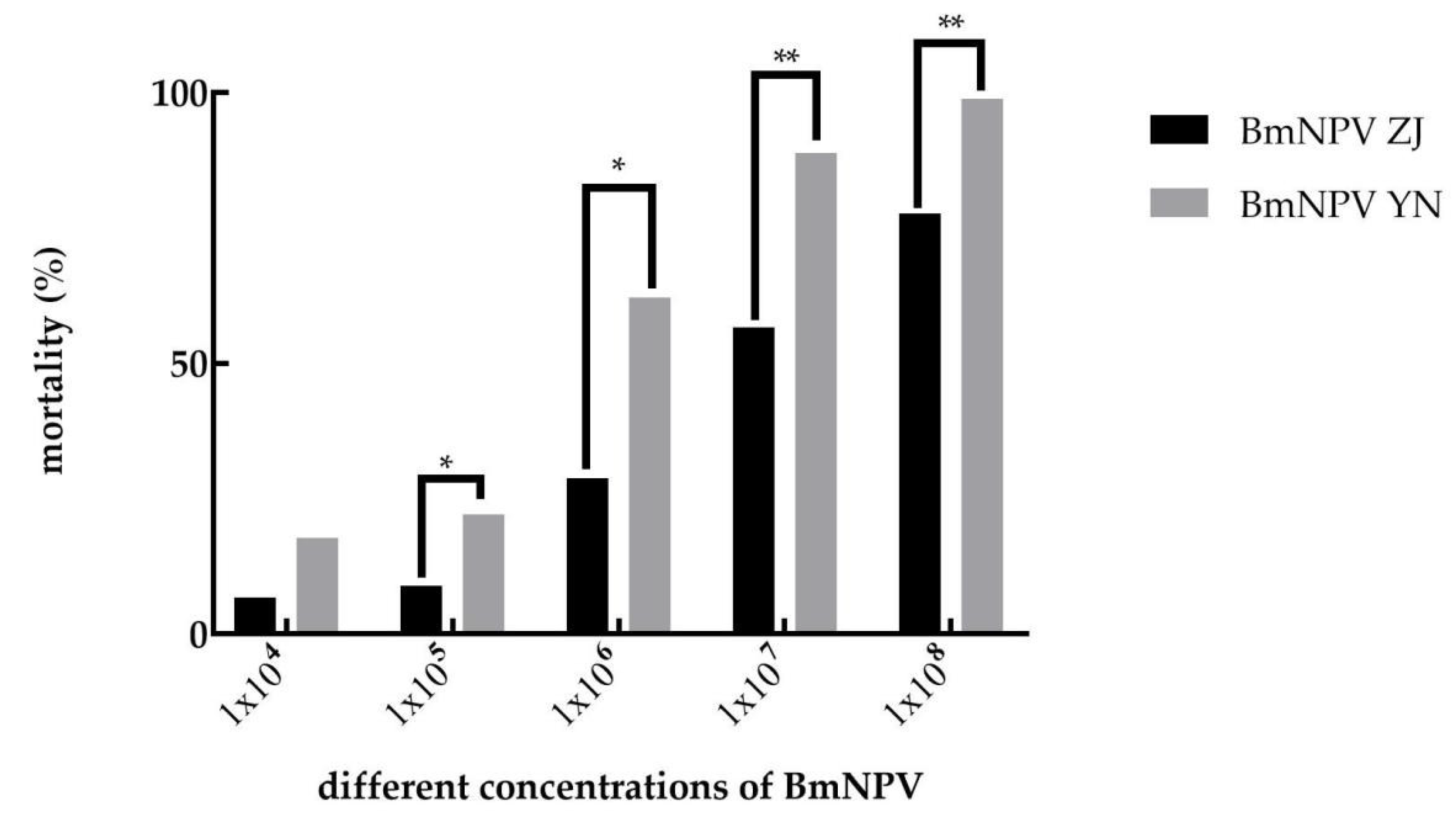
3.1. The Pathogenicity of BmNPV ZJ and BmNPV YN to B. mori
3.2. The Morphology and Size of BmNPV ZJ and BmNPV YN
3.3. The Relative Expression of Genes of BmNPV ZJ and BmNPV YN in the Midgut of Baiyu
3.4. Structural Characteristics of Virus Genomes
3.4.1. Genome Characteristics of BmNPV ZJ
3.4.2. Genome Characteristics of BmNPV YN
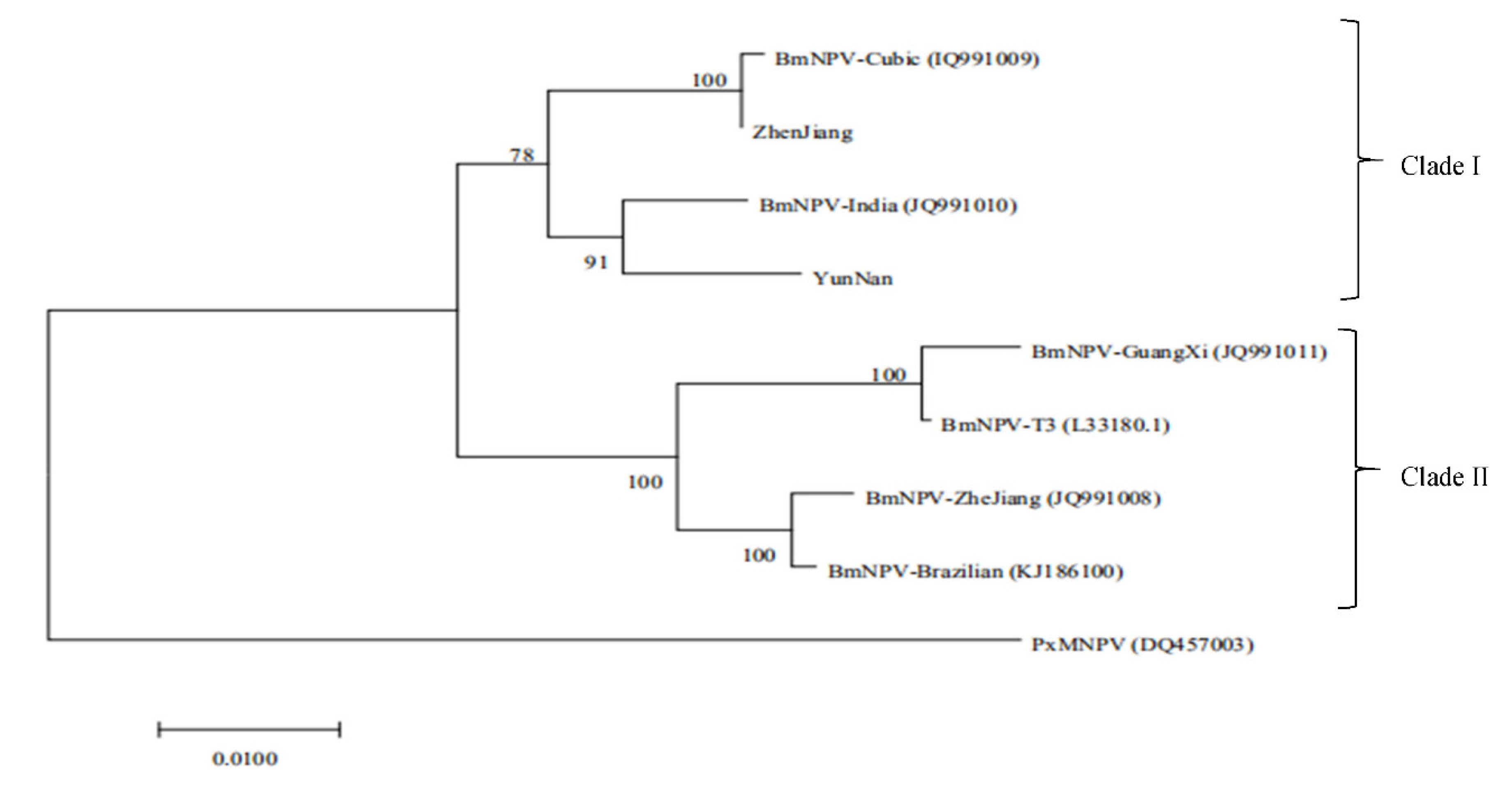
3.4.3. Genome Comparison of BmNPV
3.5. Detection and Analysis of Single Nucleotide Polymorphism and InDels in the Two Viral Genomes
3.6. Differential Genes Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yuan, M.J.; Wu, W.B.; Liu, C.; Wang, Y.J.; Hu, Z.Y.; Yang, K.; Pang, Y. A highly conserved baculovirus gene p48 (ac103) is essential for BV production and ODV envelopment. Virology 2008, 379, 87–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maeda, S.; Majima, K. Molecular cloning and physical mapping of the genome of Bombyx mori nuclear polyhedrosis virus. J. Gen. Virol. 1990, 71, 1851. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Q. Summary table of nationalsericulture production in 2019. Chinses Seric. 2020, 41, 70–72. [Google Scholar]
- Xu, A.Y.; Qian, H.Y.; Sun, P.J.; Liu, M.Z.; Lin, C.Q.; Li, G.; Li, L.; Zhang, Y.H.; Zhao, G.D. Breeding of a new silkworm variety “Huakang 3” with resistance to Bombyx mori Nucleopolyhedrovirus disease. Sci. Seric. 2019, 45, 201–211. [Google Scholar]
- Xu, A.Y.; Lin, C.Q.; Qian, H.Y.; Sun, P.J.; Zhang, Y.H.; Liu, M.Z.; Li, L. Breeding of a new silkworm variety “Huakang 2” with tolerance to Bombyx mori nucleopolyhedrovirus disease. Sci Seric. 2013, 39, 275–282. [Google Scholar]
- Shi, M.N.; Bi, L.H.; Gu, J.D.; Fei, M.H.; Qi, G.J.; Wei, B.Y.; Huang, J.T.; Huang, L.L.; Su, H.M.; Meng, Y.Y. Breeding of a new highly resistant nuclear polyhedrosis disease silkworm variety Guican N2. Guangxi Seric. 2012, 49, 1–12. [Google Scholar]
- Xu, A.Y.; Lin, C.Q.; Qian, H.Y.; Sun, P.J.; Zhang, Y.H.; Liu, M.Z.; Li, L. Brief introduction of new varieties of silkworm resistant to BmNPV. In Proceedings of the Tenth Domestic (Tussah) Silkworm Genetics and Breeding and Seed Breeding, Jilin, China, 19 July 2013; Chinese Silkworm Society: Jilin, China, 2014; pp. 184–185. [Google Scholar]
- Zang, Y.L.; Hunang, L.; Wang, Y.W.; Wang, H.L.; Gao, H.J. A preliminary report on the feeding of silkworm varieties of “Huakang 1” and “Huakang 2” with tolerance to BmNPV disease in Taian. Hebei J. For. Orchard. Res. 2014, 29, 435–438. [Google Scholar]
- Hu, S.Y.; Luo, C.B.; Sun, Y.P.; Yang, S.T.; Jiang, H.; Yang, B.; Yu, W.Z.; Huang, G.H. A brief report on feeding test of new silkworm variety Huakang 2 in Qingzhen city. Chinses Seric. 2016, 24–27. [Google Scholar] [CrossRef]
- Luo, P. Discussion on the prevention and control methods of silkworm nuclear polyhedrosis virus disease in subtropical regions. Chinses Seric. 2017, 38, 59–62,67. [Google Scholar]
- Shi, M.N.; Tang, L.; Huang, H.Y.; Wei, T.X.; Pan, Z.X.; Tang, T.X.; Pan, Z.X.; Qi, G.J.; Pu, Y.X.; Mo, Y.X. Application of new silkworm variety ‘Guican N2’. Guangxi Seric. 2015, 52, 27–31. [Google Scholar]
- Tang, F.F.; Zhang, Y.H.; Shao, Y.L.; Zhu, F.; Bai, X.R. Virulence and phylogenetic analysis of Bombyx mori nucleopolyhedrovirus isolates from Yunnan, southwestern China. Acta Entomol. Sin. 2018, 61, 42–51. [Google Scholar]
- Fu, J.Y.; Xi, Y.; Tang, M.J.; Yin, K.S. Study on the relationship between virulence and genetic structure of four wild isolates of Euproctis pseudoconspersa nuclear polyhedrosis virus. J. Tea Sci. 2011, 31, 289–294. [Google Scholar]
- Fuxa, J.R. Ecology of insect nucleopolyhedroviruses. Agric. Ecosyst. Environ. 2004, 103, 27–43. [Google Scholar] [CrossRef]
- Tang, F.F.; Shao, X.L.; Zhong, J.; Zhang, Y.H.; Huang, P.; Dong, Z.P.; Liao, P.F.; Bai, X.R. A preliminary study on molecular identification of Bombyx mori nucleopolyhedrovirus strains. Sci. Seric. 2014, 1030–1035. [Google Scholar] [CrossRef]
- Zhou, J.B. Identification of Thai Strain of Bombyx mori Nuclear Polyhedrosis Virus and Analysis of bro Gene Family. Master’s Thesis, Anhui Agricultural University, Hefei, China, 2012. [Google Scholar]
- Xu, Y.P.; Cheng, R.L.; Xi, Y.; Zhang, C.X. Genomic diversity of Bombyx mori nucleopolyhedrovirus strains. Genomics 2013, 102, 63–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, J.C.; Jin, F.L.; Xu, X.Y.; Tan, P.C. Comparative study on median lethal dose of BmNPV to B. mori in different regions. Guangdong Seric. 2001, 035, 35–37. [Google Scholar]
- Bai, X.R.; Ran, R.F.; Dong, Z.P.; Dong, J.H.; Huang, P. Study on virulence of BmNPV to Bombyx mori in different areas of Yunnan. Southwest China J. Agric. Sci. 2010, 23, 2098–2101. [Google Scholar]
- Wang, X.; Huang, X.H.; Jiang, M.G.; Tang, L.; Dong, G.Q.; Huang, S.H.; Shi, M.N.; Pan, Z.X. Epidemic factors of Bombyx mori hemolymph-type septic disease in Guangxi and its molecular phylogenetic analysis. J. South. Agric. 2020, 051, 669–676. [Google Scholar]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Bioinf. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Massouras, A.; Hens, K.; Gubelmann, C.; Uplekar, S.; Decouttere, F.; Rougemont, J.; Cole, S.T.; Deplancke, B. Primer-initiated sequence synthesis to detect and assemble structural variants. Nat. Methods 2010, 7, 485–486. [Google Scholar] [CrossRef] [PubMed]
- Kang, W.; Suzuki, M.; Zemskov, E.; Okano, K.; Maeda, S. Characterization of Baculovirus Repeated Open Reading Frames (bro) in Bombyx moriNucleopolyhedrovirus. J. Virol. 1999, 73, 10339–10345. [Google Scholar] [CrossRef] [Green Version]
- Mckenna, A.; Hanna, M.; Banks, E.; Sivachenko, A.; Cibulskis, K.; Kernytsky, A.; Garimella, K.; Altshuler, D.; Gabriel, S.; Daly, M. The Genome Analysis Toolkit: A MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010, 20, 1297–1303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cingolani, P. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 2012, 6, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.B.; Li, X.Q.; De-Eknamkul, W.; Suraporn, S.; Xu, J.P. Identification of a new Bombyx mori nucleopolyhedrovirus and analysis of its bro gene family. Virus Genes 2012, 44, 539–547. [Google Scholar] [CrossRef]
- Herniou, W.A.; Olszewski, J.A.; Cory, J.S.; O’Reilly, D.R. The genome sequence and evolution of baculoviruses. Annu. Rev. Entomol. 2003, 48, 211–234. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.Y.; Li, G.; Zhao, G.D.; Liu, M.Z.; Sun, P.J.; Xu, A.Y. Identification of new silkworm vrieties resistant to nuclear polyhedrosis Virus to BmNPV. Chinses Seric. 2017, 38, 15–22. [Google Scholar]
- Huang, C.; Fan, X.H.; Jiang, Y.H.; Song, D.Z.; Gao, L.Q.; Huang, Q.G.; Lai, Z.P. Anti-tumor effect of Newcastle disease virus strain D817 against nude mouse xenografts of human colon carcinoma. Zhonghua Zhong Liu Za Zhi Chin. J. Oncol. 2009, 31, 490. [Google Scholar]
- Wang, Y.Y.; Lu, J.; Liu, H.; Ye, R. Comparison of the infectivity of three virus strains of the genus A-coronavirus to cells derived from different hosts. China Sci. 2013. Available online: http://www.paper.edu.cn/releasepaper/content/201301-1114 (accessed on 26 September 2021).
- Lu, W.Q.; Wei, P.W. Changes and Comparison of virus concentrations of six SMV strains in different soybean varieties. Soybean Sci. 1991, 6, 27–34. [Google Scholar]
- Ma, X.C. Sequence Analysis of Genomes from Three Nucleopolyhedroviruses. Ph.D. Thesis, Zhejiang University, Hangzhou, China, 2006. [Google Scholar]



| Gene | Primer Sequences | Length/(bp) |
|---|---|---|
| actin-3 | F: 5′-CGGCTACTCGTTCACTACC-3′ | 147 |
| R: 5′-CCGTCGGGAAGTTCGTAAG-3′ | ||
| ie-1 | F: 5′-GGACGAATACTTGGACGAT-3′ | 237 |
| R: 5′-GAGAACCTGTTGGAATTGTAG-3′ | ||
| p143 | F: 5′-GCACGGCAATACTTATCATC-3′ | 120 |
| R: 5′-TGAGCACCAACAATAGTCC-3′ | ||
| vp39 | F: 5′-ACACGGAGGAATTGAGATT-3′ | 116 |
| R: 5′-GATGTCACTGCTTCTTATCG-3′ | ||
| polh | F: 5′-CTACAAGTTCCTCGCTCAA-3′ | 163 |
| R: 5′-CTCGCTGTGGATGTTCAT-3′ |
| Quality Control Items | SNP | InDel |
|---|---|---|
| QualByDepth | ≥2.0 | ≥2.0 |
| RMSMappingQuality | ≥40.0 | - |
| FisherStrand | ≤60.0 | ≤200.0 |
| StrandOddsRatio | ≤3.0 | ≤10.0 |
| MappingQualityRankSumTest | ≥−12.5 | ≥−12.5 |
| ReadPosRankSumTest | ≥−8.0 | ≥−8.0 |
| Variety | Strain | Regression Equation | LC50 | 95% Confidence Value | Slope of Regression Line/SE |
|---|---|---|---|---|---|
| Baiyu | BmNPV ZJ | y = −4.267 + 0.627x | 6.45 × 106 | 3.88 × 106–1.14 × 107 | 0.056 |
| Baiyu | BmNPV YN | y = −4.426 + 0.796x | 3.62 × 105 | 1.98 × 105–6.45 × 105 | 0.063 |
| Items | BmNPV ZJ | BmNPV YN | BmNPV T3 |
|---|---|---|---|
| Size (base) | 135,895 | 143,180 | 128,413 |
| G + C content (%) | 40.39 | 39.71 | 40 |
| Protein coding genes | 141 | 145 | 136 |
| Min length (base) | 111 | 111 | 61 |
| Max length (base) | 2964 | 4050 | 1222 |
| Average length (base) | 808.23 | 882.52 | 852.154 |
| Total coding gene (base) | 113,961 | 127,965 | 115,893 |
| Coding ratio (%) | 83.86 | 89.37 | 90.25 |
| Reference Strain | Nucleotide Homology/(%) | Reference Strain | Amino Acid Sequence/(%) | ||
|---|---|---|---|---|---|
| BmNPV ZJ (N) | BmNPV YN (N) | BmNPV ZJ (AA) | BmNPV YN (AA) | ||
| Brazilian | 94.3 | 94.2 | Brazilian | 93.7 | 92.5 |
| Cubic | 99.6 | 95.7 | Cubic | 99.7 | 96.3 |
| Guangxi | 92.1 | 93.3 | Guangxi | 91.7 | 92.2 |
| India | 96.8 | 97.3 | India | 96.6 | 97.1 |
| T3 | 95.9 | 94.1 | T3 | 92.0 | 92.5 |
| Zhejiang | 95.5 | 94.3 | Zhejiang | 94.0 | 92.8 |
| Zhenjiang | - | 95.9 | Zhenjiang | - | 95.9 |
| Yunnan | 96.9 | - | Yunnan | 96.6 | - |
| Type | SNP Number | Region |
|---|---|---|
| Transitions | 693 | |
| Transversions | 195 | |
| Mutations in coding region | 736 | |
| Intergenic mutation | 151 | |
| Synonymous mutation | 487 | CDS |
| Missense mutation | 247 | CDS |
| Nonsense mutation | 2 | CDS |
| Other mutations that could not be accurately judged | 6 |
| Type | InDel Number | Region | |
|---|---|---|---|
| Mutations in coding region | 66 | ||
| Intergenic mutation | 64 | ||
| Codon mutation | Code insertion | 19 | CDS |
| Code deletion | 19 | CDS | |
| Frameshift mutation | 25 | CDS | |
| Other mutations that could not be accurately judged | 6 | ||
| Type | BmNPV ZJ Gene Number | BmNPV YN Gene Number | Gene Name | Mutation Type | Biological Function | Description |
|---|---|---|---|---|---|---|
| Core | PROKKA 00060 | PROKKA 00011 | pif-2 | Transversions | Viral infection | The composition of the membrane, necessary for oral infection |
| PROKKA 00082 | PROKKA 00117 | p74 (pif-0) | Transversions | Viral infection | It is related to the life cycle of the virus and participation in the oral infection of the virus | |
| PROKKA 00108 | PROKKA 00135 | pif-1 | Transitions and transversions | Viral infection | The composition of the membrane, related to oral infections | |
| PROKKA 00113 | PROKKA 00108 | pif-5 (odv-e56) | InDel | Viral infection | Determines the virus host range, related to oral infections | |
| PROKKA 00009 | PROKKA 00056 | Vlf-1 | Transversions and InDel | Replication, transcription | Late gene expression | |
| PROKKA 00020 | PROKKA 00067 | dna pol | Transitions and transversions | Replication, transcription | The catalytic activity, replication of the viral genome, and host DNA polymerase cannot replace viral enzymes in this process | |
| PROKKA 00035 | PROKKA 00082 | lef-8 | Transitions | Replication, transcription | Late gene expression | |
| PROKKA 00043 | PROKKA 00090 | p47 | Transitions | Replication, transcription | Regulation of viral transcription | |
| PROKKA 00067 | PROKKA 00018 | lef-1 | InDel and transversions | Replication, transcription | Encoding DNA promoter and interacting with LEF-2 | |
| PROKKA 00073 | PROKKA 00024 | lef-2 | Transitions | Replication, transcription | Virus replication and late gene expression | |
| PROKKA 00092 | PROKKA 00036 | lef-5 | Transitions | Replication, transcription | Regulation of viral transcription | |
| PROKKA 00095 | PROKKA 00033 | AcMNPV orf103 | Transitions | Replication, transcription | Virus replication, influences virus particle formation | |
| PROKKA 00114 | PROKKA 00109 | ie-1 | Transitions | Replication, transcription | The essential transactivated protein that initiates viral DNA replication and bro promoter transcription | |
| PROKKA 00123 | PROKKA 00045 | lef-4 | Transitions and transversions | Replication, transcription | Regulation of viral transcription | |
| PROKKA 00128 | PROKKA 00040 | dna hel/p143 | Transversions | Replication, transcription | DNA helicase activity, host domain determinant | |
| PROKKA 00132 | PROKKA 00126 | p24 | Transitions | Replication, transcription | Regulation of viral transcription | |
| PROKKA 00003 | PROKKA 00050 | p95 | Transitions | Structural protein | Composition of BV and ODV | |
| PROKKA 00006 | PROKKA 00053 | gp41 | Transitions and transversions | Structural protein | Exists only in ODV, determining the manner and the ability of the virus to invade the host | |
| PROKKA 00008 | PROKKA 00055 | AcMNPV orf78 | Transitions | Structural protein | Related to BV production and M-ODV formation | |
| PROKKA 00093 | PROKKA 00035 | p40 | Transitions, transversions, and InDel | Structural protein | Includes body virus envelope components related to specific infection of host cells | |
| PROKKA 00100 | PROKKA 00143 | AcMNPV orf109 | Transversions | Structural protein | Participates in viral nucleocapsid assembly | |
| PROKKA 00122 | PROKKA 00046 | vp39 | Transitions and InDel | Structural protein | Related to virus infection | |
| Non -core | PROKKA 00101 | PROKKA 00142 | AcMNPV orf110 | Transitions | Viral infection | Related to oral infections |
| PROKKA 00024 | PROKKA 00071 | fp25K (ac61) | InDel | Auxiliary function | Involved in BV and ODV formation, implicated in host degradation after death | |
| PROKKA 00058 | PROKKA 00009 | pkip | Transitions | Auxiliary function | Related to the BV nucleocapsid component | |
| PROKKA 00066 | PROKKA 00017 | ecdysteroid UDP-glucosyl transferase (egt) | Transversions | Auxiliary function | Hinders larvae molting and pupation | |
| PROKKA 00070 | PROKKA 00021 | pk1 | Transversions and InDel | Auxiliary function | Regulation of polyhedrin gene promoter activity | |
| PROKKA 00078 | PROKKA 00029 | ptp | Transitions | Auxiliary function | BV components, essential components for effective infection of larvae brain tissue | |
| PROKKA 00086 | PROKKA 00129 | chitinase A | Transitions | Auxiliary function | Related to virus transmission | |
| PROKKA 00140 | PROKKA 00128 | viral cathepsin-like protein (v-cath) | Transversions | Auxiliary function | Related to host liquefaction and degradation | |
| PROKKA 00016 | PROKKA 00063 | AcMNPV orf69 | Transitions | Replication, transcription | Late gene expression | |
| PROKKA 00018 | PROKKA 00065 | lef-3 | Transitions | Replication, transcription | SS-DNA binding and destruction of helical stability | |
| PROKKA 00031 | PROKKA 00078 | lef-10 | Transitions | Replication, transcription | Regulation of viral transcription | |
| PROKKA 00042 | PROKKA 00089 | lef-12 | Transitions | Replication, transcription | Late gene transcirition | |
| PROKKA 00052 | PROKKA 00002 | bro-a | Transitions and transversions | Replication, transcription | DNA binding protein, complementary to BRO-C | |
| PROKKA 00054 | PROKKA 00005 | lef-6 | Transitions and transversions | Replication, transcription | Virus replication and late gene expression, affecting host cell apoptosis | |
| PROKKA 00057 | PROKKA 00008 | dbp (DNA binding protein) | InDel | Replication, transcription | SS-DNA binding protein co-localized with ie-1 and lef-3 in viral replication mechanism | |
| PROKKA 00069 | PROKKA 00020 | bm (br) orf-4 | Transitions and transversions | Replication, transcription | Early gene expression of virus | |
| PROKKA 00071 | PROKKA 00022 | orf1629 | Transitions and transversions | Replication, transcription | Virus replication | |
| PROKKA 00076 | PROKKA 00027 | Bro-b | Transitions, transversions, and InDel | Replication, transcription | DNA binding protein | |
| PROKKA 00077 | PROKKA 00028 | Bro-d | Transitions, transversions, and InDel | Replication, transcription | Virus replication and gene expression regulation | |
| PROKKA 00080 | PROKKA 00105 | pe38 | Transitions and transversions | Replication, transcription | Virus replication and gene expression regulation | |
| PROKKA 00090 | PROKKA 00038 | Bro-c | Transitions, transversions, and InDel | Replication, transcription | DNA binding protein, complementary to BRO-A | |
| PROKKA 00091 | PROKKA 00037 | 39k | InDel | Replication, transcription | Virus replication and gene expression regulation | |
| PROKKA 00097 | PROKKA 00030 | he65 | Transitions and transversions | Replication, transcription | Virus replication | |
| PROKKA 00111 | PROKKA 00106 | ie-2 | InDel | Replication, transcription | Virus replication and gene expression regulation | |
| PROKKA 00120 | PROKKA 00115 | ie-0 | Transitions | Replication, transcription | Regulation of viral transcription | |
| PROKKA 00121 | PROKKA 00116 | me53 | Transitions and transversions | Replication, transcription | Related to BV and ODV production | |
| PROKKA 00021 | PROKKA 00068 | gp37 | Transitions | Structural protein | The formation of auxiliary components of polyhedra is involved in the transport of virus particles in the cell | |
| PROKKA 00037 | PROKKA 00084 | odv-e66 | Transitions and transversions | Structural protein | Participation in BV and ODV morphogenesis and oral infection | |
| PROKKA 00038 | PROKKA 00085 | bmnpvcubigcp037 | InDel | Structural protein | Replication of the virus, regulation gof the transport of virus particles | |
| PROKKA 00065 | PROKKA 00016 | Bv/odv-e26 | Transitions | Structural protein | Related to BV and ODV envelopes | |
| PROKKA 00096 | PROKKA 00032 | vp80 | Transitions | Structural protein | Required for virus replication, BV production, and nucleocapsid maturation | |
| PROKKA 00135 | PROKKA 00123 | AcMNPV orf132 | Transitions and transversions | Structural protein | Involved in BV and ODV formation | |
| PROKKA 00139 | PROKKA 00118 | p10 | Transitions | Structural protein | Partcipates in the morphogenesis of viral polyhedra and promotes the release of polyhedra from infected insect cells | |
| PROKKA 00015 | PROKKA 00062 | iap2 | Transitions | Apoptosis | Cell apoptosis inhibiting factor | |
| PROKKA 00055 | PROKKA 00006 | iap1 | Transitions, transversions, and InDel | apoptosis | Induction of apoptosis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, H.; Zhang, B.; Zheng, X.; Sun, J.; Guo, H.; Li, G.; Zhao, G.; Xu, A.; Qian, H. Pathogenicity Detection and Genome Analysis of Two Different Geographic Strains of BmNPV. Insects 2021, 12, 890. https://doi.org/10.3390/insects12100890
Guo H, Zhang B, Zheng X, Sun J, Guo H, Li G, Zhao G, Xu A, Qian H. Pathogenicity Detection and Genome Analysis of Two Different Geographic Strains of BmNPV. Insects. 2021; 12(10):890. https://doi.org/10.3390/insects12100890
Chicago/Turabian StyleGuo, Huimin, Benzheng Zhang, Xin Zheng, Juan Sun, Huiduo Guo, Gang Li, Guodong Zhao, Anying Xu, and Heying Qian. 2021. "Pathogenicity Detection and Genome Analysis of Two Different Geographic Strains of BmNPV" Insects 12, no. 10: 890. https://doi.org/10.3390/insects12100890
APA StyleGuo, H., Zhang, B., Zheng, X., Sun, J., Guo, H., Li, G., Zhao, G., Xu, A., & Qian, H. (2021). Pathogenicity Detection and Genome Analysis of Two Different Geographic Strains of BmNPV. Insects, 12(10), 890. https://doi.org/10.3390/insects12100890






