Simple Summary
Methods to rear Osmia bees to pollinate fruit trees have been developed in various parts of the world. These bees are excellent pollinators but evidence that their populations can be sustained in orchards and their use results in increased fruit production is scarce. We released an Osmia cornuta population at one end of an almond orchard. Then, we surveyed the pollinators visiting the almond flowers and measured fruit set in trees located at increasing distances from the nesting stations. We found that fruit production was higher in the trees that received more Osmia visits. Importantly, this result was obtained against a strong background of honeybees, which were 10 times more abundant than Osmia. The Osmia population obtained at the end of the flowering period was 1.28 larger than the population initially released. Our study demonstrates that Osmia populations can be sustained in orchard environments and that even a small population of a highly effective pollinator may have a significant impact on fruit set. Our results are encouraging for the use of Osmia populations and for the implementation of measures to promote wild pollinators in agricultural environments.
Abstract
Osmia spp. are excellent orchard pollinators but evidence that their populations can be sustained in orchard environments and their use results in increased fruit production is scarce. We released an Osmia cornuta population in an almond orchard and measured its population dynamics, as well as visitation rates and fruit set at increasing distances from the nesting stations. Honeybees were 10 times more abundant than O. cornuta. However, the best models relating fruit set and bee visitation included only O. cornuta visitation, which explained 41% and 40% of the initial and final fruit set. Distance from the nesting stations explained 27.7% and 22.1% of the variability in initial and final fruit set. Of the 198 females released, 99 (54.4%) established and produced an average of 9.15 cells. Female population growth was 1.28. By comparing our results with those of previous O. cornuta studies we identify two important populational bottlenecks (female establishment and male-biased progeny sex ratios). Our study demonstrates that even a small population of a highly effective pollinator may have a significant impact on fruit set. Our results are encouraging for the use of Osmia managed populations and for the implementation of measures to promote wild pollinators in agricultural environments.
1. Introduction
Approximately three-quarters of the world’s crops benefit from animal pollination [1], and a significant part of this pollination service is provided by wild pollinators [2,3,4,5]. However, the current context of agricultural intensification, characterized by low crop diversity, increased crop field size, loss of semi-natural habitats, and increased pesticide use is clearly detrimental to pollinator abundance and diversity [6,7,8,9]. As a result, wild pollinators are notoriously scarce in many agricultural landscapes [10,11,12]. For this reason, the general perception is that wild pollinator populations are insufficient to provide adequate levels of pollination in intensively farmed areas, and populations of managed pollinators are usually introduced to enhance pollination services.
Fruit trees are highly dependent on pollinator visitation because they bloom for a short period of time in spring when weather conditions are often suboptimal for insect activity and because most cultivars are self-incompatible. For this reason, honey bee hives are usually introduced in orchards at a rate of 2–6 hives per ha, with each hive containing several thousands of foragers [13,14,15]. However, honey bees are only fully active at temperatures above 12–14 °C [16]. In addition, because they have long foraging ranges [17,18,19], and are highly generalist foragers, they often visit other flower species [14]. Finally, honey bees are not very effective fruit tree pollinators, mainly due to their low visit legitimacy (many of the visits result in no contact between the bee and the stigmas; [20,21,22,23,24,25]). These shortcomings, along with the risks associated with relying on a single species, have prompted the search for alternative pollinators, and methods to manage various Osmia species as orchard pollinators have been developed in different parts of the world [26,27]. These Osmia species are only active for a couple of months in spring and fly under marginal weather conditions [16]. In addition, they have short foraging ranges [28,29,30] and show a strong preference for fruit tree pollen [21,23,31,32,33,34,35]. Finally, Osmia visit legitimacy on fruit three flowers is close to 100%, and fruit set in flowers receiving a single visit is similar to fruit set in hand-pollinated flowers [20,21,23,24,25,36]. For these reasons, population densities recommended for orchard pollination with Osmia spp. are as low as 1250–2000 bees per ha (with a 1.6 male/female ratio) [26]. However, even if the pollinating effectiveness of Osmia spp. has been amply documented, evidence that the use of Osmia populations results in increased fruit production is still scarce [11,23,37,38].
The use of a managed pollinator is only sustainable if population levels can be maintained from year to year. Ideally, populations should be able to grow on site during the flowering period of the target crop. If this is not possible, population losses can be compensated by rearing populations under artificial conditions, as done with bumblebee colonies used for crop pollination [39]. However, attempts to mass-rear Osmia populations under artificial conditions have not been successful [40,41]. Therefore, to ensure the sustainability of Osmia spp. as managed pollinators it is essential to understand the dynamics of populations introduced in orchards. There are various factors potentially limiting the growth of Osmia populations released in orchards. These factors include: (a) Winter mortality (some of the cocoons introduced in the orchard contain individuals that are either dead or too weak to emerge); (b) low female establishment (some of the females that have successfully emerged out of their cocoon may be predated, too weak to start nesting activities, or disperse and nest away from the release site); (c) low fecundity (nesting females provision a low number of cells and therefore lay few eggs); (d) progeny mortality (a part of the progeny does not reach the adult stage due to either developmental failure or parasitism); (e) male-biased progeny sex ratio (nesting females produce a high proportion of males, which are less costly to produce than females [42]).
In this study, we released a population of the European species Osmia cornuta in an almond orchard. Almond pollination is particularly challenging because almonds bloom very early in the year (February-March) and have an unusually high bearing capacity (as many as 40% of the flowers may bear fruit) [43]. We measured female establishment and population growth, as well as flower visitation rates and fruit set at increasing distances from the nesting site. Our first objective was to assess whether the O. cornuta population had an impact on fruit production. Given the short foraging range of O. cornuta, we expected fruit set to be negatively correlated to distance from the nesting site. Our second objective was to establish whether the O. cornuta population could be increased on site and, by comparing our results with those of previous studies, to identify the main factors limiting population growth.
2. Materials and Methods
The study was conducted in an almond orchard in Vila-Seca (Tarragona, NE Spain). The orchard measured 0.5 ha and had 5 rows of the main cultivar Ferragnes intermixed with 4 rows of the pollinizer cultivar Cristomorto (both cultivars are self-incompatible) (Figure A1). Each row had 16 trees. The orchard was located within a matrix of farmland, including cereal fields, fallow land, and a mixture of olive, carob, and hazelnut orchards. There were no honey bee hives in the orchard or its surroundings (at least within a 500 m radius).
In early February 1992, prior to almond bloom, we set up 3 nesting stations for O. cornuta at one edge of the orchard (Figure A1). Nesting stations were made with wooden boxes with the front side open held 1.5 m above the ground on four metal fence posts. Each station contained 15 perforated solid wood blocks with 25 inserted paper straws (length: 15 cm; inside diameter: 8 mm). On 22 February, before the orchard started to bloom, we placed 198 females and 360 males (M/F sex ratio: 1.8) within their cocoons in open cardboard boxes inside the nesting stations. These cocoons had been wintered at 4 °C since October 1991.
To determine the number of females that established at the nesting stations, we inspected each nesting cavity with an otoscope. This was done at night, when females were roosting inside their nests, seven times during the nesting period (approximately twice a week).
During peak bloom, we conducted pollinator counts on the central row of Ferragnes. An observer slowly walked around each tree and noted all pollinators seen visiting the flowers. Total observation time was 10 h and 40 min (40 min × 16 trees).
To assess fruit set (% of flowers that set fruit) at increasing distances (0–80 m) from the O. cornuta nesting shelters, we marked all trees in 3 Ferragnes rows (48 trees), including the row used in the pollinator counts (Figure A1). Before bloom we tagged 4 branches on each tree and counted the number of flower buds on each branch (mean ± SE = 264.4 ± 1.6 flower buds per branch). In April, we counted the number of initiated fruits on each branch (initial fruit set). Following the natural fruit drop, fruits were counted again in June when they had reached their final size (final fruit set).
To obtain a measure of maximum potential fruit set, we marked 5 additional Ferragnes trees (Figure A1) on which we tagged 4 branches and counted flower buds as described above. These trees were checked daily and newly opened flowers were hand-pollinated with Cristomorto pollen. Initial and final fruit set was assessed as described above.
After petal fall, when O. cornuta nesting activity had ceased, we removed nesting materials and kept them in the laboratory. In September, when development was completed, we pulled out paper straws and analyzed the contents of each nest. We quantified the number of male and female offspring produced (female cocoons are typically larger than male cocoons and are found in the inner cells of the nest [42]). We also recorded offspring mortality.
Statistical Analysis
Initial and final fruit set were analyzed separately. To analyze whether fruit set declined with distance from the O. cornuta nesting stations, we used Linear Mixed Models (LMMs) with a tree as a random factor. We considered a linear relationship (model with distance as the only explicative variable) and a quadratic relationship (model with distance and distance2). We then used a model inference approach [44] to select the best-fit model based on AICc values using maximum likelihood criteria. Models with ΔAICc < 2 were considered equal to the best model [44]. We then run a LMM model with REML to obtain unbiased parameter estimates. We calculated a likelihood-ratio-based R2 of the best models as a measure of explanatory power.
To analyze whether fruit set was related to O. cornuta and/or A. mellifera visitation, we conducted a model selection procedure with LM models, testing all possible explanatory variable combinations through a multi-model inference approach with the ‘dredge’ function (‘MuMin’ package, [45]). We again selected the best models based on AICc values. Following model selection, we used a model averaging procedure (with averaged variable coefficients) based on AICc. This was done with the ‘model.avg’ function (‘MunMin’ package), which yields model-averaged estimates of variable coefficients and p-values for each explanatory variable. Conditional average and full average approaches yielded almost identical results. We show only conditional average results. Finally, we calculated the adjusted-R2 of the best models (containing significant explanatory variables) as a measure of explanatory power.
Percent initial and final fruit set were arcsin-transformed. The distribution of residuals was visually inspected for homoscedasticity and the normality assumption was tested with the Shapiro test. All analyses were conducted with the ‘nlme’ [46] and MuMin packages in R [47]. All means are followed by standard error (SE).
3. Results
Maximum temperatures during the days following the release of the O. cornuta population (22 February) ranged between 14 and 18 °C. The first females engaged in nesting activities were observed on 28 February. Of the 198 females released, 182 emerged out of their cocoons (91.9 % winter survival). The maximum number of females established in the nesting stations was counted on March 3 (99 females; 54.4% of the emerged females).
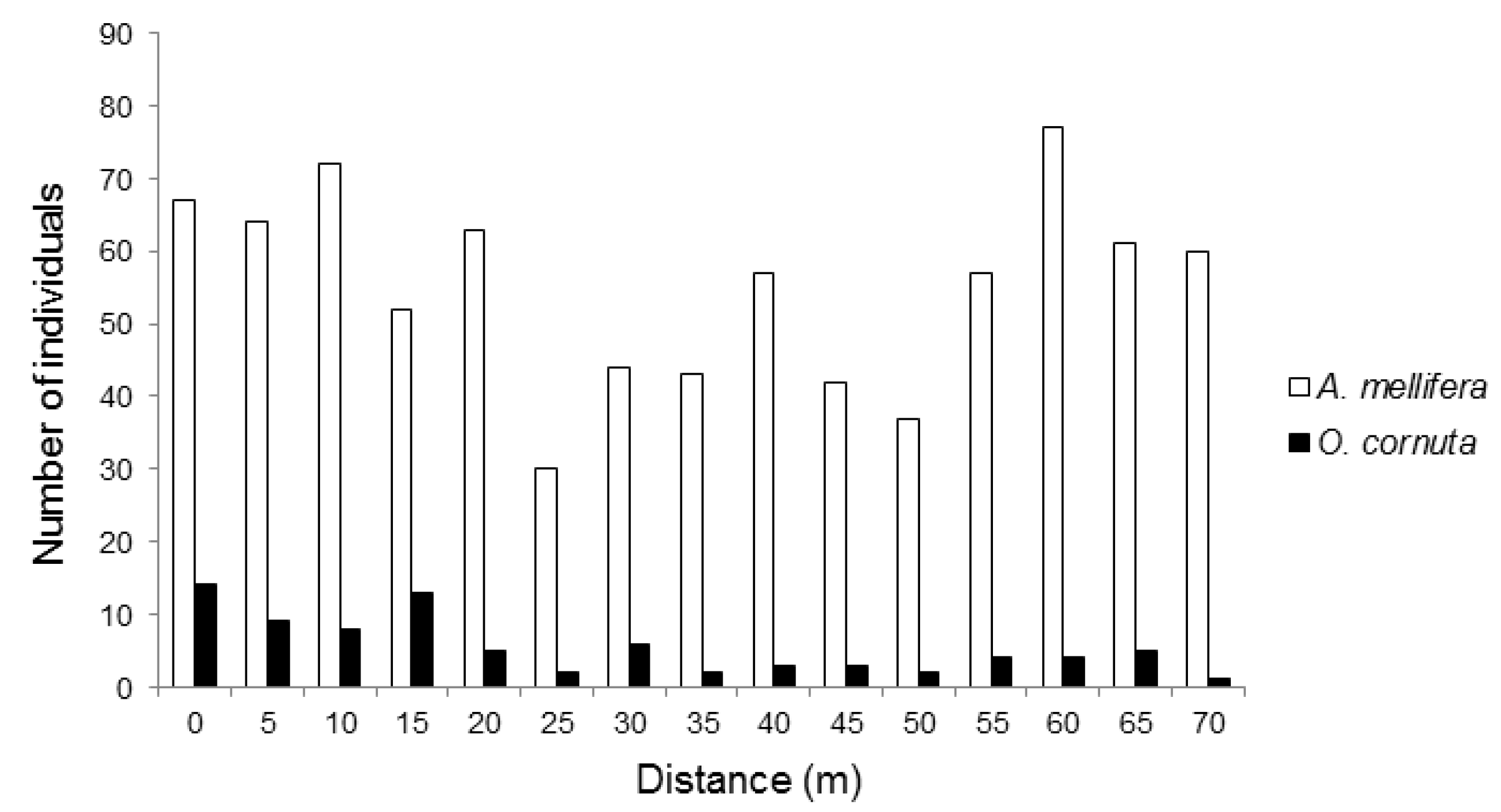
We recorded 1114 pollinators visiting the almond flowers. Although there were no hives in sight, the most frequent pollinator was, by far, Apis mellifera (74.1% of the visits recorded), followed by O. cornuta (7.3%). Other visitors included various flies (16.2%), hoverflies (4.11%, Eristalis tenax, Eupeodes sp.), and wild bees (1.1%, Andrena nigroaenea, Andrena sp., Eucera sp., Bombus terrestris, Xylocopa violacea). Most (70.4%) O. cornuta visitation occurred within 30 m from the nesting stations (Figure 1). Apis mellifera visitation followed an irregular pattern across the orchard and tended to be higher towards the two ends of the orchard (Figure 1).
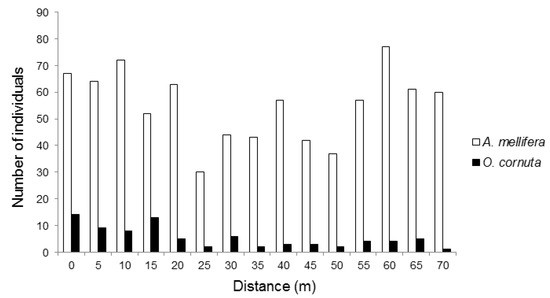
Figure 1.
Number Apis mellifera and Osmia cornuta individuals recorded visiting almond flowers at increasing distances from the O. cornuta nesting stations.
Almond bloom was over by 23 March. At that time, O. cornuta females that were still alive foraged mostly on Diplotaxis erucoides (Brassicaceae), a common weed in the surroundings of the orchard. By 29 March O. cornuta nesting activity had ceased. The number of nests produced was 203. These nests contained 253 female and 653 male cells. Offspring mortality was 7.1%. Most of this mortality was due to unknown causes (5.3%), and the rest to parasitism by the cleptoparasitic mite Chaetodactylus osmiae (1.8%). The live female population recovered was 241, and the live male population 601 (M/F sex ratio: 2.5).
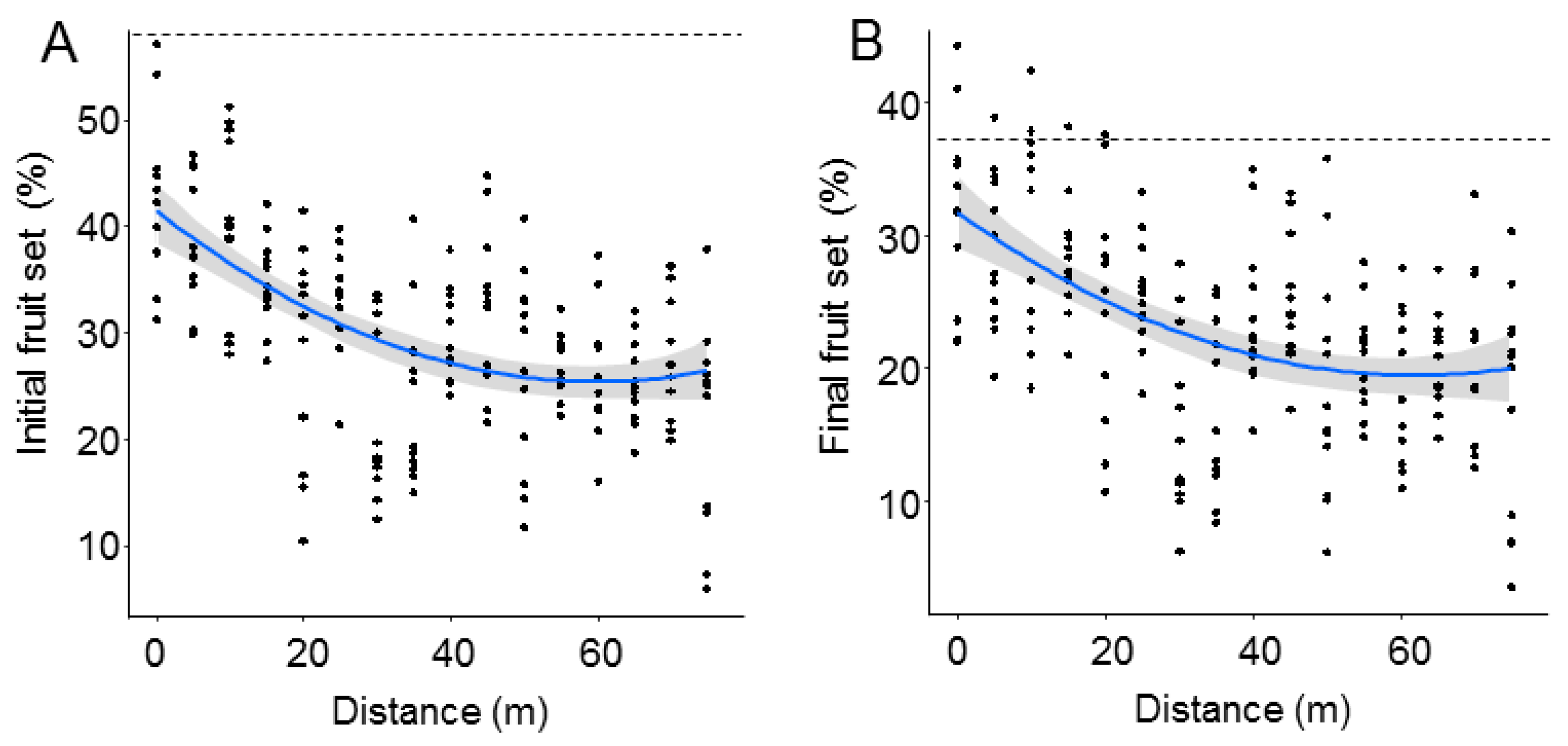
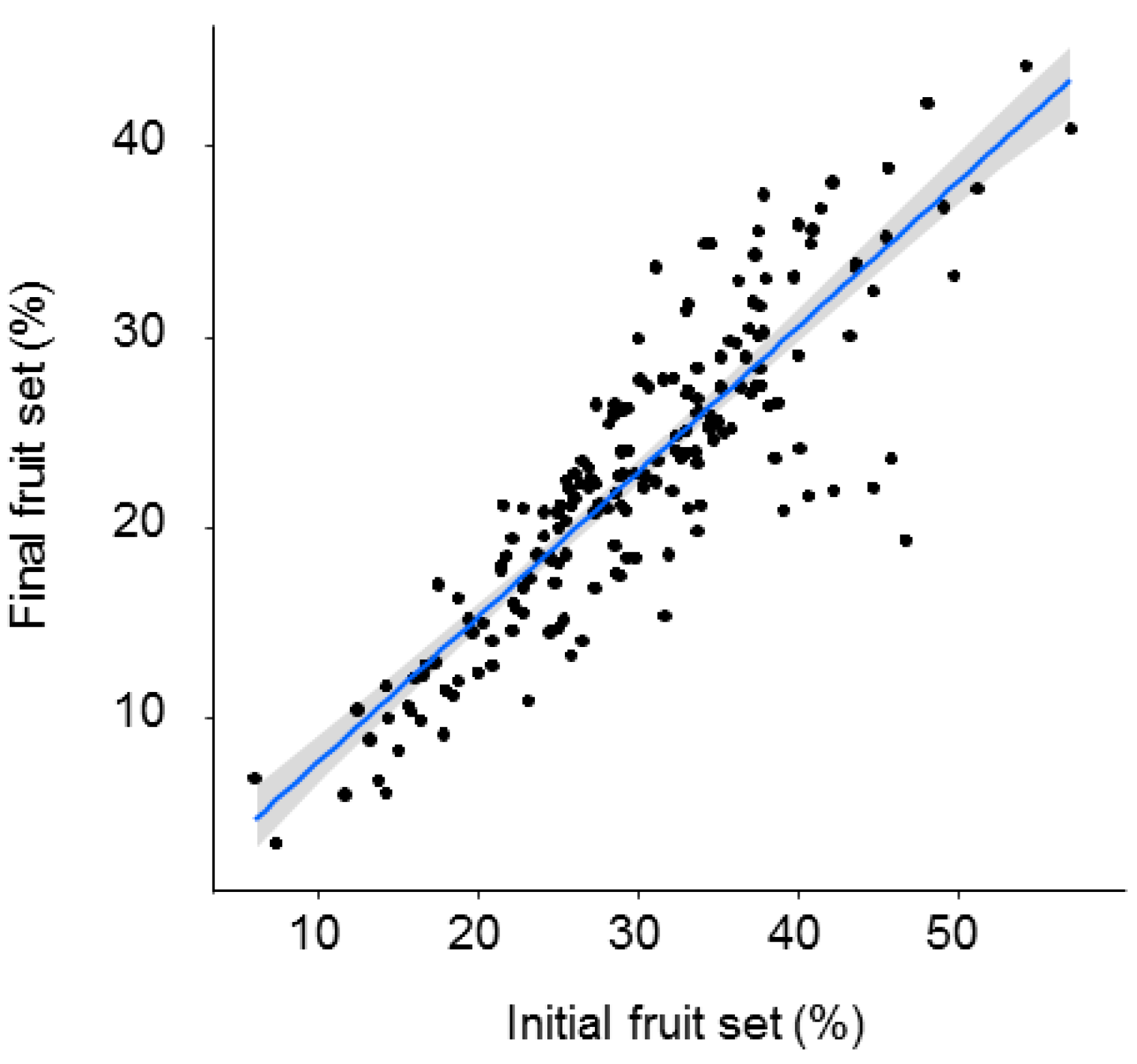
Both initial and final fruit set significantly declined with distance from the O. cornuta nesting stations (Figure 2). The quadratic model provided the best fit, with distance explaining 27.7% of the variability in initial fruit set and 22.1% of the variability in final fruit set (Table 1). Importantly, the random factor tree also had a strong effect on fruit set (initial: 38.7%; final: 37.1%). The linear models yielded similar results (Table A1 and Figure A2 in Appendix A). Final fruit set was strongly related to initial fruit set (Pearson’s r = 0.88; p < 0.0001; Figure 3).
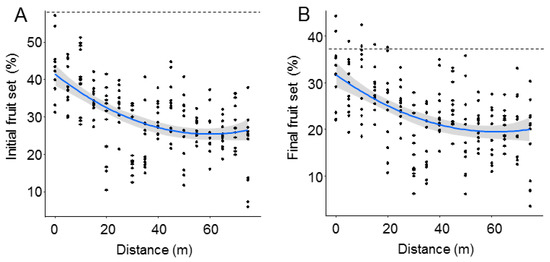
Figure 2.
Initial (A) and final (B) fruit set (% of flowers setting fruit in April and June, respectively) at increasing distances from the Osmia cornuta nesting shelters. Each dot represents a branch. Broken lines indicate mean fruit set in hand-pollinated trees (five trees; four branches per tree). The gray bands represent 95% confidence intervals.

Table 1.
Output of Linear Mixed Model (LMM) relating initial fruit (A) and final fruit set (B) to distance from the Osmia cornuta nesting shelters. Parameter- (t) and p-values are provided for the best-fitted models based on AICc selection. R2m and R2c are the marginal and conditional R2 of the best-fitted models.
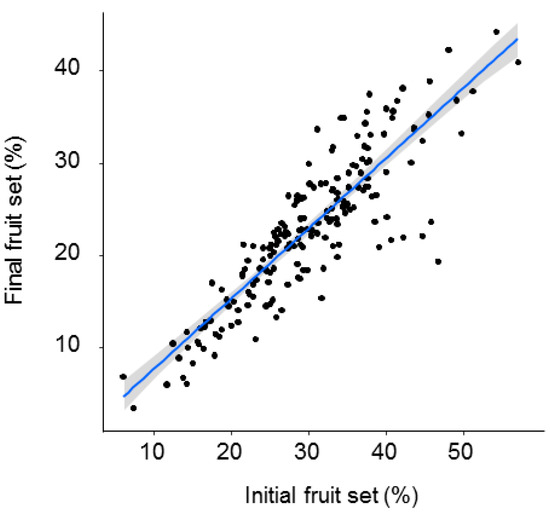
Figure 3.
Relationship between initial (April) and final (June) fruit set (% of flowers setting fruit). Each dot represents a branch. The gray band represents the 95% confidence interval.
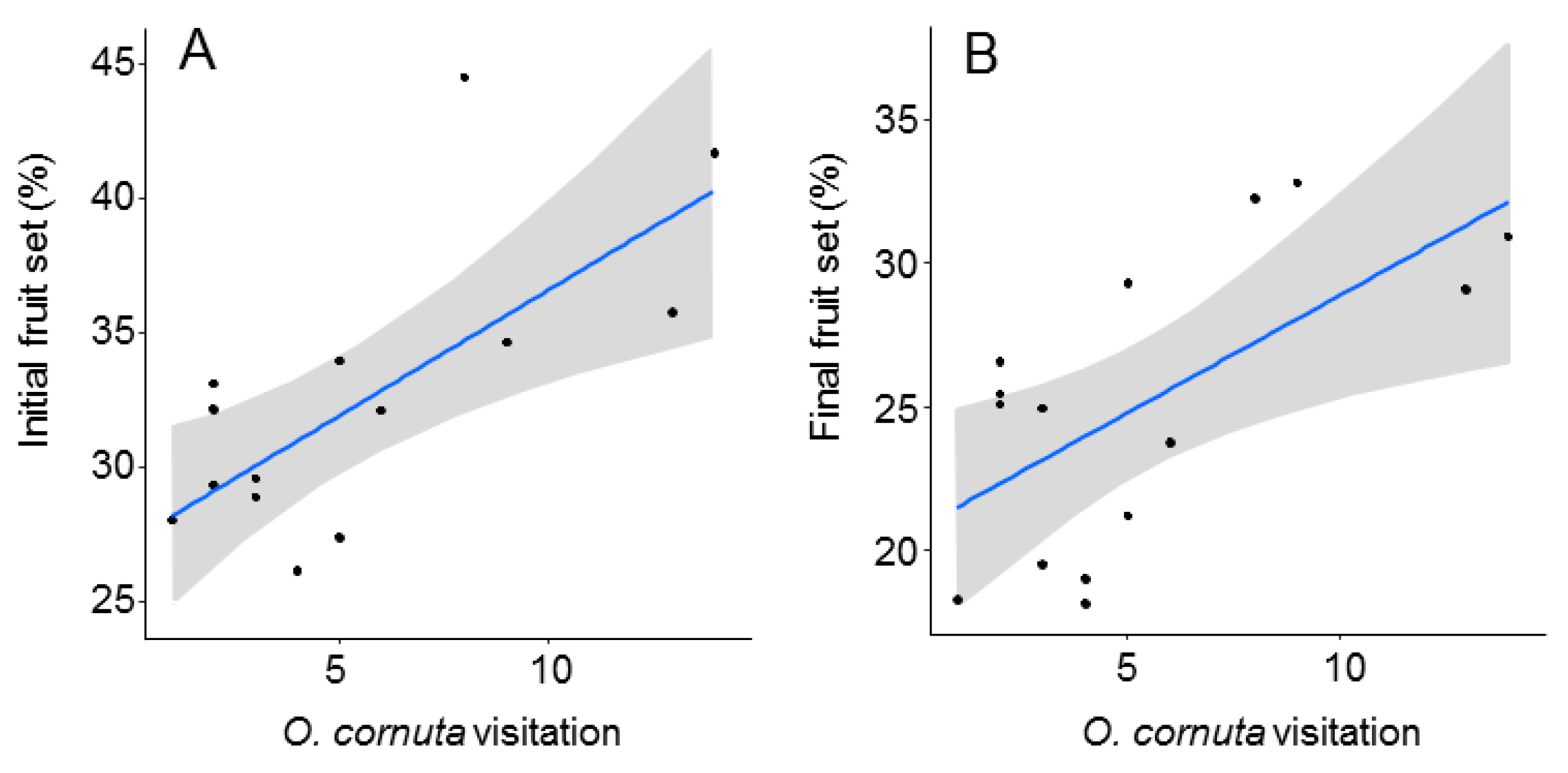
The best models analyzing the effect of O. cornuta and A. mellifera visitation on fruit set included only O. cornuta visitation, which explained 41% of the initial fruit set and 40% of the final fruit set (Table 2; Figure 4). Even then, the levels of fruit set obtained across the orchard were lower than those obtained in the five hand-pollinated trees (initial fruit set: 57.8 ± 2.0; final fruit set: 36.9 ± 1.8%) (Figure 2), indicating that pollination services could still be increased.

Table 2.
Output of LM model averaging relating initial (A) and final (B) fruit set to Osmia cornuta and Apis mellifera visitation. Estimated coefficients and their p-values are provided. Adjusted R2 of the best-fitted model (containing only O. cornuta visitation as predictor variable) are provided.
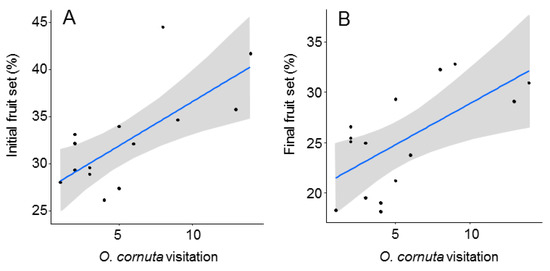
Figure 4.
Relationship between Osmia cornuta visitation and initial (A) and final (B) fruit set (% of flowers setting fruit in April and June, respectively). Each dot represents a tree. Gray bands represent 95% confidence intervals.
4. Discussion
Our first objective was to assess whether the O. cornuta population had an impact on almond production. Most of the O. cornuta observed were recorded within 30 m from the nesting stations. Although Osmia females are able to locate their nest from distances as far as 500–1800 m [28,29], populations established in orchards concentrate most of their foraging within 50 m of the nesting sites [23,30]. The negative relationship between fruit set and distance from the nesting stations closely paralleled the distribution of O. cornuta across the orchard. The contribution of O. cornuta to pollination service was further confirmed by the analysis identifying O. cornuta visitation (but not A. mellifera visitation) as a significant predictor of initial and final fruit set. This result is remarkable given that A. mellifera visitation was 10 times higher than O. cornuta visitation. Apis mellifera is consistently reported as the dominant pollinator species in commercial orchards (e.g., [11,12,48]). However, its contribution to pollination service is strongly limited by its low per-visit pollination effectiveness on fruit tree flowers [20,21,22,23,24,25]. Our results are limited to a single orchard and a single year. However, they are in line with a previous study that found a significant impact of O. cornuta visitation on final seed-set in a pear orchard in which A. mellifera was 7 times more abundant [23]. These results are also in agreement with studies showing yield increases in orchards pollinated with other Osmia species [11,37,38]. Our study also shows that initial fruit set (commonly used as a proxy of pollination service) is a good predictor of final fruit set in almonds.
Our second objective was to establish whether the O. cornuta population could be increased on site, and to identify the main factors limiting population growth. We released 198 females of which 182 emerged out of their cocoons (8.1% winter mortality). This winter mortality is similar to values obtained in other O. cornuta populations managed for orchard pollination (Table 3). Of the 182 females that emerged, 99 (54.4%) established in the nesting stations. Failure to establish can be caused by lack of vigor of emerging females [49], predation [50,51,52], and dispersal of pre-nesting females [53], but attributing a relative weight to each of these three factors is not easy. At any rate, the percent establishment obtained in our study was similar to values obtained with other O. cornuta populations released in orchards (Table 3). The 99 females that established in the nesting stations produced 906 cells (an average of 9.15 cells/female). This fecundity is again close to fecundity in other O. cornuta populations released in orchards (Table 3). Of these 906 cells, 253 contained female progeny (2.6 M/F sex ratio). This is considerably higher than that the sex ratio of the population released (1.8), but similar to sex ratios obtained from other O. cornuta populations released in orchards (Table 3). Developmental mortality (5.3%) and parasitism (1.8%) were also similar to mortality levels obtained in previous studies (Table 3). Ultimately, the female population recovered was 1.28 times larger than the female population released, providing evidence that Osmia populations can be sustained in orchard environments ([26] and references therein, [54]).

Table 3.
Population parameters of Osmia cornuta populations released in orchards. All means followed by standard error (SE).
The results of Table 3 allow us to identify two important bottlenecks in the dynamics of O. cornuta populations managed for orchard pollination. The first one occurs in the establishment phase, during which the effective female population is reduced by ca. 50%. Previous studies have shown that Osmia establishment can be enhanced by releasing populations within their natal nests, rather than as loose cocoons [33,55,59,60,61]. This result is probably mediated by olfactory nest cues that enhance the tendency of females to re-nest in their natal nesting site (philopatry; [61]). In relation to this, an olfactory attractant similar to that developed for Osmia lignaria [54] could potentially enhance establishment of O. cornuta populations released in orchards. The second important bottleneck is the production of a male-biased sex ratio. In O. cornuta, the sex ratio of populations trap-nested in semi-natural areas is 1.5 ± 0.06 (n = 4 populations) [62,63], a figure that closely matches the theoretically optimal sex ratio based on male and female body weights (1.7) [42]. However, progeny sex ratios obtained from populations released in orchards are consistently higher (ca. 2.5; Table 3). In other words, in populations nesting in orchards, a considerable fraction of the parental investment is devoted to the production of surplus males. The causes underlying these differences in sexual allocation are unclear, but a greater proportion of female cells in managed populations could be obtained by increasing the diameter [56,64,65], or the length [55,65] of the nesting cavities offered in orchard operations. A more balanced sex ratio (closer to 1.7) would increase the reproductive and pollinating potential of populations recovered from orchards.
5. Conclusions
Our study demonstrates that even a small population of a highly effective pollinator may have a significant impact on crop pollination service and fruit set. Our results are encouraging not only for the use of Osmia spp. as managed pollinators but also for the implementation of measures to protect wild pollinator communities in orchard environments. In addition to Osmia spp., various species of Andrena, Eucera and Bombus are highly effective fruit tree pollinators [5,22,24,25,36]. In the current scenario of pollinator declines, agri-environmental measures to enhance wild populations of these valuable pollinators could have important economic returns in terms of enhanced pollination service and fruit yields.
Supplementary Materials
The following are available online at https://www.mdpi.com/2075-4450/12/1/56/s1, dataset. Table S1: Fruit set vs. Distance; Table S2: Osmia vs. fset row1.
Author Contributions
J.B., conceived the research. J.B., S.O.-C., and N.V. conducted the study. S.O.-C. analyzed the data. J.B., S.O.-C., N.V., and F.S. wrote the manuscript. J.B. secured funding. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the DGICYT (project AGR. 91-0988-CO3) and the Spanish MEC (FPI grant to NV).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are available in the Supplementary Materials.
Acknowledgments
We are grateful to J. Calzadilla and M. A. Escolano for their help with field work and nest analysis.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Table A1.
Output of Linear Mixed Model (LMM) relating initial fruit (A) and final fruit set (B) to distance from the Osmia cornuta nesting stations considering a linear fit. R2m and R2c are the marginal and conditional R2 of the best-fitted models.
Table A1.
Output of Linear Mixed Model (LMM) relating initial fruit (A) and final fruit set (B) to distance from the Osmia cornuta nesting stations considering a linear fit. R2m and R2c are the marginal and conditional R2 of the best-fitted models.
| (A) Initial Fruit Set | ||
|---|---|---|
| t-Value | p-Value | |
| (Intercept) | 32.03 | <0.0001 |
| Distance | −4.73 | <0.0001 |
| R2m: 0.24; R2c: 0.66 | ||
| (B) Final Fruit Set | ||
| t-Value | p-Value | |
| (Intercept) | 29.22 | <0.0001 |
| Distance | −4.31 | 0.0001 |
| R2m: 0.19; R2c: 0.59 | ||
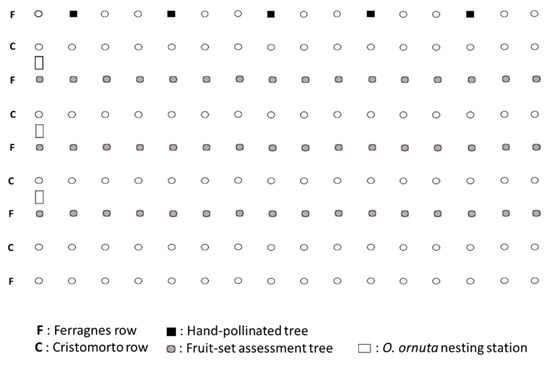
Figure A1.
Structure of the study orchard and location of the Osmia cornuta nesting stations.
Figure A1.
Structure of the study orchard and location of the Osmia cornuta nesting stations.
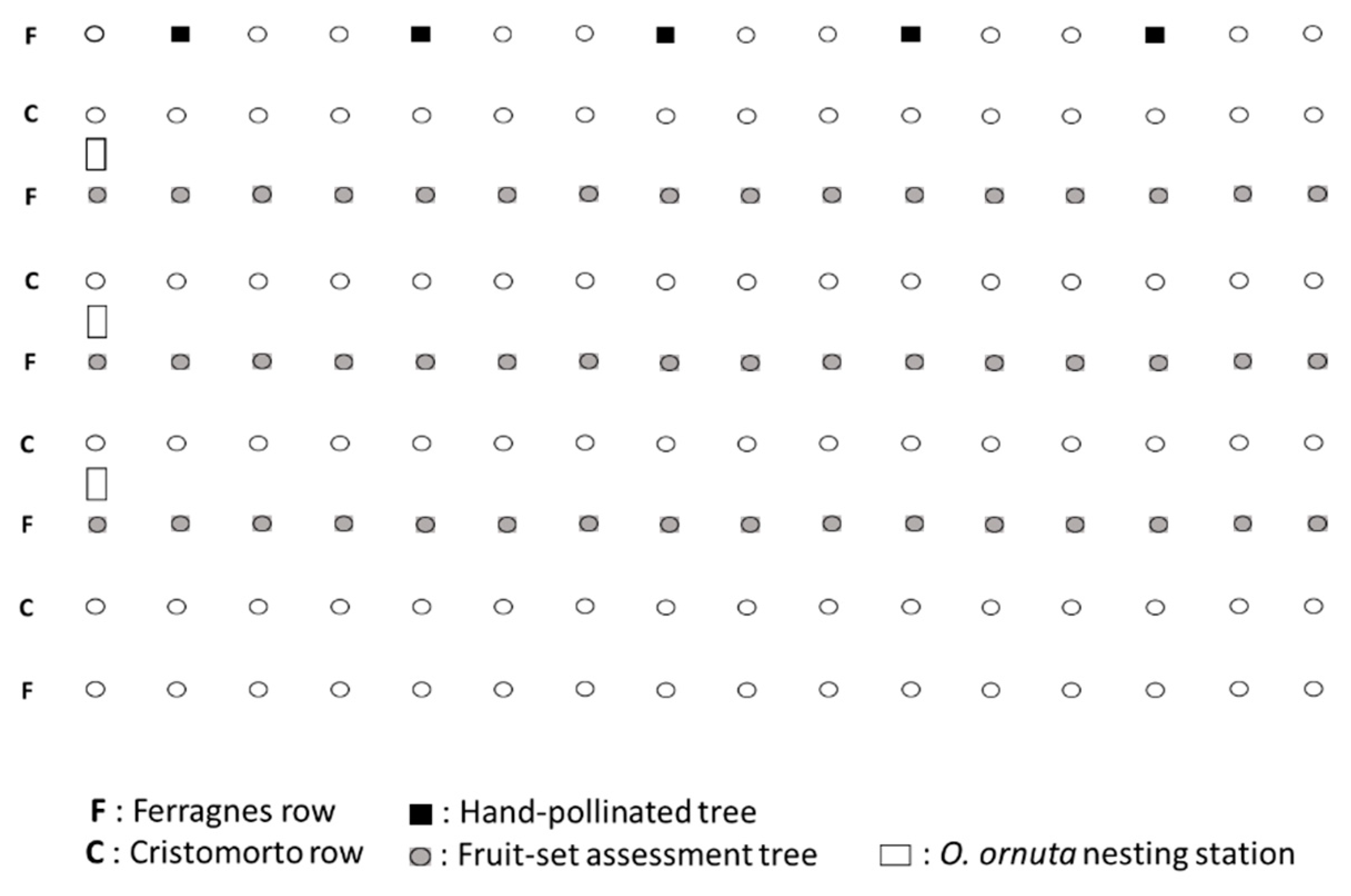
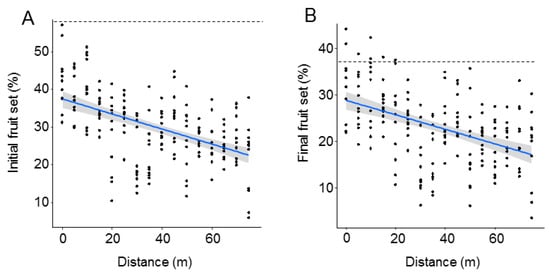
Figure A2.
Initial (A) and final (B) fruit set (% of flowers setting fruit in April and June, respectively) at increasing distances from the Osmia cornuta nesting shelters. Each dot represents a branch. Broken lines indicate mean fruit set in hand-pollinated trees (5 trees; 4 branches per tree). The gray bands represent 95% confidence intervals.
Figure A2.
Initial (A) and final (B) fruit set (% of flowers setting fruit in April and June, respectively) at increasing distances from the Osmia cornuta nesting shelters. Each dot represents a branch. Broken lines indicate mean fruit set in hand-pollinated trees (5 trees; 4 branches per tree). The gray bands represent 95% confidence intervals.
References
- Klein, A.-M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B 2007, 274, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Winfree, R.; Williams, N.M.; Gaines, H.; Ascher, J.S.; Kremen, C. Wild bee pollinators provide the majority of crop visitation across land-use gradients in New Jersey and Pennsylvania, USA. J. Appl. Ecol. 2008, 45, 793–802. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Steffan-Dewenter, I.; Winfree, R.; Aizen, M.A.; Bommarco, R.; Cunningham, S.A.; Kremen, C.; Carvalheiro, L.G.; Harder, L.D.; Afik, O.; et al. Wild Pollinators enhance fruit set of crops regardless of honey bee abundance. Science 2013, 339, 1608–1611. [Google Scholar] [CrossRef] [PubMed]
- Hevia, V.; Bosch, J.; Azcárate, F.M.; Fernández, E.; Rodrigo, A.; Barril-Graells, H.; González, J.A. Bee diversity and abundance in a livestock drove road and its impact on pollination and seed set in adjacent sunflower fields. Agric. Ecosyst. Environ. 2016, 232, 336–344. [Google Scholar] [CrossRef]
- Park, M.G.; Raguso, R.A.; Losey, J.E.; Danforth, B.N. Per-visit pollinator performance and regional importance of wild Bombus and Andrena (Melandrena) compared to the managed honey bee in New York apple orchards. Apidologie 2016, 47, 145–160. [Google Scholar] [CrossRef]
- Klein, A.-M.; Brittain, C.; Hendrix, S.D.; Thorp, R.; Williams, N.; Kremen, C. Wild pollination services to California almond rely on semi-natural habitat. J. Appl. Ecol. 2012, 49, 723–732. [Google Scholar] [CrossRef]
- Woodcock, B.A.; Bullock, J.M.; Shore, R.F.; Heard, M.S.; Pereira, M.G.; Redhead, J.; Ridding, L.; Dean, H.; Sleep, D.; Henrys, P.; et al. Country-specific effects of neonicotinoid pesticides on honey bees and wild bees. Science 2017, 356, 1393–1395. [Google Scholar] [CrossRef] [PubMed]
- Hass, A.L.; Kormann, U.G.; Tscharntke, T.; Clough, Y.; Bosem Baillod, A.; Sirami, C.; Fahrig, L.; Martin, J.-L.; Baudry, J.; Bertrand, C.; et al. Landscape configurational heterogeneity by small-scale agriculture, not crop diversity, maintains pollinators and plant reproduction in Western Europe. Proc. R. Soc. B 2018, 285, 20172242. [Google Scholar] [CrossRef] [PubMed]
- Sirami, C.; Gross, N.; Baillod, A.B.; Bertrand, C.; Carrié, R.; Hass, A.; Henckel, L.; Miguet, P.; Vuillot, C.; Alignier, A.; et al. Increasing crop heterogeneity enhances multitrophic diversity across agricultural regions. Proc. Natl. Acad. Sci. USA 2019, 116, 16442–16447. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, C.M.; Lonsdorf, E.; Neel, M.C.; Williams, N.M.; Ricketts, T.H.; Winfree, R.; Bommarco, R.; Brittain, C.; Burley, A.L.; Cariveau, D.; et al. A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecol. Lett. 2013, 16, 584–599. [Google Scholar] [CrossRef] [PubMed]
- Pitts-Singer, T.L.; Artz, D.R.; Peterson, S.S.; Boyle, N.K.; Wardell, G.I. Examination of a managed pollinator strategy for almond production using Apis mellifera (Hymenoptera: Apidae) and Osmia lignaria (Hymenoptera: Megachilidae). Environ. Entomol. 2018, 47, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Roquer-Beni, L.; Alins, G.; Arnan, X.; Boreux, V.; García, D.; Hambäck, P.A.; Happe, A.-K.; Klein, A.-M.; Miñarro, M.; Mody, K.; et al. Management-dependent effects of pollinator functional diversity on apple pollination services: A response-effect trait approach. (Unpublished; Manuscript in Preparation).
- McGregor, S.E. Insect Pollination of Cultivated Crop Plants; Agricultural Research Service: Washington, DC, USA, 1976; p. 411.
- Free, J.B. Insect Pollination of Crops, 2nd ed.; Academic Press: London, UK, 1993. [Google Scholar]
- Delaplane, K.S.; Mayer, D.R. Crop Pollination by Bees; CABI: Wallingford, UK, 2000. [Google Scholar]
- Vicens, N.; Bosch, J. Weather-dependent pollinator activity in an apple orchard, with special reference to Osmia cornuta and Apis mellifera (Hymenoptera: Megachilidae and Apidae). Environ. Entomol. 2000, 29, 413–420. [Google Scholar] [CrossRef]
- Visscher, P.K.; Seeley, T.D. Foraging strategy of honeybee colonies in temperate deciduous forest. Ecology 1982, 63, 1790–1801. [Google Scholar] [CrossRef]
- Beekman, M.; Ratnieks, F.L.W. Long-range foraging by the honey-bee, Apis mellifera L. Funct. Ecol. 2000, 14, 490–496. [Google Scholar] [CrossRef]
- Hagler, J.R.; Mueller, S.; Teuber, L.R.; Machtley, S.A.; Van Deynze, A. Foraging range of honey bees, Apis mellifera, in alfalfa seed production fields. J. Insect Sci. 2011, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bosch, J.; Blas, M. Foraging behaviour and pollinating efficiency of Osmia cornuta and Apis mellifera on almond (Hymenoptera, Megachilidae and Apidae). Appl. Entomol. Zool. 1994, 29, 1–9. [Google Scholar] [CrossRef]
- Vicens, N.; Bosch, J. Pollinating efficacy of Osmia cornuta and Apis mellifera (Hymenoptera: Megachilidae, Apidae) on ‘Red Delicious’ apple. Environ. Entomol. 2000, 29, 235–240. [Google Scholar] [CrossRef]
- Thomson, J.D.; Goodell, K. Pollen removal and deposition by honeybee and bumblebee visitors to apple and almond flowers. J. Appl. Ecol. 2001, 38, 1032–1044. [Google Scholar] [CrossRef]
- Monzón, V.H.; Bosch, J.; Retana, J. Foraging behavior and pollinating effectiveness of Osmia cornuta (Hymenoptera: Megachilidae) and Apis mellifera (Hymenoptera: Apidae) on “Comice” pear. Apidologie 2004, 35, 575–585. [Google Scholar] [CrossRef]
- Eeraerts, M.; Vanderhaegen, R.; Smagghe, G.; Meeus, I. Pollination efficiency and foraging behaviour of honey bees and non-Apis bees to sweet cherry. Agric. For. Entomol. 2020, 22, 75–82. [Google Scholar] [CrossRef]
- Roquer-Beni, L.; Arnan, X.; Rodrigo, A.; Bosch, J. What Makes a Good Pollinator? Relationship between Pollinator Traits and Pollination Effectiveness in Apple Flowers. (Unpublished; Manuscript in Preparation).
- Bosch, J.; Kemp, W.P. Developing and establishing bee species as crop pollinators: The example of Osmia spp. (Hymenoptera: Megachilidae) and fruit trees. Bull. Entomol. Res. 2002, 92, 3–16. [Google Scholar] [PubMed]
- Sédivy, C.; Dorn, S. Towards a sustainable management of bees of the subgenus Osmia (Megachilidae; Osmia) as fruit tree pollinators. Apidologie 2014, 45, 88–105. [Google Scholar] [CrossRef]
- Vicens, N.; Bosch, J. Nest site orientation and relocation of populations of the orchard pollinator Osmia cornuta (Hymenoptera: Megachilidae). Environ. Entomol. 29, 69–75. [CrossRef]
- Guédot, C.; Bosch, J.; Kemp, W.P. Effect of three-dimension and color contrast on nest localization performance of two solitary bees (Hymenoptera: Megachilidae). J. Kansas Entomol. Soc. 2007, 80, 90–104. [Google Scholar] [CrossRef]
- Biddinger, D.; Joshi, N.; Rajotte, E.; Halbrendt, N. An immunomarking method to determine the foraging patterns of Osmia cornifrons and resulting fruit set in a cherry orchard. Apidologie 2013, 44, 738–749. [Google Scholar] [CrossRef]
- Torchio, P.F. Use of Osmia lignaria Say (Hymenoptera: Apoidea: Megachilidae) as a pollinator in an apple and prune orchard. J. Kansas Entomol. Soc. 1976, 49, 475–482. [Google Scholar]
- Torchio, P.F. Field experiments with Osmia lignaria propinqua Cresson as a pollinator in almond orchards: II, 1976 studies (Hymenoptera: Megachilidae). J. Kansas Entomol. Soc. 1981, 54, 824–836. [Google Scholar]
- Maeta, Y. Comparative studies on the biology of the bees of the genus Osmia in Japan, with special reference to their management for pollination of crops (Hymenoptera, Megachilidae). Bull. Tohoku Natl. Agric. Exp. Stn. 1978, 57, 1–221. (In Japanese) [Google Scholar]
- Márquez, J.; Bosch, J.; Vicens, N. Pollens collected by wild and managed populations of the potential orchard pollinator Osmia cornuta (Latr.) (Hym., Megachilidae). J. Appl. Entomol. 1994, 117, 353–359. [Google Scholar] [CrossRef]
- Maccagnani, B.; Ladurner, E.; Santi, F.; Burgio, G. Osmia cornuta (Hymenoptera, Megachilidae) as a pollinator of pear (Pyrus communis): Fruit- and seed-set. Apidologie 2003, 34, 207–216. [Google Scholar] [CrossRef][Green Version]
- Garratt, M.P.D.; Breeze, T.D.; Boreux, V.; Fountain, M.T.; McKerchar, M.; Webber, S.M.; Coston, D.J.; Jenner, N.; Dean, R.; Westbury, D.B.; et al. Apple pollination: Demand depends on variety and supply depends on pollinator identity. PLoS ONE 2016, 11, e0153889. [Google Scholar] [CrossRef] [PubMed]
- Bosch, J.; Kemp, W.P.; Trostle, G.E. Bee population returns and cherry yields in an orchard pollinated with Osmia lignaria (Hymenoptera: Megachilidae). J. Econ. Entomol. 2006, 99, 408–413. [Google Scholar] [CrossRef]
- Sheffield, C.S. Pollination, seed set and fruit quality in apple: Studies with Osmia lignaria (Hymenoptera: Megachilidae) in the Annapolis Valley, Nova Scotia, Canada. J. Pollinat. Ecol. 2014, 12. [Google Scholar] [CrossRef]
- Velthuis, H.H.W.; van Doorn, A. A century of advances in bumblebee domestication and the economic and environmental aspects of its commercialization for pollination. Apidologie 2006, 37, 421–451. [Google Scholar] [CrossRef]
- van der Steen, J.J.M. Indoor rearing of the solitary bee Osmia rufa L. Proc. Exp. Appl. Entomol. NEV Amst. 1997, 8, 81–84. [Google Scholar]
- Ladurner, E.; Maccagnani, B.; Tesoriero, D.; Nepi, M.; Felicioli, A. Laboratory rearing of Osmia cornuta Latreille (Hymenoptera Megachilidae) on artificial diet. Boll. Dell’ist. Entomol. Univ. Bologna 1999, 53, 133–146. [Google Scholar]
- Bosch, J.; Vicens, N. Body size as an estimator of production costs in a solitary bee. Ecol. Entomol. 2002, 27, 129–137. [Google Scholar] [CrossRef]
- Kester, D.E.; Griggs, W.H. Fruit setting in the almond: The effect of cross-pollinating various percentages of flowers. Proc. Am. Soc. Hortic. Sci. 1959, 74, 206–213. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Model Selection and Inference: A Practical Information-Theoretic Approach, 1st ed.; Springer: New York, NY, USA, 2002. [Google Scholar] [CrossRef]
- Barton, K. ‘MuMIn’: Multi-Model Inference. R Package Version 1.43.17. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 11 January 2021).
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. ‘nlme’: Linear and Nonlinear Mixed Effects Models. R Package Version 3.1-151. Available online: https://CRAN.R-project.org/package=nlme (accessed on 11 January 2021).
- R Core Team. R: A Language and Environment for Statistical Computing, Version 4.0.1; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Melin, A.; Rouget, M.; Midgley, J.J.; Donadson, J.S. Pollination ecosystem services in South African agricultural systems. S. Afr. J. Sci. 2014, 110, 1–9. [Google Scholar] [CrossRef]
- Sgolastra, F.; Arnan, X.; Pitts-Singer, T.L.; Maini, S.; Kemp, W.P.; Bosch, J. Pre-wintering conditions and post-winter performance in a solitary bee: Does diapause impose an energetic cost on reproductive success? Ecol. Entomol. 2016, 41, 201–210. [Google Scholar] [CrossRef]
- Felicioli, A. Le osmie. In Api e Impollinazione; Pinzauti, M., Ed.; Giunta Regionale Toscana: Firenze, Italy, 2000; pp. 159–188. [Google Scholar]
- Krunić, M.; Stanisaviljević, L.Ž. The Biology of European Orchard bee Osmia Cornuta (Latr.) (Hymenoptera: Megachilidae); Faculty of Biology University of Belgrade: Belgrade, Serbia, 2006. [Google Scholar]
- Bosch, J.; Kemp, W.P. How to Manage the Blue Orchard Bee: As an Orchard Pollinator; Sustainable Agriculture Network: Beltsville, MD, USA, 2001. [Google Scholar]
- Bosch, J.; Vicens, N. Relationship between body size, provisioning rate, longevity and reproductive success in females of the solitary bee Osmia cornuta. Behav. Ecol. Sociobiol. 2006, 60, 26–33. [Google Scholar] [CrossRef]
- Boyle, N.K.; Pitts-Singer, T.L. The effect of nest box distribution on sustainable propagation of Osmia lignaria (Hymenoptera: Megachilidae) in commercial tart cherry orchards. J. Insect Sci. 2016, 17, 41. [Google Scholar] [CrossRef]
- Bosch, J. Osmia cornuta Latr. (Hymenoptera, Megachilidae) as a potential pollinator in almond orchards: Releasing methods and nest-hole length. J. Appl. Entomol. 1994, 117, 151–157. [Google Scholar] [CrossRef]
- Bosch, J. Improvement of field management of Osmia cornuta Latr. for almond pollination (Hymenoptera, Megachilidae). Apidologie 1994, 25, 71–83. [Google Scholar] [CrossRef]
- Bosch, J. Parasitism in wild and managed populations of the almond pollinator Osmia cornuta Latr. (Hymenoptera, Megachilidae). J. Apicult. Res. 1992, 31, 77–82. [Google Scholar] [CrossRef]
- Bosch, J. Comparison of nesting materials for the orchard pollinator Osmia cornuta Latr. (Hymenoptera, Megachilidae). Entomol. Gen. 1995, 19, 285–289. [Google Scholar] [CrossRef]
- Torchio, P.F. Field experiments with the pollinator species, Osmia lignaria propinqua Cresson (Hymenoptera: Megachilidae) in apple orchards: III, 1977 studies. J. Kansas Entomol. Soc. 1984, 57, 517–521. [Google Scholar]
- Torchio, P.F. Field experiments with the pollinator species, Osmia lignaria propinqua Cresson in apple orchards: V, 1979–1980, methods of introducing bees, nesting success, seed counts, fruit yields (Hymenoptera: Megachilidae). J. Kansas Entomol. Soc. 1985, 58, 448–464. [Google Scholar]
- Pitts-Singer, T.L. Olfactory response of megachilid bees, Osmia lignaria, Megachile rotundata, and M. pugnata, to individual cues from old nest cavities. Environ. Entomol. 2007, 36, 402–408. [Google Scholar] [CrossRef]
- Vicens, N.; Bosch, J.; Blas, M. Análisis de los nidos de algunas Osmia (Hymenoptera, Megachilidae) nidificantes en cavidades preestablecidas. Orsis 1993, 8, 41–52. [Google Scholar]
- Bosch, J.; Vicens, N. Sex allocation in the solitary bee Osmia cornuta: Do females behave in agreement with Fisher’s theory? Behav. Ecol. Sociobiol. 2005, 59, 124–132. [Google Scholar] [CrossRef]
- Tepedino, V.J.; Torchio, P.F. The influence of nest-hole selection on sex ratio and progeny size in Osmia lignaria propinqua (Hymenoptera: Megachilidae). Ann. Entomol. Soc. Am. 1989, 82, 355–360. [Google Scholar] [CrossRef]
- Seidelmann, K.; Bienasch, A.; Pröhl, F. The impact of nest tube dimensions on reproduction parameters in a cavity nesting solitary bee, Osmia bicornis (Hymenoptera: Megachili-dae). Apidologie 2016, 47, 114–122. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).