Beyond the Decline of Wild Bees: Optimizing Conservation Measures and Bringing Together the Actors
Simple Summary
Abstract
1. Introduction
2. Why Should We Conserve Wild Bees?
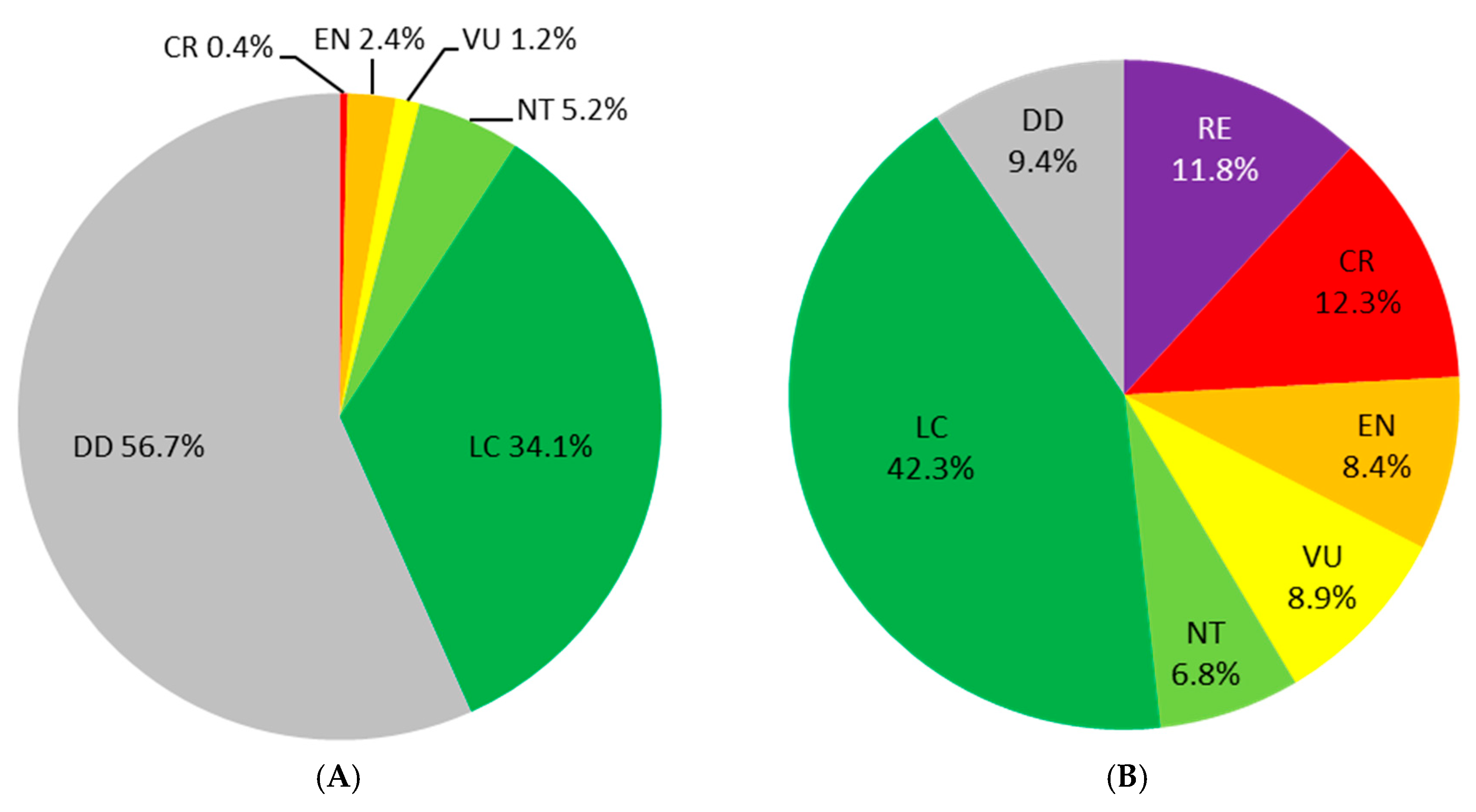
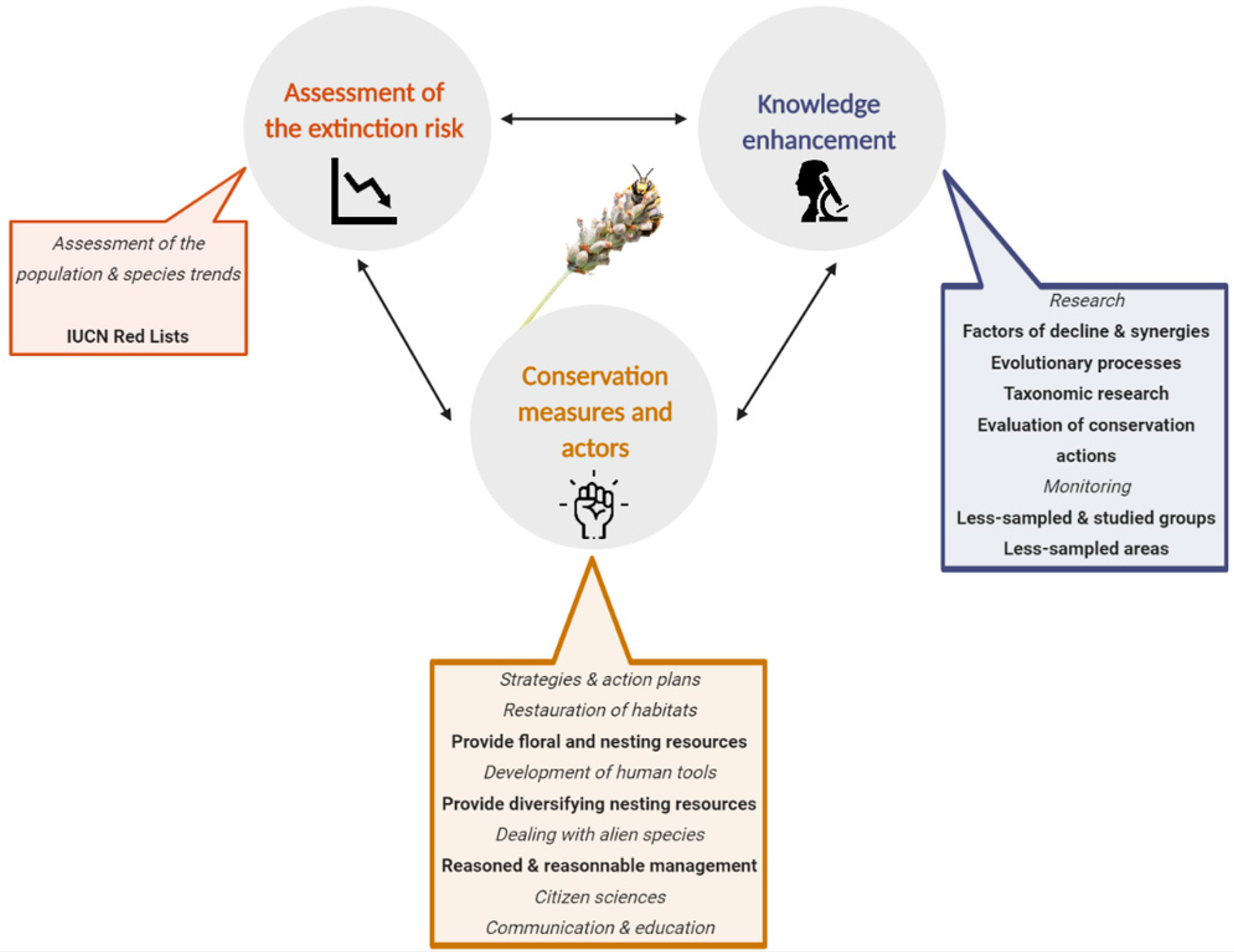
3. Preliminary Steps: Assessment of the Extinction Risk and Knowledge Enhancement
4. Conservation Measures and Actors
4.1. Protection and Restoration of Wild Bee Habitats
4.2. Conservation Measures in Anthropogenic Habitats: The Examples of Urban and Agricultural Areas
4.3. Providing Nesting Resources
4.4. Dealing with Invasive Alien Species
4.5. Conserving Wild Bees: An Action for All
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Underwood, E.; Darwin, G.; Gerritsen, E. Pollinator Initiatives in EU Member States: Success Factors and Gaps. Report for European Commission under Contract for Provision of Technical Support Related to Target 2 of the EU Biodiversity Strategy to 2020–Maintaining and Restoring Ecosystems and Their Services; Institute for European Environmental Policy: Brussels, Belgium, 2017; ENV.B.2/SER/2016/0018. [Google Scholar]
- Wilson, J.S.; Forister, M.L.; Carril, O.M. Interest exceeds understanding in public support of bee conservation. Front. Ecol. Environ. 2017, 15, 460–466. [Google Scholar] [CrossRef]
- Schatz, B.; Drossart, M.; Henry, M.; Geslin, B.; Allier, F.; Savajol, C.; Bellanger, S.; Michez, D. A boom of convergent information about the urgency to conserve pollinators. Acta Oecol. 2020, 105, (under major revision). [Google Scholar]
- Hart, A.G.; Sumner, S. Marketing insects: Can exploiting a commercial framework promote undervalued insect species? Insect Conserv. Diver. 2020, 13, 214–218. [Google Scholar] [CrossRef]
- Royal Mail. Royal Mail Issues Special Stamps Illustrating the Beauty of British Bees. Available online: https://www.royalmailgroup.com/en/press-centre/press-releases/royal-mail/royal-mail-issues-special-stamps-illustrating-the-beauty-of-british-bees/ (accessed on 20 April 2020).
- Didham, R.K.; Basset, Y.; Collins, C.M.; Leather, S.R.; Littlewood, N.; Menz, M.H.M.; Müller, J.; Packer, L.; Sauners, M.E.; Schönrogge, K.; et al. Interpreting insect declines: Seven challenges and a way forward. Insect Conserv. Diver. 2020, 13, 103–114. [Google Scholar] [CrossRef]
- Harvey, J.A.; Heinen, R.; Armbrecht, I.; Basset, Y.; Baxter-Gilbert, J.H.; Bezemer, T.M.; Böhm, M.; Bommarco, R.; Borges, P.A.V.; Cardoso, P.; et al. International scientists formulate a roadmap for insect conservation and recovery. Nat. Ecol. Evol. 2020, 4, 174–176. [Google Scholar] [CrossRef] [PubMed]
- van Klink, R.; Bowler, D.E.; Gongalsky, K.B.; Swengel, A.B.; Gentile, A.; Chase, J.M. Meta-analysis reveals declines in terrestrial but increases in freshwater insect abundances. Science 2020, 368, 417–420. [Google Scholar] [PubMed]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef]
- Goulson, D.; Nicholls, E.; Botias, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef]
- Gärdenfors, U. Classifying threatened species at national versus global levels. Trends Ecol. Evol. 2001, 16, 511–516. [Google Scholar]
- Primack, R.B.; Sarrazin, F.; Lecomte, J. Biologie de la Conservation; Dunord: Paris, France, 2012. [Google Scholar]
- IUCN. Guidelines for Appropriate Uses of IUCN Red List Data. Incorporating, as Annexes, the 1) Guidelines for Reporting on Proportion Threatened Species (ver. 1.1); 2) Guidelines on Scientific Collecting of Threatened Species (ver. 1.0); and 3) Guidelines for the Appropriate Use of the IUCN Red List by Business (ver. 1.0.). Version 3. Adopted by the IUCN Red List Committee, 3rd ed.; IUCN: Gland, Switzerland, 2016; p. 32. [Google Scholar]
- Nieto, A.; Roberts, S.P.; Kemp, J.; Rasmont, P.; Kulhmann, M.; García Criado, M.; Biesmeijer, J.C.; Bogusch, P.; Dathe, H.H.; De la Rúa, P.; et al. European Red List of Bees; Publication Office of the European Union: Luxembourg, 1998. [Google Scholar]
- Drossart, M.; Rasmont, P.; Vanormelingen, P.; Dufrêne, M.; Folschweiller, M.; Pauly, A.; Vereecken, N.J.; Vray, S.; Zambra, E.; D’Haeseleer, J.; et al. Belgian Red List of Bees; Belgian Science Policy 2018 (BRAIN-be–(Belgian Research Action through Interdisciplinary Networks); Presse Universitaire de l’Université de Mons: Mons, Belgium, 2018; p. 140. [Google Scholar]
- Ollerton, J.; Winfree, R.; Tarrant, S. How many flowering plants are pollinated by animals? Oikos 2011, 120, 321–326. [Google Scholar] [CrossRef]
- Willmer, P. Pollination and Floral Ecology; Princeton University Press: Princeton, NJ, USA, 2011; p. 840. [Google Scholar]
- IPBES. The Assessment Report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services on Pollinators, Pollination and Food Production; Potts, S.G., Imperatriz-Fonseca, V.L., Ngo, H.T., Eds.; Secretariat of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services: Bonn, Germany, 2016; p. 552. [Google Scholar]
- Potts, S.G.; Imperatriz-Fonseca, V.; Ngo, N.T.; Aizen, M.A.; Biesmeijer, J.C.; Breeze, T.D.; Dicks, L.V.; Garibaldi, L.A.; Hill, R.; Settele, J.; et al. Safeguarding pollinators and their values to human well-being. Nature 2016, 540, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.M.; Vaissiere, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Aizen, M.A.; Garibaldi, L.A.; Cunningham, S.A.; Klein, A.M. Long-term global trends in crop yield and production reveal no current pollination shortage but increasing pollinator dependency. Curr. Biol. 2008, 18, 1572–1575. [Google Scholar] [CrossRef] [PubMed]
- Gallai, N.; Salles, J.M.; Settele, J.; Vaissière, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Lautenbach, S.; Seppelt, R.; Liebscher, J.; Dormann, C.F. Spatial and temporal trends of global pollination benefit. PLoS ONE 2012, 7, e35954. [Google Scholar] [CrossRef] [PubMed]
- Eilers, E.J.; Kremen, C.; Greenleaf, S.S.; Garber, A.L.; Klein, A.M. Contribution of pollinator-mediated crops to nutrients in the human food supply. PLoS ONE 2011, 6, e21363. [Google Scholar] [CrossRef]
- Smith, M.R.; Singh, G.M.; Mozaffarian, D.; Myers, S.S. Effects of decreases of animal pollinators on human nutrition and global health: A modelling analysis. Lancet 2015, 386, 1964–1972. [Google Scholar] [CrossRef]
- Bauer, D.M.; Wings, I.S. The macroeconomic cost of catastrophic pollinator declines. Ecol. Econ. 2016, 126, 1–13. [Google Scholar] [CrossRef]
- Gibbs, J.; Elle, E.; Bobiwash, K.; Haapalainen, T.; Isaacs, R. Contrasting pollinators and pollination in native and non-native regions of highbush blueberry production. PLoS ONE 2016, 11, e0158937. [Google Scholar] [CrossRef]
- Rader, R.; Bartomeus, I.; Garibaldi, L.; Garratt, M.P.D.; Howlett, B.; Cunningham, S.A.; Mayfield, M.M.; Arthur, A.D.; Andersson, G.K.S.; Bommarco, R.; et al. Non-bee insects are important contributors to global crop pollination. Proc. Natl. Acad. Sci. USA 2016, 113, 146–151. [Google Scholar] [CrossRef]
- Packard, S.; Mutel, F.C. The Tallgrass Restoration Handbook: For Prairies, Savannas, and Woodlands; Society for Ecological Restoration by Island Press: Washington, DC, USA, 1997. [Google Scholar]
- Brittain, C.; Kremen, C.; Klein, A.M. Biodiversity buffers pollination in changing environmental conditions. Glob. Change Biol. 2013, 19, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, B. “Majoring” and “Minoring” by foraging bumblebees, Bombus vagans: An experimental analysis. Ecology 1979, 60, 245–255. [Google Scholar] [CrossRef]
- King, M.J.; Buchmann, S.L. Floral sonication by bees: Mesosomal vibration by Bombus and Xylocopa, but not Apis (Hymenoptera: Apidae), ejects pollen from poricidal anthers. J. Kans. Entomol. Soc. 2003, 76, 295–305. [Google Scholar]
- Fründ, J.; Dormann, C.F.; Holzschuh, A.; Tscharntke, T. Bee diversity effects on pollination depend on functional complementarity and niche shifts. Ecology 2013, 94, 2042–2045. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Carvalhiero, L.G.; Leonhardt, S.D.; Aizen, M.A.; Blaauw, B.R.; Isaacs, R.; Kuhlmann, M.; Kleijn, D.; Klein, A.M.; Kremen, C.; et al. From research to action: Enhancing crop yield through wild pollinators. Front. Ecol. Environ. 2014, 12, 439–447. [Google Scholar] [CrossRef]
- Isaacs, R.; Williams, N.; Ellis, J.; Pitts-Singer, L.; Bommarco, R.; Vaughan, M. Integrated crop pollination: Combining strategies to ensure stable and sustainable yields of pollination-dependent crops. Basic Appl. Ecol. 2017, 22, 44–60. [Google Scholar] [CrossRef]
- Grab, H.; Branstetter, M.G.; Amon, N.; Urban-Mead, K.R.; Park, M.G.; Gibbs, J.; Blitzer, E.J.; Poveda, K.; Loeb, G.; Danforth, B.N. Agriculturally dominated landscapes reduce bee phylogenetic diversity and pollination services. Science 2019, 363, 282–284. [Google Scholar] [CrossRef]
- Martins, K.T.; Gonzalez, A.; Lechowicz, M.J. Pollination services are mediated by bee functional diversity and landscape context. Agric. Ecosyst. Environ. 2015, 200, 12–20. [Google Scholar] [CrossRef]
- Säumel, I.; Weber, F.; Kowarik, I. Toward livable and healthy urban streets: Roadside vegetation provides ecosystem services where people live and more. Environ. Sci. Policy 2016, 62, 24–33. [Google Scholar] [CrossRef]
- Cummings, A.R.; Read, J.M. Drawing on traditional knowledge to identify and describe ecosystem services associated with Northern Amazon’s multiple-use plants. Integrative J. Biodivers. Sci. Ecosyst. Serv. Manag. 2016, 12, 39–56. [Google Scholar] [CrossRef]
- Massaro, F.C.; Brooks, P.R.; Wallace, H.M.; Nsengiyumva, V.; Narokai, L.; Russel, F.D. Effect of Australian propolis from stingless bees (Tetragonula carbonaria) on pre-contracted human and porcine isolated arteries. PLoS ONE 2013, 8, e81297. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, S.D.; Kwaiser, K.S.; Heard, S.B. Bee communities (Hymenoptera: Apoidea) of small Iowa hill prairies are as diverse and rich as those of large prairie reserve. Biodivers. Conserv. 2010, 19, 1699–1709. [Google Scholar] [CrossRef]
- Ollerton, J. Pollinator Diversity: Distribution, Ecological Function, and Conservation. Annu. Rev. Ecol. Evol. Syst. 2017, 48, 353–376. [Google Scholar] [CrossRef]
- Valiente-Banuet, A.; Aizen, M.A.; Alcantara, J.M.; Arroyo, K.; Cocucci, A.; Galetti, M.; Garcia, M.B.; Garcia, D.; Gomez, J.M.; Jordano, P.; et al. Beyond species loss: The extinction of ecological interactions in a changing world. Funct. Ecol. 2015, 29, 299–307. [Google Scholar] [CrossRef]
- Jacquemin, F.; Violle, C.; Munoz, F.; Mahy, G.; Rasmont, P.; Roberts, S.P.; Vray, S.; Dufrêne, M. Loss of pollinator specialization revealed by historical opportunistic data: Insights from network-based analysis. PLoS ONE 2020, 15, e0235890. [Google Scholar] [CrossRef]
- Waser, N.M.; Ollerton, J. Plant-Pollinator Interactions: From Specialization to Generalization; University of Chicago Press: Chicago, IL, USA, 2006. [Google Scholar]
- Cane, J.; Sipes, S. Floral Specialization by Bees: Analytical Methods and a Revised Lexicon for Oligolecty. In Plant-Pollinator Interactions: From Specialization to Generalization; Waser, O., Ed.; University of Chicago: Chicago, IL, USA, 2006; pp. 99–122. [Google Scholar]
- Muller, A.; Kuhlmann, M. Pollen hosts of western palaearctic bees of the genus Colletes (Hymenoptera: Colletidae): The Asteraceae paradox. Biol. J. Linn. Soc. 2008, 95, 719–733. [Google Scholar] [CrossRef]
- Dötterl, S.; Vereecken, N.J. The chemical ecology and evolution of bee-flower interactions: A review and perspectives. Can. J. Zool. 2010, 88, 668–697. [Google Scholar] [CrossRef]
- Biesmeijer, J.C.; Roberts, S.P.M.; Reemer, M.; Ohlemüller, R.; Edwards, M.; Peeters, T.; Schaffers, A.P.; Potts, S.G.; Kleukers, R.; Thomas, C.D.; et al. Parallel Declines in Pollinators and Insect-Pollinated Plants in Britain and the Netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef]
- Roger, N.; Moerman, R.; Carvalheiro, L.G.; Aguirre-Guitierrez, J.; Jacquemart, A.L.; Kleijn, D.; Lognay, G.; Moquet, L.; Quinet, M.; Rasmont, P.; et al. Impact of pollen resources drift on common bumblebees in NW Europe. Glob. Chang. Biol. 2017, 23, 68–76. [Google Scholar] [CrossRef]
- Diaz, S.; Purvis, A.; Cornelissen, J.H.C.; Mace, G.M.; Donoghue, M.J.; Ewers, R.M.; Jordano, P.; Pearse, W.D. Functional traits, the phylogeny of function and ecosystem service vulnerability. Ecol. Evol. 2013, 3, 2958–2975. [Google Scholar] [CrossRef]
- Aizen, M.; Sabatino, M.; Tylianakis, J.M. Specialization and rarity predict non-random loss of interactions from mutualist networks. Science 2012, 335, 1486–1489. [Google Scholar] [CrossRef] [PubMed]
- Dirzo, R.; Youg, H.S.; Galetti, M.; Ceballos, G.; Isaac, N.J.B.; Collen, B. Defaunation in the Anthropocene. Science 2014, 345, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Powney, G.D.; Carvell, C.; Edwards, M.; Morris, R.K.A.; Roy, H.E.; Woodcock, B.A.; Isaac, N.J.B. Widespread losses of pollinating insects in Britain. Nat. Commun. 2019, 10, 1018. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.S.; Pilgrim, J.D.; Lamoreux, J.F.; Hoffmann, M.; Brooks, T.M. The value of the IUCN Red List for conservation. Trends Ecol. Evol. 2006, 21, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Westrich, P.; Frommer, U.; Mandery, K.; Riemann, H.; Ruhnke, H.; Saure, C.; Voith, J. Rote Liste und Gesamtartenliste der Bienen (Hymenoptera, Apidae) Deutschlands (5. Fassung, Dezember 2011). Rote List. Gefährdeter Tiere Pflanz. Pilze Dtschl. 2011, 3, 371–416. [Google Scholar]
- Reemer, M. Basisrapport voor de Rode Lijst Bijen; EIS Kenniscentrum Insecten: Leiden, The Netherlands, 2018; p. 174. [Google Scholar]
- Hatfield, R.; Colla, S.; Jepsen, S.; Richardson, L.L.; Thorp, R.W.; Jordan, S.F. IUCN Assessments for North American Bombus ssp. for the North American IUCN Bumble Bee Specialist Group. The Xerces Society for Invertebrate Conservation. Available online: https://xerces.org/sites/default/files/publications/14-065.pdf (accessed on 18 August 2020).
- Forister, M.L.; Pelton, E.M.; Black, S.H. Declines in insect abundance and diversity: We know enough to act now. Conserv. Sci. Pract. 2019, 1, e80. [Google Scholar] [CrossRef]
- Nicholson, C.C.; Egan, P.A. Natural hazard threats to pollinators and pollination. Glob. Chang. Biol. 2019, 26, 380–391. [Google Scholar] [CrossRef]
- Meeus, I.; Pisman, M.; Smagghe, G.; Piot, N. Interaction effects of different drivers of wild bee decline and their influence on host-pathogen dynamics. Curr. Opin. Insect. Sci. 2018, 26, 136–141. [Google Scholar] [CrossRef]
- Bartomeus, I.; Stavert, J.R.; Ward, D.; Aguado, O. Historical collections as a tool for assessing the global pollination crisis. Philos. Trans. R. Soc. B 2019, 374, 20170389. [Google Scholar] [CrossRef]
- Frankham, R.; Ballou, J.D.; Briscoe, D.A. A Primer of Conservation Genetics, Illustrated, Reprint; Cambridge University Press: Cambridge, UK, 2004. [Google Scholar]
- Epps, C.W.; Keyghobadi, N. Landscape genetics in a changing world: Disentangling historical and contemporary influences and inferring change. Mol. Ecol. 2015, 24, 6021–6040. [Google Scholar] [CrossRef]
- Woodard, S.H.; Lozier, J.D.; Goulson, D.; Williams, P.H.; Strange, J.P.; Jha, S. Molecular tools and bumble bees: Revealing hidden details of ecology and evolution in a model system. Mol. Ecol. 2015, 24, 2916–2936. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Uribe, M.M.; Soro, A.; Jha, S. Conservation genetics of bees: Advances in the application of molecular tools to guide bee pollinator conservation. Conserv. Genet. 2017, 18, 501–506. [Google Scholar] [CrossRef]
- Lozier, J.D.; Zayed, A. Bee conservation in the age of genomics. Conserv. Genet. 2016, 18, 713–729. [Google Scholar] [CrossRef]
- Zayed, A.; Packer, L. Complementary sex determination substantially increases extinction proneness of haplodiploid populations. Proc. Natl. Acad. Sci. USA 2005, 102, 10742–10746. [Google Scholar] [CrossRef]
- Packer, J.G.; Delean, S.; Kueffer, C.; Prider, J.; Abley, K.; Farcelli, J.M.; Carthew, S.M. Native faunal communities depend on habitat from non-native plants in novel but not in natural ecosystems. Biodivers. Conserv. 2016, 25, 503–523. [Google Scholar] [CrossRef]
- Cerna, K.; Munclinger, P.; Vereecken, N.J.; Straka, J. Mediterranean lineage endemism, cold-adapted paleodemographic dynamics and recent changes in population size in two solitary bees of the genus Anthophora. Conserv. Genet. 2017, 18, 521–538. [Google Scholar] [CrossRef]
- Koch, J.B.; Looney, C.; Sheppard, W.S.; Stange, J.P. Patterns of population genetic structure and diversity across bumble bee communities in the Pacific Northwest. Conserv. Genet. 2017, 18, 507–520. [Google Scholar] [CrossRef]
- Lecocq, T.; Gérard, M.; Michez, D.; Dellicour, S. Conservation genetics of European bees: New insights from the continental scale. Conserv. Genet. 2017, 18, 585–596. [Google Scholar] [CrossRef]
- Lecocq, T.; Michez, D.; Gérard, M.; Vereecken, N.J.; Delangre, J.; Rasmont, P.; Vray, S.; Dufrêne, M.; Mardulyn, P.; Dellicour, S. Divergent geographic patterns of genetic diversity among wild bees: Conservation implications. Divers. Distrib. 2018, 24, 1860–1868. [Google Scholar] [CrossRef]
- Winfree, R.; Bartomeus, I.; Cariveau, D.P. Native pollinators in anthropogenic habitats. Annu. Rev. Ecol. Evol. Syst. 2011, 42, 1–22. [Google Scholar] [CrossRef]
- Belsky, J.; Joshi, N.K. Impact of Biotic and Abiotic Stressors on Managed and Feral Bees. Insects 2019, 10, 233. [Google Scholar] [CrossRef] [PubMed]
- Cely-Santos, M.; Philpott, S.M. Local and landscape habitat influences on bee diversity in agricultural landscapes in Anolaima, Colombia. J. Insect Conserv. 2019, 23, 133–146. [Google Scholar] [CrossRef]
- Grover, S.N.; Miller, J.E.D.; Damschen, E. Indirect Effects of Landscape Spatial Structure and Plant Species Richness on Pollinator Diversity in Ozark Glades. Castanea 2017, 82, 24–31. [Google Scholar] [CrossRef]
- Le Féon, V.; Schermann-Legionnet, A.; Delettre, Y.R.; Aviron, S.; Billeter, R.; Bugter, R.; Hendrickx, F.; Burel, F. Intensification of agriculture, landscape composition and wild bee communities: A large scale study in four European countries. Agric. Ecosyst. Environ. 2010, 137, 143–150. [Google Scholar] [CrossRef]
- Goulson, D.; Stout, J.C.; Kells, A.R. Do exotic bumblebees and honey bees compete with native flower-visiting insects in Tasmania. J. Insect. Conserv. 2002, 6, 179–189. [Google Scholar] [CrossRef]
- Bryne, A.; Fitzpatrick, U. Bee conservation policy at the global, regional and national levels. Apidologie 2009, 40, 194–210. [Google Scholar] [CrossRef]
- Stout, J.C.; Morales, C.L. Ecological Impacts of invasive alien species on bees. Apidologie 2009, 40, 388–409. [Google Scholar] [CrossRef]
- European Union. The EU Biodiversity Strategy to 2020; Office of the European Union: Luxembourg, 2011; 28p, ISBN 978-92-79-20762-4. [Google Scholar]
- Belgian National Focal Point to the Convention on Biological Diversity (Belgian NFP-CBD). Biodiversity 2020–Update of Belgium’s National Biodiversity Strategy; Royal Belgian Institute of Natural Sciences: Brussels, Belgium, 2020; p. 148. [Google Scholar]
- The Heinz Center. Pollinators and the State Wildlife Action Plans: Voluntary Guidance for State; Wildlife Agencies: Washington, DC, USA, 2013; p. 20. [Google Scholar]
- Gadoum, S.; Roux-Fouillet, J.M. Plan National D’actions « France Terre de Pollinisateurs » Pour la Préservation des Abeilles et des Insectes Pollinisateurs Sauvages; Office Pour les Insectes et leur Environnement–Ministère de l’Écologie, du Développement Durable et de l’Énergie: Paris, France, 2016; p. 136. [Google Scholar]
- Pollinator Plan Steering Group. All-Ireland Pollinator Plan 2015–2020; National Biodiversity Data Centre Series No. 3; National Biodiversity Data Centre: Waterford, Ireland, 2015; 48p, ISSN 2009-6844. [Google Scholar]
- Saunders, M.E.; Smith, T.J.; Rader, R. Bee conservation: Key role of managed bees. Sciences 2018, 360, 381–389. [Google Scholar]
- Turo, K.J.; Gardiner, M.M. From potential to practical: Conserving bees in urban public green spaces. Front. Ecol. Environ. 2019, 17, 167–175. [Google Scholar] [CrossRef]
- Sobral-Souza, T.; Vancine, M.H.; Ribeiro, M.C.; Lima-Ribeiro, M. Efficiency of protected areas in Amazon and atlantic forest conservation: A spatial temporal view. Acta Oecologica 2018, 87, 1–7. [Google Scholar] [CrossRef]
- Krechemer, F.S.; Marchioro, C.A. Past, present and future distributions of bumblebees in South America: Identifying priority species and areas for conservation. J. Appl. Ecol. 2020. [Google Scholar] [CrossRef]
- Tonietto, R.K.; Larkin, D.J. Habitat restoration benefits wild bees: A meta-analysis. J. Appl. Ecol. 2018, 55, 582–590. [Google Scholar] [CrossRef]
- Svensson, B.; Lagerlöf, J.G.; Svensson, B.G. Habitat preferences of nest-seeking bumble bees (Hymenoptera: Apidae) in an agricultural landscape. Agr. Ecosyst. Environ. 2000, 77, 247–255. [Google Scholar] [CrossRef]
- Carrié, R.; Andrieu, E.; Cunningham, S.A.; Lentini, P.E.; Loreau, M.; Ouin, A. Relationships among ecological traits of wild bee communities along gradients of habitat amount and fragmentation. Ecography 2017, 40, 85–97. [Google Scholar] [CrossRef]
- Ferreira, P.A.; Boscolo, D.; Carvalheiro, L.G.; Biesmeijer, J.C.; Rocha, P.L.B.; Viana, B.F. Responses of bees to habitat loss in fragmented landscapes of Brazilian atlantic rainforest. Landsc. Ecol. 2015, 30, 2067–2078. [Google Scholar] [CrossRef]
- Folschweiller, M.; Drossart, M.; D’Haeseleer, J.; Marescaux, Q.; Rey, G.; Rousseau-Piot, J.S.; Barbier, Y.; Dufrêne, M.; Hautekeete, N.; Jacquemin, F.; et al. Plan d’action transfrontalier en faveur des pollinisateurs sauvages. In Projet Interreg V SAPOLL–Sauvons nos Pollinisateurs–Samenwerken voor Pollinators; Université de Mons: Mons, Belgium, 2019; p. 147. ISBN 978-2-87325-118-5. [Google Scholar]
- Hall, D.M.; Steiner, R. Insect pollinator conservation policy innovations at subnational levels: Lessons for lawmakers. Environ. Sci. Policy 2019, 93, 118–128. [Google Scholar] [CrossRef]
- United Nations. United Nations, Department of Economic and Social Affairs. Available online: https://www.un.org/development/desa/publications/2018-revision-of-world-urbanization-prospects.html (accessed on 14 August 2020).
- Kennedy, C.M.; Lonsdorf, E.; Neel, M.C.; Williams, N.M.; Ricketts, T.H.; Winfree, R.; Bommarco, R.; Brittain, C.; Burley, A.L.; Cariveau, D.; et al. A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecol. Lett. 2013, 16, 584–599. [Google Scholar] [CrossRef]
- Kohler, F.; Verhulst, J.; Van Klink, R.; Kleijn, D. At what spatial scale do high-quality habitats enhance the diversity of forbs and pollinators in intensively farmed landscapes? J. Appl. Ecol. 2008, 45, 753–762. [Google Scholar] [CrossRef]
- Terzo, M.; Rasmont, P. Abeilles Sauvages, Bourdons et Autres Insectes Pollinisateurs; Les Livrets de l’Agriculture, 14, DGA, Ministère de la Région Wallonne: Namur, Belgium, 2007; p. 64. [Google Scholar]
- Mader, E.; Shepherd, M.; Vaughan, M.; Black, S.H.; LeBuhn, G. Attracting native pollinators: Protecting North America’s bees and butterflies. In The Xerces Society Guide; Storey Publishing: North Adams, MA, USA, 2011; p. 372. [Google Scholar]
- Nowakowski, M.; Pywell, R.F. Habitat Creation and Management for Pollinators; Centre for Ecology & Hydrology: Wallingford, UK, 2016; p. 77. [Google Scholar]
- Gosselin, M.; Moerman, R.; Terzo, M.; Vereecken, N.; Rasmont, P. Abeilles sauvages, bourdons et autres insectes pollinisateurs. In Collection Agrinature no 9; Service Public de Wallonie: Namur, Belgium, 2018; p. 155. [Google Scholar]
- Cramner, L.; McCollin, D.; Ollerton, J. Landscape structure influences pollinator movements and directly affects plant reproductive success. Oikos 2012, 121, 562–568. [Google Scholar] [CrossRef]
- Morandin, L.A.; Winston, M.L.; Franklin, M.T.; Abbott, V.A. Lethal and sub-lethal effects of Spinosad on bumble bees (Bombus impatiens Cresson). Pest Manag. Sci. 2005, 61, 619–626. [Google Scholar] [CrossRef]
- Hall, D.M.; Camilo, G.R.; Tonietto, R.K.; Ollerton, J.; Ahrné, K.; Arduser, M.; Ascher, J.S.; Baldock, K.C.R.; Fowler, R.; Frankie, G.; et al. The city as a refuge for insect pollinators. Conserv. Biol. 2017, 31, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Crone, E.E.; Brown, L.M.; Hodgson, J.A.; Lutscher, F.; Schultz, C.B. Faster movement in nonhabitat matrix promotes range shifts in heterogeneous landscapes. Ecology 2019, 100, e02701. [Google Scholar] [CrossRef] [PubMed]
- Senapathi, D.; Goddard, M.A.; Kunin, W.E.; Baldock, K.C.R. Landscape impacts on pollinator communities in temperate systems: Evidence and knowledge gaps. Funct. Ecol. 2017, 31, 26–37. [Google Scholar] [CrossRef]
- Osborne, J.L.; Martin, A.P.; Shortall, C.R.; Todd, A.D.; Goulson, D.; Knight, M.E.; Hale, R.J.; Sanderson, R.A. Quantifying and comparing bumblebee nest densities in gardens and countryside habitats: Bumblebee nest survey in gardens and countryside. J. Appl. Ecol. 2008, 45, 784–792. [Google Scholar] [CrossRef]
- Fortel, L.; Henry, M.; Guilbaud, L.; Guirao, A.L.; Kuhlmann, M.; Mouret, H.; Rollin, O.; Vaissiere, B.E. Decreasing abundance, increasing diversity and changing structure of the wild bee community (Hymenoptera: Anthophila) along an urbanization gradient. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Givetash, L. Bees are Dying at an Alarming Rate. Amsterdam May Have the Answer. NBC News, 7 September 2018. Available online: https://www.nbcnews.com/news/world/bees-are-dying-alarming-rate-amsterdammay-have-answer-n897856 (accessed on 15 July 2020).
- Greenleaf, S.S.; Williams, N.M.; Winfree, R.; Kremen, C. Bee foraging ranges and their relationship to body size. Oecologia 2007, 153, 589–596. [Google Scholar] [CrossRef]
- Warzecha, D.; Diekötter, T.; Wolters, V.; Jauker, F. Intraspecific body size increases with habitat fragmentation in wild bee pollinators. Landsc. Ecol. 2016, 31, 1449–1455. [Google Scholar] [CrossRef]
- Gérard, M.; Martinet, B.; Maebe, K.; Marshall, L.; Smagghe, G.; Vereecken, N.J.; Vray, S.; Rasmont, P.; Michez, D. Shift in size of bumblebee queens over the last century. Glob. Chang. Biol. 2019, 26, 1185–1195. [Google Scholar] [CrossRef]
- La Vie Sauvage Emprunte Aussi nos Routes. Available online: http://biodiversite.wallonie.be/fr/la-vie-sauvage.html?IDC=3649 (accessed on 14 August 2020).
- François, D.; Le Féon, V. Abeilles sauvages et dépendances vertes routières, Pourquoi et comment développer la capacité d’accueil des dépendances vertes routières en faveur des abeilles sauvages. In Ouvrages Scientifiques, OSI2; Ifsttar: Marne-la-Vallée, France, 2017; p. 120. ISBN 978-2-85782-733-7. [Google Scholar]
- Kleijn, D.; Baquero, R.A.; Clough, Y.; Diaz, M.; De Esteban, J.; Fernandez, F.; Gabriel, D.; Herzog, F.; Holzschuh, A.; Jöhl, R.; et al. Mixed biodiversity benefits of agri-environment schemes in five European countries. Ecol. Lett. 2006, 9, 243–254. [Google Scholar] [CrossRef]
- Kohler, F.; Verhulst, J.; Knop, E.; Herzog, F.; Kleijn, D. Indirect effects of grassland extensification schemes on pollinators in two contrasting European countries. Biol. Conserv. 2007, 135, 302–307. [Google Scholar] [CrossRef]
- Holzschuh, A.; Steffan-Dewenter, I.; Kleijn, D.; Tscharntke, T. Diversity of flower-visiting bees in cereal fields: Effects of farming system, landscape composition and regional context. J. Appl. Ecol. 2007, 44, 41–49. [Google Scholar] [CrossRef]
- Rundlöf, M.; Nilsson, H.; Smith, H.G. Interacting effects of farming practice and landscape context on bumble bees. Biol. Conserv. 2008, 22, 343–349. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. “Crop Yields”. Published online at OurWorldInData.org. Available online: https://ourworldindata.org/crop-yields (accessed on 14 August 2020).
- Lane, I.; Portman, Z.M.; Cariveau, D.P. Floral resource diversity drives bee community diversity in prairie restorations along an agricultural landscape gradient. J. Appl. Ecol. 2020. [Google Scholar] [CrossRef]
- Albrecht, M.; Duelli, P.; Muller, C.; Kleijn, D.; Schmid, B. Effects of a Swiss agri-environment scheme on pollinator communities and seed set of plants in nearby intensely managed farmland. J. Appl. Ecol. 2007, 44, 813–822. [Google Scholar] [CrossRef]
- Grass, I.; Albrecht, J.; Jauker, F.; Diekötter, T.; Warzecha, D.; Wolters, V.; Farwig, N. Much more than bees–Wildflower plantings support highly diverse flower-visitor communities from complex to structurally simple agricultural landscapes. Agric. Ecosyst. Environ. 2016, 225, 45–53. [Google Scholar] [CrossRef]
- Cole, L.J.; Kleijn, D.; Dicks, L.V.; Stout, J.C.; Potts, S.G.; Albrecht, M.; Balzan, M.V.; Bartomeus, I.; Bebeli, P.J.; Bevk, D.; et al. A critical analysis of the potential for EU Common Agricultural Policy measures to support wild pollinators on farmland. J. Appl. Ecol. 2020, 57, 681–694. [Google Scholar] [CrossRef]
- Kleijn, D.; Berendse, F.; Smit, R.; Gilissen, N. Agri-environment schemes do not effectively protect biodiversity in Dutch agricultural landscapes. Nature 2001, 413, 723–725. [Google Scholar] [CrossRef]
- Terzo, M.; Rasmont, P. MALVAS: Méthodes Agroenvironnementales Liées à la Valorisation des Abeilles Sauvages. Université de Mons-Hainaut; Direction Générale de l’Agriculture de la Région Wallonne: Mons, Namur, Belgium, 2007; p. 77. [Google Scholar]
- Wood, T.J.; Holland, J.M.; Goulson, D. Pollinator-friendly management does not increase the diversity of farmland bees and wasps. Biol. Conserv. 2015, 187, 120–126. [Google Scholar] [CrossRef]
- Wood, T.J.; Holland, J.M.; Hughes, W.O.; Goulson, D. Targeted agri-environment schemes significantly improve the population size of common farmland bumblebee. Mol. Ecol. 2015, 24, 1668–1680. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Farrell, L.M.; Patch, H.M.; Grozinger, C.M.; Tooker, J.F. Consistent pollen nutritional intake drives bumble bee (Bombus impatiens) colony growth and reproduction across different habitats. Ecol. Evol. 2018, 8, 5765–5776. [Google Scholar] [CrossRef]
- Batáry, P.; Dicks, L.V.; Kleijn, D.; Sutherland, W.J. The role of agri-environment schemes in conservation and environmental management. Conserv. Biol. 2015, 29, 1006–1016. [Google Scholar] [CrossRef] [PubMed]
- Geppert, C.; Hass, A.; Földesi, R.; Donko, B.; Akter, A.; Tscharntke, T.; Batary, P. Agri-environment schemes enhance pollinator richness and abundance but bumblebee reproduction depends on field size. J. Appl. Ecol. 2020. [Google Scholar] [CrossRef]
- Wood, T.J.; Holland, J.M.; Goulson, D. Providing foraging resources for solitary bees on farmland: Current schemes for pollinators benefit a limited suite of species. J. Appl. Ecol. 2017, 54, 323–333. [Google Scholar] [CrossRef]
- Mallinger, R.E.; Franco, J.G.; Prischmann-Voldseth, D.A.; Prasifka, J.R. Annual cover crops for managed and wild bees: Optimal plant mixtures depend on pollinator enhancement goals. Agric. Ecosyst. Environ. 2019, 273, 107–116. [Google Scholar] [CrossRef]
- Nichols, R.N.; Goulson, D.; Holland, J.M. The best wildflowers for wild bees. J. Insect. Conserv. 2019, 23, 1–12. [Google Scholar] [CrossRef]
- Hoehn, P.; Tscharntke, T.; Tylianakis, J.M.; Steffan-Dewenter, I. Functional diversity of bee pollinators increases crop yield. Proc. R. Soc. B Biol. Sci. 2008, 275, 2283–2291. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Steffan-Dewenter, I.; Winfree, R.; Aizen, M.A.; Bommarco, R.; Cunningham, S.A.; Kremen, C.; Cavalheiro, L.G.; Harder, L.D.; Afik, O.; et al. Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 2013, 339, 1608–1611. [Google Scholar] [CrossRef]
- Morse, R.A.; Calderone, N.W. The value of honey bees as pollinators of U.S. crops in 2000. Bee Cult. 2000, 128, 1–15. [Google Scholar]
- Sumner, D.A.; Boriss, H. Bee-conomics and the leap in pollination fees. Agr. Resour. Econ. Update 2006, 9, 9–11. [Google Scholar]
- Velthuis, H.H.; Van Doorn, A. A century of advances in bumblebee domestication and the economic and environmental aspects of its commercialization for pollination. Apidologie 2006, 37, 421–451. [Google Scholar] [CrossRef]
- Goulson, D. Bumblebees: Behaviour and Ecology; Oxford University Press: Oxford, UK, 2009; p. 317. [Google Scholar]
- Lye, G.C.; Park, K.; Osborne, J.; Holland, J.M.; Goulson, D. Assessing the value of Rural Stewardship schemes for providing foraging resources and nesting habitat for bumblebee queens (Hymenoptera: Apidae). Biol. Conserv. 2009, 142, 2023–2032. [Google Scholar] [CrossRef]
- DEFRA. The National Pollinator Strategy: For Bees and other Pollinators in England. Defra Publ.PB14221. Available online: https://www.gov.uk/government/publications/national-pollinator-strategy-for-bees-and-other-pollinators-in-england (accessed on 21 April 2020).
- Gérard, M.; Vanderplanck, M.; Wood, T.J.; Michez, D. Global warming and plant-pollinator mismatches. Emerg. Top. Life Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tuell, J.K.; Fiedler, A.K.; Landis, D.; Isaacs, R. Visitation by wild and managed bees (Hymenoptera: Apoidea) to eastern US native plants for use in conservation programs. Environ. Entomol. 2008, 37, 707–718. [Google Scholar] [CrossRef]
- Quinet, M.; Mabeluanga, T.; Moquet, L.; Jacquemart, A.L. Introduction of new tools to improve pollination in European pear orchards. Sci. Hortic. 2016, 213, 5–12. [Google Scholar] [CrossRef]
- Moquet, L.; Vanderplanck, M.; Moerman, R.; Quinet, M.; Roger, N.; Michez, D.; Jacquemart, A.L. Bumblebees depend on ericaceous species to survive in temperate heathlands. Insect Conserv. Diver. 2017, 10, 78–93. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Tooker, J.F.; Grozinger, C.M.; Patch, H.M. Bee nutrition and floral resource restoration. Curr. Opin. Insect Sci. 2015, 10, 133–141. [Google Scholar] [CrossRef]
- Filipiak, M. A better understanding of bee nutritional ecology is needed to optimize conservation strategies for wild bees-the application of ecological stoichiometry. Insects 2018, 9, 85. [Google Scholar] [CrossRef]
- Gresty, C.E.A.; Clare, E.; Devey, D.S.; Cowan, R.S.; Csiba, L.; Malakasi, P.; Lewis, O.T.; Willis, K.T. Flower preferences and pollen transport networks for cavity-nesting solitary bees: Implications for the design of agri-environment schemes. Ecol. Evol. 2018, 15, 7574–7587. [Google Scholar] [CrossRef]
- Bukovinszky, T.; Rikken, I.; Evers, S.; Wäckers, F.L.; Biesmeijer, J.C.; Prins, H.H.T.; Kleijn, D. Effects of pollen species composition on the foraging behaviour and offspring performance of the mason bee Osmia bicornis (L.). Basic Appl. Ecol. 2017, 18, 21–30. [Google Scholar] [CrossRef]
- Filipiak, M.; Kuszewska, K.; Asselman, M.; Denisow, B.; Stawiarz, E.; Woyciechowski, M.; Weiner, J. Ecological stoichiometry of the honeybee: Pollen diversity and adequate species composition are needed to mitigate limitations imposed on the growth and development of bees by pollen quality. PLoS ONE 2017, 12, e0183236. [Google Scholar] [CrossRef]
- Filipiak, M. Key pollen host plants provide balanced diets for wild bee larvae: A lesson for planting flower strips and hedgerows. J. Appl. Ecol. 2019, 56, 1410–1418. [Google Scholar] [CrossRef]
- Sterner, E.W.; Elser, J.J. Ecological stoichiometry: Overview. In Encyclopedia of Ecology; Jorgensen, S.E., Fath, B.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 1101–1116. [Google Scholar]
- Griffiths, J.; Nicholls, E.; Goulson, D. Companion planting to attract pollinators increases the yield and quality of strawberry fruit in gardens and allotments. Ecol. Entomol. 2020. [Google Scholar] [CrossRef]
- Tosi, S.; Nieh, J.C. Lethal and sublethal synergistic effects of a new systemic pesticide, flupyradifurone (Sivanto), on honeybees. Proc. R. Soc. B Biol. Sci. 2019, 286, 2019433. [Google Scholar] [CrossRef] [PubMed]
- Gradish, A.E.; van der Steen, J.; Scott-Dupree, C.D.; Cabrera, A.R.; Cutler, G.C.; Goulson, D.; Klein, O.; Lehmann, D.M.; Luckmann, J.; O’Neill, B.; et al. Comparison of pesticide exposure in honey bees (Hymenoptera: Apidae) and Bumble Bees (Hymenoptera: Apidae): Implications for Risk Assessments. Env. Entomol. 2019, 48, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Biddinger, D.J.; Robertson, J.L.; Mullin, C.; Frazier, J.; Ashcraft, S.A.; Rajotte, E.G.; Joshi, N.K.; Vaughn, M. Comparative toxicities and synergism of apple orchard pesticides to Apis mellifera (L.) and Osmia cornifrons (Radoszkowski). PLoS ONE 2013, 8, e72587. [Google Scholar] [CrossRef] [PubMed]
- Kremen, C.; Iles, A.; Bacon, C. Diversified farming systems: An agroecological, systems-based alternative to modern industrial agriculture. Ecol. Soc. 2012, 17, 44. [Google Scholar] [CrossRef]
- Isman, M.B. Plant essential oils for pest and disease management. Crop. Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Tripathi, A.K.; Upadhyay, S.; Bhuiyan, M.; Bhattacharya, P.R. A review on prospects of essential oils as biopesticide in insect-pest management. J. Pharmacog. Phytother. 2009, 1, 52–63. [Google Scholar]
- Biddinger, D.; Rajotte, E.G.; Joshi, N.K. Integrating pollinator health into tree fruit IPM—A case study of Pennsylvania apple production. In The Pollination of Cultivated Plants: A Compendium for Practitioners, 2nd ed.; FAO: Rome, Italy, 2018; Volume 1, pp. 69–83. [Google Scholar]
- Dicks, L.V.; Viana, B.; Bommarco, R.; Brosi, B.; Arizmendi, M.C.; Cummingham, S.A.; Galetto, L.; Hill, R.; Lopes, A.V.; Pires, C.; et al. Ten policies for pollinators. Science 2016, 354, 975–976. [Google Scholar] [CrossRef]
- Egan, P.A.; Dicks, L.V.; Hokkanen, H.M.T.; Stenberg, J.A. Delivering Integrated Pest and Pollinator Management (IPPM). Trends Plant. Sci. 2020, 25, 577–589. [Google Scholar] [CrossRef]
- Biddinger, D.J.; Rajotte, E.G. Integrated pest and pollinator management–adding a new dimension to an accepted paradigm. Curr. Opin. Insect Sci. 2015, 10, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Fortel, L.; Henry, M.; Guilbaud, L.; Mouret, H.; Vaissiere, B.E. Use of human-made nesting structures by wild bees in an urban environment. J. Insect. Conserv. 2016, 20, 239–253. [Google Scholar] [CrossRef]
- Cane, J.H. A native ground-nesting bee (Nomia melanderi) sustainably managed to pollinate alfalfa across an intensively agricultural landscape. Apidologie 2008, 39, 315–323. [Google Scholar] [CrossRef]
- Sardiñas, H.S.; Kremen, C. Evaluating nesting microhabitat for ground-nesting bees using emergence traps. Basic Appl. Ecol. 2014, 15, 161–168. [Google Scholar] [CrossRef]
- Fortel, L. Ecologie et Conservation des Abeilles Sauvages le Long d’un Gradient D’urbanisation. Ph.D. Thesis, Université d’Avignon et des Pays du Vaucluse, Avignon, France, 2014. [Google Scholar]
- Maclvor, J.S.; Packer, L. ‘Bee Hotels’ as Tools for Native Pollinator Conservation: A Premature Verdict? PLoS ONE 2015, 10, e0122126. [Google Scholar]
- Geslin, B.; Gachet, S.; Deschamps-Cottin, M.; Flacher, F.; Ignace, B.; Knoploch, C.; Meineri, E.; Robles, C.; Ropars, L.; Schurr, L.; et al. Bee hotels host a high abundance of exotic bees in an urban context. Acta Oecol. 2020, 105, 103556. [Google Scholar] [CrossRef]
- Wcislo, W.T. Parasitism rates in relation to nest site in bees and wasps (Hymenoptera: Apoidea). J. Insect. Conserv. 1996, 9, 643–656. [Google Scholar] [CrossRef]
- Cane, J.H. Soils of ground-nesting bees (Hymenoptera: Apoidea): Texture, moisture, cell depth and climate. J. Kansas Entomol. Soc. 1991, 64, 406–413. [Google Scholar]
- Potts, S.G.; Vulliamy, B.; Roberts, S.; O’Toole, C.; Dafni, A.; Ne’eman, G.; Willmer, P. Role of nesting resources in organising diverse bee communities in a Mediterranean landscape. Ecol. Entomol. 2005, 30, 78–85. [Google Scholar] [CrossRef]
- Kleijn, D.; Raemakers, I. A Retrospective analysis of pollen host plan use by stable and declining bumble bee species. Ecology 2008, 89, 1811–1823. [Google Scholar] [CrossRef]
- Davis, E.S.; Kelly, R.; Maggs, C.A.; Stout, J.C. Contrasting impacts of highly invasive plant species on flower-visiting insect communities. Biodivers. Conserv. 2018, 27, 2069–2085. [Google Scholar] [CrossRef]
- Vanderhoeven, S.; Braquart, E.; Gregoire, J.C.; Mahy, G. Les espèces exotiques envahissantes. In Dossier Scientifique Réalisé dans le Cadre de l’élaboration du Rapport Analytique 2006–2007 sur l’état de l’environnement Wallon; FUSAGx, CRNFBGx et ULB, Belgium: Brussels, Belgium, 2006; p. 42. [Google Scholar]
- Burke, M.J.; Grime, J.P. An experimental study of plant community invasibility. Ecology 1996, 77, 776–790. [Google Scholar] [CrossRef]
- Meerts, P.; Dassonville, N.; Vanderhoeven, S.; Chapuis-Lardy, L.; Koutika, L.S.; Jacquemart, A.L. Les Plantes Exotiques Envahissantes et Leurs Impacts. In Biodiversité: Etat, Enjeux et Perspectives; De Boeck Universités: Bruxelles, Belgium, 2004; p. 238. [Google Scholar]
- Alpert, P.; Bone, E.; Holzapfel, C. Invasiveness, invasibility and the role of environmental stress in the spread of non-native plants. Perspect. Plant. Ecol. 2000, 3, 52–66. [Google Scholar] [CrossRef]
- Lake, J.C.; Leishman, M.R. Invasion success of exotic plants in natural ecosystems: The role of disturbance, plant attributes and freedom from herbivores. Biol. Conserv. 2004, 117, 215–226. [Google Scholar] [CrossRef]
- Robinson, R.A.; Sutherland, W.J. Postwar changes in arable farming and biodiversity in Great Britain. J. Appl. Ecol. 2002, 39, 157–176. [Google Scholar] [CrossRef]
- Rasmont, P. La régression massive des espèces d’abeilles sauvages et de bourdons d’Europe: Un effet de la perturbation mondiale du cycle de l’azote. Actes Colloq. Insectes Biodiversité 2006, 6, 43–60. [Google Scholar]
- Lambdon, P.W.; Pyšek, P.; Basnou, C.; Hejda, M.; Arianoutsou, M.; Essl, F.; Essl, F.; Vojtěch, J.; Pergl, J.; Winter, M.; et al. Alien flora of Europe: Species diversity, temporal trends, geographical patterns and research needs. Preslia 2008, 80, 101–149. [Google Scholar]
- Hobbs, R.J.; Higgs, E.S.; Hall, C. Novel Ecosystems: Intervening in the New Ecological World Order; John Wiley & Sons: New York, NY, USA, 2013. [Google Scholar]
- Drossart, M.; Michez, D.; Vanderplanck, M. Invasive plants as potential food resource for native pollinators: A case study with two invasive species and a generalist bumble bee. Sci. Rep. UK 2017, 7, 16242. [Google Scholar] [CrossRef]
- Jachuła, J.; Denisow, B.; Strzałkowska-Abramek, M. Does an invader have a bright side? Floral reward in two Solidago species. J. Apicult. Res. 2020. [Google Scholar] [CrossRef]
- Hanley, M.E.; Awbi, A.J.; Franco, M. Going native? Flower use by bumblebees in English urban gardens. Ann. Bot. Lond. 2014, 113, 799–806. [Google Scholar] [CrossRef]
- Sivakoff, F.; Prajzner, S.P.; Gardiner, M.M. Unique bee community assembly within vacant lots and urban farms results from variation in surrounding landscape urbanization intensity. Sustainability 2018, 10, 1926. [Google Scholar] [CrossRef]
- Müller, A.; Diener, S.; Schnyder, S.; Stutz, K.; Sedivy, C.; Dorn, S. Quantitative pollen requirements of solitary bees: Implications for bee conservation and the evolution of bee-flower relationships. Biol. Conserv. 2006, 130, 604–615. [Google Scholar] [CrossRef]
- Chittka, L.; Schürkens, S. Successful invasion of a floral market. Nature 2001, 411, 653. [Google Scholar] [CrossRef] [PubMed]
- Van Kleunen, M.; Weber, E.; Fischer, M. A meta-analysis of trait differences between invasive and non-invasive plant species. Ecol. Lett. 2010, 13, 235–245. [Google Scholar] [CrossRef]
- Moroń, D.; Lenda, M.; Skórka, P.; Szentgyörgyi, H.; Settele, J.; Woyciechowski, M. Wild pollinator communities are negatively affected by invasion of alien goldenrods in grassland landscapes. Biol. Conserv. 2009, 142, 1322–1332. [Google Scholar] [CrossRef]
- Wagner, D.L.; Van Driesche, R.G. Threats posed to rare or endangered insects by invasions of non-native species. Annu. Rev. Entomol. 2010, 55, 547–568. [Google Scholar] [CrossRef]
- Lowenstein, D.M.; Minor, E.S. Diversity in flowering plants and their characteristics: Integrating humans as a driver of urban floral resources. Urban. Ecosyst. 2016, 19, 1735–1748. [Google Scholar] [CrossRef]
- Majewska, A.A.; Altizer, S. Planting gardens to support insect pollinators. Conserv. Biol. 2018, 34, 15–25. [Google Scholar] [CrossRef]
- Boltovskoy, D.; Sylvester, F.; Paolucci, E.M. Invasive species denialism: Sorting out facts, beliefs, and definitions. Ecol. Evol. 2018, 8, 11190–11198. [Google Scholar] [CrossRef]
- Traveset, A.; Richardson, D.M. Biological invasions as disruptors of plant reproductive mutualisms. Trends Ecol. Evol. 2006, 21, 208–216. [Google Scholar] [CrossRef]
- Morales, C.L. Introduccion de abejorros (Bombus) no nativos: Causas, consecuencias ecologicas y perspectivas. Ecol. Austral 2007, 17, 51–65. [Google Scholar]
- Arca, M.; Papachristoforou, A.; Mougel, F.; Rortais, A.; Monceau, K.; Bonnard, O.; Tardy, P.; Thiéry, D.; Silvain, J.F.; Arnold, G. Defensive behaviour of Apis mellifera against Vespa velutina in France: Testing whether European honeybees can develop an effective collective defence against a new predator. Behav. Process. 2014, 106, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Keeling, M.J.; Franklin, D.N.; Datta, S.; Brown, M.A.; Budge, G.E. Predicting the spread of the Asian hornet (Vespa velutina) following its incursion into Great Britain. Sci. Rep. UK 2017, 7, 6240. [Google Scholar] [CrossRef] [PubMed]
- Laurino, D.; Lioy, S.; Carisio, L.; Manino, A.; Porporato, M. Vespa velutina: An Alien Driver of Honey Bee Colony Losses. Diversity 2020, 12, 5. [Google Scholar] [CrossRef]
- Sutherland, W.J.; Barnard, P.; Broad, S.; Clout, M.; Connor, B.; Côté, I.M.; Dicks, L.V.; Doran, H.; Entwistle, A.C.; Esmail, N.; et al. Horizon scan of emerging issues for global conservation and biological diversity. Trends Ecol. Evol. 2016, 32, 31–42. [Google Scholar] [CrossRef]
- Kanbe, Y.; Okada, I.; Yoneda, M.; Goka, K.; Tsuchida, K. Interspecific mating of the introduced bumblebee Bombus terrestris and the native Japanese bumblebee Bombus hypocrita sapporoensis results in inviable hybrids. Naturwissenschaften 2008, 95, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Aizen, M.A.; Smith-Ramirez, C.; Morales, C.L.; Vieli, L.; Saez, A.; Barahona-Segovia, R.M.; Arbetman, M.P.; Montalva, J.; Garibaldi, L.A.; Inouye, D.W.; et al. Coordinated species importation policies are needed to reduce serious invasions globally: The case of alien bumblebees in South America. J. Appl. Ecol. 2019, 56, 100–106. [Google Scholar] [CrossRef]
- Le Féon, V.; Aubert, M.; Genoud, D.; Andrieu-Ponel, V.; Westrich, P.; Geslin, B. Range expansion of the Asian native giant resin bee Megachile sculpturalis (Hymenoptera, Apoidea, Megachilidae) in France. Ecol. Evol. 2018, 8, 1534–1542. [Google Scholar] [CrossRef]
- Blackburn, T.M.; Essl, F.; Evans, T.; Hulme, P.E.; Jeschke, J.M.; Kühn, I.; Kumschick, S.; Marková, Z.; Mrugała, A.; Nentwig, W.; et al. A unified classification of alien species based on the magnitude of their environmental impacts. PLoS Biol. 2014, 12, e1001850. [Google Scholar] [CrossRef]
- Saunders, M.E.; Janes, J.; O’Hanlon, J. Semantics of the insect decline narrative; recommendations for communicating insect conservation to peer and public audiences. Insect Conserv. Diver. 2020, 13, 211–213. [Google Scholar] [CrossRef]
- Saunders, M.E.; Janes, J.; O’Hanlon, J. Moving on from the insect apocalypse narrative: Engaging with evidence-based insect conservation. BioScience 2019. [Google Scholar] [CrossRef]
- Gardiner, T.; Didham, R.K. Glowing, glowing, gone? Monitoring long term trends in glow-worm numbers in south-east England. Insect Conserv. Diver. 2020, 13, 162–174. [Google Scholar] [CrossRef]
- Ubach, A.; Paramo, F.; Gutierrez, C.; Stefanescu, C. Vegetation encroachment drives changes in the composition of butterfly assemblages and species loss in Mediterranean ecosystems. Insect Conserv. Diver. 2020, 13, 151–161. [Google Scholar] [CrossRef]
- Duchenne, F.; Thébault, E.; Michez, D.; Elias, M.; Drake, M.; Persson, M.; Rousseau-Piot, J.S.; Pollet, M.; Vanormelingen, P.; Fontaine, C. Phenological shifts alter the seasonal structure of pollinator assemblages in Europe. Nat. Ecol. Evol. 2020, 4, 115–121. [Google Scholar] [CrossRef]
- Dickinson, J.L.; Zuckerber, B.; Bonter, D.N. Citizen science as an ecological research tool: Challenges and benefits. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 149–172. [Google Scholar] [CrossRef]
- Ward, D.F. Understanding sampling and taxonomic biases recorded by citizen scientists. J. Insect. Conserv. 2014, 18, 753–756. [Google Scholar] [CrossRef]
- Silva, A.; Minor, E.S. Adolescents’ experience and knowledge of, and attitudes toward, bees: Implications and recommendations for conservation. Anthrozoos 2017, 30. [Google Scholar] [CrossRef]
- Schonfelder, M.L.; Bogner, F.X. Individual perception of bees: Between perceived danger and willingness to protect. PLoS ONE 2017, 12, e0180168. [Google Scholar] [CrossRef]
- Soulé, M.E. What is conservation biology? BioScience 1985, 35, 727–734. [Google Scholar]
- Schmitt, S.; Maréchaux, I.; Chave, J.; Fischer, F.J.; Piponiot, C.; Traissac, S.; Hérault, B. Functional diversity improves tropical forest resilience: Insights from a long-term virtual experiment. J. Ecol. 2020, 108, 831–843. [Google Scholar] [CrossRef]
- Wood, T.J.; Michez, D.; Paxton, R.J.; Drossart, M.; Neumann, P.; Gérard, M.; Vanderplanck, M.; Barraud, A.; Martinet, B.; Leclercq, N.; et al. Managed honey bees as a radar for wild bee decline? Apidologie 2020. [Google Scholar] [CrossRef]
- Urbanowicz, C.; Muniz, P.A.; McArt, S.H. Honey bees and wild pollinators differ in their preference for and use of introduced floral resources. Ecol. Evol. 2020, 10, 6741–6751. [Google Scholar] [CrossRef] [PubMed]
- Myers, N. Biodiversity and the Precautionary Principle. Ambio 1993, 22, 74–79. [Google Scholar]
- Miller, J.R. Biodiversity conservation and the extinction of experience. Trends Ecol. Evol. 2005, 20, 430–434. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drossart, M.; Gérard, M. Beyond the Decline of Wild Bees: Optimizing Conservation Measures and Bringing Together the Actors. Insects 2020, 11, 649. https://doi.org/10.3390/insects11090649
Drossart M, Gérard M. Beyond the Decline of Wild Bees: Optimizing Conservation Measures and Bringing Together the Actors. Insects. 2020; 11(9):649. https://doi.org/10.3390/insects11090649
Chicago/Turabian StyleDrossart, Maxime, and Maxence Gérard. 2020. "Beyond the Decline of Wild Bees: Optimizing Conservation Measures and Bringing Together the Actors" Insects 11, no. 9: 649. https://doi.org/10.3390/insects11090649
APA StyleDrossart, M., & Gérard, M. (2020). Beyond the Decline of Wild Bees: Optimizing Conservation Measures and Bringing Together the Actors. Insects, 11(9), 649. https://doi.org/10.3390/insects11090649




