The Role of Flies as Pollinators of Horticultural Crops: An Australian Case Study with Worldwide Relevance
Abstract
1. Introduction
2. Search Strategy and Selection Criteria
3. Fly Taxa Associated with Horticultural Crops
4. Target Horticultural Crops
4.1. Avocado (Persea americana)
4.2. Mango (Mangifera indica)
4.3. Lychee (Litchi chinensis)
4.4. Berries (Blueberries, Strawberries and Raspberries)
4.5. Vegetable Seed Crops
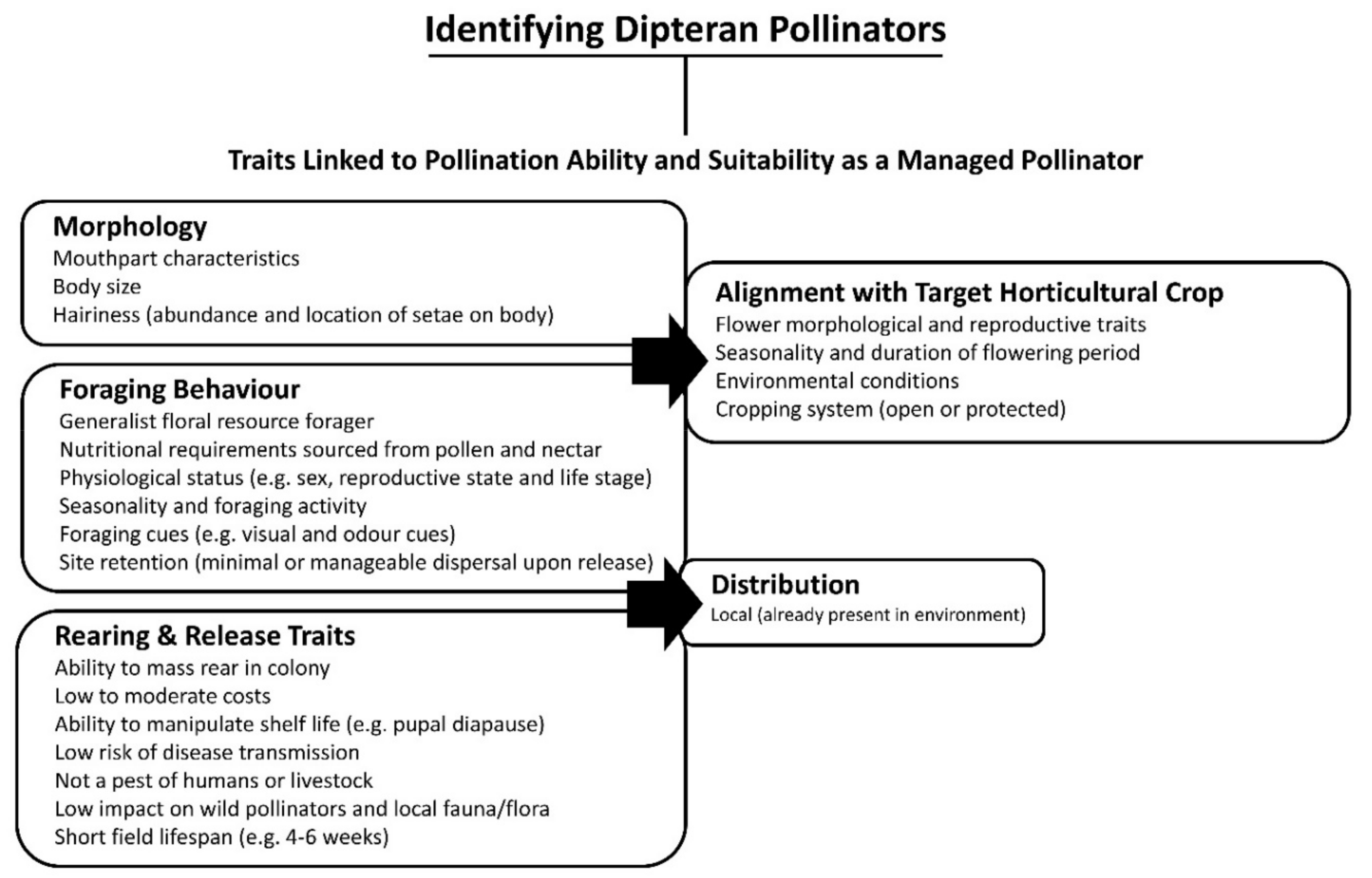
5. Identified Dipteran Pollinators
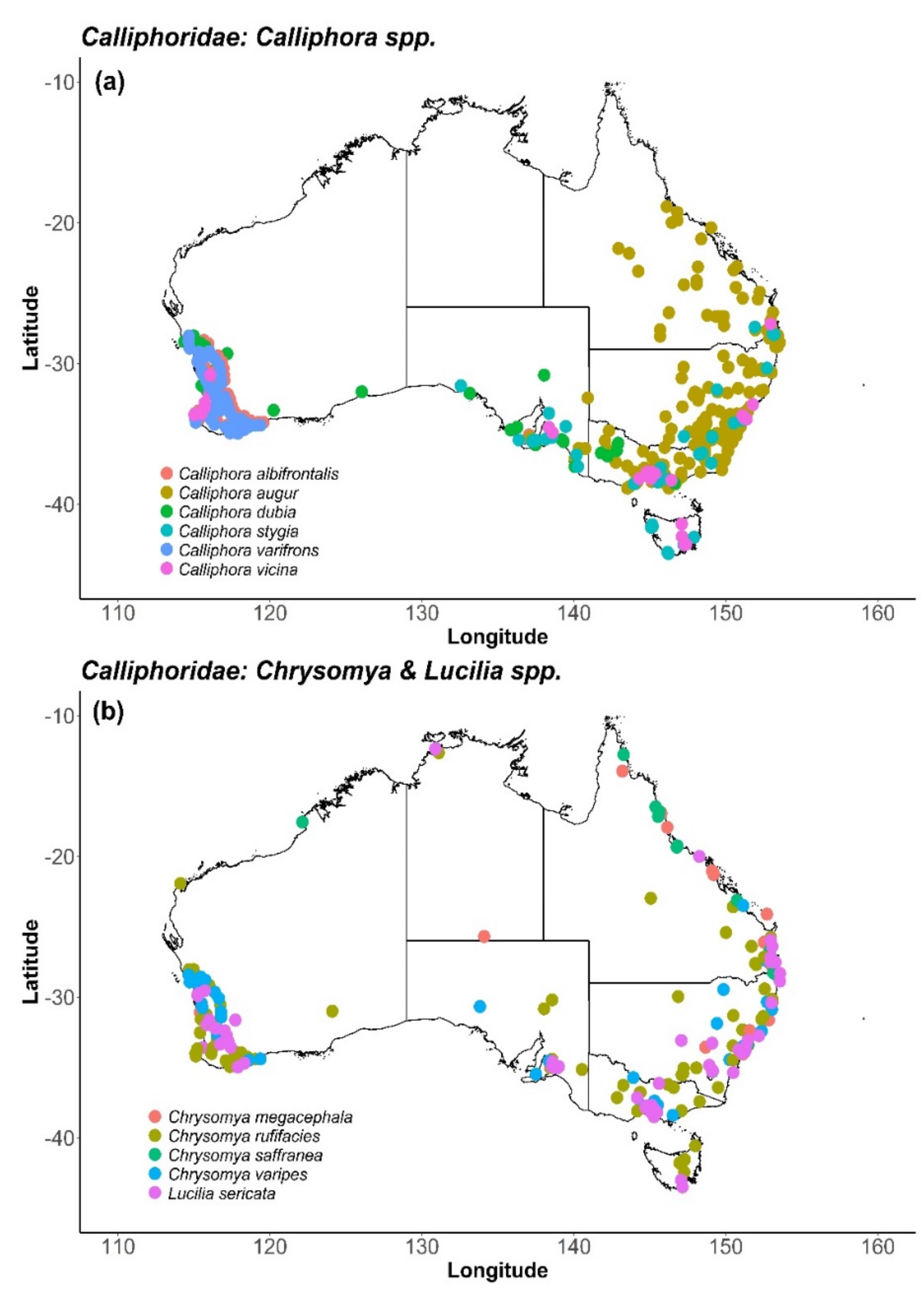
6. Calliphoridae (Blow Flies)
6.1. Morphology
6.2. Distribution and Foraging Behaviour
6.3. Rearing and Release
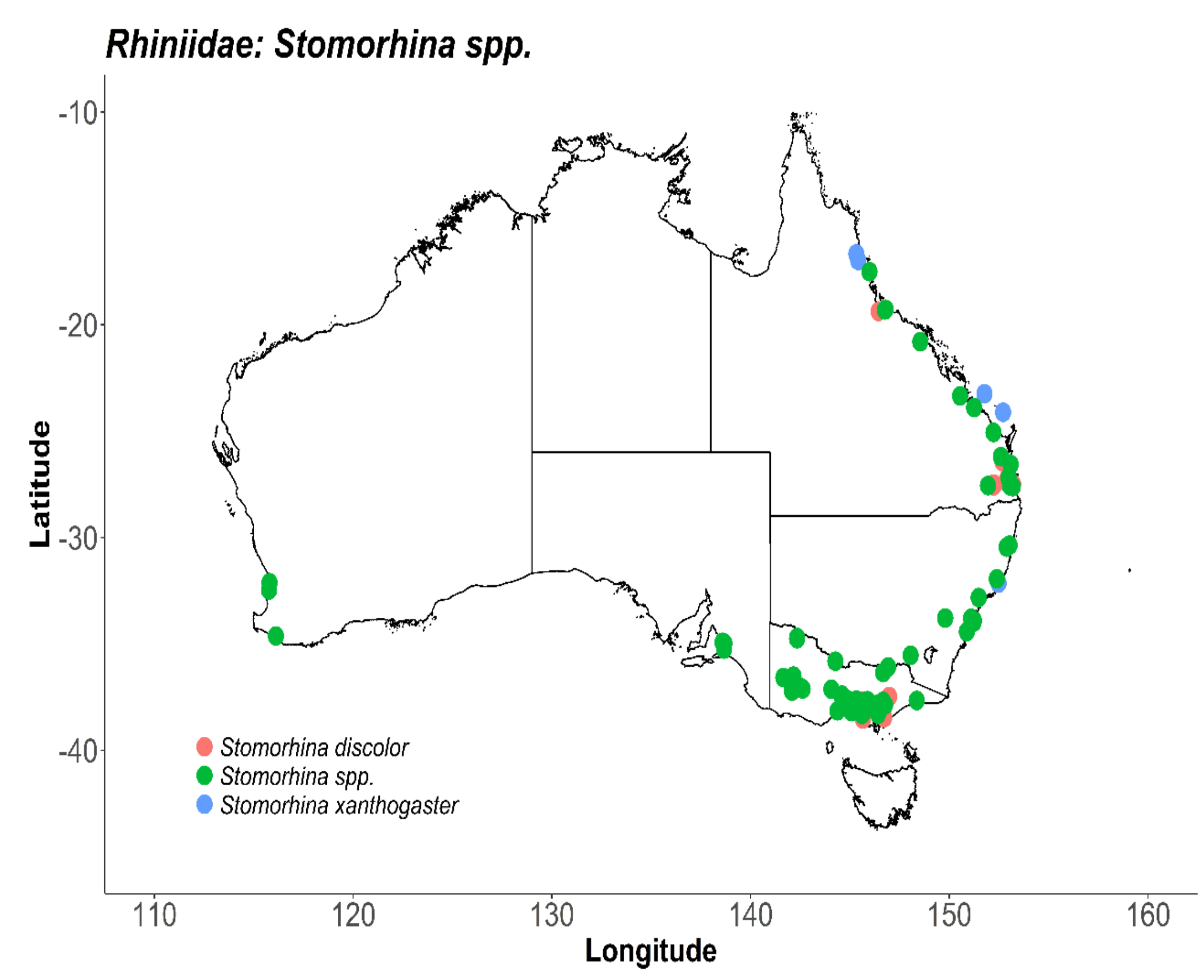
7. Rhiniidae (Nose Flies)
7.1. Morphology
7.2. Distribution and Foraging Behaviour
7.3. Rearing and Release
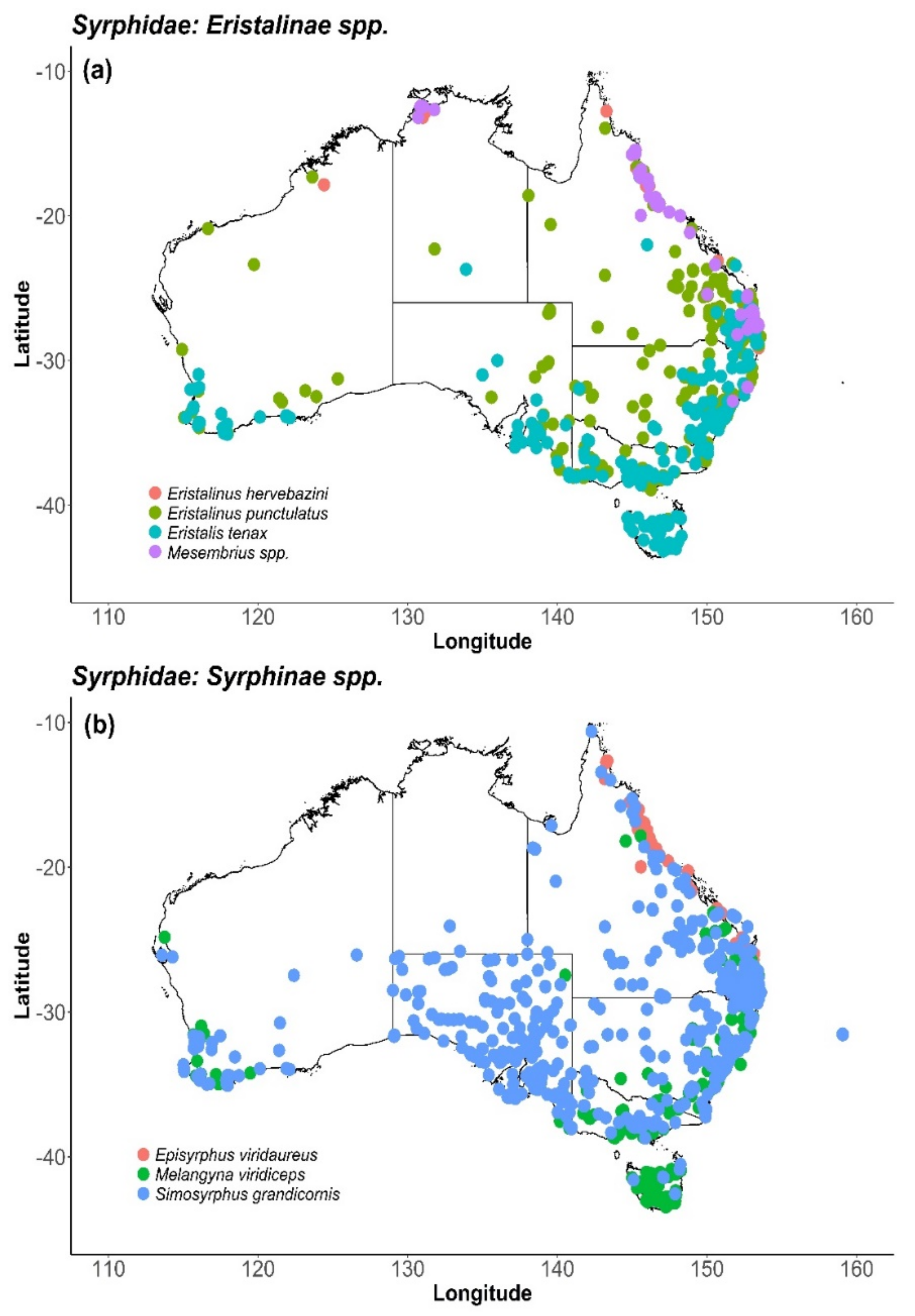
8. Syrphidae (Hover Flies)
8.1. Morphology
8.2. Distribution and Foraging Behaviour
8.3. Rearing and Release
9. The Future of Managed Flies for Crop Pollination
10. Commercial Pollination Services
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Aizen, M.A.; Aguiar, S.; Biesmeijer, J.C.; Garibaldi, L.A.; Inouye, D.W.; Jung, C.; Martins, D.J.; Medel, R.; Morales, C.L.; Ngo, H.; et al. Global agricultural productivity is threatened by increasing pollinator dependence without a parallel increase in crop diversification. Glob. Chang. Biol. 2019, 25, 3516–3527. [Google Scholar] [CrossRef]
- Khan, M.; Yogi, M.K. Insect Crop Pollinators. In Industrial Entomology; Omkar, Ed.; Springer: Singapore, 2017; pp. 397–412. [Google Scholar]
- Hafi, A.; Millist, N.; Morley, K.; Caley, P.; Buetre, B. A Benefit-Cost Framework for Responding to an Incursion of Varroa Destructor; ABARES Research Report 12.5; Australian Government, Department of Agricultre, Fisheries and Forestry: Canberra, ACT, Australia, 2012. [Google Scholar]
- Free, J.B. Insect Pollination of Crops; Academic press: London, UK, 1993. [Google Scholar]
- Larson, B.M.H.; Kevan, P.G.; Inouye, D.W. Flies and flowers: Taxonomic diversity of anthophiles and pollinators. Can. Entomol. 2001, 133, 439–465. [Google Scholar] [CrossRef]
- Ollerton, J.; Winfree, R.; Tarrant, S. How many flowering plants are pollinated by animals? Oikos 2011, 120, 321–326. [Google Scholar] [CrossRef]
- Rader, R.; Cunningham, S.A.; Howlett, B.G.; Inouye, D.W. Non-bee insects as visitors and pollinators of crops: Biology, ecology, and management. Annu. Rev. Entomol. 2020, 65, 391–407. [Google Scholar] [CrossRef]
- Kendall, D.A. The viability and compatibility of pollen on insects visiting apple blossom. J. Appl. Ecol. 1973, 10, 847–853. [Google Scholar] [CrossRef]
- Williams, R.R.; Wilson, D. Towards Regulated Cropping: A Report of Recent Fruit-Set Experiments in British Orchards; Grower Books: London, UK, 1970. [Google Scholar]
- Albano, S.; Salvado, E.; Duarte, S.; Mexia, A.; Borges, P.A.V. Pollination effectiveness of different strawberry floral visitors in Ribatejo, Portugal: Selection of potential pollinators. Part 2. Adv. Hortic. Sci. 2009, 23, 246–253. [Google Scholar]
- Jauker, F.; Wolters, V. Hover flies are efficient pollinators of oilseed rape. Oecologia 2008, 156, 819. [Google Scholar] [CrossRef] [PubMed]
- Orford, K.A.; Vaughan, I.P.; Memmott, J. The forgotten flies: The importance of non-syrphid Diptera as pollinators. Proc. R. Soc. B Biol. Sci. 2015, 282, 20142934. [Google Scholar] [CrossRef]
- Ssymank, A.; Kearns, C.A.; Pape, T.; Thompson, F.C. Pollinating flies (Diptera): A major contribution to plant diversity and agricultural production. Biodiversity 2008, 9, 86–89. [Google Scholar] [CrossRef]
- Rader, R.; Howlett, B.G.; Cunningham, S.A.; Westcott, D.A.; Newstrom-Lloyd, L.E.; Walker, M.K.; Teulon, D.A.J.; Edwards, W. Alternative pollinator taxa are equally efficient but not as effective as the honeybee in a mass flowering crop. J. Appl. Ecol. 2009, 46, 1080–1087. [Google Scholar] [CrossRef]
- Rader, R.; Howlett, B.G.; Cunningham, S.A.; Westcott, D.A.; Edwards, W. Spatial and temporal variation in pollinator effectiveness: Do unmanaged insects provide consistent pollination services to mass flowering crops? J. Appl. Ecol. 2012, 49, 126–134. [Google Scholar] [CrossRef]
- Stavert, J.R.; Pattemore, D.E.; Bartomeus, I.; Gaskett, A.C.; Beggs, J.R. Exotic flies maintain pollination services as native pollinators decline with agricultural expansion. J. Appl. Ecol. 2018, 55, 1737–1746. [Google Scholar] [CrossRef]
- Rader, R.; Bartomeus, I.; Garibaldi, L.A.; Garratt, M.P.D.; Howlett, B.G.; Winfree, R.; Cunningham, S.A.; Mayfield, M.M.; Arthur, A.D.; Andersson, G.K.S.; et al. Non-bee insects are important contributors to global crop pollination. Proc. Natl. Acad. Sci. USA 2016, 113, 146–151. [Google Scholar] [CrossRef]
- Norris, K. The bionomics of blow flies. Annu. Rev. Entomol. 1965, 10, 47–68. [Google Scholar] [CrossRef]
- Heine, E. Observations on the pollination of New Zealand flowering plants. Trans. Proc. R. Soc. N. Z. 1937, 67, 133–148. [Google Scholar]
- Stavert, J.R.; Liñán-Cembrano, G.; Beggs, J.R.; Howlett, B.G.; Pattemore, D.E.; Bartomeus, I. Hairiness: The missing link between pollinators and pollination. PeerJ 2016, 4, e2779. [Google Scholar] [CrossRef]
- Ireland, S.; Turner, B. The effects of larval crowding and food type on the size and development of the blowfly, Calliphora vomitoria. Forensic Sci. Int. 2006, 159, 175–181. [Google Scholar] [CrossRef]
- Inouye, D.W.; Larson, B.M.; Ssymank, A.; Kevan, P.G. Flies and flowers III: Ecology of foraging and pollination. J. Pollinat. Ecol. 2015, 16, 115–133. [Google Scholar] [CrossRef]
- Heath, A.C.G. Beneficial aspects of blowflies (Diptera: Calliphoridae). N. Z. Entomol. 1982, 7, 343–348. [Google Scholar] [CrossRef]
- Woodcock, T. Pollination in the Agricultural Landscape: Best Management Practices for Crop Pollination; NSERC-CANPOLIN; University of Guelph: Guelph, ON, Canada, 2012; Volume 113. [Google Scholar]
- Skevington, J.H.; Dang, P.T. Exploring the diversity of flies (Diptera). Biodiversity 2002, 3, 3–27. [Google Scholar] [CrossRef]
- Raguso, R.A. Don’t forget the flies: Dipteran diversity and its consequences for floral ecology and evolution. Appl. Entomol. Zool. 2020, 55, 1–7. [Google Scholar] [CrossRef]
- Howlett, B.G.; Walker, M.K.; Newstrom-Lloyd, L.E.; Donovan, B.J.; Teulon, D.A.J. Window traps and direct observations record similar arthropod flower visitor assemblages in two mass flowering crops. J. Appl. Entomol. 2009, 133, 553–564. [Google Scholar] [CrossRef]
- Rader, R.; Edwards, W.; Westcott, D.A.; Cunningham, S.A.; Howlett, B.G. Diurnal effectiveness of pollination by bees and flies in agricultural Brassica rapa: Implications for ecosystem resilience. Basic Appl. Ecol. 2013, 14, 20–27. [Google Scholar] [CrossRef]
- Gaffney, A.; Allen, G.R.; Brown, P.H. Insect visitation to flowering hybrid carrot seed crops. N. Z. J. Crop Hortic. Sci. 2011, 39, 79–93. [Google Scholar] [CrossRef]
- Faulkner, G. Blowflies as pollinators of brassica crops. Commercial Grower 1962, 3457, 807–809. [Google Scholar]
- Faulkner, G.J.; Hinton, W.L. F1 hybrid brussels sprout seed: An assessment of production methods and their economic viability. Hortic. Res. 1980, 20, 49–59. [Google Scholar]
- Howlett, B. Optimising Pollination of Macadamia and Avocado in Australia; Horticulture Innovation Australia, Final Report, Project: MT13060; Horticulture Innovation Australia Limited: Sydney, NSW, Australia, 2017. [Google Scholar]
- Mann, N.A. Intensive Berry Production Using Greenhouses, Substrates and Hyrdoponics. Is This This Way Forward; Nuffield Australia: North Sydney, NSW, Australia, 2014. [Google Scholar]
- Saeed, S.; Naqqash, M.N.; Jaleel, W.; Saeed, Q.; Ghouri, F. The effect of blow flies (Diptera: Calliphoridae) on the size and weight of mangos (Mangifera indica L.). PeerJ 2016, 4, e2076. [Google Scholar] [CrossRef]
- Saeed, S.; Sajjad, A.; Kwon, O.; Kwon, Y.J. Fidelity of Hymenoptera and Diptera pollinators in onion (Allium cepa L.) pollination. Entomol. Res. 2008, 38, 276–280. [Google Scholar] [CrossRef]
- Vithanage, H.I.M.V. Insect pollination of avocado and macadamia. ActaHortic 1986, 175, 97–102. [Google Scholar] [CrossRef]
- Willcox, B.K.; Howlett, B.G.; Robson, A.J.; Cutting, B.; Evans, L.; Jesson, L.; Kirkland, L.; Jean-Meyzonnier, M.; Potdevin, V.; Saunders, M.E.; et al. Evaluating the taxa that provide shared pollination services across multiple crops and regions. Sci. Rep. 2019, 9, 13538. [Google Scholar] [CrossRef]
- Spurr, C. Identification and Management of Factors Limiting Hybrid Carrot Seed Production in Australia. Ph.D. Thesis, University of Tasmania, Tasmania, Australia, 2003. [Google Scholar]
- Clement, S.L.; Hellier, B.C.; Elberson, L.R.; Staska, R.T.; Evans, M.A. Flies (Diptera: Muscidae: Calliphoridae) are efficient pollinators of Allium ampeloprasum L. (Alliaceae) in field cages. J. Econ. Entomol. 2007, 100, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Howlett, B.G. Hybrid carrot seed crop pollination by the fly Calliphora vicina (Diptera: Calliphoridae). J. Appl. Entomol. 2012, 136, 421–430. [Google Scholar] [CrossRef]
- Howlett, B.G.; Evans, L.J.; Pattemore, D.E.; Nelson, W.R. Stigmatic pollen delivery by flies and bees: Methods comparing multiple species within a pollinator community. Basic Appl. Ecol. 2017, 19, 19–25. [Google Scholar] [CrossRef]
- Cook, D.F.; Deyl, R.A.; Mickan, B.S.; Howse, E.T. Yield of southern highbush blueberry (Vaccinium corymbosum) using the fly Calliphora albifrontalis (Diptera: Calliphoridae) as a pollinator. Aust. J. Entomol. 2020. [Google Scholar] [CrossRef]
- Anderson, D.; Sedgley, M.; Short, J.; Allwood, A. Insect pollination of mango in northern Australia. Aust. J. Agric. Res. 1982, 33, 541–548. [Google Scholar] [CrossRef]
- Fajardo, A.; Medina, J.R.; Opina, O.S.; Cervancia, C.R. Insect pollinators and floral visitors of mango (Mangifera indica L. cv. Carabao). Philipp. Agric. Sci. 2008, 91, 372–382. [Google Scholar]
- Pérez-Balam, J.; Quezada-Euán, J.; Alfaro-Bates, R.; Medina, S.; McKendrick, L.; Soro, A.; Paxton, R.J. The contribution of honey bees, flies and wasps to avocado (Persea americana) pollination in southern Mexico. J. Pollinat. Ecol. 2012, 8, 42–47. [Google Scholar] [CrossRef]
- Rajan, V.V.; Reddy, P.V.R. A dead heat in pollination race: A comparative evaluation of the efficiency of a fly (Chrysomya megacephala) and a bee (Apis florea) in mango pollination. J. Entomol. Zool. Stud. 2019, 7, 1087–1091. [Google Scholar]
- Ish-Am, G.; Barrientos-Priego, F.; Castañeda-Vildozola, A.; Gazit, S. Avocado (Persea americana Mill.) pollinators in its region of origin. Rev. Chapingo Ser. Hortic. 1999, 5, 137–143. [Google Scholar]
- Das, R.; Jha, S.; Halder, A. Insect pollinators of litchi with special reference to foraging behaviour of honey bees. J. Pharmacogn. Phytochem. 2019, 8, 396–401. [Google Scholar]
- Herrmann, J.D.; Beye, H.; de la Broise, C.; Hartlep, H.; Diekötter, T. Positive effects of the pollinators Osmia cornuta (Megachilidae) and Lucilia sericata (Calliphoridae) on strawberry quality. Arthropod-Plant Inter. 2019, 13, 71–77. [Google Scholar] [CrossRef]
- Dag, A.; Gazit, S. Mango pollinators in Israel. J. Appl. Hortic. 2000, 2, 39–43. [Google Scholar] [CrossRef]
- Gaffney, A.; Bohman, B.; Quarrell, S.R.; Brown, P.H.; Allen, G.R. Frequent insect visitors are not always pollen carriers in hybrid carrot pollination. Insects 2018, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Rader, R.; Edwards, W.; Westcott, D.A.; Cunningham, S.A.; Howlett, B.G. Pollen transport differs among bees and flies in a human-modified landscape. Divers. Distrib. 2011, 17, 519–529. [Google Scholar] [CrossRef]
- Albano, S.; Salvado, E.; Borges, P.A.V.; Mexia, A. Floral visitors, their frequency, activity rate and Index of Visitation Rate in the strawberry fields of Ribatejo, Portugal: Selection of potential pollinators. Part 1. Adv. Hortic. Sci. 2009, 23, 238–245. [Google Scholar]
- Bohart, G.E.; Nye, W.P.; Hawthorn, L.R. Onion pollination as affected by different levels of pollinator activity. Utah Agricultural Experiment Station Bulletin 1970, 482, 460. [Google Scholar]
- Gervais, A.; Chagnon, M.; Fournier, V. Diversity and pollen loads of flower flies (Diptera: Syrphidae) in cranberry crops. Ann. Entomol. Soc. Am. 2018, 111, 326–334. [Google Scholar] [CrossRef]
- Jarlan, A.; De Oliveiha, D.; Gingras, J. Effects of Eristalis tenax (Diptera: Syrphidae) pollination on characteristics of greenhouse sweet pepper fruits. J. Econ. Entomol. 1997, 90, 1650–1654. [Google Scholar] [CrossRef]
- Jarlan, A.; De Oliveira, D.; Gingras, J. Pollination by Eristalis tenax (Diptera: Syrphidae) and seed set of greenhouse sweet pepper. J. Econ. Entomol. 1997, 90, 1646–1649. [Google Scholar] [CrossRef]
- Howlett, B.; Gee, M. The potential management of the drone fly (Eristalis tenax) as a crop pollinator in New Zealand. N. Z. Plant Prot. 2019, 72, 221–230. [Google Scholar] [CrossRef]
- Bezuidenhout, M.; du Toit, E.; Robbertse, P. Finding the Best Polliniser for ‘Hass’ Avocado and the Effects of Bees as Pollinators. South African Growers Assocaition Year Book 2017, 40, 117–122. [Google Scholar]
- Papademetriou, M.K. A study of the viability of avocado pollen under natural conditions. Calif. Avocado Soc. Yearb. 1975, 58, 54–56. [Google Scholar]
- Dixon, J. Avocado Pollination, Best Practice Guidelines; Avocado Industry Council: Sydney, NSW, Australia, 2006; p. 12. [Google Scholar]
- Ish-Am, G.; Lahav, E. Evidence for a major role of honeybees (Apis mellifera) rather than wind during avocado (Persea americana Mill.) pollination. J. Hortic. Sci. Biotechnol. 2011, 86, 589–594. [Google Scholar] [CrossRef]
- Wysoki, M.; van den Berg, M.A.; Ish-Am, G.; Gazit, S.; Peña, J.E.; Waite, G.K. Pests and pollinators of avocado. In Tropical Fruit Pests and Pollinators, Biology, Economic Importance, Natural Enemies and Control; CABI Publishing: Wallingford, Oxfordshire, UK, 2002; pp. 223–293. [Google Scholar]
- Ish-Am, G.; Eisikowitch, D. Low attractiveness of avocado (Persea americana Mill.) flowers to honeybees (Apis mellifera L.) limits fruit set in Israel. J. Hortic. Sci. Biotechnol. 1998, 73, 195–204. [Google Scholar] [CrossRef]
- Kavazos, C.R.J.; Wallman, J.F. Community composition of carrion-breeding blowflies (Diptera: Calliphoridae) along an urban gradient in south-eastern Australia. Landsc. Urban Plan. 2012, 106, 183–190. [Google Scholar] [CrossRef]
- Kearns, C.A.; Inouye, D.W. Fly pollination of Linum lewish (Linaceae). Am. J. Bot. 1994, 81, 1091–1095. [Google Scholar] [CrossRef]
- Evans, L.; Goodwin, R.; Howlett, B. The role of insect pollinators in avocado (Persea americana) pollination in New Zealand and Australia. In Proceedings of the VII World Avocado Congress, Cairns, Australia, 5–9 September 2011; pp. 280–286. [Google Scholar]
- Carvalheiro, L.G.; Seymour, C.L.; Veldtman, R.; Nicolson, S.W. Pollination services decline with distance from natural habitat even in biodiversity-rich areas. J. Appl. Ecol. 2010, 47, 810–820. [Google Scholar] [CrossRef]
- Sharma, S.; Abbas, S.R.; Shukia, R.P.; Sharma, S. An easy and quick method of breeding flies for pollination of mango blossoms. Insect Environ. 1998, 4, 76–77. [Google Scholar]
- Sung, I.H.; Lin, M.Y.; Chang, C.H.; Cheng, A.S.; Chen, W.S.; Ho, K.K. Pollinators and their behaviors on mango flowers in southern Taiwan. Formos. Entomol 2006, 26, 161–170. [Google Scholar]
- Huda, A.N.; Salmah, M.R.C.; Hassan, A.A.; Hamdan, A.; Razak, M.N.A. Pollination services of mango flower pollinators. J. Insect Sci. 2015, 15, 113. [Google Scholar] [CrossRef]
- Finch, J.T.D. Fishing for flies. Mango Matters 2020, 38, 20–21. [Google Scholar]
- Stern, R.A.; Gazit, S. Pollen viability in lychee. J. Am. Soc. Hortic. Sci. 1998, 123, 41–46. [Google Scholar] [CrossRef]
- Dag, A. Interaction between pollinators and crop plants: The Israeli experience. Isr. J. Plant Sci. 2009, 57, 231–242. [Google Scholar] [CrossRef]
- Stern, R.A.; Gazit, S. The reproductive biology of the lychee. Hortic. Rev. 2002, 28, 393–453. [Google Scholar]
- Danka, R.G.; Sampson, B.J.; Villa, J.D. Association between density of foraging bees and fruit set in commercial fields of rabbiteye blueberries (Ericales: Ericaceae) in Louisiana and Mississippi. J. Econ. Entomol. 2019, 112, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
- Bushmann, S.L.; Drummond, F.A. Abundance and Diversity of Wild Bees (Hymenoptera: Apoidea) Found in Lowbush Blueberry Growing Regions of Downeast Maine. Environ. Entomol. 2015, 44, 975–989. [Google Scholar] [CrossRef] [PubMed]
- Moisan-Deserres, J.; Girard, M.; Chagnon, M.; Fournier, V. Pollen loads and specificity of native pollinators of lowbush blueberry. J. Econ. Entomol. 2014, 107, 1156–1162. [Google Scholar] [CrossRef]
- Chagnon, M.; Gingras, J.; DeOliveira, D. Complementary aspects of strawberry pollination by honey and indigenous bees (Hymenoptera). J. Econ. Entomol. 1993, 86, 416–420. [Google Scholar] [CrossRef]
- Campbell, J.W.; O’Brien, J.; Irvin, J.H.; Kimmel, C.B.; Daniels, J.C.; Ellis, J.D. Managed bumble bees (Bombus impatiens) (Hymenoptera: Apidae) caged with blueberry bushes at high density did not increase fruit set or fruit weight compared to open pollination. Environ. Entomol. 2017, 46, 237–242. [Google Scholar] [CrossRef]
- Malagodi-Braga, K.S.; Kleinert, A.d.M.P. Could Tetragonisca angustula Latreille (Apinae, Meliponini) be effective as strawberry pollinator in greenhouses? Aust. J. Agric. Res. 2004, 55, 771–773. [Google Scholar] [CrossRef]
- Roselino, A.; Santos, S.; Hrncir, M.; Bego, L. Differences between the quality of strawberries (Fragaria × ananassa) pollinated by the stingless bees Scaptotrigona aff. depilis and Nannotrigona testaceicornis. Genet. Mol. Res. 2009, 8, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Hodgkiss, D.; Brown, M.; Fountain, M. Syrphine hoverflies are effective pollinators of commercial strawberry. J. Pollinat. Ecol. 2018, 22, 55–66. [Google Scholar] [CrossRef]
- Andrikopoulos, C.J.; Cane, J.H. Comparative pollination efficacies of five bee species on raspberry. J. Econ. Entomol. 2018, 111, 2513–2519. [Google Scholar] [CrossRef] [PubMed]
- Madrid, M.; Beaudry, R. Chapter 16.2—Small fruits: Raspberries, blackberries, blueberries. In Controlled and Modified Atmospheres for Fresh and Fresh-Cut Produce; Gil, M.I., Beaudry, R., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 335–346. [Google Scholar]
- Howlett, B.G.; Donovan, B.J.; McCallum, J.A.; Newstrom, L.E.; Teulon, D.A.J. Between and within field variability of New Zealand indigenous flower visitors to onions. N. Z. Plant Prot. 2005, 58, 213–218. [Google Scholar] [CrossRef]
- Brown, P.; Spurr, C.; Gracie, A.; Geard, A. Pollination and Seed Development in Hybrid Vegetable Seed Crops; Final Report VG03084; Horticulture Australia Limited: North Sydney, NSW, Australia, 2006. [Google Scholar]
- Erickson, E.; Peterson, C. Asynchrony of floral events and other differences in pollinator (Apis mellifera) foraging stimuli between fertile and male-sterile carrot inbreds. J. Am. Soc. Hortic. Sci. 1979, 104, 639–643. [Google Scholar]
- Delaplane, K.S.; Mayer, D.R. Crop Pollination by Bees; CABI: Cambridge, UK, 2000. [Google Scholar]
- Rodet, G.; Torre Grossa, J.P.; Bonnet, A. Foraging behavior of Apis Mellifera L. on male-sterile and male-fertile inbred lines of carrot (Daucus carota L.) in gridded enclosures. ActaHortic 1991, 288, 371–375. [Google Scholar] [CrossRef]
- Dyer, F.C.; Gould, J.L. Honey bee orientation: A backup system for cloudy days. Science 1981, 214, 1041–1042. [Google Scholar] [CrossRef]
- Hall, M.; Jones, J.; Rocchetti, M.; Wright, D.; Rader, R. Bee visitation and fruit quality in berries under protected cropping vary along the length of polytunnels. bioRxiv 2019, 722041. [Google Scholar] [CrossRef]
- Bohart, G.E.; Nye, W.P. Insect pollinators of carrots in Utah. Utah Agric. Exp. Stn. Bull. 1960, 419, 16. [Google Scholar]
- Currah, L.; Ockendon, D.J. Pollination activity by blowflies and honeybees on onions in breeders’ cages. Ann. Appl. Biol. 1984, 105, 167–176. [Google Scholar] [CrossRef]
- Jones, H.; Emsweller, S. The use of flies as onion pollinators. Proc. Am. Soc. Hortic. Sci. 1934, 31, 160–164. [Google Scholar]
- Wilson, R.; Widrlechner, M.; Reitsma, K. Pollination methods for maintaining carrot germplasm collections. FAO/IBPGR Plant Genet. Resour. Newsl. 1991, 85, 1. [Google Scholar]
- Burden, H. Tasmanian farmer produces cauliflower seed by breeding flies. In Tasmanian Country Hour; Australian Braodcasting Corporation: Sydney, Australia, 2015. [Google Scholar]
- Phillips, B.B.; Williams, A.; Osborne, J.L.; Shaw, R.F. Shared traits make flies and bees effective pollinators of oilseed rape (Brassica napus L.). Basic Appl. Ecol. 2018, 32, 66–76. [Google Scholar] [CrossRef]
- Winfree, R.; Williams, N.M.; Dushoff, J.; Kremen, C. Native bees provide insurance against ongoing honey bee losses. Ecol. Lett. 2007, 10, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M. Higher pollinator effectiveness by specialist than generalist flower-visitors of unspecialized Knautia arvensis (Dipsacaceae). Oecologia 2005, 146, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Kudo, G.; Cooper, E. When spring ephemerals fail to meet pollinators: Mechanism of phenological mismatch and its impact on plant reproduction. Proc. R. Soc. B Biol. Sci. 2019, 286, 20190573. [Google Scholar] [CrossRef]
- Mizunaga, Y.; Kudo, G. A linkage between flowering phenology and fruit-set success of alpine plant communities with reference to the seasonality and pollination effectiveness of bees and flies. Oecologia 2017, 185, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Brodie, B.; Gries, R.; Martins, A.; VanLaerhoven, S.; Gries, G. Bimodal cue complex signifies suitable oviposition sites to gravid females of the common green bottle fly. Entomol. Exp. Appl. 2014, 153, 114–127. [Google Scholar] [CrossRef]
- Mohr, R.; Tomberlin, J. Development and validation of a new technique for estimating a minimum postmortem interval using adult blow fly (Diptera: Calliphoridae) carcass attendance. Int. J. Leg. Med. 2014, 129, 851–859. [Google Scholar] [CrossRef]
- Ellis, C.R.; Feltham, H.; Park, K.; Hanley, N.; Goulson, D. Seasonal complementary in pollinators of soft-fruit crops. Basic Appl. Ecol. 2017, 19, 45–55. [Google Scholar] [CrossRef]
- Primack, R.B. Variability in New Zealand montane and alpine pollinator assemblages. N. Z. J. Ecol. 1978, 1, 66–73. [Google Scholar]
- Evans, L.J.; Cutting, B.T.; Jochym, M.; Janke, M.A.; Felman, C.; Cross, S.; Jacob, M.; Goodwin, M. Netted crop covers reduce honeybee foraging activity and colony strength in a mass flowering crop. Ecol. Evol. 2019, 9, 5708–5719. [Google Scholar] [CrossRef] [PubMed]
- Aak, A.; Knudsen, G.K. Sex differences in olfaction-mediated visual acuity in blowflies and its consequences for gender-specific trapping. Entomol. Exp. Appl. 2011, 139, 25–34. [Google Scholar] [CrossRef]
- Kostal, V.; Finch, S. Preference of the cabbage root fly, Delia radicum (L.), for coloured traps: Influence of sex and physiological status of the flies, trap background and experimental design. Physiol. Entomol. 1996, 21, 123–130. [Google Scholar] [CrossRef]
- Raguso, R.A. Flowers as sensory billboards: Progress towards an integrated understanding of floral advertisement. Curr. Opin. Plant Biol. 2004, 7, 434–440. [Google Scholar] [CrossRef]
- Weiss, M.R. Floral colour changes as cues for pollinators. Nature 1991, 354, 227. [Google Scholar] [CrossRef]
- Brodie, B.S.; Smith, M.A.; Lawrence, J.; Gries, G. Effects of floral scent, color and pollen on foraging decisions and oocyte development of common green bottle flies. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Yan, G.; Liu, S.; Schlink, A.C.; Flematti, G.R.; Brodie, B.S.; Bohman, B.; Greeff, J.C.; Vercoe, P.E.; Hu, J.; Martin, G.B. Behavior and electrophysiological response of gravid and non-gravid Lucilia cuprina (Diptera: Calliphoridae) to carrion-associated compounds. J. Econ. Entomol. 2018, 111, 1958–1965. [Google Scholar] [CrossRef]
- Wall, R.; Fisher, P. Visual and olfactory cue interaction in resource-location by the blowfly, Lucilia sericata. Physiol. Entomol. 2001, 26, 212–218. [Google Scholar] [CrossRef]
- McAlpine, J.F.; Peterson, B.V.; Shewell, G.E.; Teskey, H.J.; Vockeroth, J.R.; Wood, D.M. Manual of Nearctic Diptera; Agriculture Canada: Ottawa, QC, Canada, 1981; Volume 1, pp. 675–1332. [Google Scholar]
- MacFarlane, R.P. Notes on insects of the Chatham Islands. N. Zeal. Entomol. 1979, 7, 64–70. [Google Scholar] [CrossRef]
- Graham-Smith, G.S. Further observations on the anatomy and function of the proboscis of the blow-fly, Calliphora erythrocephala L. Parasitology 1930, 22, 47–115. [Google Scholar] [CrossRef]
- Lutz, L.; Williams, K.A.; Villet, M.H.; Ekanem, M.; Szpila, K. Species identification of adult African blowflies (Diptera: Calliphoridae) of forensic importance. Int. J. Leg. Med. 2018, 132, 831–842. [Google Scholar] [CrossRef] [PubMed]
- Kandori, I. Diverse visitors with various pollinator importance and temporal change in the important pollinators of Geranium thunbergii (Geraniaceae). Ecol. Res. 2002, 17, 283–294. [Google Scholar] [CrossRef]
- Li, X.; Yang, Y.; Li, G.; Li, H.; Wang, Q.; Wan, L. The effect of dietary fat levels on the size and development of Chrysomya megacephala (Diptera: Calliphoridae). J. Insect Sci. 2014, 14, 174. [Google Scholar] [CrossRef]
- Saunders, D.S.; Wheeler, I.; Kerr, A. Survival and reproduction of small blow flies (Calliphora vicina; Diptera: Calliphoridae) produced in severely overcrowded short-day larval cultures. Eur. J. Entomol. 1999, 96, 19–22. [Google Scholar]
- Shingleton, A.W.; Frankino, W.A.; Flatt, T.; Nijhout, H.F.; Emlen, D.J. Size and shape: The developmental regulation of static allometry in insects. BioEssays 2007, 29, 536–548. [Google Scholar] [CrossRef]
- Smith, J.L.; Palermo, N.A.; Theobald, J.C.; Wells, J.D. Body size, rather than male eye allometry, explains Chrysomya megacephala (Diptera: Calliphoridae) activity in low light. J. Insect Sci. 2015, 15, 133. [Google Scholar] [CrossRef][Green Version]
- Hu, Y.; Yuan, X.; Zhu, F.; Lei, C. Development time and size-related traits in the oriental blowfly, Chrysomya megacephala along a latitudinal gradient from China. J. Therm. Biol. 2010, 35, 366–371. [Google Scholar] [CrossRef]
- Stoffolano, J.G.; Gonzalez, E.Y.; Sanchez, M.; Kane, J.; Velázquez, K.; Oquendo, A.L.; Sakolsky, G.; Schafer, P.; Yin, C.M. Relationship between size and mating success in the blow fly Phormia regina (Diptera: Calliphoridae). Ann. Entomol. Soc. Am. 2000, 93, 673–677. [Google Scholar] [CrossRef]
- Jander, U.; Jander, R. Allometry and resolution of bee eyes (Apoidea). Arthropod Struct. Dev. 2002, 30, 179–193. [Google Scholar] [CrossRef]
- Sukontason, K.L.; Chaiwong, T.; Piangjai, S.; Upakut, S.; Moophayak, K.; Sukontason, K. Ommatidia of blow fly, house fly, and flesh fly: Implication of their vision efficiency. Parasitol. Res. 2008, 103, 123–131. [Google Scholar] [CrossRef]
- Willmer, P.G.; Unwin, D.M. Field analyses of insect heat budgets: Reflectance, size and heating rates. Oecologia 1981, 50, 250–255. [Google Scholar] [CrossRef]
- Colquhoun, E. HAL Report AH98002: Horticultural Productivity and Sustainability Project; Horticulture Australia Ltd.: Sydney, Australia, 2000. [Google Scholar]
- Wells, J.D. Chrysomya megacephala (Diptera: Calliphoridae) has reached the continental United States: Review of its biology, pest status, and spread around the world. J. Med. Entomol. 1991, 28, 471–473. [Google Scholar] [CrossRef]
- Atlas of Living Australia. 2020. Available online: https://www.ala.org.au/ (accessed on 10 March 2020).
- Sanei-Dehkordi, A.; Khamesipour, A.; Akbarzadeh, K.; Akhavan, A.A.; Rassi, Y.; Oshaghi, M.A.; Miramin-Mohammadi, A.; Eskandari, S.E.; Rafinejad, J. Experimental colonization and life table of the Calliphora vicina (Robineau-Desvoidy)(Diptera: Calliphoridae). J. Entomol. Zool. Stud. 2014, 2, 45–48. [Google Scholar]
- Donovan, S.E.; Hall, M.J.R.; Turner, B.D.; Moncrieff, C.B. Larval growth rates of the blowfly, Calliphora vicina, over a range of temperatures. Med. Vet. Entomol. 2006, 20, 106–114. [Google Scholar] [CrossRef]
- Davies, L. Lifetime reproductive output of Calliphora vicina and Lucilia sericata in outdoor caged and field populations; flight vs. egg production? Med. Vet. Entomol. 2006, 20, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Vinogradova, E.B. Effect of food and temperature on the reproduction of the blowfly, Calliphora vicina R.-D. (Diptera, Calliphoridae), a popular model object in biological research. Entomol. Rev. 2009, 89, 137–142. [Google Scholar] [CrossRef]
- Williams, H.; Richardson, A.M.M. Growth energetics in relation to temperature for larvae of four species of necrophagous flies (Diptera: Calliphoridae). Aust. J. Ecol. 1984, 9, 141–152. [Google Scholar] [CrossRef]
- Anderson, G.S. Minimum and maximum development rates of some forensically important Calliphoridae (Diptera). J. Forensic Sci. 2000, 45, 824–832. [Google Scholar] [CrossRef]
- Hulbert, A.J.; Usher, M.J.; Wallman, J.F. Food consumption and individual lifespan of adults of the blowfly, Calliphora stygia: A test of the ‘rate of living’ theory of aging. Exp. Gerontol. 2004, 39, 1485–1490. [Google Scholar] [CrossRef]
- Mackerras, M.J. Observations on the life-histories, nutritional requirements and fecundity of blowflies. Bull. Entomol. Res. 1933, 24, 353–362. [Google Scholar] [CrossRef]
- O’Flynn, M.A. The succession and rate of development of blowflies in carrion in southern Queensland and the application of these data to forensic entomology. Aust. J. Entomol. 1983, 22, 137–148. [Google Scholar] [CrossRef]
- Ujvari, B.; Wallman, J.; Madsen, T.; Whelan, M.; Hulbert, A.J. Experimental studies of blowfly (Calliphora stygia) longevity: A little dietary fat is beneficial but too much is detrimental. Comp. Biochem. Physiol. Part A 2009, 154, 383–388. [Google Scholar] [CrossRef]
- Dear, J.P. Calliphoridae (Insecta: Diptera). Fauna New Zealand 1986, 8, 88. [Google Scholar]
- Callinan, A. Aspects of the ecology of Calliphora Augur (Fabricius) (Diptera: Calliphoridae), a native Australian blowfly. Aust. J. Zool. 1980, 28, 679–684. [Google Scholar] [CrossRef]
- Norris, K.R. The ecology of sheep blowflies in Australia. In Biogeography and Ecology in Australia; Keast, A., Crocker, R.L., Christian, C.S., Eds.; Springer: Dordrecht, The Netherlands, 1959; pp. 514–544. [Google Scholar] [CrossRef]
- Levot, G. Sheep blowflies; State of New South Wales Primefact 485, NSW Department of Primary Industries: Sydney, NSW, Australia, 2009. [Google Scholar]
- Monzu, N. Coexistence of Carrion Breeding Calliphoridae (Diptera) in Western Australia; University of Western Australia: Perth, Australia, 1977. [Google Scholar]
- Morris, B. Description of the life history stages of Calliphora nociva Hardy (Diptera: Calliphoridae). Aust. J. Entomol. 1991, 30, 79–82. [Google Scholar] [CrossRef]
- Cook, D.F.; Dadour, I.R. Larviposition in the ovoviviparous blowfly Calliphora dubia. Med. Vet. Entomol. 2011, 25, 53. [Google Scholar] [CrossRef]
- Voss, S.C.; Cook, D.F.; Wei-Feng, H.; Dadour, I.R. Survival and development of the forensically important blow fly, Calliphora varifrons (Diptera: Calliphoridae) at constant temperatures. Forensic Sci. Int. 2014, 10, 314–321. [Google Scholar] [CrossRef]
- Cook, D.F.; Voss, S.C.; Dadour, I.R. The laying of live larvae by the blowfly Calliphora varifrons (Diptera: Calliphoridae). Forensic Sci. Int. 2012, 223, 44–46. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.T.; Shiao, S.F. Oviposition preferences of two forensically important blow fly species, Chrysomya megacephala and C. rufifacies (Diptera: Calliphoridae), and implications for postmortem interval estimation. J. Med. Entomol. 2012, 49, 424–435. [Google Scholar] [CrossRef]
- Gruner, S.; Slone, D.; Capinera, J. Forensically important Calliphoridae (Diptera) associated with pig carrion in rural north-central Florida. J. Med. Entomol. 2007, 44, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Gabre, R.; Adham, F.; Chi, H. Life table of Chrysomya megacephala (Fabricius) (Diptera: Calliphoridae). Acta Oecologica 2005, 27, 179–183. [Google Scholar] [CrossRef]
- Badenhorst, R.; Villet, M. The uses of Chrysomya megacephala (Fabricius, 1794)(Diptera: Calliphoridae) in forensic entomology. Forensic Sci. Res. 2018, 3, 2–15. [Google Scholar] [CrossRef]
- Wijesundara, D. The life history and bionomics of Chrysomyia megacephala (Fab.). Ceylon J. Sci. 1957, 25, 169–185. [Google Scholar]
- Yang, Y.Q.; Li, X.B.; Shao, R.Y.; Lyu, Z.; Li, H.W.; Li, G.P.; Xu, L.Z.; Wan, L.H. Developmental times of Chrysomya megacephala (Fabricius) (Diptera: Calliphoridae) at constant temperatures and applications in forensic entomology. J. Forensic Sci. 2016, 61, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Nelson, L.A.; Dowton, M.; Wallman, J.F. Thermal attributes of Chrysomya species. Entomol. Exp. Appl. 2009, 133, 260–275. [Google Scholar] [CrossRef]
- Baumgartner, D. Review of Chrysomya rufifacies (Diptera: Calliphoridae). J. Med. Entomol. 1993, 30, 338–352. [Google Scholar] [CrossRef]
- Byrd, J.H.; Butler, J.F. Effects of temperature on Chrysomya rufifacies (Diptera: Calliphoridae) development. J. Med. Entomol. 1997, 34, 353–358. [Google Scholar] [CrossRef]
- Roy, D.N.; Siddons, L.B. On the life history and bionomics of Chrysomyia rufifacies Macq.(order Diptera, family Calliphoridae). Parasitology 1939, 31, 442–447. [Google Scholar] [CrossRef]
- Abd-AlGalil, F.; Zambare, S.; Khan, L.; Mali, K. Effect of seasonal temperature variations on the life cycle duration of forensically important calliphorid fly, Chrysomya saffranea (Bigot, 1877). J. Forensic Res. 2017, 8, 364–369. [Google Scholar] [CrossRef]
- Levot, G.W.; Brown, K.R.; Shipp, E. Larval growth of some calliphorid and sarcophagid Diptera. Bull. Entomol. Res. 1979, 69, 469–475. [Google Scholar] [CrossRef]
- Lutz, L.; Verhoff, M.A.; Amendt, J. Environmental factors influencing flight activity of forensically important female blow flies in Central Europe. Int. J. Leg. Med. 2019, 133, 1267–1278. [Google Scholar] [CrossRef]
- Hayes, E.J.; Wall, R.; Smith, K.E. Mortality rate, reproductive output, and trap response bias in populations of the blowfly Lucilia sericata. Ecol. Entomol. 1999, 24, 300–307. [Google Scholar] [CrossRef]
- Grassberger, M.; Reiter, C. Effect of temperature on Lucilia sericata (Diptera: Calliphoridae) development with special reference to the isomegalen and isomorphen-diagram. Forensic Sci. Int. 2001, 120, 32–36. [Google Scholar] [CrossRef]
- Tarone, A.M.; Picard, C.J.; Spiegelman, C.; Foran, D.R. Population and temperature effects on Lucilia sericata (Diptera: Calliphoridae) body size and minimum development time. J. Med. Entomol. 2011, 48, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Wall, R. The reproductive output of the blowfly Lucilia sericata. J. Insect Physiol. 1993, 39, 743–750. [Google Scholar] [CrossRef]
- Kamal, A.S. Comparative study of thirteen species of sarcosaprophagous Calliphoridae and Sarcophagidae (Diptera) I. Bionomics. Ann. Entomol. Soc. Am. 1958, 51, 261–270. [Google Scholar] [CrossRef]
- Roe, A.; Higley, L.G. Development modeling of Lucilia sericata (Diptera: Calliphoridae). PeerJ 2015, 3, e803. [Google Scholar] [CrossRef]
- Dear, J. A revision of Australian Rhiniinae (Diptera: Calliphoridae). Aust. J. Zool. 1977, 25, 779–826. [Google Scholar] [CrossRef]
- Campoy, A.; Sáez, L.; Pérez-Bañón, C.; Rojo, S. Demography and population parameters of two species of eristaline flower flies (Diptera, Syrphidae, Eristalini). J. Appl. Entomol. 2019, 144, 133–143. [Google Scholar] [CrossRef]
- Nicholas, S.; Thyselius, M.; Holden, M.; Nordstrom, K. Rearing and long-term maintenance of Eristalis tenax hoverflies for research studies. J. Vis. Exp. 2018, 135, e57711. [Google Scholar] [CrossRef] [PubMed]
- Klecka, J.; Hadrava, J.; Biella, P.; Akter, A. Flower visitation by hoverflies (Diptera: Syrphidae) in a temperate plant-pollinator network. PeerJ 2018, 6, e6025. [Google Scholar] [CrossRef] [PubMed]
- Manpoong, N.S.; Firake, D.M.; Behere, G.T.; Rajesh, T. Biological attributes and feeding potential of three dominant predators of Lipaphis erysimi (Kaltenbach). J. Biol. Control 2016, 30, 190–194. [Google Scholar] [CrossRef]
- Soleyman-Nezhadiyan, E. The ecology of Melangyna viridiceps and Simosyrphus grandicornis (Diptera: Syrphidae) and Their Impact on Populations of the Rose Aphid, Macrosiphum rosae. Ph.D. Thesis, University of Adelaide, Adelaide, Australia, 1996. [Google Scholar]
- Soleyman-Nezhadiyan, E.; Laughlin, R. Voracity of larvae, rate of development in eggs, larvae and pupae, and flight seasons of adults of the hoverflies Melangyna viridiceps Macquart and Symosyrphus grandicornis Macquart (Diptera: Syrphidae). Aust. J. Entomol. 1998, 37, 243–248. [Google Scholar] [CrossRef]
- Pérez-Bañón, C.; Juan, A.; Petanidou, T.; Marcos-García, M.A.; Crespo, M.B. The reproductive ecology of Medicago citrina (Font Quer) Greuter (Leguminosae): A bee-pollinated plant in Mediterranean islands where bees are absent. Plant Syst. Evol. 2003, 241, 29–46. [Google Scholar] [CrossRef]
- Thomson, G.M. The Naturalisation of Animals and Plants in New Zealand; Cambridge University Press: London, UK, 1922. [Google Scholar]
- Wallman, J.F.; Leys, R.; Hogendoorn, K. Molecular systematics of Australian carrion-breeding blowflies (Diptera: Calliphoridae) based on mitochondrial DNA. Invertebr. Syst. 2005, 19, 1. [Google Scholar] [CrossRef]
- Doucet, D.; Walker, V.K.; Qin, W. The bugs that came in from the cold: Molecular adaptations to low temperatures in insects. Cell. Mol. Life Sci. 2009, 66, 1404–1418. [Google Scholar] [CrossRef]
- Corbet, S.; Fussell, M.; Ake, R.; Fraser, A.; Gunson, C.; Savage, A.; Smith, K. Temperature and the pollinating activity of social bees. Ecol. Entomol. 1993, 18, 17–30. [Google Scholar] [CrossRef]
- Vicens, N.; Bosch, J. Weather-dependent pollinator activity in an apple orchard, with special reference to Osmia cornuta and Apis mellifera (Hymenoptera: Megachilidae and Apidae). Environ. Entomol. 2000, 29, 413–420. [Google Scholar] [CrossRef]
- Norris, K.R. Daily patterns of flight activity of blowflies (Calliphoridae: Diptera) in the Canberra district as indicated by trap catches. Aust. J. Zool. 1966, 14, 835–853. [Google Scholar] [CrossRef]
- Guillot, F.S.; Brown, H.E.; Broce, A.B. Behavior of sexually active male screwworm flies. Ann. Entomol. Soc. Am. 1978, 71, 199–201. [Google Scholar] [CrossRef]
- Graham-Smith, G.S. Observations on the habits and parasites of common flies. Parasitology 1916, 8, 441–544. [Google Scholar] [CrossRef]
- Ullerich, F.H. Analysis of sex determination in the monogenic blowfly Chrysomya rufifacies by pole cell transplantation. Mol. Gen. Genet. 1984, 193, 479–487. [Google Scholar] [CrossRef]
- Browne, L.B. Physiologically induced changes in resource-oriented behavior. Annu. Rev. Entomol. 1993, 38, 1–23. [Google Scholar] [CrossRef]
- Sherman, R.A.; Wyle, F.A. Low-cost, low-maintenance rearing of maggots in hospitals, clinics, and schools. Am. J. Trop. Med. Hyg. 1996, 54, 38–41. [Google Scholar] [CrossRef]
- Firoozfar, F.; Moosa-Kazemi, H.; Baniardalani, M.; Abolhassani, M.; Khoobdel, M.; Rafinejd, J. Mass rearing of Lucilia sericata Meigen (Diptera: Calliphoridae). Asian Pac. J. Trop. Biomed. 2011, 1, 54–56. [Google Scholar] [CrossRef]
- Gurney, W.; Woodhill, A. Investigations on sheep blowflies. Sci. Bull. Dep. Agric. New South Wales 1926, 27, 1–19. [Google Scholar]
- Tsuda, Y.; Hayashi, T.; Higa, Y.; Hoshino, K.; Kasai, S.; Tomita, T.; Kurahashi, H.; Kobayashi, M. Dispersal of a blow fly, Calliphora nigribarbis, in relation to the dissemination of highly pathogenic avian influenza virus. Jpn. J. Infect. Dis. 2009, 62, 294–297. [Google Scholar]
- Anderson, P.; Shipp, E.; Anderson, J.; Dobbie, W. Population maintenance of Lucilia cuprina (Wiedemann) in the arid zone. Aust. J. Zool. 1988, 36, 241–249. [Google Scholar] [CrossRef]
- Pereira, R.; Yuval, B.; Liedo, P.; Teal, P.E.A.; Shelly, T.E.; McInnis, D.O.; Hendrichs, J. Improving sterile male performance in support of programmes integrating the sterile insect technique against fruit flies. J. Appl. Entomol. 2013, 137, 178–190. [Google Scholar] [CrossRef]
- Moophayak, K.; Meeinkuirt, W. Predominance of blow flies (Diptera: Calliphoridae) among insects visiting flowers of Buchanania lanzan (Sapindales: Anacardiaceae). Appl. Ecol. Environ. Res. 2017, 15, 651–659. [Google Scholar] [CrossRef]
- Bharti, M.; Bharti, H. Association and impact of ectoparasitic blowflies (Diptera: Calliphoridae) on Himalayan ants of genus Myrmica. Insect. Soc. 2016, 63, 477–480. [Google Scholar] [CrossRef]
- Gilbert, F.S. Foraging ecology of hoverflies: Morphology of the mouthparts in relation to feeding on nectar and pollen in some common urban species. Ecol. Entomol. 1981, 6, 245–262. [Google Scholar] [CrossRef]
- Holloway, B.A. Pollen-feeding in hover-flies (Diptera: Syrphidae). N. Z. J. Zool. 1976, 3, 339–350. [Google Scholar] [CrossRef]
- Colless, D.; McAlpine, D. Chapter 39. Diptera (Flies); CSIRO: Melbourne, Australia, 1991; Volume 2, pp. 717–786. [Google Scholar]
- Ferguson, E. Revision of Australian Syrphidae (Diptera). Part I. Proc. Linn. Soc. New South Wales 1926, 51, 137–183. [Google Scholar]
- Finch, J.T.D.; Cook, J.M. Flies on vacation: Evidence for the migration of Australian Syrphidae (Diptera). Ecol. Entomol. 2020. [Google Scholar] [CrossRef]
- Pinheiro, L.A.; Torres, L.; Raimundo, J.; Santos, S.A.P. Effect of floral resources on longevity and nutrient levels of Episyrphus balteatus (Diptera: Syrphidae). Biol. Control 2013, 67, 178–185. [Google Scholar] [CrossRef]
- Haslett, J.R. Interpreting patterns of resource utilization: Randomness and selectivity in pollen feeding by adult hoverflies. Oecologia 1989, 78, 433–442. [Google Scholar] [CrossRef]
- Hannah, L.; Dyer, A.G.; Garcia, J.E.; Dorin, A.; Burd, M. Psychophysics of the hoverfly: Categorical or continuous color discrimination? Curr. Zool. 2019, 65, 483–492. [Google Scholar] [CrossRef]
- Primante, C.; Dötterl, S. A syrphid fly uses olfactory cues to find a non-yellow flower. J. Chem. Ecol. 2010, 36, 1207–1210. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.G.V. An introduction to the immature stages of British flies: Diptera larvae, with notes on eggs, puparia and pupae. Handb. Identif. Br. Insects 1989, 10, 1–280. [Google Scholar]
- Hondelmann, P.; Poehling, H.M. Diapause and overwintering of the hoverfly Episyrphus balteatus. Entomol. Exp. Appl. 2007, 124, 189–200. [Google Scholar] [CrossRef]
- Desek, J.; Laska, P. Overwinteing and spring emergence of some common species of aphidophagous syrphids (Syrphidae, Diptera). Folia Fac. Sci. Nat. Univers. Purkyn. Brun. Biológia 1974, 43, 71–75. [Google Scholar]
- Raymond, L.; Sarthou, J.P.; Plantegenest, M.; Gauffre, B.; Ladet, S.; Vialatte, A. Immature hoverflies overwinter in cultivated fields and may significantly control aphid populations in autumn. Agric. Ecosyst. Environ. 2014, 185, 99–105. [Google Scholar] [CrossRef]
- Pinheiro, L.A.; Torres, L.M.; Raimundo, J.; Santos, S.A.P. Effects of pollen, sugars and honeydew on lifespan and nutrient levels of Episyrphus balteatus. BioControl 2015, 60, 47–57. [Google Scholar] [CrossRef]
- Pastor, B.; Velasquez, Y.; Gobbi, P.; Rojo, S. Conversion of organic wastes into fly larval biomass: Bottlenecks and challenges. J. Insects Food Feed 2015, 1, 179–193. [Google Scholar] [CrossRef]
- Ganda, H.; Zannou-Boukari, E.; Kenis, M.; Chrysostome, C.; Mensah, G. Potentials of animal, crop and agri-food wastes for the production of fly larvae. J. Insects Food Feed 2019, 5, 59–67. [Google Scholar] [CrossRef]
- Van Huis, A. Manure and flies: Biodegradation and/or bioconversion? J. Insects Food Feed 2019, 5, 55–58. [Google Scholar] [CrossRef]
| Fly Pollinator | Crops A | Literature |
|---|---|---|
| Anthomyiidae (Flower Flies) | ||
| Delia spp. | PK | [16] |
| Delia platura | PK | [14,27,28] |
| Anthomyia punctipennis | CT, ON, PK | [14], [16], [27] |
| Bombyliidae (Bee Flies) | ||
| Comptosia ocellata | CT | [29] |
| Calliphoridae (Blow Flies) | AV, BL, BS, CT, MC, MG, ON | [29,30,31,32,33,34,35,36,37,38] |
| Calliphora spp. | AV, MC, MG, ON | [35,36,37] |
| Calliphora vicina | AV, CT, LC, LK, ON, PK | [14,27,39,40] |
| Calliphora stygia | AV, ON, PK | [14], [16], [27], [32], [41] |
| Calliphora augur | AV | [32], [37] |
| Calliphora albifrontalis | AV, BL | [42] |
| Chrysomya spp. | MG | [37,43,44] |
| Chrysomya megacephala | MG | [43,45,46,47] |
| Chrysomya saffranea | MG | [43] |
| Chrysomya rufifacies | AV, MG | [32,43] |
| Chrysomya varipes | AV | [36] |
| Lucilia sericata | AV, CE, LY, MG, ON, PK, ST | [14,27,32,48,49,50] |
| Lucilia cuprina | AV | [32] |
| Muscidae (House Flies) | [29,37,51] | |
| Musca domestica | LK, MG, ON | [27], [35], [39], [43], [50] |
| Hydrotaea rostrata | ON, PK | [14,27] |
| Spilogona spp. | PK | [28] |
| Rhiniidae (Nose Flies) | AV | [32,37] |
| Stomorhina discolour | AV, MC, MG | [32], [37] |
| Stomorhina xanthogaster | MG | [43] |
| Sarcophagidae (Flesh Flies) | MG, ON | [35,37,43] |
| Oxysarcodexia varia | ON, PK | [14], [16], [27] |
| Stratiomyidae (Soldier Flies) | [37,52] | |
| Odontomyia spp. | PK | [14,27,28] |
| Odontomyia atrovirens | PK | [52] |
| Syrphidae (Hover Flies) | AV, LY, MC, MG, ON, ST | [10,13,14,28,37,47,52,53,54,55] |
| Eristalis spp (Drone Flies) | MG | [43,44] |
| Eristalinus hervebazini B | MG | [43] |
| Eristalis tenax | CB, CT, ON, PE, PK | [14,15,16,27,28,29,38,41,51,55,56,57,58] |
| Melangyna spp. | CT, ON, PK | [14,27,29,51] |
| Mesembrius bengalensis | MG | [43] |
| Simosyrphus grandicornis | AV | [32] |
| Tachinidae (Bristle Flies) | AV, ON, PK | [14,27,32,37,43,52] |
| Tabanidae (Horse Flies) | ||
| Scaptia spp. | PK ON | [14,27] |
| Fly Pollinator | Size (mm) | Eggs/♀ | Lifespan | DT | DP | ATR | Literature |
|---|---|---|---|---|---|---|---|
| Calliphoridae (Blow flies) | |||||||
| Calliphora vicina | 10–14 | 111–250 | 40 (F) | 15 | 4–25 | [132,133,134,135,136] | |
| Calliphora stygia (N) | 8–13 | 85 | 35 (L) | 18 | 10 | 7–24 | [136,137,138,139,140,141,142] |
| Calliphora augurVIV (N) | 11 | 67 (F) | 13 | 8 | 11–26 | [139,140,143,144,145] | |
| Calliphora dubiaVIV (N) | 11 | 50–80 | 19 | [145,146,147,148] | |||
| Calliphora albifrontalis (N) | 20 | [146] | |||||
| Calliphora varifronsVIV (N) | 33.4 ± 1.0 | 19 | 12 | 6–32 | [146,149,150] | ||
| Chrysomya megacephala | 8–9 | 223.7 ± 2.4 | 54–105 (L) | 19 | 6 | [124,140,151,152,153,154,155,156,157] | |
| Chrysomya rufifacies (N) | 9 | 210 | 24 (L) | 12 | 5 | 13–28 | [139,140,145,146,158,159,160] |
| Chrysomya saffranea (N) | 12 | 18–33 | [140,157,161] | ||||
| Chrysomya varipes (N) | 6 | 11 | 5 | 20–28 | [140,162] | ||
| Lucilia sericata | 10–14 | 225 ± 7 | 30 (F) 40–59 (L) | 13 | 6 | 10–30 | [139,163,164,165,166,167,168,169] |
| Rhiniidae (Nose Flies) | |||||||
| Stomorhina discolour | 4.5–6.5 | [170] | |||||
| Stomorhina xanthogaster | 9.5–11 | [170] | |||||
| Syrphidae (Hover Flies) | |||||||
| Eristalinus hervebazini (N) | |||||||
| Eristalinus punctulatus (N) | |||||||
| Eristalis tenax | 14.52 | 191.4 ± 75.7 | 23.4 ± 2.4 (L) | 22.6 ± 0.2 | 5–30 | [171,172,173] | |
| Episyrphus viridaureus | 45.0 ± 16.8 | 14.0 ± 1.5 (L) | 29–30 | 7 | [174] | ||
| Melangyna viridiceps (N) | 288.0 ± 34.0 | 32.9 ± 1.6 (L) | 17.8 | 7.1 | [175,176] | ||
| Mesembrius bengalensis | |||||||
| Simosyrphus grandicornis | 307.9 ± 23.2 | 19.9 ± 1.4 (L) | 17.4 | 8.2 | [175,176] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cook, D.F.; Voss, S.C.; Finch, J.T.D.; Rader, R.C.; Cook, J.M.; Spurr, C.J. The Role of Flies as Pollinators of Horticultural Crops: An Australian Case Study with Worldwide Relevance. Insects 2020, 11, 341. https://doi.org/10.3390/insects11060341
Cook DF, Voss SC, Finch JTD, Rader RC, Cook JM, Spurr CJ. The Role of Flies as Pollinators of Horticultural Crops: An Australian Case Study with Worldwide Relevance. Insects. 2020; 11(6):341. https://doi.org/10.3390/insects11060341
Chicago/Turabian StyleCook, David F, Sasha C Voss, Jonathan T D Finch, Romina C Rader, James M Cook, and Cameron J Spurr. 2020. "The Role of Flies as Pollinators of Horticultural Crops: An Australian Case Study with Worldwide Relevance" Insects 11, no. 6: 341. https://doi.org/10.3390/insects11060341
APA StyleCook, D. F., Voss, S. C., Finch, J. T. D., Rader, R. C., Cook, J. M., & Spurr, C. J. (2020). The Role of Flies as Pollinators of Horticultural Crops: An Australian Case Study with Worldwide Relevance. Insects, 11(6), 341. https://doi.org/10.3390/insects11060341






