An Insight into the Role of Trissolcus mitsukurii as Biological Control Agent of Halyomorpha halys in Northeastern Italy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling Sites and Laboratory Study
2.2. Insect Identification and Molecular Analysis
3. Results
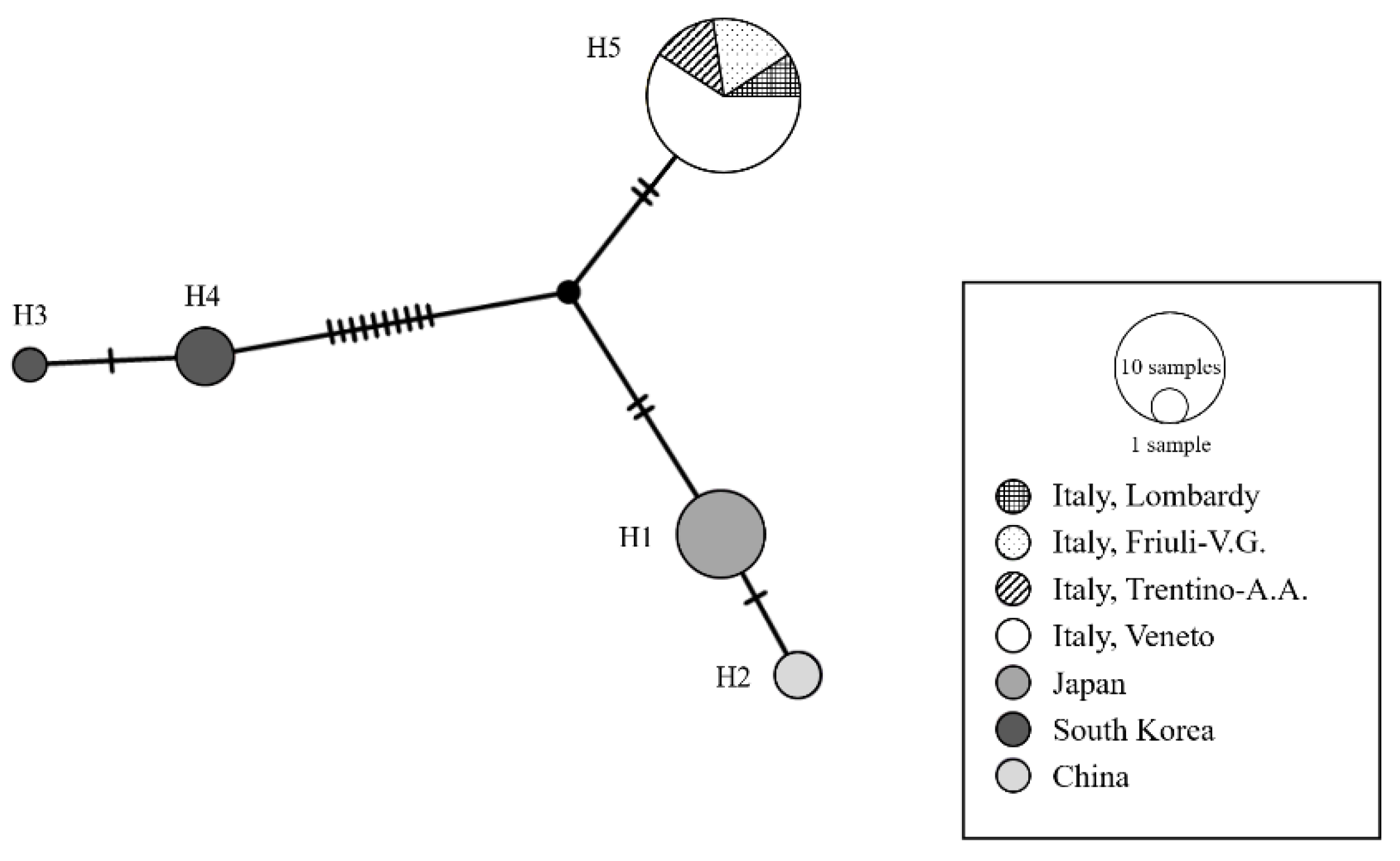
Molecular Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoebeke, E.R.; Carter, M.E. Halyomorpha halys (Stål) (Heteroptera: Pentatomidae): A polyphagous plant pest from Asia newly detected in North America. Proc. Entomol. Soc. Wash. 2003, 105, 225–237. [Google Scholar]
- Wermelinger, B.; Wyniger, D.; Forster, B. First records of an invasive bug in Europe: Halyomorpha halys Stål (Heteroptera: Pentatomidae), a new pest on woody ornamentals and fruit trees? Mitt. Schweiz. Entomol. Ges. 2008, 81, 1–8. [Google Scholar] [CrossRef]
- Faúndez, E.I.; Rider, D.A. The brown marmorated stink bug Halyomorpha halys (Stål, 1855) (Heteroptera: Pentatomidae) in Chile. Arq. Entomolóxicos 2017, 17, 305–307. [Google Scholar]
- Haye, T.; Gariepy, T.; Hoelmer, K.; Rossi, J.P.; Streito, J.C.; Tassus, X.; Desneux, N. Range expansion of the invasive brown marmorated stink bug, Halyomorpha halys: An increasing threat to field, fruit and vegetable crops worldwide. J. Pest. Sci. 2015, 88, 665–673. [Google Scholar] [CrossRef]
- Leskey, T.C.; Nielsen, A.L. Impact of the invasive brown marmorated stink bug in North America and Europe: History, biology, ecology, and management. Annu. Rev. Entomol. 2018, 63, 599–618. [Google Scholar] [CrossRef] [Green Version]
- Moore, L.C.; Tirello, P.; Scaccini, D.; Toews, M.D.; Duso, C.; Pozzebon, A. Characterizing damage potential of the brown marmorated stink bug (Hemiptera: Pentatomidae) in cherry orchards. Entomol. Gen. 2019, 39, 271–283. [Google Scholar] [CrossRef]
- Nielsen, A.L.; Hamilton, G.C. Life history of the invasive species Halyomorpha halys (Hemiptera: Pentatomidae) in northeastern United States. Ann. Entomol. Soc. Am. 2009, 102, 608–616. [Google Scholar] [CrossRef] [Green Version]
- Inkley, D.B. Characteristics of home invasion by the brown marmorated stink bug (Hemiptera: Pentatomidae). J. Entomol. Sci. 2012, 47, 125–130. [Google Scholar] [CrossRef]
- Leskey, T.C.; Hamilton, G.C.; Nielsen, A.L.; Polk, D.F.; Rodriguez-Saona, C.; Bergh, J.C.; Herbert, D.A.; Kuhar, T.P.; Pfeiffer, D.; Dively, G.P.; et al. Pest status of the brown marmorated stink bug, Halyomorpha halys in the USA. Outlooks Pest Manag. 2012, 23, 218–226. [Google Scholar] [CrossRef] [Green Version]
- Bariselli, M.; Bugiani, R.; Maistrello, L. Distribution and damage caused by Halyomorpha halys in Italy. Bull. OEPP 2016, 46, 332–334. [Google Scholar] [CrossRef]
- Bosco, L.; Moraglio, S.T.; Tavella, L. Halyomorpha halys, a serious threat for hazelnut in newly invaded areas. J. Pest Sci. 2018, 91, 661–670. [Google Scholar] [CrossRef]
- Maistrello, L.; Dioli, P.; Vaccari, G.; Nannini, R.; Bortolotti, P.; Caruso, S.; Costi, E.; Montermini, A.; Casoli, L.; Bariselli, M. Primi rinvenimenti in Italia della cimice esotica Halyomorpha halys, una nuova minaccia per la frutticoltura. Atti Giornate Fitopatol. CLUEB 2014, 2014, 283–288. [Google Scholar]
- Maistrello, L.; Dioli, P.; Dutto, M.; Volani, S.; Pasquali, S.; Gilioli, G. Tracking the spread of sneaking aliens by integrating crowdsourcing and spatial modelling: The Italian invasion of Halyomorpha halys. BioScience 2018, 68, 979–989. [Google Scholar] [CrossRef] [Green Version]
- Unterthurner, M.; Zelger, A.; Fischnaller, S.; Walcher, M.; Haye, T. Die marmorierte baumwanze im visier. Obstbau Weinbau 2017, 53, 5–11. (In German) [Google Scholar]
- Cianferoni, F.; Graziani, F.; Dioli, P.; Ceccolini, F. Review of the occurrence of Halyomorpha halys (Hemiptera: Pentatomidae) in Italy, with an update of its European and world distribution. Biologia 2018, 73, 599–607. [Google Scholar] [CrossRef]
- Candian, V.; Pansa, M.G.; Briano, R.; Peano, C.; Tedeschi, R.; Tavella, L. Exclusion nets: A promising tool to prevent Halyomorpha halys from damaging nectarines and apples in NW Italy. Bull. Insectol. 2018, 71, 21–30. [Google Scholar]
- Candian, V.; Pansa, M.G.; Santoro, K.; Spadaro, D.; Tavella, L.; Tedeschi, R. Photoselective exclusion netting in apple orchards: Effectiveness against pests and impact on beneficial arthropods, fungal diseases and fruit quality. Pest Manag. Sci. 2020, 76, 179–187. [Google Scholar] [CrossRef]
- Lee, D.H.; Short, B.D.; Joseph, S.V.; Bergh, J.C.; Leskey, T.C. Review of the biology, ecology, and management of Halyomorpha halys (Hemiptera: Pentatomidae) in China, Japan, and the Republic of Korea. Environ. Entomol. 2013, 42, 627–641. [Google Scholar] [CrossRef]
- Abram, P.K.; Hoelmer, K.A.; Acebes-Doria, A.; Andrews, H.; Beers, E.H.; Bergh, J.C.; Bessin, R.; Biddinger, D.; Botch, P.; Buffington, M.L.; et al. Indigenous arthropod natural enemies of the invasive brown marmorated stink bug in North America and Europe. J. Pest Sci. 2017, 90, 1009–1020. [Google Scholar] [CrossRef]
- Arakawa, R.; Namura, Y. Effects of temperature on development of three Trissolcus spp. (Hymenoptera: Scelionidae), egg parasitoids of the brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). Entomol. Sci. 2002, 5, 215–218. [Google Scholar]
- Arakawa, R.; Miura, M.; Fujita, M. Effects of host species on the body size, fecundity, and longevity of Trissolcus mitsukurii (Hymenoptera: Scelionidae), a solitary egg parasitoid of stink bugs. Appl. Entomol. Zool. 2004, 39, 177–181. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.Q.; Yao, Y.X.; Qiu, L.F.; Li, Z.X. A new species of Trissolcus (Hymenoptera: Scelionidae) parasitizing eggs of Halyomorpha halys (Heteroptera: Pentatomidae) in China with comments on its biology. Ann. Entomol. Soc. Am. 2009, 102, 39–47. [Google Scholar] [CrossRef]
- Talamas, E.J.; Herlihy, M.V.; Dieckhoff, C.; Hoelmer, K.A.; Buffington, M.; Bon, M.-C.; Weber, D.C. Trissolcus japonicus (Ashmead) (Hymenoptera, Scelionidae) emerges in North America. J. Hymenopt. Res. 2015, 43, 119–128. [Google Scholar] [CrossRef] [Green Version]
- Stahl, J.; Tortorici, F.; Pontini, M.; Bon, M.-C.; Hoelmer, K.; Marazzi, C.; Tavella, L.; Haye, T. First discovery of adventive populations of Trissolcus japonicus in Europe. J. Pest Sci. 2018, 92, 371–379. [Google Scholar] [CrossRef] [Green Version]
- Moraglio, S.T.; Tortorici, F.; Pansa, M.G.; Castelli, G.; Pontini, M.; Scovero, S.; Visentin, S.; Tavella, L. A 3-year survey on parasitism of Halyomorpha halys by egg parasitoids in northern Italy. J. Pest Sci. 2020, 93, 183–194. [Google Scholar] [CrossRef]
- Sabbatini Peverieri, G.; Talamas, E.; Bon, M.C.; Marianelli, L.; Bernardinelli, I.; Malossini, G.; Benvenuto, L.; Roversi, P.F.; Hoelmer, K.A. Two Asian egg parasitoids of Halyomorpha halys (Stål) (Hemiptera, Pentatomidae) emerge in northern Italy: Trissolcus mitsukurii (Ashmead) and Trissolcus japonicus (Ashmead) (Hymenoptera, Scelionidae). J. Hymenopt. Res. 2018, 67, 37–53. [Google Scholar] [CrossRef]
- Gariepy, T.D.; Bruin, A.; Haye, T.; Milonas, P.; Vétek, G. Occurrence and genetic diversity of new populations of Halyomorpha halys in Europe. J. Pest Sci. 2015, 88, 451–460. [Google Scholar] [CrossRef]
- Morrison, W.R., III; Milonas, P.; Kapantaidaki, D.E.; Cesari, M.; Di Bella, E.; Guidetti, R.; Haye, T.; Maistrello, L.; Moraglio, S.T.; Piemontese, L.; et al. Attraction of Halyomorpha halys (Hemiptera: Pentatomidae) haplotypes in North America and Europe to baited traps. Sci. Rep. UK 2017, 7, 16941. [Google Scholar] [CrossRef]
- Cesari, M.; Maistrello, L.; Piemontese, L.; Bonini, R.; Dioli, P.; Lee, W.; Park, C.-G.; Partsinevelos, G.K.; Rebecchi, L.; Guidetti, R. Genetic diversity of the brown marmorated stink bug Halyomorpha halys in the invaded territories of Europe and its patterns of diffusion in Italy. Biol. Invasions 2018, 20, 1073–1092. [Google Scholar] [CrossRef]
- Bin, F.; Vinson, S.B. Efficacy assessment in egg parasitoids (Hymenoptera); proposal for a unified terminology. In Trichogramma and Other Parasitoids, Proceedings of the 3rd International Symposium on Le Colloques de l’INRA, San Antonio, TX, USA, 23–27 September 1990; Wajnberg, E., Vinson, S.B., Eds.; INRA: Paris, France, 1991; Volume 56, pp. 175–179. [Google Scholar]
- SAS Institute. PROC User’s Manual, 6th ed.; SAS Institute: Cary, NC, USA, 2016; Available online: http://support.sas.com/documentation/cdl/en/indbug/68442/PDF/default/indbug.pdf (accessed on 30 August 2019).
- Tillman, G.; Toews, M.; Blaauw, B.; Sial, A.; Cottrell, T.; Talamas, E.; Buntin, D.; Joseph, S.; Balusu, R.; Fadamiro, H.; et al. Parasitism and predation of sentinel eggs of the invasive brown marmorated stink bug, Halyomorpha halys (Stål) (Hemiptera: Pentatomidae), in the southeastern US. Biol. Control 2020, 145, 104247. [Google Scholar] [CrossRef]
- Morrison, W.R., III; Mathews, C.R.; Leskey, T.C. Frequency, efficiency, and physical characteristics of predation by generalist predators of brown marmorated stink bug (Hemiptera: Pentatomidae) eggs. Biol. Control 2016, 97, 120–130. [Google Scholar] [CrossRef] [Green Version]
- Jones, A.L.; Jennings, D.E.; Hooks, C.R.; Shrewsbury, P.M. Field surveys of egg mortality and indigenous egg parasitoids of the brown marmorated stink bug, Halyomorpha halys, in ornamental nurseries in the mid-Atlantic region of the USA. J. Pest Sci. 2017, 90, 1159–1168. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, M.A.; Kononova, S.V. Telenominae of the Fauna of the USSR (Hymenoptera, Scelionidae, Telenominae); Nauka—Leningradskoe Otdelenie: Leningrad, Russia, 1983. (In Russian) [Google Scholar]
- Talamas, E.J.; Buffington, M.L.; Hoelmer, K. Revision of Palearctic Trissolcus Ashmead (Hymenoptera, scelionidae). J. Hymenopt. Res. 2017, 56, 3–185. [Google Scholar] [CrossRef] [Green Version]
- Askew, R.R.; Nieves-Aldrey, J.L. Further observations on Eupelminae (Hymenoptera, Chalcidoidea, Eupelmidae) in the Iberian Peninsula and Canary Islands, including descriptions of new species. Graellsia 2004, 60, 27–39. [Google Scholar] [CrossRef]
- Sabbatini Peverieri, G.; Mitroiu, M.-D.; Bon, M.-C.; Balusu, R.; Benvenuto, L.; Bernardinelli, I.; Fadamiro, H.; Falagiarda, M.; Fusu, L.; Grove, E.; et al. Surveys of stink bug egg parasitism in Asia, Europe and North America, morphological taxonomy, and molecular analysis reveal the Holarctic distribution of Acroclisoides sinicus (Huang & Liao) (Hymenoptera, Pteromalidae). J. Hymenopt. Res. 2019, 74, 123–151. [Google Scholar] [CrossRef] [Green Version]
- Patwary, M.U.; Kenchington, E.L.; Bird, C.J.; Zouros, E. The use of random amplified polymorphic DNA markers in genetic studies of the sea scallop Placopecten magellanicus (Gmelin, 1791). J. Shellfish Res. 1994, 13, 547–553. [Google Scholar]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotech. 1994, 3, 294–299. [Google Scholar]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Barcode of Life Data System. Available online: http://www.barcodinglife.org (accessed on 30 August 2019).
- EMBOSS. Available online: http://www.ebi.ac.uk/Tools/emboss/transeq/index.html (accessed on 30 August 2019).
- Leigh, J.W.; Bryant, D. POPART: Full-feature software for haplotype network construction. Methods Ecol. Evol. 2015, 6, 1110–1116. [Google Scholar] [CrossRef]
- Mita, T.; Nishimoto, H.; Shimizu, N.; Mizutani, N. Occurrence of Trissolcus basalis (Hymenoptera, Platygastridae), an egg parasitoid of Nezara viridula (Hemiptera, Pentatomidae), in Japan. Appl. Entomol. Zool. 2015, 50, 27–31. [Google Scholar] [CrossRef]
- Elton, C.S. The Ecology of Invasion by Plants and Animals; Melthuen Publishing: London, UK, 1958. [Google Scholar]
- Costi, E.; Haye, T.; Maistrello, L. Surveying native egg parasitoids and predators of the invasive Halyomorpha halys in Northern Italy. J. Appl. Entomol. 2019, 143, 299–307. [Google Scholar] [CrossRef]
- Haye, T.; Fischer, S.; Zhang, J.; Gariepy, T. Can native egg parasitoids adopt the invasive brown marmorated stink bug, Halyomorpha halys (Heteroptera: Pentatomidae), in Europe? J. Pest Sci. 2015, 88, 693–705. [Google Scholar] [CrossRef]
- Tortorici, F.; University of Torino, Grugliasco, Italy; Tavella, L.; University of Torino, Grugliasco, Italy. Results of project BIOHALY, 2017–2020. Personal communication.
- Cesari, M.; Maistrello, L.; Ganzerli, F.; Dioli, P.; Rebecchi, L.; Guidetti, R. A pest alien invasion in progress: Potential pathways of origin of the brown marmorated stink bug Halyomorpha halys populations in Italy. J. Pest Sci. 2015, 88, 1–7. [Google Scholar] [CrossRef]
- Gariepy, T.D.; Haye, T.; Fraser, H.; Zhang, J. Occurrence, genetic diversity, and potential pathways of entry of Halyomorpha halys in newly invaded areas of Canada and Switzerland. J. Pest Sci. 2014, 87, 17–28. [Google Scholar] [CrossRef]
- Kapantaidaki, D.E.; Evangelou, V.I.; Morrison, W.R., III; Leskey, T.C.; Brodeur, J.; Milonas, P. Halyomorpha halys (Hemiptera: Pentatomidae) genetic diversity in North America and Europe. Insects 2019, 10, 174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dieckhoff, C.; Tatman, K.M.; Hoelmer, K.A. Natural biological control of Halyomorpha halys by native egg parasitoids: A multi-year survey in northern Delaware. J. Pest Sci. 2017, 90, 1143–1158. [Google Scholar] [CrossRef]
- Konopka, J.K.; Haye, T.; Gariepy, T.D.; McNeil, J.N. Possible coexistence of native and exotic parasitoids and their impact on control of Halyomorpha halys. J. Pest Sci. 2017, 90, 1119–1125. [Google Scholar] [CrossRef]
- Falagiarda, M.; Laimburg Research Centre, Auer, South Tyrol, Italy; Pozzebon, A.; University of Padova, Legnaro, Italy; Scaccini, D.; University of Padova, Legnaro, Italy; Tirello, P.; University of Padova, Legnaro, Italy. Results of monitoring of Halyomorpha halys parasitoids. Personal communication, 2018. [Google Scholar]
- Johnson, N.F. Revision of Australasian Trissolcus species (Hymenoptera: Scelionidae). Invertebr. Taxon. 1991, 5, 211–239. [Google Scholar] [CrossRef]
- Ryu, J.; Hirashima, Y. Taxonomic studies on the genus Trissolcus Ashmead of Japan and Korea (Hymenoptera, Scelionidae). J. Fac. Agric. Kyushu Univ. 1984, 29, 35–58. [Google Scholar]
- Watanabe, C. On five scelionid egg-parasites of some pentatomid and coreid bugs from Shikoku, Japan (Hymenoptera: Proctotrupoidea). Trans. Shikoku Entomol. Soc. 1951, 2, 17–26. [Google Scholar]
- Faraci, F.; Vlach, R.; Heteroptera, M. Checklist delle Specie della Fauna Italiana; Minelli, A., Ruffo, S., La Posta, S., Eds.; Calderini: Bologna, Italy, 1995; Volume 41, pp. 1–56. [Google Scholar]
- Fischnaller, S.; Messner, M. Wanzen im Südtiroler Apfelanbau. Obstbau Weinbau 2018, 54, 24–28. (In German) [Google Scholar]
- Charles, J.G.; Avila, G.A.; Hoelmer, K.A.; Hunt, S.; Gardner-Gee, R.; MacDonald, F.; Davis, V. Experimental assessment of the biosafety of Trissolcus japonicus in New Zealand, prior to the anticipated arrival of the invasive pest Halyomorpha halys. BioControl 2019, 64, 367–379. [Google Scholar] [CrossRef]
- Haye, T.; Moraglio, S.T.; Stahl, J.; Visentin, S.; De Gregorio, T.; Tavella, L. Fundamental host range of Trissolcus japonicus in Europe. J. Pest Sci. 2020, 93, 171–182. [Google Scholar] [CrossRef] [Green Version]
- Lara, J.R.; Pickett, C.H.; Kamiyama, M.T.; Figueroa, M.; Romo, M.; Cabanas, C.; Bazurto, V.; Strode, V.; Briseno, K.; Lewis, M.; et al. Physiological host range of Trissolcus japonicus in relation to Halyomorpha halys and other pentatomids from California. BioControl 2019, 64, 513–528. [Google Scholar] [CrossRef]
- Roversi, P.F.; Binazzi, F.; Marianelli, L.; Costi, E.; Maistrello, L.; Sabbatini Peverieri, G. Searching for native egg-parasitoids of the invasive alien species Halyomorpha halys Stål (Heteroptera Pentatomidae) in Southern Europe. Redia 2016, 99, 63–70. [Google Scholar] [CrossRef]
- Noyes, J.S. Universal Chalcidoidea Database; Natural History Museum: London, UK, 2018. Available online: https://www.nhm.ac.uk/our-science/data/chalcidoids/database/ (accessed on 30 August 2019).
- Stahl, J.M.; Babendreier, D.; Haye, T. Using the egg parasitoid Anastatus bifasciatus against the invasive brown marmorated stink bug in Europe: Can non-target effects be ruled out? J. Pest Sci. 2018, 91, 1005–1017. [Google Scholar] [CrossRef] [Green Version]
- Stahl, J.M.; Babendreier, D.; Haye, T. Life history of Anastatus bifasciatus, a potential biological control agent of the brown marmorated stink bug in Europe. Biol. Control 2019, 129, 178–186. [Google Scholar] [CrossRef]
- Stahl, J.M.; Babendreier, D.; Marazzi, C.; Caruso, S.; Costi, E.; Maistrello, L.; Haye, T. Can Anastatus bifasciatus be used for augmentative biological control of the brown marmorated stink bug in fruit orchards? Insects 2019, 10, 108. [Google Scholar] [CrossRef] [Green Version]
- Talamas, E.J.; Johnson, N.F.; Buffington, M.L. Key to Nearctic species of Trissolcus Ashmead (Hymenoptera, Scelionidae), natural enemies of native and invasive stink bugs (Hemiptera, Pentatomidae). J. Hymenopt. Res. 2015, 43, 45–111. [Google Scholar] [CrossRef] [Green Version]
- Balusu, R.R.; Cottrell, T.E.; Talamas, E.J.; Toews, M.D.; Blaauw, B.R.; Sial, A.A.; Buntin, D.G.; Vinson, E.L.; Fadamiro, H.Y.; Tillman, G.P. New record of Trissolcus solocis (Hymenoptera: Scelionidae) parasitising Halyomorpha halys (Hemiptera: Pentatomidae) in the United States of America. Biodivers. Data J. 2019, 7, e30124. [Google Scholar] [CrossRef]
- Clarke, A.R.; Seymour, J.E. Two species of Acroclisoides Girault and Dodd (Hymenoptera: Pteromalidae) parasitic on Trissolcus basalis (Wollaston) (Hymenoptera: Scelionidae), a parasitoid of Nezara viridula (L.) (Hemiptera: Pentatomidae). Aust. J. Entomol. 1992, 31, 299–300. [Google Scholar] [CrossRef]
- Gariepy, T.D.; Haye, T.; Zhang, J. A molecular diagnostic tool for the preliminary assessment of host–parasitoid associations in biological control programmes for a new invasive pest. Mol. Ecol. 2014, 23, 3912–3924. [Google Scholar] [CrossRef] [PubMed]
- Grissell, E.E.; Smith, D.R. First report of Acroclisoides Girault and Dodd (Hymenoptera: Pteromalidae) in the western Hemisphere, with description of a new species. Proc. Entomol. Soc. Wash. 2006, 108, 923–929. [Google Scholar]
- Abram, P.K.; Brodeur, J.; Burte, V.; Boivin, G. Parasitoid-induced host egg abortion: An underappreciated component of biological control services provided by egg parasitoids. Biol. Control 2016, 98, 52–60. [Google Scholar] [CrossRef]
- Vinson, S.B. Physiological interactions between egg parasitoids and their hosts. In Biological Control with Egg Parasitoids; Wajnberg, E., Hassan, S.A., Eds.; CAB International: Berkshire, UK, 1994; pp. 201–217. [Google Scholar]
- Ogburn, E.C.; Walgenbach, J.F. Effects of insecticides used in organic agriculture on Anastatus reduvii (Hymenoptera: Eupelmidae) and Telenomus podisi (Hymenoptera: Scelionidae), egg parasitoids of pestivorous stink bugs. J. Econ. Entomol. 2019, 112, 108–114. [Google Scholar] [CrossRef]
- Konopka, J.K.; Poinapen, D.; Gariepy, T.; Holdsworth, D.W.; McNeil, J.N. Timing of failed parasitoid development in Halyomorpha halys eggs. Biol. Control 2020, 141, 104124. [Google Scholar] [CrossRef]


| Site Number | Region | Province | Coordinates, Altitude (m a.s.l.) | Survey Period | Host Plants | Habitat Description |
|---|---|---|---|---|---|---|
| 1 | Friuli-Venezia Giulia | Pordenone | 45.975556 N 12.451667 E, 98 m | August (2018) | Ficus carica (on leaves) | Organic orchard (Ficus carica) |
| 2 | Friuli-Venezia Giulia | Udine | 46.032500 N 13.226944 E, 92 m | August (2018) | Glycine max (on leaves) | Experimental farm with soybean, other raw crops, vineyards and orchards |
| 3 | Trentino-Alto Adige | Bolzano | 46.362028 N 11.298500 E, 224 m | September and October (2018) | Acer spp. (on leaves and fruits), Ailanthus altissima (on leaves) and linden (on leaves) | Urban area (parking zone) with maple trees (Acer platanoides, Acer negundo and Acer pseudoplatanus), ailanthus (Ailanthus altissima) and linden (Tilia platyphyllos) |
| 4 | Veneto | Padua | 45.621319 N 11.719279 E, 43 m | June to November (2017–2018) | Actinidia deliciosa (on leaves and fruits) | Small organic orchard (Actinidia deliciosa), near urban area |
| 5 | Veneto | Padua | 45.632120 N 11.799309 E, 40 m | June to November (2017–2018) | Actinidia deliciosa (on leaves and fruits) | Small organic orchard (Actinidia deliciosa), near urban area |
| 6 | Veneto | Padua | 45.646779 N 11.740218 E, 50 m | August to October (2016), June to November (2017) | Actinidia deliciosa (on leaves and fruits) | Small organic orchard (Actinidia deliciosa), near urban area |
| 7 | Veneto | Padua | 45.580714 N 11.787064 E, 27 m | August to October (2016), June to November (2017–2018) | Actinidia deliciosa (on leaves and fruits) | Organic orchard (Actinidia deliciosa), near urban area |
| 8 | Veneto | Treviso | 45.715497 N 11.939604 E, 61 m | August to October (2016), June to November (2017–2018) | Actinidia deliciosa, Prunus spp., Vitis vinifera (on leaves) | Conventional farm with orchard (Actinidia deliciosa, Malus domestica, Prunus avium, Prunus persica, Prunus spp.), small vineyard and vegetables, with hedges |
| 9 | Veneto | Treviso | 45.760649 N 12.007810 E, 94 m | June to November (2017–2018) | Vitis vinifera (on leaves) | Conventional farm with vineyard and orchard (Actinidia deliciosa, Malus domestica, Prunus persica) |
| 10 | Veneto | Treviso | 45.795083 N 12.440934 E, 17 m | June to October (2017) | Vitis vinifera (on leaves) | Conventional farm with vineyard |
| 11 | Veneto | Vicenza | 45.75157 N 11.68244 E, 100 m | August to October (2016–2017) | Olea europaea and Vitis vinifera (on leaves) | Conventional farm with Olea europaea and Vitis vinifera |
| Site Number | Total n Egg Masses | Total n Eggs | Mean (± SE) of Eggs/Egg Mass | n of Parasitized Egg Masses | Parasitoid Species | n of Parasitoid Specimens | Relative Abundance per Site (%) a | Exploitation Efficiency (%, Mean ± SE) per Egg Mass (n of Egg Masses) b | n of Egg Masses with More Than a Parasitoid Species |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | 28 | 28 | 1 | Trissolcus mitsukurii | 24 | 100 | 85.71 (1) | |
| 2 | 2 | 55 | 27.5 (± 0.7) | 1 | Trissolcus basalis | 1 | 100 | 3.57 (1) | |
| 3 c | 63 | 1520 | 24.1 (± 0.2) | 22 | Acroclisoides sinicus | 14 | 10.4 | 100 (1) | 2 (A. bifasciatus and T. mitsukurii) |
| Anastatus bifasciatus | 66 | 49.3 | 31.1 ± 10.4 (11) | ||||||
| Trissolcus mitsukurii | 54 | 40.3 | 43.5 ± 16.4 (5) | ||||||
| 4 | 10 | 271 | 27.1 (± 0.1) | 1 | Trissolcus mitsukurii | 26 | 100 | 100 (1) | |
| 5 | 9 | 241 | 26.8 (± 0.1) | 2 | Anastatus bifasciatus | 8 | 25.8 | 28.6 (1) | |
| Trissolcus mitsukurii | 23 | 74.2 | 85.2 (1) | ||||||
| 6 | 22 | 583 | 26.5 (± 0.1) | 3 | Anastatus bifasciatus | 18 | 40.0 | 27.1 ± 8.6 (2) | 1 (A. bifasciatus and T. mitsukurii) |
| Trissolcus mitsukurii | 27 | 60.0 | 32.3 ± 13.8 (2) | ||||||
| 7 | 32 | 838 | 26.2 (± 0.1) | 2 | Anastatus bifasciatus | 13 | 59.1 | 46.4 (1) | |
| Trissolcus mitsukurii | 9 | 40.9 | 34.6 (1) | ||||||
| 8 | 82 | 2173 | 26.5 (± 0.2) | 8 | Acroclisoides sinicus | 9 | 5.6 | 23.1 (1) | 1 (A. bifasciatus and T. mitsukurii) |
| Anastatus bifasciatus | 6 | 3.7 | 10.7 (1) | ||||||
| Trissolcus basalis | 5 | 3.1 | 17.9 (1) | ||||||
| Trissolcus mitsukurii | 142 | 87.7 | 53.7 ± 12.0 (6) | ||||||
| 9 | 19 | 513 | 27.0 (± 0.1) | 3 | Acroclisoides sinicus | 6 | 8.5 | 22.2 (1) | 1 (A. sinicus and T. mitsukurii) |
| Trissolcus mitsukurii | 65 | 91.5 | 80.6 ± 7.8 (3) | ||||||
| 10 | 5 | 137 | 27.4 (± 0.2) | 1 | Trissolcus kozlovi | 2 | 100 | 7.1 (1) | |
| 11 | 6 | 168 | 28.0 (± 0.1) | 2 | Anastatus bifasciatus | 2 | 7.7 | 7.4 (1) | |
| Trissolcus mitsukurii | 24 | 92.3 | 82.8 (1) |
| Index a | Species | Year | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | 2017 | 2018 | ||||||||||||||
| Index | χ2 | df | p-Value | Index | χ2 | df | p-Value | Index | χ2 | df | p-Value | |||||
| Discovery efficiency (%) | Trissolcus basalis | 7.7 | a | 4.02 | 2 | 0.1343 | n.a. | - | 0.79 | 1 | 0.3730 | n.a. | - | 1.17 | 1 | 0.2798 |
| Trissolcus mitsukurii | 30.8 | a | 17.3 | a | 13.2 | a | ||||||||||
| Anastatus bifasciatus | 7.4 | a | 4.8 | a | 16.1 | a | ||||||||||
| Exploitation efficiency (%) | Trissolcus basalis | 17.9 | a | 5.47 | 2 | 0.0648 | n.a. | - | 29.47 | 1 | <0.0001 | n.a. | - | 8.68 | 1 | 0.0032 |
| Trissolcus mitsukurii | 46.6 | a | 70.2 | a | 52.7 | a | ||||||||||
| Anastatus bifasciatus | 38.0 | a | 10.7 | b | 37.9 | b | ||||||||||
| Parasitoid impact (%) | Trissolcus basalis | 1.5 | b | 61.68 | 2 | <0.0001 | n.a. | - | 60.38 | 1 | <0.0001 | n.a. | - | 11.31 | 1 | 0.0008 |
| Trissolcus mitsukurii | 15.1 | a | 9.6 | a | 7.7 | a | ||||||||||
| Anastatus bifasciatus | 3.3 | b | 0.1 | b | 3.5 | b | ||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scaccini, D.; Falagiarda, M.; Tortorici, F.; Martinez-Sañudo, I.; Tirello, P.; Reyes-Domínguez, Y.; Gallmetzer, A.; Tavella, L.; Zandigiacomo, P.; Duso, C.; et al. An Insight into the Role of Trissolcus mitsukurii as Biological Control Agent of Halyomorpha halys in Northeastern Italy. Insects 2020, 11, 306. https://doi.org/10.3390/insects11050306
Scaccini D, Falagiarda M, Tortorici F, Martinez-Sañudo I, Tirello P, Reyes-Domínguez Y, Gallmetzer A, Tavella L, Zandigiacomo P, Duso C, et al. An Insight into the Role of Trissolcus mitsukurii as Biological Control Agent of Halyomorpha halys in Northeastern Italy. Insects. 2020; 11(5):306. https://doi.org/10.3390/insects11050306
Chicago/Turabian StyleScaccini, Davide, Martina Falagiarda, Francesco Tortorici, Isabel Martinez-Sañudo, Paola Tirello, Yazmid Reyes-Domínguez, Andreas Gallmetzer, Luciana Tavella, Pietro Zandigiacomo, Carlo Duso, and et al. 2020. "An Insight into the Role of Trissolcus mitsukurii as Biological Control Agent of Halyomorpha halys in Northeastern Italy" Insects 11, no. 5: 306. https://doi.org/10.3390/insects11050306
APA StyleScaccini, D., Falagiarda, M., Tortorici, F., Martinez-Sañudo, I., Tirello, P., Reyes-Domínguez, Y., Gallmetzer, A., Tavella, L., Zandigiacomo, P., Duso, C., & Pozzebon, A. (2020). An Insight into the Role of Trissolcus mitsukurii as Biological Control Agent of Halyomorpha halys in Northeastern Italy. Insects, 11(5), 306. https://doi.org/10.3390/insects11050306







