Screening of Helicoverpa armigera Mobilome Revealed Transposable Element Insertions in Insecticide Resistance Genes
Simple Summary
Abstract
1. Introduction
2. Material and Methods
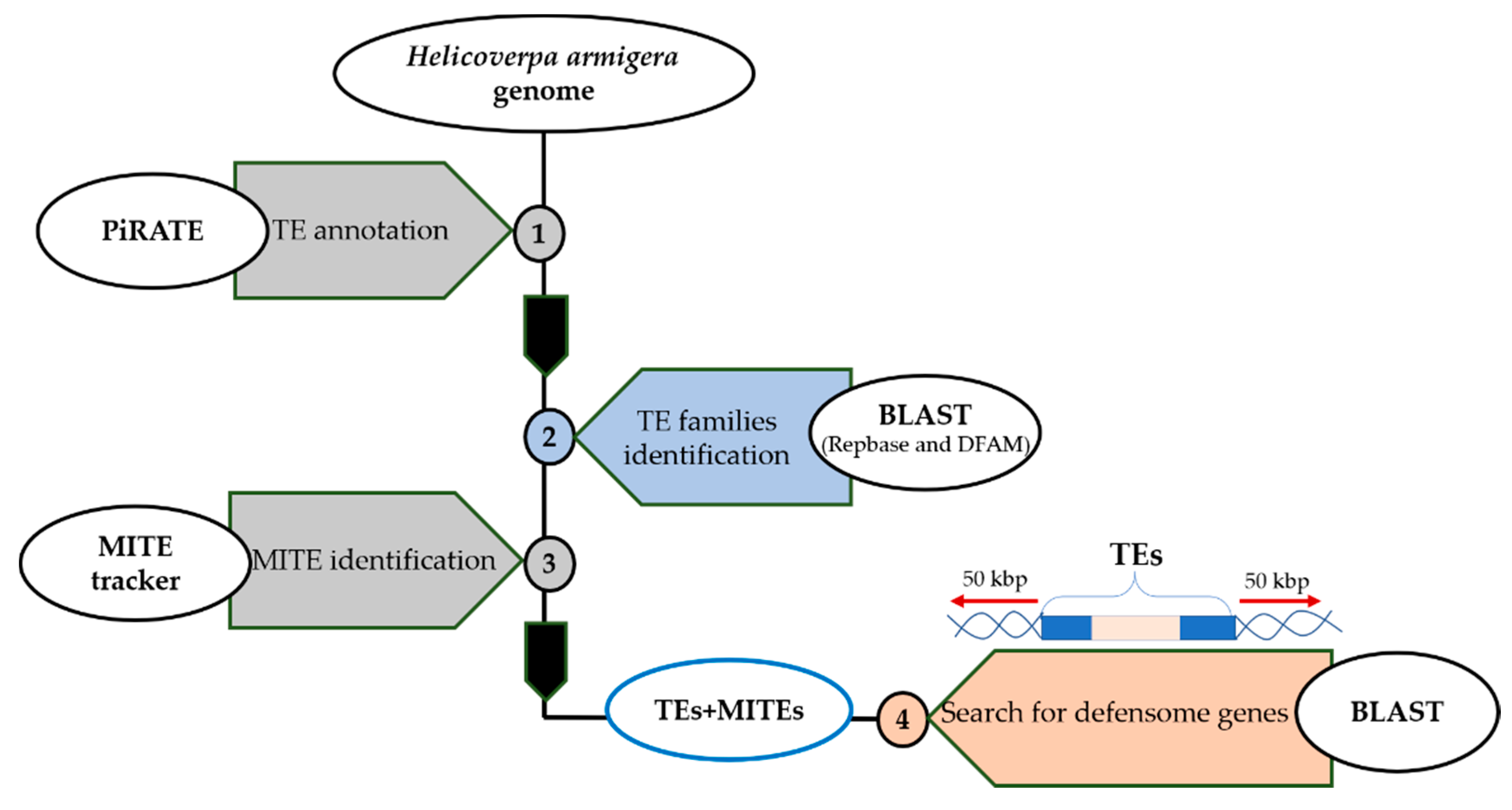
2.1. Mobilome Annotation
2.2. Search for TE Insertions in Defensome Genes
3. Results
3.1. TEs Annotation of the H. armigera Genome
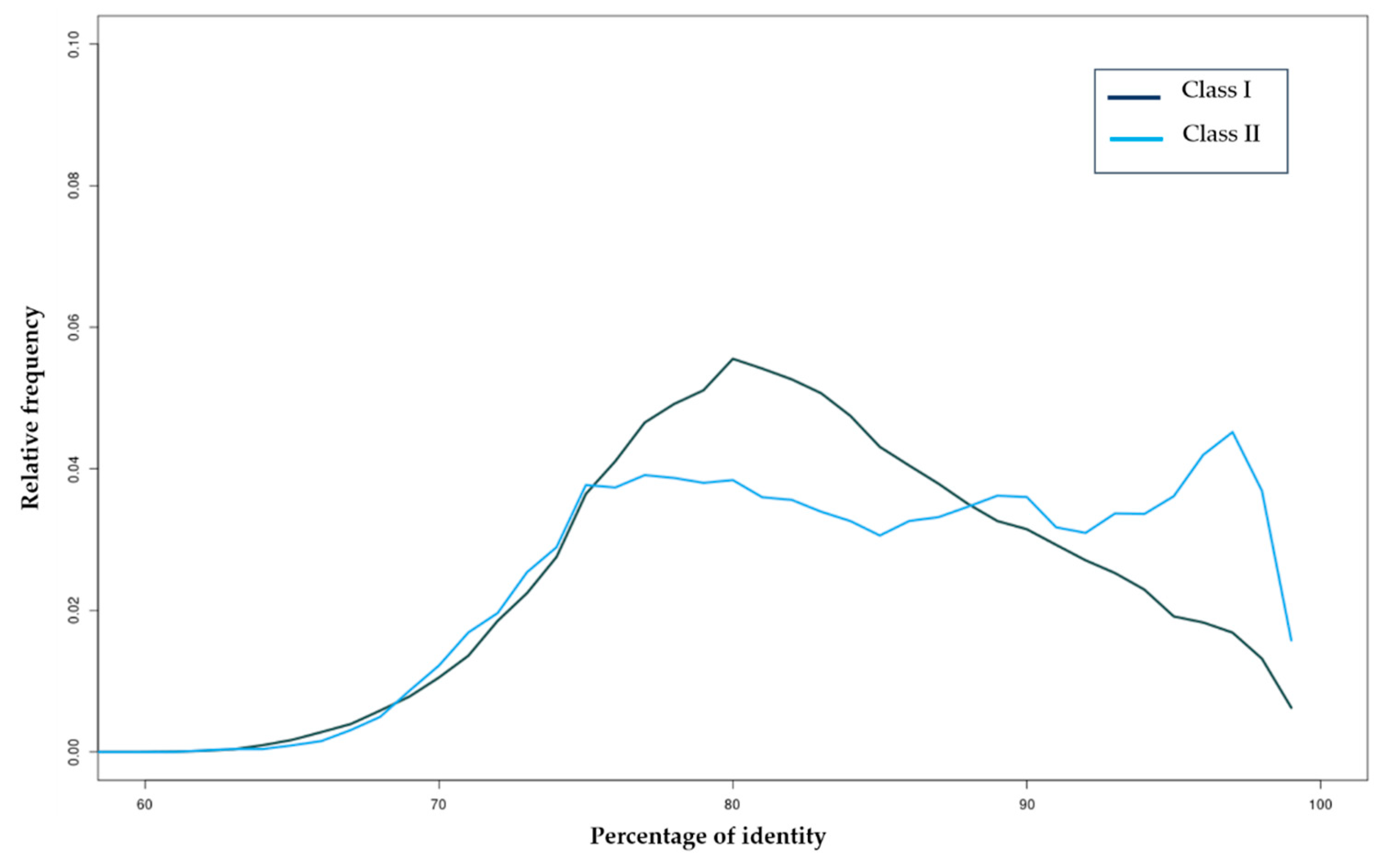
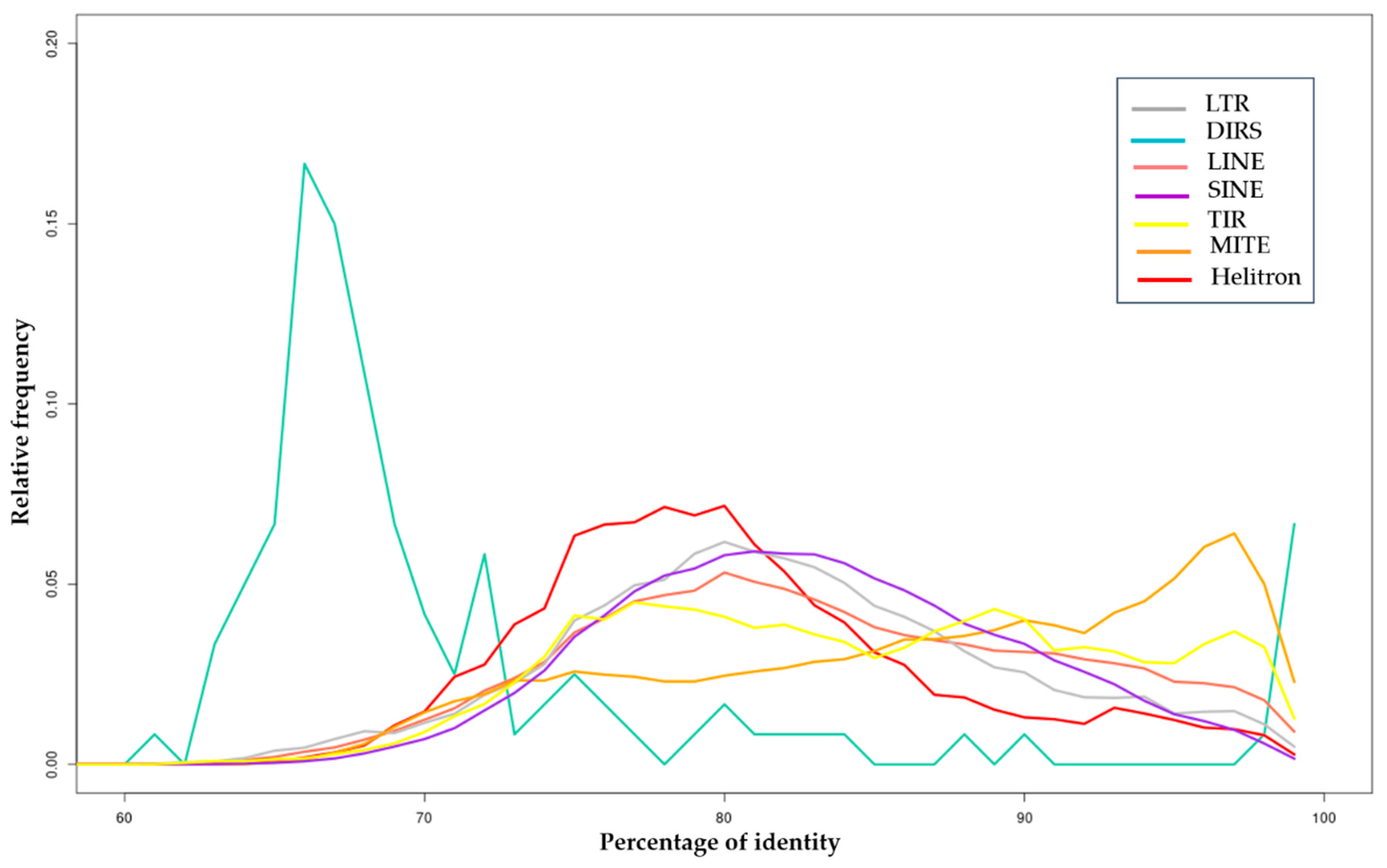
3.1.1. Class I Retrotransposons
3.1.2. Class II Transposons
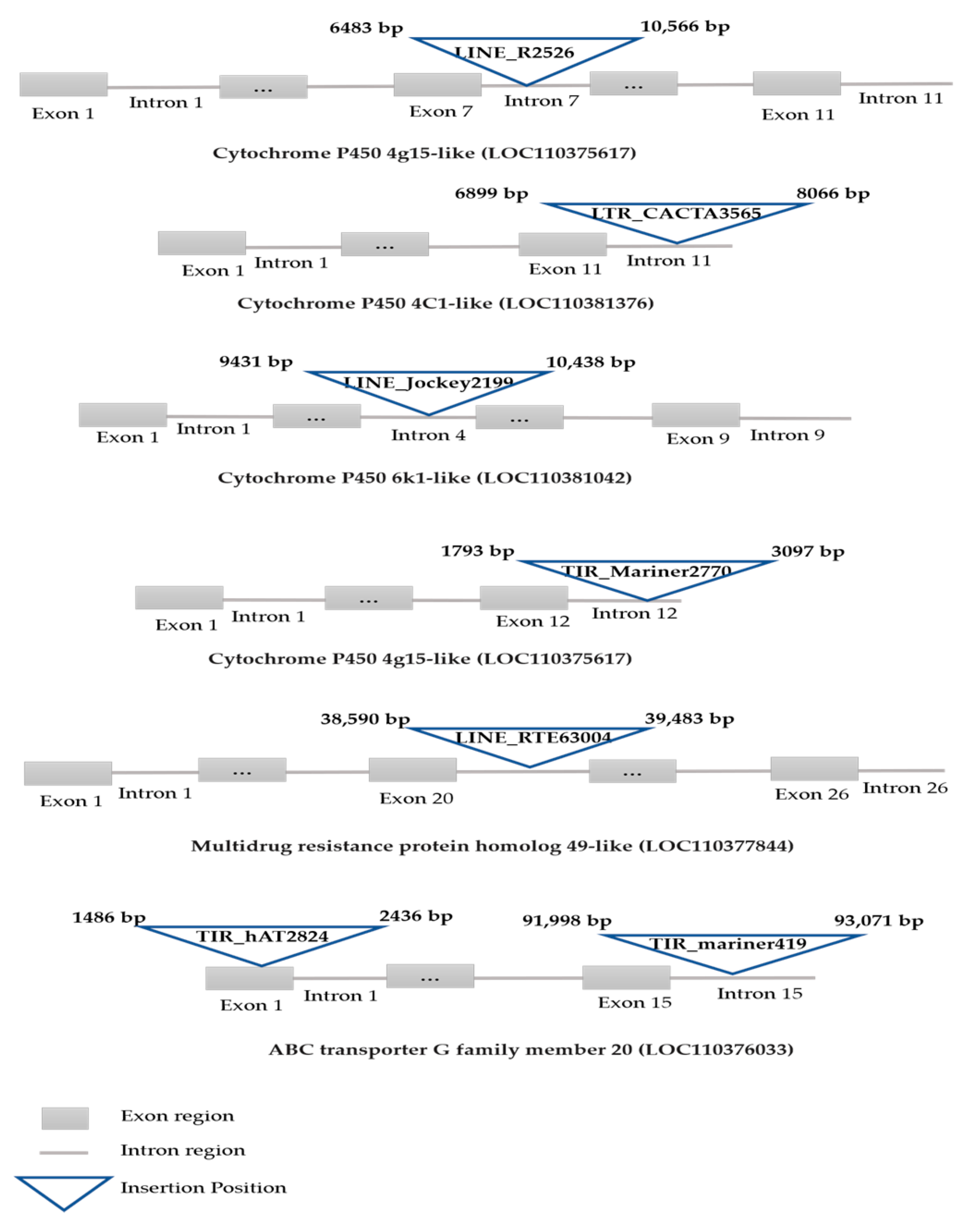
3.2. TE Insertions Scanning in Defensome Genes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Burgio, G.; Ravaglia, F.; Maini, S.; Bazzocchi, G.G.; Masetti, A.; Lanzoni, A. Mating Disruption of Helicoverpa armigera (Lepidoptera: Noctuidae) on Processing Tomato: First Applications in Northern Italy. Insects 2020, 11, 206. [Google Scholar] [CrossRef] [PubMed]
- Tembrock, L.R.; Timm, A.E.; Zink, F.A.; Gilligan, T.M. Phylogeography of the Recent Expansion of Helicoverpa armigera (Lepidoptera: Noctuidae) in South America and the Caribbean Basin. Ann. Entomol. Soc. Am. 2019, 112, 388–401. [Google Scholar] [CrossRef]
- Sarate, P.J.; Tamhane, V.A.; Kotkar, H.M.; Ratnakaran, N.; Susan, N.; Gupta, V.S.; Giri, A.P. Developmental and digestive flexibilities in the midgut of a polyphagous pest, the cotton bollworm, Helicoverpa armigera. J. Insect Sci. 2012, 12, 42. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.P.; Reigada, C.; Diniz, A.J.F.; Parra, J.R.P. Potential of Two Trichogrammatidae species for Helicoverpa armigera control. Neotrop. Entomol. 2019, 48, 966–973. [Google Scholar] [CrossRef]
- Ansari, M.S.; Moraiet, M.A.; Ahmad, S. Insecticides: Impact on the Environment and Human Health. In Environmental Deterioration and Human Health: Natural and Anthropogenic Determinants; Malik, A., Grohmann, E., Akhtar, R., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 99–123. [Google Scholar] [CrossRef]
- Srinivas, R.; Udikeri, S.S.; Jayalakshmi, S.K.; Sreeramulu, K. Identification of factors responsible for insecticide resistance in Helicoverpa armigera. Comp. Biochem. Physiol. 2004, 137, 261–269. [Google Scholar] [CrossRef]
- Sarwar, M.; Salman, M. Insecticides Resistance in Insect Pests or Vectors and Development of Novel Strategies to Combat Its Evolution. IJBBE 2015, 1, 344–351. [Google Scholar]
- Sampath, P.; Murukarthick, J.; Izzah, N.K.; Lee, J.; Choi, H.I.; Shirasawa, K.; Choi, B.S.; Liu, S.; Nou, I.S.; Yang, T.J. Genome-Wide Comparative Analysis of 20 Miniature Inverted-Repeat Transposable Element Families in Brassica rapa and B. oleracea. PLoS ONE 2014, 9, e94499. [Google Scholar] [CrossRef]
- De Marco, L.; Sassera, D.; Epis, S.; Mastrantonio, V.; Ferrari, M.; Ricci, I.; Comandatore, F.; Bandi, C.; Porretta, D.; Urbanelli, S. The choreography of the chemical defensome response to insecticide stress: Insights into the Anopheles stephensi transcriptome using RNA-Seq. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Dermauw, W.; van Leeuwen, T. The ABC gene family in arthropods: Comparative genomics and role in insecticide transport and resistance. Insect Biochem. Mol. Biol. 2014, 45, 89–110. [Google Scholar] [CrossRef]
- Su, H.; Gao, Y.; Liu, Y.; Li, X.; Liang, Y.; Dai, X.; Xu, Y.; Zhou, Y.; Wang, H. Comparative transcriptome profiling reveals candidate genes related to insecticide resistance of Glyphodes pyloalis. Bull. Entomol. Res. 2020, 110, 57–67. [Google Scholar] [CrossRef]
- Mérel, V.; Boulesteix, M.; Fablet, M.; Vieira, C. Transposable elements in Drosophila. Mob. DNA 2020, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, C.A. Transposable Element Content in Non-Model Insect Genomes. Master’s Thesis, Mississippi State University, Starkwell, MS, USA, 2020. [Google Scholar]
- Chénais, B.; Caruso, A.; Hiard, S.; Casse, N. The impact of transposable elements on eukaryotic genomes: From genome size increase to genetic adaptation to stressful environments. Gene 2012, 509, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Bourgeois, Y.; Boissinot, S. On the Population Dynamics of Junk: A Review on the Population Genomics of Transposable Elements. Genes 2019, 10, 419. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, C.E.W. Arms Race between Plants and Animals: Biotransformation System in Stress Ecology: Environmental Stress as Ecological Driving Force and Key Player in Evolution; Springer: New York, NY, USA, 2012; pp. 61–105. [Google Scholar]
- Le Goff, G.; Hilliou, F. Resistance evolution in Drosophila: The case of CYP6G1. Pest Manag. Sci. 2017, 73, 493–499. [Google Scholar] [CrossRef]
- Catania, F.; Kauer, M.O.; Daborn, P.J.; Yen, J.L.; French-Constant, R.H.; Schlötterer, C. World-wide survey of an Accord insertion and its association with DDT resistance in Drosophila melanogaster. Mol. Ecol. 2004, 13, 2491–2504. [Google Scholar] [CrossRef]
- Schlenke, T.A.; Begun, D.J. Strong selective sweep associated with a transposon insertion in Drosophila simulans. Proc. Natl. Acad. Sci. USA 2004, 101, 1626–1631. [Google Scholar] [CrossRef]
- Chen, S.; Li, X. Transposable elements are enriched within or in close proximity to xenobiotic-metabolizing cytochrome P450 genes. BCM Evol. Biol. 2007, 7, 46. [Google Scholar] [CrossRef]
- Finnegan, D.J. Eukaryotic transposable elements and genome evolution. Trends Genet. 1989, 5, 103–107. [Google Scholar] [CrossRef]
- Wicker, T.; Sabot, F.; Hua-Van, A.; Bennetzen, J.L.; Capy, P.; Chalhoub, B.; Flavell, A.; Leroy, P.; Morgante, M.; Panaud, O.; et al. An unified classification system for eukaryotic transposable elements. Nat. Rev. Genet. 2007, 8, 973–982. [Google Scholar] [CrossRef]
- Kapitonov, V.V.; Jurka, J. Rolling-Circle Transposons in Eukaryotes. Proc. Natl. Acad. Sci. USA 2001, 98, 8714–8719. [Google Scholar] [CrossRef]
- Pritham, E.J.; Putliwala, T.; Feschotte, C. Mavericks, a novel class of giant transposable elements widespread in eukaryotes and related to DNA viruses. Gene 2007, 390, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Kuang, H.; Padmanabhan, C.; Li, F.; Kamei, A.; Bhaskar, P.B.; Ouyang, S.; Jiang, J.; Robin Buell, C.; Baker, B. Identification of miniature inverted-repeat transposable elements (MITEs) and biogenesis of their SiRNAs in the Solanaceae: New functional implications for MITEs. Genome Res. 2009, 19, 42–56. [Google Scholar] [CrossRef] [PubMed]
- Schrader, L.; Schmitz, J. The impact of transposable elements in adaptive evolution. Mol. Ecol. 2019, 28, 1537–1549. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Torchiana, F.; Bombarely, A. Guideline for genome transposon annotation derived from evaluation of popular TE identification tools. Preprints 2020, 2020080275. [Google Scholar] [CrossRef]
- Berthelier, J.; Casse, N.; Daccord, N.; Jamilloux, V.; Saint-Jean, B.; Carrier, G. A transposable element annotation pipeline and expression analysis reveal potentially active elements in the microalga Tisochrysis lutea. BMC Genom. 2018, 19, 1–14. [Google Scholar] [CrossRef]
- Pearce, S.L.; Clarke, D.F.; East, P.D.; Elfekih, S.; Gordon, K.H.J.; Jermiin, L.S.; McGaughran, A.; Oakeshott, J.G.; Papanikolaou, A.; Perera, O.P.; et al. Genomic innovations, transcriptional plasticity and gene loss underlying the evolution and divergence of two highly polyphagous and invasive Helicoverpa pest species. BMC Biol. 2017, 15, 1–30. [Google Scholar] [CrossRef]
- Smit, A.F.A.; Hubley, R.; Green, P. Repeat Masker. Unpublished Data. Current Version: Open-4.09 (Dfam:3.0 only) 1996. Available online: http://www.repeatmasker.org (accessed on 15 April 2019).
- Eddy, S.R. Multiple alignment using hidden Markov models. ISMB 1996, 3, 114–120. [Google Scholar]
- Ellinghaus, D.; Kurtz, S.; Willhoeft, U. LTR harvest, an efficient and flexible software for de novo detection of LTR retrotransposons. BMC Bioinform. 2008, 9, 18. [Google Scholar] [CrossRef]
- Rho, M.; Tang, H. MGEScan-Non-LTR: Computational identification and classification of autonomous non-LTR retrotransposons in eukaryotic genomes. Nucleic Acids Res. 2009, 37, e143. [Google Scholar] [CrossRef]
- Wenke, T.; Döbel, T.; Sörensen, T.R.; Junghans, H.; Weisshaar, B.; Schmidta, T. Targeted Identification of Short Interspersed Nuclear Element Families Shows Their Widespread Existence and Extreme Heterogeneity in Plant Genomes. Plant Cell 2011, 23, 3117–3128. [Google Scholar] [CrossRef]
- Flutre, T.; Duprat, E.; Feuillet, C.; Quesneville, H. Considering Transposable Element Diversification in de Novo Annotation Approaches. PLoS ONE 2011, 6, e16526. [Google Scholar] [CrossRef] [PubMed]
- Price, A.L.; Jones, N.C.; Pevzner, P.A. De novo identification of repeat families in large genomes. Bioinformatics 2005, 21 (Suppl. 1), i351–i358. [Google Scholar] [CrossRef] [PubMed]
- Goubert, C.; Modolo, L.; Vieira, C.; Moro, C.V.; Mavingui, P.; Boulesteix, M. De Novo Assembly and Annotation of the Asian Tiger Mosquito (Aedes albopictus) Repeatome with DnaPipeTE from Raw Genomic Reads and Comparative Analysis with the Yellow Fever Mosquito (Aedes aegypti). GBE 2015, 7, 1192–1205. [Google Scholar] [CrossRef] [PubMed]
- Hoede, C.; Arnoux, S.; Moisset, M.; Chaumier, T.; Inizan, O.; Jamilloux, V.; Quesneville, H. PASTEC: An Automatic Transposable Element Classification Tool. PLoS ONE 2014, 9, e91929. [Google Scholar] [CrossRef]
- Quesneville, H.; Bergman, C.M.; Andrieu, O.; Autard, D.; Nouaud, D.; Ashburner, M.; Anxolabehere, D. Combined Evidence Annotation of Transposable Elements in Genome Sequences. PLoS Comput. Biol. 2005, 1, 0166–0175. [Google Scholar] [CrossRef]
- Daccord, N.; Celton, J.M.; Linsmith, G.; Becker, C.; Choisne, N.; Schijlen, E.; van de Geest, H.; Bianco, L.; Micheletti, D.; Velasco, R.; et al. High-quality de novo assembly of the apple genome and methylome dynamics of early fruit development. Nat. Genet. 2017, 49, 1099–1106. [Google Scholar] [CrossRef]
- Crescente, J.M.; Zavallo, D.; Helguera, M.; Vanzetti, L.S. MITE Tracker: An accurate approach to identify miniature inverted-repeat transposable elements in large genomes. BMC Bioinform. 2018, 19, 348. [Google Scholar] [CrossRef]
- Rognes, T.; Flouri, T.; Nichols, B.; Quince, C.; Mahé, F. VSEARCH: A versatile open source tool for metagenomics. PeerJ 2016, 4, 2584. [Google Scholar] [CrossRef]
- Goerner-Potvin, P.; Bourque, G. Computational tools to unmask transposable elements. Nat. Rev. Genet. 2018, 19, 688–704. [Google Scholar] [CrossRef]
- D’Alençon, E.; Sezutsu, H.; Legeai, F.; Permal, E.; Bernard-Samain, S.; Gimenez, S.; Gagneur, C.; Cousserans, F.; Shimomura, M.; Brun-Barale, A.; et al. Extensive synteny conservation of holocentric chromosomes in lepidoptera despite high rates of local genome rearrangements. Proc. Natl. Acad. Sci. USA 2010, 107, 7680–7685. [Google Scholar] [CrossRef]
- Sun, Z.C.; Wu, M.; Miller, T.A.; Han, Z.J. PiggyBac-like elements in cotton bollworm, Helicoverpa armigera (Hübner). Insect Mol. Biol. 2008, 17, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Miller, T.A.; Park, Y. Identification of mariner-like elements belonging to the Cecropia subfamily in two closely related Helicoverpa species. Insect Sci. 2011, 18, 619–628. [Google Scholar] [CrossRef]
- Zhu, W.; Yu, R.; Wu, H.; Zhang, X.; Liu, Y.; Zhu, K.Y.; Zhang, J.; Ma, E. Identification and characterization of two CYP9A genes associated with pyrethroid detoxification in Locusta migratoria. Pestic. Biochem. Physiol. 2016, 132, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Cheng, T.; Wu, Y.; Yang, X.; Feng, Q.; Mita, K. Genome-wide patterns of copy number variations in Spodoptera litura. Genomics 2019, 111, 1231–1238. [Google Scholar] [CrossRef]
- Langfield, K.L. Characterization of Neonicotinoid Resistance in the Cotton Aphid, Aphis gossypii from Australian Cotton. Ph.D. Thesis, University of Technology Sydney, Sydney, Australia, April 2017. [Google Scholar]
- Kim, J.I.; Kwon, M.; Kim, G.H.; Kim, S.Y.; Lee, S.H. Two mutations in nAChR beta subunit is associated with imidacloprid resistance in the Aphis gossypii. J. Asia Pac. Entomol. 2015, 18, 291–296. [Google Scholar] [CrossRef]
- Greene, B.; Wako’, R.; Hake, S. Mutator Insertions in an Intron of the Maize Knotted1 Gene Result in Dominant Suppressible Mutations. Genetics 1994, 138, 1275–1285. [Google Scholar]
| Class | Order | Superfamily | Total Sequences 1 | Full-Length Copies 2 | TE Percentage | Copy Number 3 |
|---|---|---|---|---|---|---|
| Class I | LTR | Gypsy | 241 | 181 | 14,876 | |
| Bel pao | 155 | 97 | 1792 | |||
| Copia | 77 | 67 | 223 | |||
| Total LTR | 473 | 345 | 5.55% | 16,891 | ||
| DIRS | DIRS | 45 | 38 | 149 | ||
| Total DIRS | 45 | 38 | 0.53% | 149 | ||
| LINE | Jockey | 716 | 480 | 44,432 | ||
| RTE | 604 | 543 | 46,458 | |||
| I | 172 | 93 | 16,520 | |||
| R2 | 169 | 101 | 10,614 | |||
| Total LINE | 1661 | 1217 | 19.49% | 118,024 | ||
| SINE | tRNA | 1797 | 1120 | 51,581 | ||
| 5S | 4 | - | 0 | |||
| Total SINE | 1801 | 1120 | 21.13% | 51,581 | ||
| Total Class I | 3980 | 2720 | 46.71% | 186,645 | ||
| Class II Subclass I | TIR | hAT | 120 | 105 | 6267 | |
| Mariner | 106 | 74 | 2284 | |||
| Piggybac | 41 | 32 | 6670 | |||
| Transib | 40 | 32 | 188 | |||
| P | 15 | 12 | 6103 | |||
| CACTA | 14 | 11 | 201 | |||
| Mutator | 6 | 4 | 477 | |||
| PIF_harbinger | 6 | 5 | 90 | |||
| Merlin | 1 | 1 | 30 | |||
| Total TIR | 349 | 276 | 4.09% | 22,310 | ||
| Class II Subclass II | Crypton | Crypton | 1 | 1 | 3 | |
| Helitron | Helitron | 4 | 4 | 595 | ||
| Maverick | Maverick | 2 | 2 | 14 | ||
| MITEs | MITEs | 4185 | 130 | 49.11% | 26,565 | |
| Total classII | 4541 | 413 | 53.29% | 49,487 | ||
| Total TEs | 8521 | 3133 | 100 | 236,132 | ||
| Superfamily | TSD | Number of MITEs | MITE Length (bp) | TIR Length (bp) |
|---|---|---|---|---|
| Tc1/mariner | TA | 1817 | 50–360 | 10–21 |
| PIF-Harbinger | TWA | 1368 | 55–685 | 15–32 |
| CACTA | 2–3 bp | 250 | 78–775 | 10–26 |
| Piggybac | TTAA | 93 | 50–800 | 15–31 |
| hAT | TNNNNA | 20 | 56–260 | 17–29 |
| Transib | CNNNG | 16 | 83–386 | 13–27 |
| Maverick | 6 bp | 6 | 50–800 | 10–24 |
| Other | - | 615 | 50–800 | 11–35 |
| Gene Family | Gene Name | Gene Length (bp) | Inserted Element Name | TE Length (bp) | Insertion Position |
|---|---|---|---|---|---|
| Cytochrome P450 (CYP450) | 4g15-like (LOC110375617) | 14,974 | LINE_R2_526 | 4083 | 6483–10,566 |
| 4C1-like (LOC113006340) | 39,707 | LINE_RTE17512 | 620 | 5655–6274 | |
| LINE_RTE84107 | 740 | 7021–7763 | |||
| 4C1-like (LOC110381376) | 8332 | LTR_CACTA3565 | 1179 | 6899–8066 | |
| 6k1-like (LOC110381042) | 15,958 | LINE_jockey2199 | 1629 | 9431–10,438 | |
| Glutathione S-transferase (GST) | GST 1-like (LOC110371343) | 4001 | TIR_Mariner2770 | 1304 | 1793–3097 |
| ATP binding cassette (ABC) transporter | ABC-G member 20 (LOC110376033) | 96,146 | TIR_Mariner419 | 1085 | 91,998–93,071 |
| TIR_hAT2824 | 951 | 1486–2436 | |||
| ABC-C1 (Multidrug resistance protein homolog 49-like MRP1) (LOC110377844) | 48,302 | LINE_RTE63004 | 894 | 38,590–39,483 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
KLAI, K.; CHÉNAIS, B.; ZIDI, M.; DJEBBI, S.; CARUSO, A.; DENIS, F.; CONFAIS, J.; BADAWI, M.; CASSE, N.; MEZGHANI KHEMAKHEM, M. Screening of Helicoverpa armigera Mobilome Revealed Transposable Element Insertions in Insecticide Resistance Genes. Insects 2020, 11, 879. https://doi.org/10.3390/insects11120879
KLAI K, CHÉNAIS B, ZIDI M, DJEBBI S, CARUSO A, DENIS F, CONFAIS J, BADAWI M, CASSE N, MEZGHANI KHEMAKHEM M. Screening of Helicoverpa armigera Mobilome Revealed Transposable Element Insertions in Insecticide Resistance Genes. Insects. 2020; 11(12):879. https://doi.org/10.3390/insects11120879
Chicago/Turabian StyleKLAI, Khouloud, Benoît CHÉNAIS, Marwa ZIDI, Salma DJEBBI, Aurore CARUSO, Françoise DENIS, Johann CONFAIS, Myriam BADAWI, Nathalie CASSE, and Maha MEZGHANI KHEMAKHEM. 2020. "Screening of Helicoverpa armigera Mobilome Revealed Transposable Element Insertions in Insecticide Resistance Genes" Insects 11, no. 12: 879. https://doi.org/10.3390/insects11120879
APA StyleKLAI, K., CHÉNAIS, B., ZIDI, M., DJEBBI, S., CARUSO, A., DENIS, F., CONFAIS, J., BADAWI, M., CASSE, N., & MEZGHANI KHEMAKHEM, M. (2020). Screening of Helicoverpa armigera Mobilome Revealed Transposable Element Insertions in Insecticide Resistance Genes. Insects, 11(12), 879. https://doi.org/10.3390/insects11120879








