Strategic Approach, Advances, and Challenges in the Development and Application of the SIT for Area-Wide Control of Aedes albopictus Mosquitoes in Reunion Island
Abstract
Simple Summary
Abstract
1. Introduction
2. Opportunities for Establishing a Viable SIT Strategy
3. Strategic Objectives Proposed for Establishing the SIT Feasibility Program in Reunion Island
4. Challenges in the Development and Pilot Testing of SIT on Reunion Island—10 Years of Research and Development
5. Pre-Release Entomological Field Studies
5.1. Field Site Selection and Characterization
5.2. Pre-Release Entomological Surveys
5.3. Baseline Fecundity and Fertility in Wild Ae. albopictus Populations
5.4. Seasonal Monitoring of Adult Ae. albopictus Population Density and Behavior
6. Colonization and Mass-Rearing of Local Strain of Aedes albopictus
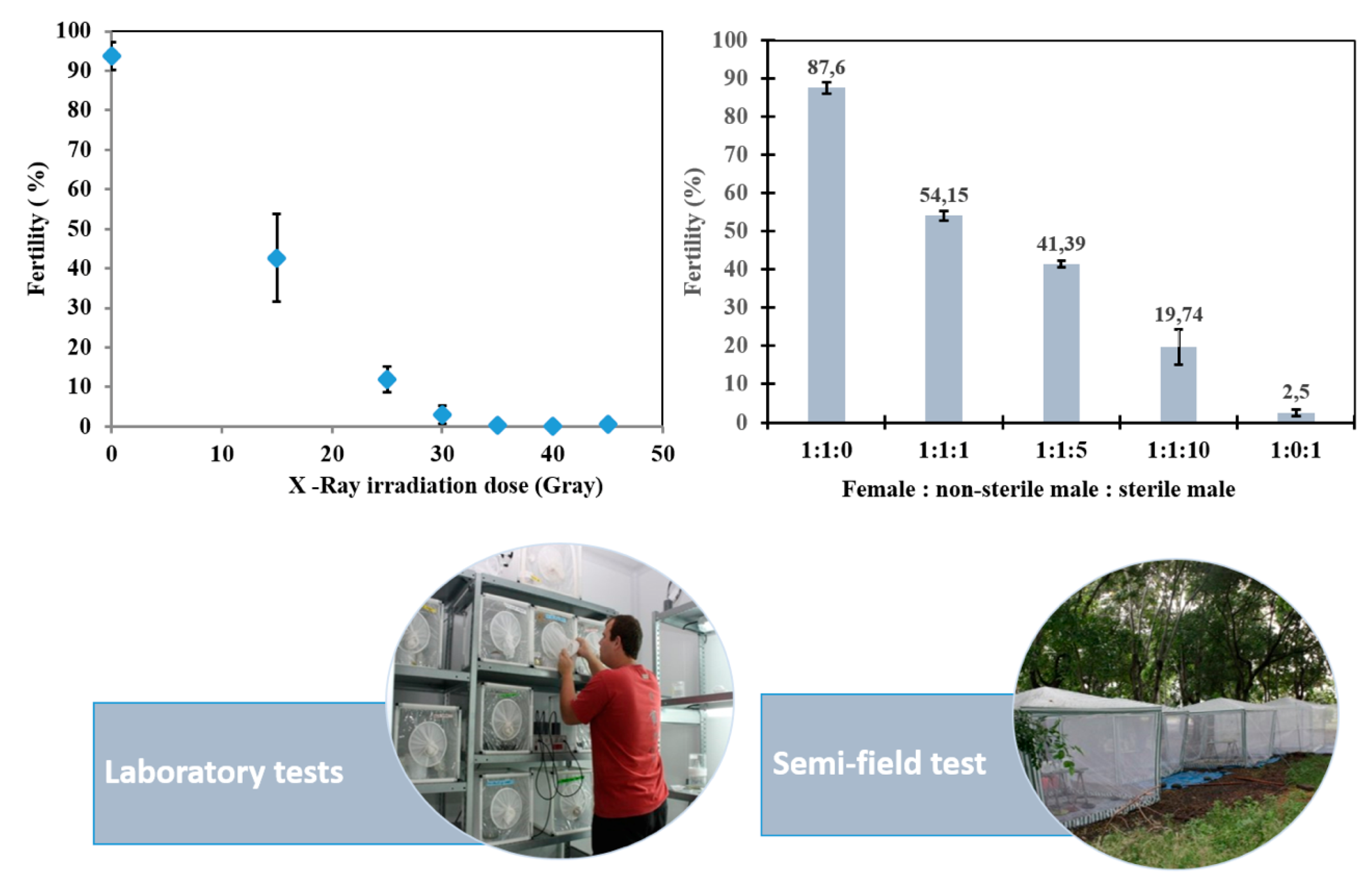
6.1. Sex Separation and Irradiation of Males of Laboratory-Reared Aedes albopictus
6.2. Effect of Irradiation on Ae. albopictus Male Quality
6.3. Assessing the Field Behavior and Mating Capacity of Sterile Ae. albopictus Males
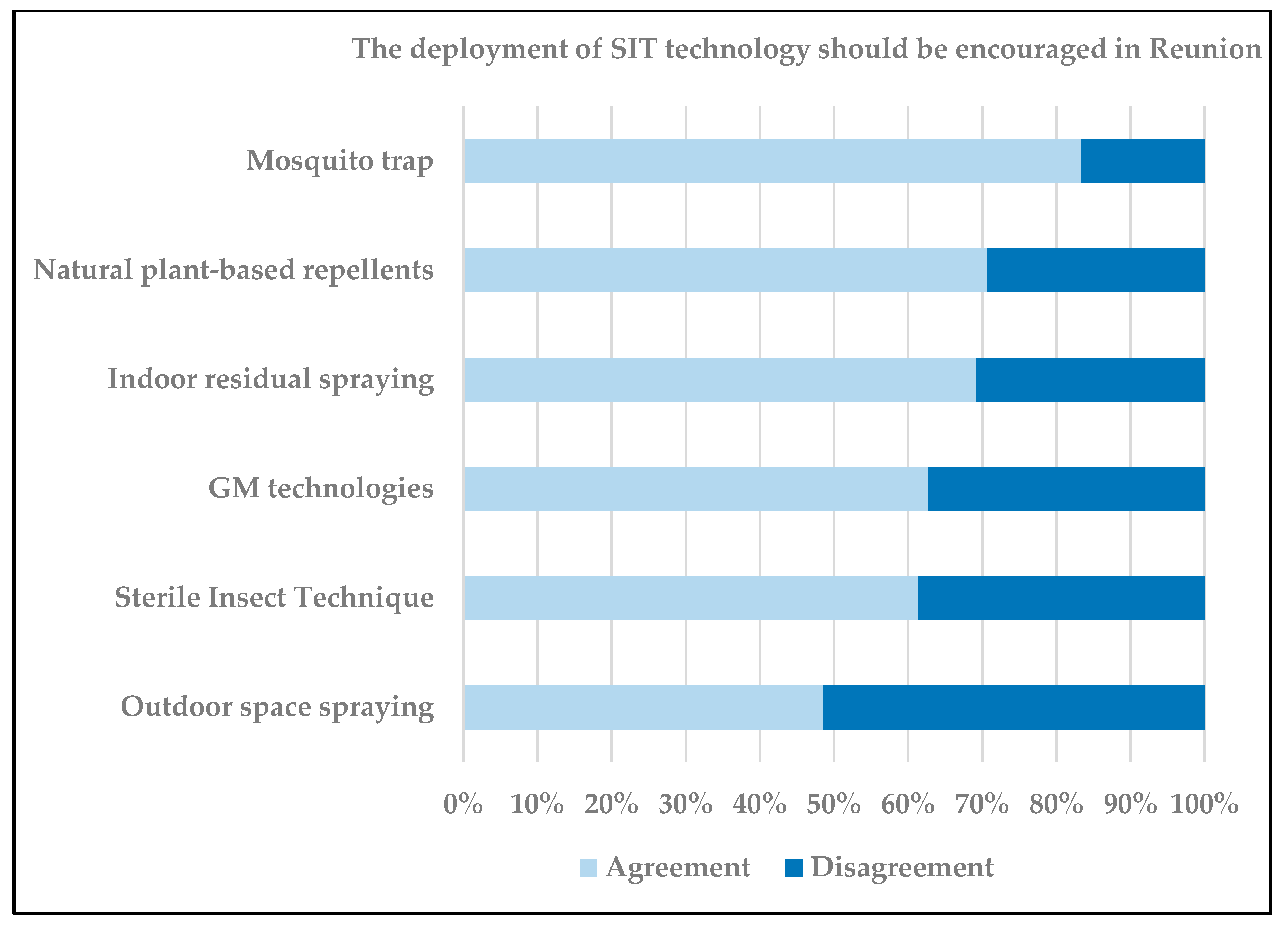
7. Communication, Social, and Regulatory Challenges for SIT Testing in Reunion Island
8. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benedict, M.Q.; Levine, R.S.; Hawley, W.A.; Lounibos, L.P. Spread of the tiger: Global risk of invasion by the mosquito Aedes albopictus. Vector Borne Zoonotic Dis. 2007, 7, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef]
- Messina, J.P.; Brady, O.J.; Golding, N.; Kraemer, M.U.G.; Wint, G.R.W.; Ray, S.E.; Pigott, D.M.; Shearer, F.M.; Johnson, K.; Earl, L.; et al. The current and future global distribution and population at risk of dengue. Nat. Microbiol. 2019, 4, 1508–1515. [Google Scholar] [CrossRef] [PubMed]
- Perrau, J.; Catteau, C.; Michault, A.; Parain, A.; Favier, F. Fin 2006, 300000 personnes avaient été atteintes par le chikungunya. Economie de la Réunion 2007, 129, 16–17. [Google Scholar]
- Renault, P.; Solet, J.L.; Sissoko, D.; Balleydier, E.; Larrieu, S.; Filleul, L.; Lassalle, C.; Thiria, J.; Rachou, E.; de Valk, H.; et al. A major epidemic of chikungunya virus infection on Réunion Island, France, 2005–2006. Am. J. Trop. Med. Hyg. 2007, 77, 727–731. [Google Scholar] [CrossRef] [PubMed]
- CIRE, Point épidémiologique-No. 16. Saint Denis, La Reunion: Cire océan Indien. March 2018, 1–7. Available online: http://www.reunion.gouv.fr/epidemie-de-dengue-a-la-reunion-la-circulation-du-a5079.html (accessed on 28 March 2019).
- Bousses, P.; Dehecq, J.S.; Brengues, C.; Fontenille, D. Inventaire actualisé des moustiques (Diptera: Culicidae) de l’île de La Réunion, océan Indien. Bull. Soc. Pathol. Exot. 2013, 106, 113–125. [Google Scholar] [CrossRef]
- Delatte, H.; Dehecq, J.S.; Thiria, J.; Domerg, C.; Paupy, C.; Fontenille, D. Geographic distribution and developmental sites of Aedes albopictus (Diptera: Culicidae) during a chikungunya epidemic event. Vector Borne Zoonotic Dis. 2008, 8, 25–34. [Google Scholar] [CrossRef]
- Schuffenecker, I.; Iteman, I.; Michault, A.; Murri, S.; Frangeul, L.; Vaney, M.-C.; Lavenir, R.; Pardigon, N.; Reynes, J.-M.; Pettinelli, F.; et al. Genome microevolution of chikungunya viruses causing the Indian Ocean outbreak. PLoS Med. 2006, 3, e263. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, M.; Moutailler, S.; Coudrier, D.; Rousseaux, C.; Khun, H.; Huerre, M.; Thiria, J.; Dehecq, J.-S.; Fontenille, D.; Schuffenecker, I.; et al. Two Chikungunya Isolates from the Outbreak of La Reunion (IndianOcean) Exhibit Different Patterns of Infection in the Mosquito, Aedes albopictus. PLoS ONE 2007, 2, e1168. [Google Scholar] [CrossRef] [PubMed]
- Salvan, M.; Mouchet, J. Aedes albopictus et Aedes aegypti à l’Ile de La Réunion. Ann. Soc. Belg. Méd. Trop. 1994, 74, 323–326. [Google Scholar] [PubMed]
- Delatte, H.; Paupy, C.; Dehecq, J.S.; Thiria, J.; Failloux, A.B.; Fontenille, D. Aedes albopictus, vector of chikungunya and dengue viruses in Reunion Island: Biology and control. Parasite (Paris, France) 2008, 15, 3–13. [Google Scholar] [CrossRef]
- Bagny, L.; Delatte, H.; Quilici, S.; Fontenille, D. Progressive decrease in Aedes aegypti distribution in Reunion Island since the 1900s. Med. Entomol. 2009, 46, 1541–1545. [Google Scholar] [CrossRef]
- Bagny, L.; Delatte, H.; Juliano, S.A.; Fontenille, D.; Quilici, S. Ecological interactions in Aedes species on Reunion Island. Med. Vet. Entomol. 2012, 27, 387–397. [Google Scholar] [CrossRef]
- Delatte, H.; Toty, C.; Boyer, S.; Bouetard, A.; Bastien, F.; Fontenille, D. Evidence of habitat structuring Aedes albopictus populations in Réunion Island. PLoS Negl. Trop. Dis. 2013, 7, e2111. [Google Scholar] [CrossRef] [PubMed]
- Tantely, M.L.; Tortosa, P.; Alout, H.; Berticat, C.; Berthomieu, A.; Rutee, A.H.; Dehecq, J.-S.; Makoundou, P.; Labbé, P.; Pasteur, N. Insecticide resistance in Culex pipiens quinquefasciatus and Aedes albopictus mosquitoes from La Réunion Island. Insect Biochem. Mol. Biol. 2010, 40, 317–324. [Google Scholar] [CrossRef]
- Wiktelius, S.; Chiverton, P.A.; Meguenni, H.; Bennaceur, M.; Ghezal, F.; Umeh, E.-D.; Egwuatu, R.; Minja, E.; Makusi, R.; Tukahirwa, E.; et al. Effects of insecticides on non-target organisms in African agroecosystems: A case for establishing regional testing programmes. Agric. Ecosyst. Environ. 1999, 75, 121–131. [Google Scholar] [CrossRef]
- Biondi, A.; Mommaerts, V.; Smagghe, G.; Viñuela, E.; Zappalà, L.; Desneux, N. The non-target impact of spinosyns on beneficial arthropods. Pest Manag. Sci. 2012, 68, 1523–1536. [Google Scholar] [CrossRef]
- Thomas, D.D.; Donnelly, C.A.; Wood, R.J.; Alphey, L.S. Insect population control using a dominant, repressible, lethal genetic system. Science 2000, 287, 2474–2476. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, D.O.; McKemey, A.R.; Garziera, L.; Lacroix, R.; Donnelly, C.A.; Alphey, L.; Malavasi, A.; Capurro, M.L. Suppression of a field population of Aedes aegypti in Brazil by sustained release of transgenic male mosquitoes. PLoS Negl. Trop. Dis. 2015, 9, e0003864. [Google Scholar] [CrossRef] [PubMed]
- Alphey, N.; Coleman, P.G.; Donnelly, C.A.; Alphey, L. Managing insecticide resistance by mass release of engineered insects. J. Econ. Entomol. 2007, 100, 1642–1649. [Google Scholar] [CrossRef]
- Sinkins, S.P. Wolbachia and cytoplasmic incompatibility in mosquitoes. Insect Biochem. Mol. Biol. 2004, 34, 723–729. [Google Scholar] [CrossRef]
- O’Connor, L.; Plichart, C.; Sang, A.C.; Brelsfoard, C.L.; Bossin, H.C.; Dobson, S.L. Open Release of Male Mosquitoes Infected with a Wolbachia Biopesticide: Field Performance and Infection Containment. PLoS Negl. Trop. Dis. 2012, 6, e1797. [Google Scholar] [CrossRef]
- Knipling, E.F. Possibilities of insect control or eradication through the use of sexually sterile males. J. Econ. Entomol. 1955, 48, 459–469. [Google Scholar] [CrossRef]
- Knipling, E.F. Sterile-male method of population control. Science 1959, 130, 902–904. [Google Scholar] [CrossRef]
- Alphey, L.; McKemey, A.; Nimmo, D.; Oviedo, M.N.; Renaud Lacroix, R.; Matzen, K.; Beech, C. Review—Genetic control of Aedes mosquitoes. Pathog. Glob. Health 2013, 107, 170–179. [Google Scholar] [CrossRef]
- Bourtzis, K.; Lees, R.S.; Hendrichs, J.; Vreysen, M.J. More than one rabbit out of the hat: Radiation, transgenic and symbiont-based approaches for sustainable management of mosquito and tsetse fly populations. Acta Trop. 2016, 157, 115–130. [Google Scholar] [CrossRef]
- HCB Scientific Committee. Scientific Opinion of the High Council for Biotechnology concerning use of genetically modified mosquitoes for vector control in response to the referral of 12 October 2015 (Ref. HCB-2017.06.07). Paris HCB 2017, 141. Available online: http://www.hautconseildesbiotechnologies.fr (accessed on 4 November 2017).
- Calkins, C.O.; Parker, A.G. Sterile insect quality. In Sterile Insect Technique Principles and Practice in Area-Wide Integrated Pest Management; Dyck, V.A., Hendrichs, J., Robinson, A.S., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 269–296. [Google Scholar] [CrossRef]
- Enkerlin, E.R. Impact of fruit fly control programmes using the sterile insect technique. In Sterile Insect Technique: Principles and Practice in Area-Wide Integrated Pest Management; : Dyck, V.A., Hendrichs, J., Robinson, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 651–676. [Google Scholar] [CrossRef]
- Hendrichs, J.; Vreysen, M.J.B.; Enkerlin, W.R.; Cayol, J.P. Strategic options in using sterile insects for area-wide integrated pest management. In Sterile Insect Technique; Dyck, V.A., Hendrichs, J., Robinson, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 563–600. [Google Scholar] [CrossRef]
- Klassen, W.; Curtis, C.F. History of the sterile insect technique. In Sterile Insect Technique; Dyck, V.A., Hendrichs, J., Robinson, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 3–36. [Google Scholar] [CrossRef]
- Dame, D.A.; Curtis, C.F.; Benedict, M.Q.; Robinson, A.S.; Knols, B.G. Historical applications of induced sterilisation in field populations of mosquitoes. Malar. J. 2009, 8 (Suppl. 2), S2. [Google Scholar] [CrossRef]
- Weidhaas, D.E.; Schmidt, C.H.; Seabrook, E.L. Field studies on the release of sterile males for the control of Anopheles quadrimaculatus. Mosq. News 1962, 22, 283–290. [Google Scholar]
- Weidhaas, D.E.; Breeland, S.G.; Lofgren, C.S.; Dame, D.A.; Kaiser, R. Release of chemosterilized males for the control of Anopheles albimanus in El Salvador. IV. Dynamics of the test population. Am. J. Trop. Med. Hyg. 1974, 23, 298–308. [Google Scholar] [CrossRef]
- Lees, R.S.; Gilles, J.R.; Hendrichs, J.; Vreysen, M.J.; Bourtzis, K. Back to the future: The sterile insect technique against mosquito disease vectors. Curr. Opin. Insect Sci. 2015, 10, 156–162. [Google Scholar] [CrossRef]
- Bellini, R.; Calvitti, M.; Medici, A.; Carrieri, M.; Celli, G.; Maini, S. Use of the sterile insect technique against Aedes albopictus in Italy: First results of a pilot trial. In Area-wide Control of Insect Pests: From Research to Field Implementation; Vreysen, M.J.B., Robinson, A.S., Hendrichs, J., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 505–515. ISBN 1402060599, 9781402060595. [Google Scholar]
- Bellini, R.; Medici, A.; Puggioli, A.; Balestrino, F.; Carrieri, M. Pilot field trials with Aedes albopictus irradiated sterile males in Italian urban areas. J. Med. Entomol. 2013, 50, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Zhang, D.; Li, Y.; Yang, C.; Wu, Y.; Liang, X.; Liang, Y.; Pan, X.; Hu, L.; Sun, Q.; et al. Incompatible and sterile insect techniques combined eliminate mosquitoes. Nature 2019, 572, 56–61. [Google Scholar] [CrossRef]
- Bouyer, J.; Yamada, H.; Pereira, R.; Bourtzis, K.; Vreysen, M.J.B. Phased conditional approach for mosquito management using sterile insect technique. Trends Parasitol. 2020, 36, 325–336. [Google Scholar] [CrossRef]
- WHO/TDR, FAO/IAEA. Guidance Framework for Testing the Sterile Insect Technique (SIT) as a Vector Control Tool against Aedes-Borne Diseases. In WHO Technical Report; WHO: Geneva, Switzerland, 2019; p. 117. ISBN 978-92-4-000237-1. [Google Scholar]
- ANSES. Agence Nationale de Sécurité Sanitaire de L’alimentation, de L’environnement et du Travail. Avis Saisine n° «2018-SA-0136» Relatif à l’«Analyse de la Stratégie de Lutte Anti-Vectorielle (LAV) Mise en Œuvre à La Réunion Depuis 2017»; Maisons-Alfort, France, 2018; Available online: https://www.anses.fr/fr/system/files/VECTEURS2018SA0136.pdf (accessed on 11 December 2018).
- Boyer, S.; Foray, C.; Dehecq, J.S. Spatial and temporal heterogeneities of Aedes albopictus density in La Reunion Island: Rise and weakness of entomological indices. PLoS ONE 2014, 9, e91170. [Google Scholar] [CrossRef] [PubMed]
- Paupy, C.; Girod, R.; Salvan, M.; Rodhain, F.; Failloux, A.B. Population structure of Aedes albopictus from La Réunion Island (Indian Ocean) with respect to susceptibility to a dengue virus. Heredity 2001, 87, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Delatte, H.; Bagny, L.; Brengue, C.; Bouetard, A.; Paupy, C.; Fontenille, D. The invaders: Phylogeography of dengue and chikungunya viruses Aedes vectors, on the South West islands of the Indian Ocean. Infect. Genet. Evol. 2011, 11, 1769–1781. [Google Scholar] [CrossRef]
- Delatte, H.; Gimonneau, G.; Triboire, A.; Fontenille, D. Influence of temperature on immature development, survival, longevity, fecundity, and gonotrophic cycles of Aedes albopictus, vector of chikungunya and dengue in the Indian Ocean. J. Med. Entomol. 2009, 46, 33–41. [Google Scholar] [CrossRef]
- Boyer, S.; Gilles, J.; Merancienne, D.; Lemperiere, G.; Fontenille, D. Sexual performance of male Aedes albopictus. Med. Vet. Entomol. 2011, 25, 454–459. [Google Scholar] [CrossRef]
- Damiens, D.; Lebon, C.; Wilkinson, D.A.; Dijoux-Millet, D.; Le Goff, G.; Bheecarry, A.; Gouagna, L.C. Cross-mating compatibility and competitiveness among Aedes albopictus strains from distinct geographic origins-implications for future application of SIT programs in the South West Indian Ocean Islands. PLoS ONE 2016, 11, e0163788. [Google Scholar] [CrossRef]
- Lebon, C. Study of Reproductive Behavior in Aedes albopictus (Diptera: Culicidae); Report, Master Studies II-Sciences-Technologies-Health-Ecology et Ethology; University of Saint-Etienne: Saint-Etienne, France, 2011; 34p. (In French) [Google Scholar]
- Boyer, S.; Toty, C.; Jacquet, M.; Lempérière, G.; Fontenille, D. Evidence of multiple inseminations in the field in Aedes albopictus. PLoS ONE 2012, 7, e42040. [Google Scholar] [CrossRef]
- Boyer, S.; Maillot, L.; Gouagna, L.C.; Fontenille, D.; Chadee, D.; Lempérière, G. Diel activity patterns of male Aedes albopictus in the laboratory. J. Am. Mosq.Control Assoc. 2013, 29, 74–77. [Google Scholar] [CrossRef]
- Oliva, C.F.; Damiens, D.; Vreysen, M.J.; Lemperière, G.; Gilles, J. Reproductive strategies of Aedes albopictus (Diptera: Culicidae) and implications for the sterile insect technique. PLoS ONE 2013, 8, e78884. [Google Scholar] [CrossRef]
- Oliva, C.F.; Maier, M.J.; Gilles, J.; Jacquet, M.; Lemperiere, G.; Quilici, S.; Vreysen, M.J.B.; Schooneman, F.; Chadee, D.D.; Boyer, S. Effects of irradiation, presence of females, and sugar supply on the longevity of sterile males Aedes albopictus (Skuse) under semi-field conditions on Reunion Island. Acta Trop. 2013, 125, 287–293. [Google Scholar] [CrossRef]
- Damiens, D.; Tjeck, P.O.; Lebon, C.; Le Goff, G.; Gouagna, L.C. The effects of age at first mating and release ratios on the mating competitiveness of Gamma-sterilised Aedes albopictus males under semi field conditions. Vector Biol. J. 2016, 1, 29–31. [Google Scholar] [CrossRef]
- Dumont, Y.; Tchuenche, J.M. Mathematical Studies on the Sterile Insect Technique for the Chikungunya Disease and Aedes albopictus. J. Math. Biol. 2012, 65, 809–855. [Google Scholar] [CrossRef]
- Dufourd, C.; Dumont, Y. Modeling and simulations of mosquito dispersal. The case of Aedes albopictus. Proc. Biomath 2012, 1, 1209262. [Google Scholar] [CrossRef][Green Version]
- Dufourd, C.; Dumont, Y. Impact of environmental factors on mosquito dispersal in the prospect of sterile insect technique control. Comput. Math. Appl. 2013, 66, 1695–1715. [Google Scholar] [CrossRef]
- Dufourd, C.; Weldon, C.; Anguelov, R.; Dumont, Y. Parameter identification in population models for insects using trap data. Biomath 2013, 2, 1312061. [Google Scholar] [CrossRef][Green Version]
- Tran, A.; Mangeas, M.; Demarchi, M.; Roux, E.; Degenne, P.; Haramboure, M.; Le Goff, G.; Damiens, D.; Gouagna, L.-C.; Herbreteau, V.; et al. Complementarity of empirical and process-based approaches to modelling mosquito population dynamics with Aedes albopictus as an example-Application to the development of an operational mapping tool of vector populations. PLoS ONE 2020, 15, e0227407. [Google Scholar] [CrossRef]
- Thuilliez, J.; Bellia, C.; Dehecq, J.S.; Reilhes, O. Household-level expenditure on protective measures against mosquitoes on the Island of La Reeunion, France. PLoS Negl. Trop. Dis. 2014, 8, e2609. [Google Scholar] [CrossRef] [PubMed]
- Dupé, S. [Transform to control—Humans and mosquitoes in Reunion, in the era of biosecurity]. Revue D’Anthropologie des Connaissances 2015, 9, 213–236. [Google Scholar] [CrossRef]
- Ciss, M.; Bassène, M.D.; Seck, M.T.; Mbaye, A.G.; Sall, B.; Fall, A.G.; Vreysen, M.J.B.; Bouyer, J. Environmental impact of tsetse eradication in Senegal. Sci. Rep. 2019, 9, 20313. [Google Scholar] [CrossRef]
- Bouyer, J.; Lefrançois, T. Boosting the sterile insect technique to control mosquitoes. Trends Parasitol. 2014, 30, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Ellis, B.R.; Wilcox, B.A. The ecological dimensions of vector-borne disease research and control. Cadernos de Saúde Pública 2009, 25 (Suppl. 1), S155–S167. [Google Scholar] [CrossRef]
- Campbell-Lendrum, D.; Molyneux, D.; Amerasinghe, F.; Davies, C.; Fletcher, E.; Schofield, C.; Hougard, J.M.; Karen Polson, K.; Sinkins, S. Ecosystems and Vector-borne Disease Control. In Ecosystems and Human Well-being: A Framework for Assessment; Island Press: Washington, DC, USA, 2013; pp. 354–372. ISBN 9781559634021. [Google Scholar]
- Iyaloo, D.P.; Elahee, K.B.; Bheecarry, A.; Lees, R.S. Guidelines to site selection for population surveillance and mosquito control trials: A case study from Mauritius. Acta Trop. 2014, 132, S140–S149. [Google Scholar] [CrossRef]
- Lacroix, R.; Delatte, H.; Hue, T.; Dehecq, J.S.; Reiter, P. Adaptation of the BG-Sentinel trap to capture male and female Aedes albopictus mosquitoes. Med. Vet. Entomol. 2009, 23, 160–162. [Google Scholar] [CrossRef]
- Le Goff, G.; Damiens, D.; Payet, L.; Ruttee, A.H.; Jean, F.; Lebon, C.; Dehecq, J.-S.; Gouagna, L.C. Enhancement of the BG-Sentinel trap with varying number of mice for field sampling male and female Aedes albopictus mosquitoes. Parasites Vectors 2016, 9, 514. [Google Scholar] [CrossRef]
- Le Goff, G.; Damiens, D.; Ruttee, A.H.; Payet, L.; Lebon, C.; Dehecq, J.-S.; Geier, M.; Gouagna, L.C. Comparison of Efficiency of BG-Sentinel Traps Baited with Mice, Mouse-Litter, and CO2 Lures for Field Sampling of Male and Female Aedes albopictus Mosquitoes. Insects 2017, 8, 95. [Google Scholar] [CrossRef]
- Le Goff, G.; Damiens, D.; Ruttee, A.-H.; Payet, L.; Lebon, C.; Dehecq, J.-S.; Gouagna, L.C. Field evaluation of seasonal trends in relative population sizes and dispersal pattern of Aedes albopictus males in support of the design of sterile male release strategy. Parasites Vectors 2019, 12, 81. [Google Scholar] [CrossRef]
- Gouagna, L.C.; Dehecq, J.S.; Fontenille, D.; Dumont, Y.; Boyer, S. Seasonal variation in size estimates of Aedes albopictus population based on standard markrelease-recapture experiments in an urban area on Reunion Island. Acta Trop. 2015, 143, 89–96. [Google Scholar] [CrossRef]
- Haramboure, M.; Labbe, P.; Baldet, T.; Damiens, D.; Gouagna, L.C.; Bouyer, J.; Tran, A. Modelling the control of Aedes albopictus mosquitoes based on sterile males release techniques in a tropical environment. Ecol. Model. 2020, 424, 109002. [Google Scholar] [CrossRef]
- Service, M.W. Mosquito (Diptera: Culicidae) dispersal—The long and short of it. J. Med. Entomol. 1997, 34, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Vavassori, L.; Saddler, A.; Müller, P. Active dispersal of Aedes albopictus: A mark-release-recapture study using self-marking units. Parasites Vectors 2019, 12, 583. [Google Scholar] [CrossRef]
- Damiens, D.; Marquereau, L.; Lebon, C.; Le Goff, G.; Gaudillat, B.; Habchi-Hanriot, N.; Gouagna, L.C. Aedes albopictus Adult Medium Mass Rearing for SIT Program Development. Insects 2019, 10, 246. [Google Scholar] [CrossRef] [PubMed]
- Balestrino, F.; Benedict, M.Q.; Gilles, J.R. A new larval tray and rack system for improved mosquito mass rearing. J. Med. Entomol. 2012, 49, 595–605. [Google Scholar] [CrossRef]
- Balestrino, F.; Puggioli, I.A.; Bellini, R.; Petric, D.; Gilles, J.R. Mass production cage for Aedes albopictus (Diptera:Culicidae). J. Med. Entomol. 2014, 51, 155–163. [Google Scholar] [CrossRef]
- Gilles, J.R.L.; Schetelig, M.F.; Francesca, S.; Marec, F.; Capurro, M.L.; Franz, G.; Bourtzis, K. Towards mosquito sterile insect technique programmes: Exploring genetic, molecular, mechanical and behavioural methods of sex separation in mosquitoes. Acta Trop. 2014, 132, S178–S187. [Google Scholar] [CrossRef]
- Papathanos, P.A.; Bourtzis, K.; Tripet, F.; Bossin, H.; Virginio, J.F.; Capurro, M.L.; Pedrosa, M.C.; Guindo, A.; Sylla, L.; Coulibaly, M.; et al. A perspective on the need and current status of efficient sex separation methods for mosquito genetic control. Parasites Vectors 2018, 11 (Suppl. 2), 654. [Google Scholar] [CrossRef]
- Balestrino, F.; Medici, A.; Candini, G.; Carrieri, M.; Maccagnani, B.; Calvitti, M.; Maini, S.; Bellini, R. γ ray dosimetry and mating capacity studies in the laboratory on Aedes albopictus males. J. Med. Entomol. 2010, 47, 581–591. [Google Scholar] [CrossRef]
- Balestrino, F.; Puggioli, A.; Carrieri, M.; Bouyer, J.; Bellini, R. Quality control methods for Aedes albopictus sterile male production. PLoS Negl. Trop. Dis. 2017, 11, e0005881. [Google Scholar] [CrossRef]
- HCSP. Opinion on the Development of Recommendations to Authorize the Release of Sterile Mosquitoes for Vector Control Purposes; High Council of Public Health: Paris, France, 2018; 35p, Available online: https://www.hcsp.fr/explore.cgi/avisrapportsdomaine?clefr=711 (accessed on 4 May 2019). (In French)
- Bakri, A.; Mehta, K.; Lance, D.R. Sterilizing insects with ionizing radiation. In Sterile Insect Technique Principles and Practice in Area-Wide Integrated Pest Management; Dyck, V.A., Hendrichs, J., Robinson, A.S., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 233–268. [Google Scholar]
- Bakri, A.; Heather, N.; Hendrichs, J.; Ferris, I. Fifty years of radiation biology in entomology: Lessons learned from IDIDAS. Ann. Entomol. Soc. Am. 2014, 98, 112. [Google Scholar] [CrossRef]
- Bellini, R.; Balestrino, F.; Medici, A.; Gentile, G.; Veronesi, R.; Carrieri, M. Mating competitiveness of Aedes albopictus radio-sterilized males in large enclosures exposed to natural conditions. J. Med. Entomol. 2013, 50, 94–102. [Google Scholar] [CrossRef]
- Bhuyan, K.B.; Barik, T.K. Impact of High Dose of Gamma Radiation on Field Collected Aedes aegypti. J. Mosq. Res. 2016, 6, 1–6. [Google Scholar] [CrossRef]
- FAO/IAEA. Thematic Plan for the Development and Application of the Sterile Insect Technique (SIT) and Related Genetic and Biological Control Methods for Disease Transmitting Mosquitoes; Food and Agriculture Organization of the United Nations; International Atomic Energy Agency: Vienna, Austria, 2019; 93p. [Google Scholar]
- Oliva, C.F.; Damiens, D.; Benedict, M.Q. Male reproductive biology of Aedes mosquitoes. Acta Trop. 2014, 132, S12–S19. [Google Scholar] [CrossRef]
- Lebon, C.; Soupapoule, K.; Wilkinson, D.A.; Le Goff, G.; Damiens, D.; Gouagna, L.C. Laboratory evaluation of the effects of sterilizing doses of gamma-rays from Caesium-137 source on the daily flight activity and flight performance of Aedes albopictus males. PLoS ONE 2018, 13, e020223. [Google Scholar] [CrossRef]
- Tjeck, O. [Evaluation of Sexual Competitiveness as a Function of Age and Sterile:Fertile Males Ratio of Aedes albopictus, Vector of Chikungunya in Reunion]. Report, Master Classes II, Biodiversity and Tropical Ecosystems (BEST); Univerty of Reunion Island: Reunion, France, 2013; p. 30. (In French) [Google Scholar]
- Oliva, C.F.; Jacquet, M.; Gilles, J.; Lemperiere, G.; Maquart, P.O.; Quilici, S.; Schooneman, F.; Vreysen, M.J.; Boyer, S. The sterile insect technique for controlling populations of Aedes albopictus (Diptera: Culicidae) on Reunion Island: Mating vigour of sterilized males. PLoS ONE 2012, 7, e49414. [Google Scholar] [CrossRef]
- Iyaloo, D.P.; Oliva, C.; Facknath, S.; Bheecarry, A. A field cage study of the optimal age for release of radio-sterilized Aedes albopictus mosquitoes in a sterile insect technique program. Entomol. Exp. Appl. 2019, 168, 137–147. [Google Scholar] [CrossRef]
- Johnson, B.J.; Mitchell, S.N.; Paton, C.J.; Stevenson, J.; Staunton, K.M.; Snoad, N.; Beebe, N.W.; White, B.J.; Ritchie, S.A. Use of rhodamine B to mark the body and seminal fluid of male Aedes aegypti for mark-release-recapture experiments and estimating efficacy of sterile male releases. PLoS Negl. Trop. Dis. 2017, 11, e0005902. [Google Scholar] [CrossRef]
- Gaskell, G.; Bauer, M.W.; Durant, J.; Allum, N.C. Worlds apart? The reception of genetically modified foods in Europe and the US. Science 1999, 285, 384–387. [Google Scholar] [CrossRef]
- Gaskell, G.; Stares, S.; Allansdottir, A.; Allum, N.; Castro, P.; Esmer, Y.; Mejlgaard, N.; Europeans and Biotechnology in 2010. Winds of Change? A Report to the European Commission’s Directorate-General for Research. Available online: https://repositori.upf.edu/bitstream/handle/10230/21297/europeans-biotechnology-in-2010_en.pdf?sequence=1 (accessed on 2 March 2018).
- AFB. [Opinion of the Scientific Committee on a Project to Release Sterile Male Mosquitoes in Reunion for the Purpose of Vector Control]; Report of the scientific council French Agency for Biodiversity-session of April 26–27, 2018; Deliberation No. CS/2018-01; 2018; p. 4. Available online: https://www.afbiodiversite.fr/ (accessed on 7 November 2020).
- Vreysen, M.J.B. Monitoring sterile and wild insects in area-wide integrated pest management programmes. In Sterile Insect Technique. Principles and Practice in Area-Wide Integrated Pest Management; Dyck, V.A., Hendrichs, J., Robinson, A.S., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 325–361. [Google Scholar] [CrossRef]
- Mubarqui, R.L.; Perez, R.C.; Angulo Kladt, R.; Zavala, L.; Parker, A.; Seck, M.T.; Sall, B.; Bouyer, J. The smart aerial release machine, a universal system for applying the sterile insect technique. PLoS ONE 2014, 9, e103077. [Google Scholar] [CrossRef]
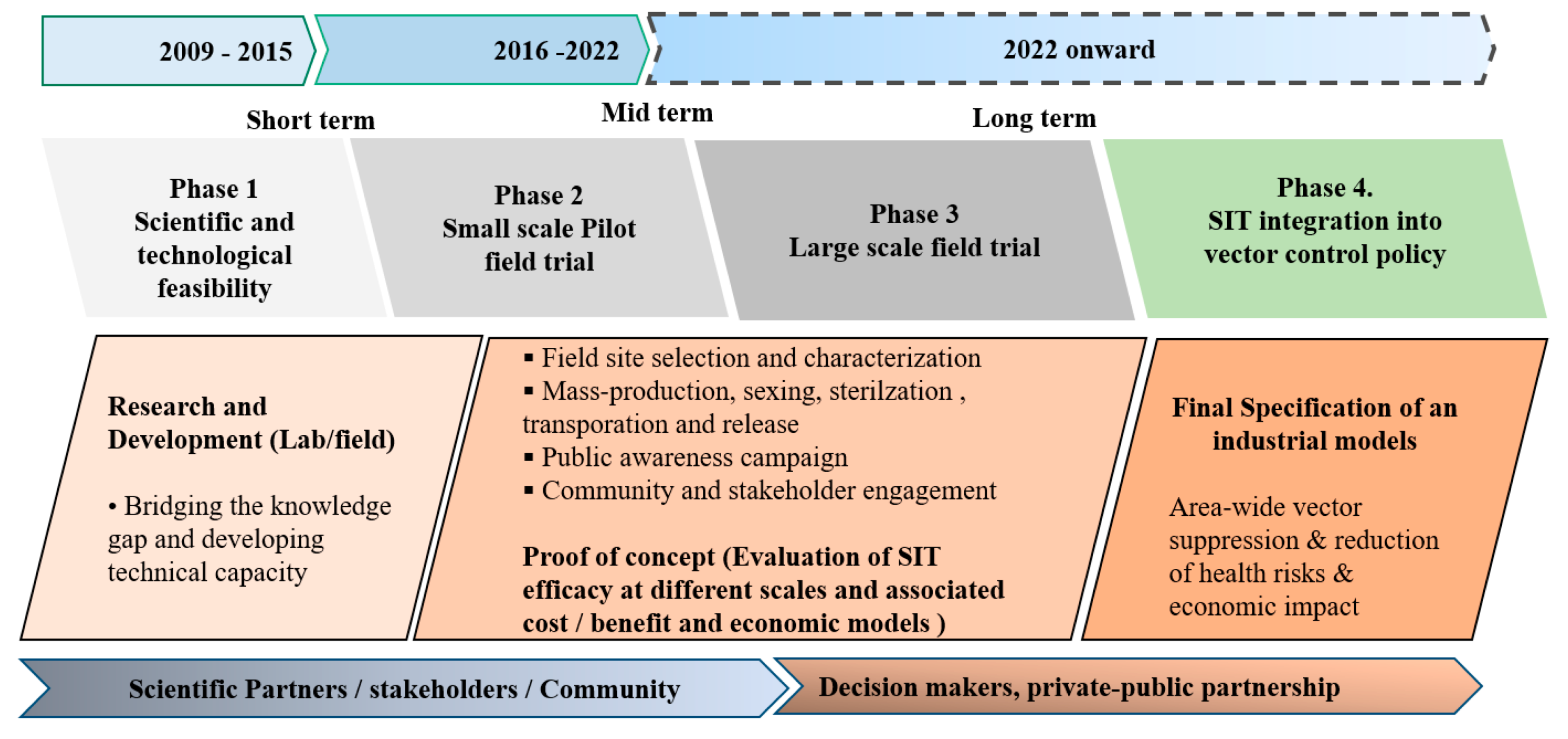

| R&D Priorities | Phase 1—Feasibility Studies (Filling the Knowledge Gap and Developing Technological Requisite) | Phase 2—Small Scale Pilot Tests Under Field Conditions (as Validated in Phase 1 Feasibility Studies) | Phase 3—Large-Scale Implementation (If Efficacy Is Proven in Pilot Test) |
|---|---|---|---|
| Biology and ecology of the target species | Refine knowledge on the ecology, biology, genetics, vector behavior, etc.,) of Aedes albopictus and Aedes aegypti | (1) Selection and characterization of pilot field sites based on entomological monitoring and studies on the behaviors of the target population. (2) Implementation of the SIT-based vector suppression in real conditions and efficacy testing. (3) Evaluation of SIT efficacy based on entomological, social, environmental and economic) indicators. | (1) Selection of the testing zones. (2) Implementation of the SIT-based vector suppression in real conditions and efficacy testing. (3) Evaluation of SIT efficacy based on entomological epidemiological, social, environmental and economic indicators. (4) Surveillance and monitoring. |
| Technological component (rearing, sexing, sterilization) | Establish the colony of the target species and improve capacity and knowledge on rearing, sexing, sterilization, and on key behavioral traits | Up scaling the mass rearing and (infrastructure construction, etc.,) and strengthening of the release strategy and quality control | Technology transfer, business model for transition to large (industrial) scale production of sterile males. Implementation tools (transport, release, etc.,). |
| Modeling and simulations | Modeling of the methodology and release of irradiated males (dispersion, dynamics, etc.,) | Models validation based on efficiency indicators (entomological, epidemiological, social, environmental and economic) parameters. Cost/efficacy analysis and model of integrated strategies | Confirm/validate the predictions of epidemiological models. Predictions to other impact indicators. Cost-benefit analysis associated with large-scale SIT application. |
| Communication and social sciences component | Refine knowledge (existing social perception, attitude and practices T0; survey of costs of existing control techniques), and social levers for the acceptability of the project. Inform the public | (1) Inform the general public and stakeholders to enhance their understanding and support. (2) Analysis of the evolution of social acceptance and engagement. (3) Cost/effectiveness analysis of combined vector control strategies. (4) Economy model for the transition to industrial production scale | Public awareness and influence of SIT-based vector control strategy on acceptability and behavior change. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouagna, L.C.; Damiens, D.; Oliva, C.F.; Boyer, S.; Le Goff, G.; Brengues, C.; Dehecq, J.-S.; Raude, J.; Simard, F.; Fontenille, D. Strategic Approach, Advances, and Challenges in the Development and Application of the SIT for Area-Wide Control of Aedes albopictus Mosquitoes in Reunion Island. Insects 2020, 11, 770. https://doi.org/10.3390/insects11110770
Gouagna LC, Damiens D, Oliva CF, Boyer S, Le Goff G, Brengues C, Dehecq J-S, Raude J, Simard F, Fontenille D. Strategic Approach, Advances, and Challenges in the Development and Application of the SIT for Area-Wide Control of Aedes albopictus Mosquitoes in Reunion Island. Insects. 2020; 11(11):770. https://doi.org/10.3390/insects11110770
Chicago/Turabian StyleGouagna, Louis Clément, David Damiens, Clélia F. Oliva, Sébastien Boyer, Gilbert Le Goff, Cécile Brengues, Jean-Sébastien Dehecq, Jocelyn Raude, Frédéric Simard, and Didier Fontenille. 2020. "Strategic Approach, Advances, and Challenges in the Development and Application of the SIT for Area-Wide Control of Aedes albopictus Mosquitoes in Reunion Island" Insects 11, no. 11: 770. https://doi.org/10.3390/insects11110770
APA StyleGouagna, L. C., Damiens, D., Oliva, C. F., Boyer, S., Le Goff, G., Brengues, C., Dehecq, J.-S., Raude, J., Simard, F., & Fontenille, D. (2020). Strategic Approach, Advances, and Challenges in the Development and Application of the SIT for Area-Wide Control of Aedes albopictus Mosquitoes in Reunion Island. Insects, 11(11), 770. https://doi.org/10.3390/insects11110770






