Fumigation Activity against Phosphine-Resistant Tribolium castaneum (Coleoptera: Tenebrionidae) Using Carbonyl Sulfide
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Fumigants
2.3. Fumigation Experiments
2.4. Gas Concentration and Sorption Measurements
2.5. Rice Quality Evaluation
2.6. Statistical Analysis
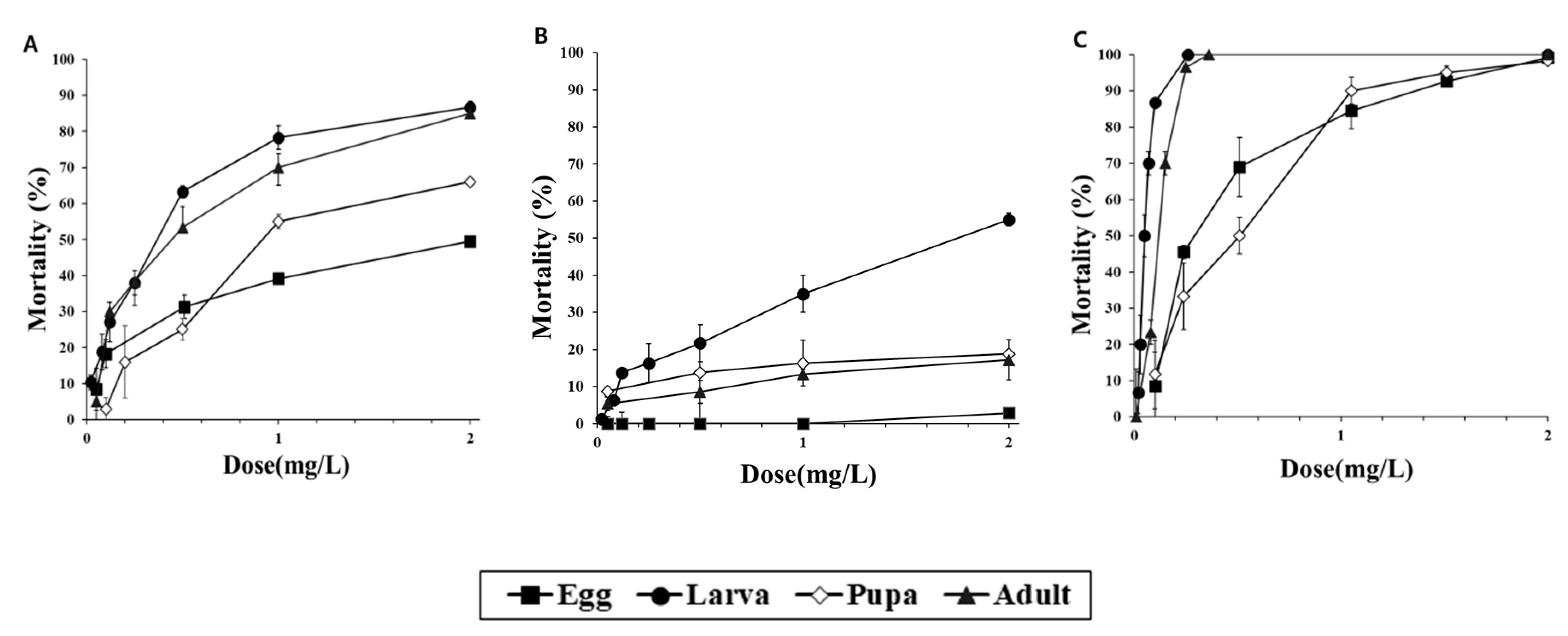
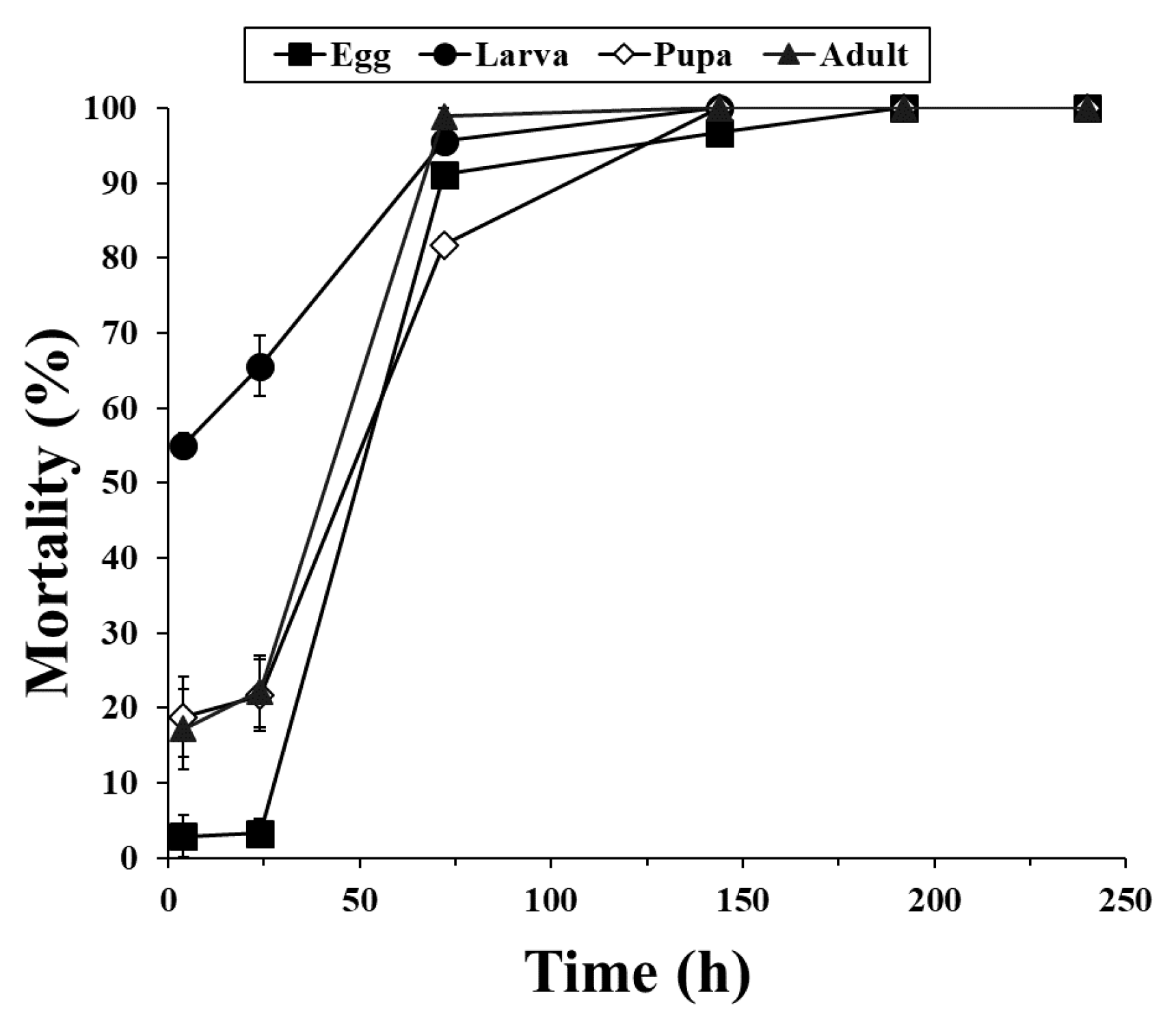
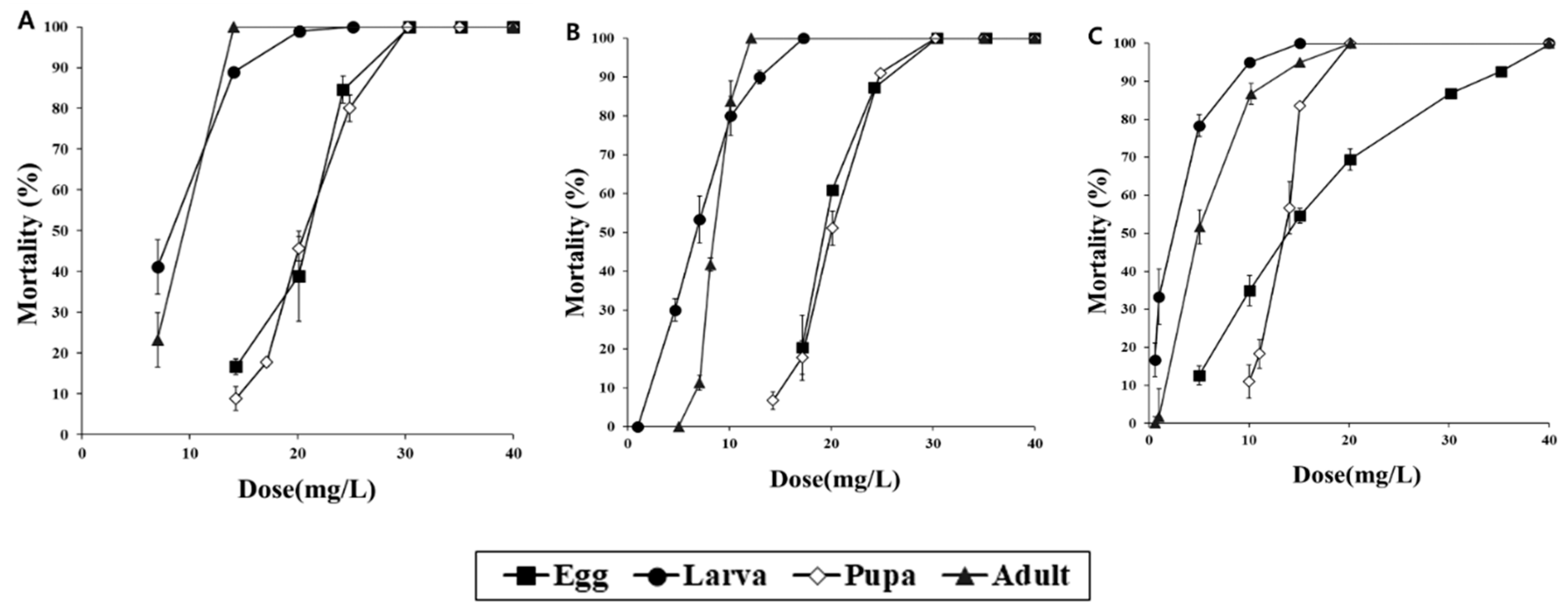
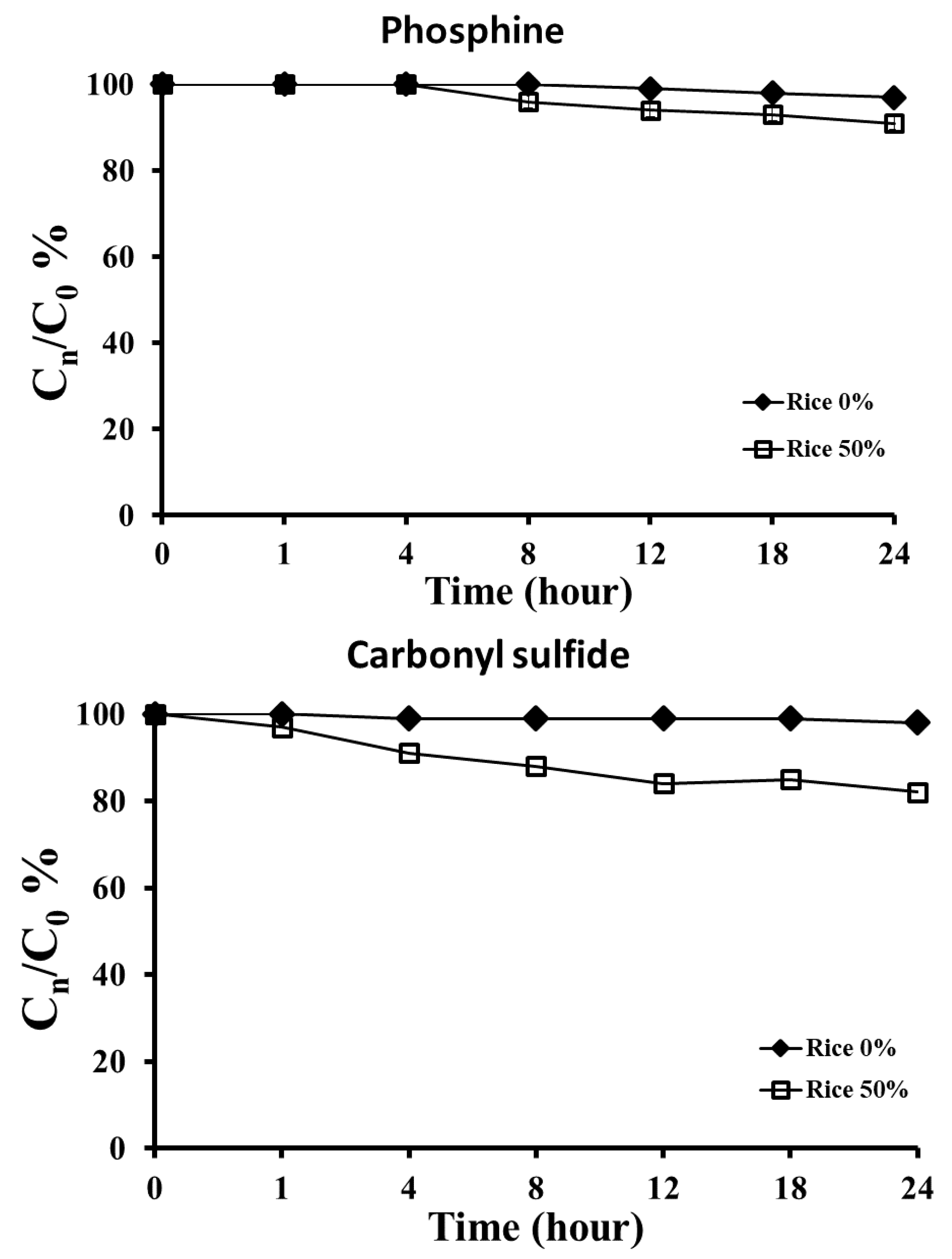
3. Results
3.1. Fumigation Activities of PH3 and COS in a 12-LDesiccator
3.2. Scale-Up Fumigation Experiment with PH3 and COS in a 0.5 m3 Fumigation Chamber
3.3. Rice Quality and Sorption of the Two Fumigants
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Follett, P.A.; Neven, L.G. Current trends in quarantine entomology. Annu. Rev. Entomol. 2006, 51, 359–385. [Google Scholar] [CrossRef] [PubMed]
- Heather, N.W.; Hallman, G.J. Agricultural warfare and bioterrorism using invasive species. In Pest Management and Phytosanitary Trade Barriers, 1st ed.; CABI Publishing: Wallingford, UK, 2008; pp. 14–19. [Google Scholar]
- Hallman, G.J. Process control in phytosanitary irradiation of fresh fruits and vegetables as a model for other phytosanitary treatment processes. Food Control 2017, 72, 372–377. [Google Scholar] [CrossRef]
- Cho, W.S.; Koo, H.N.; Yun, S.H.; Lee, J.S.; Jeong, D.H.; Kang, W.J.; Lee, S.J.; Kim, H.K.; Han, J.H.; Kwon, Y.D.; et al. Electron beam-induced sterility and inhibition of ovarian development in the sakhalin pine longicorn, Monochamus saltuarius (Coleoptera: Cerambycidae). J. Econ. Entomol. 2018, 111, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Kyung, Y.J.; Kim, H.K.; Cho, S.W.; Kim, B.S.; Yang, J.O.; Koo, H.N.; Kim, G.H. Comparison of the efficacy and phytotoxicity of phosphine and ethyl formate for controlling Pseudococcus longispinus (Hemiptera: Pseudococcidae) and Pseudococcus orchidicola on imported foliage nursery plants. J. Econ. Entomol. 2019, 112, 2149–2156. [Google Scholar] [CrossRef]
- Bond, E.J. Manual of fumigation for insect control. In FAO Plant Production and Protection Paper No. 54; FAO: Rome, Italy, 1984. [Google Scholar]
- Chaudhry, M.Q. Phosphine resistance. Pestic. Outlook 2000, 11, 88–91. [Google Scholar] [CrossRef]
- Zettler, J.L.; Arthur, F.H. Chemical control of stored product insects with fumigants and residual treatments. Crop Prot. 2000, 19, 577–582. [Google Scholar] [CrossRef]
- UNEP (United Nations Environment Programme). Handbook for the Montreal Protocol on Substances That Deplete the Ozone Layer, 7th ed.; UNEP: Nairobi, Kenya, 2006. [Google Scholar]
- Daglish, G.J. Effect of exposure period on degree of dominance of phosphine resistance in adults of Rhyzopertha dominica (Coleoptera: Bostrychidae) and Sitophilus oryzae (Coleoptera: Curculionidae). Pest Manag. Sci. 2004, 60, 822–826. [Google Scholar] [CrossRef]
- Opit, G.P.; Phillips, T.W.; Aikins, M.J. Phosphine resistance in Tribolium castaneum and Rhyzopertha dominica from stored wheat in Oklahoma. J. Econ. Entomol. 2012, 105, 1107–1114. [Google Scholar] [CrossRef]
- Konemann, C.E.; Hubhachen, Z.; Opit, G.P.; Gautam, S.; Bajracharya, N.S. Phosphine resistance in Cryptolestes ferrugineus (Coleoptera: Laemophloeidae) collected from grain storage facilities in Oklahoma, USA. J. Econ. Entomol. 2017, 110, 1377–1383. [Google Scholar] [CrossRef]
- Desmarchelier, J.M. Grain protectants: Trends and developments. In Stored Product Protection; Highley, E., Wright, E.J., Banks, H.J., Champ, B.R., Eds.; CAB International: Wallingford, UK, 1994; pp. 722–728. [Google Scholar]
- Zettler, J.L.; Leesch, J.G.; Gill, R.F.; Mackey, B.E. Toxicity of carbonyl sulfide to stored product insects. J. Econ. Entomol. 1997, 90, 832–836. [Google Scholar] [CrossRef]
- Desmarchelier, J.M.; Allen, S.E.; Ren, Y.; Moss, R.; Vu, L.T. Commercial-scale trials on the application of ethyl formate, carbonyl sulfide and carbon disulfide to wheat. In CSIRO Entomology Technical Report No. 75; CSIRO Division of Entomology: Canberra, Australia, 1998; p. 63. [Google Scholar]
- Weller, G.L.; Morton, R. Fumigation with carbonyl sulfide: A model for the interaction of concentration, time and temperature. J. Stored Prod. Res. 2001, 37, 383–398. [Google Scholar] [CrossRef]
- Weller, G.L. The role of concentration, time and temperature in determining dosage for fumigation with carbonyl sulfide. In Proceedings of the Seventh International Working Conference on Stored Product Protection, Beijing, China, 14–19 October 1998; Jin, Z., Liang, Q., Liang, Y., Tan, X., Guan, L., Eds.; Sichuan Publishing House of Science and Technology: Chengdu, China, 1999; pp. 548–553. [Google Scholar]
- Bartholomaeus, A.R.; Haritos, V.S. Review of the toxicology of carbonyl sulfide, a new grain fumigant. Food Chem. Toxicol. 2005, 43, 1687–1701. [Google Scholar] [CrossRef]
- Daglish, G.J.; Nayak, M.K.; Pavic, H.; Smith, L.W. Prevalence and potential fitness cost of weak phosphine resistance in Tribolium castaneum (Herbst) in eastern Australia. J. Stored Prod. Res. 2015, 61, 54–58. [Google Scholar] [CrossRef]
- Mutters, R.G.; Thompson, J.F. Rice Quality Handbook; UC ANR Publication: Oakland, CA, USA, 2009; p. 141. [Google Scholar]
- Bell, C.H. Fumigation in the 21st century. Crop Prot. 2000, 19, 563–569. [Google Scholar] [CrossRef]
- Fedina, T.Y.; Lewis, S.M. Effect of Tribolium castaneum (Coleoptera: Tenebrionidae) nutritional environment, sex, and mating status on response to commercial pheromone traps. J. Econ. Entomol. 2007, 100, 1924–1927. [Google Scholar] [CrossRef]
- Mathlein, R. Rearing experiments with Oryzaephilus surinamensis L. and Cryptolestes ferrugineus Steph. on grain. Natl. Swed. Inst. Plant Prot. Contrib. 1971, 15, 187–203. [Google Scholar]
- Champ, B.R.; Dyte, C.E. Report of the FAO global survey of pesticide susceptibility of stored grain pests. In FAO Plant Production and Protection Series Vol. 5; FAO: Rome, Italy, 1976. [Google Scholar]
- AFHB/ACIAR Suggested recommendations for the fumigation of grain in ASEAN region. In Principles and General Practice, Part 1; ASEAN Food Handling Bureau: Kuala Lumpur, Malaysia, 1989; p. 139.
- Khattak, S.U.; Khatoon, R.; Wahid, M. Insect attack and nutritional losses in stored dried fruits. Nucleus (Islamabad) 1999, 36, 113–116. [Google Scholar]
- Matthews, G.A. Insecticide application in stores. In Application Technology for Crop Protection; Matthews, G.A., Hislop, E.C., Eds.; CABI international: Wallingford, UK, 1993; pp. 305–315. [Google Scholar]
- Benhalima, H.; Chaudry, M.Q.; Mills, K.A.; Price, N.R. Phosphine resistance in stored-product insects collected from various grain storage facilities in Morocco. J. Stored Prod. Res. 2004, 40, 241–249. [Google Scholar] [CrossRef]
- Kaur, R.; Nayak, M.K. Developing effective fumigation protocols to manage strongly phosphine-resistant Cryptolestes ferrugineus (Stephens) (Coleoptera: Laemophloeidae). Pest Manag. Sci. 2015, 71, 1297–1302. [Google Scholar] [CrossRef]
- Koçak, E.; Schlipalius, D.; Kaur, R.; Tuck, A.; Ebert, P.; Collins, P.; Yilmaz, A. Determining phosphine resistance in rust red flour beetle, Tribolium castaneum (Herbst.)(Coleoptera: Tenebrionidae) populations from Turkey. Turk. J. Entomol. 2015, 39, 129–136. [Google Scholar] [CrossRef]
- Nayak, M.K.; Jagadeesan, R.; Kaur, R.; Daglish, G.J.; Reid, R.; Pavic, H.; Collins, P.J. Use of sulfuryl fluoride in the management of strongly phosphine-resistant insect pest populations in bulk grain storages in Australia. Indian J. Entomol. 2016, 78, 100–107. [Google Scholar] [CrossRef]
- Chen, Z.; Schlipalius, D.; Opit, G.; Subramanyam, B.; Phillips, T.W. Diagnostic molecular markers for phosphine resistance in US populations of Tribolium castaneum and Rhyzopertha dominica. PLoS ONE 2015, 10, e0121343. [Google Scholar]
- Nayak, M.K.; Falk, M.G.; Enermy, R.N.; Collins, P.J.; Joanne, H.C. An analysis of trends, frequencies and factors influencing the development of resistance to phosphine in the red flour beetle Tribolium castaneum (Herbst) in Australia. J. Stored Prod. Res. 2017, 72, 35–48. [Google Scholar] [CrossRef]
- Navarro, S.; Isikber, A.A.; Finkelman, S.; Rindner, M.; Azrieli, A.; Dias, R. Effectiveness of short exposures of propylene oxide alone and in combination with low pressure or carbon dioxide against Tribolium castaneum (Herbst)(Coleoptera:Tenebrionidae). J. Stored Prod. Res. 2004, 40, 197–205. [Google Scholar] [CrossRef]
- KOTRA. Overseas Market News; Australian Wheat Imports, Need to Understand Local Logistics System and Price Structure to Secure Stable Raw Materials (Korean). Available online: http://news.kotra.or.kr/user/globalBbs/kotranews/782/globalBbsDataView.do?setIdx=243&dataIdx=125127 (accessed on 10 October 2020).
- Kyung, Y.J.; Kim, H.K.; Lee, J.S.; Kim, B.S.; Yang, J.O.; Lee, B.H.; Koo, H.N.; Kim, G.H. Efficacy and phytotoxicity of phosphine as fumigants for Frankliniella occidentalis (Thysanoptera: Thripidae) on asparagus. J. Econ. Entomol. 2018, 111, 2644–2651. [Google Scholar] [CrossRef]
- SAS Institute. SAS User’s Guide, Statistics Version 9, 1st ed.; SAS Institute: Cary, NC, USA, 2009. [Google Scholar]
- Pimentel, M.A.G.; Faroni, L.R.D.A.; Totola, M.R.; Guedes, R.N.C. Phosphine resistance, respiration rate and fitness consequences in stored-product insects. Pest Manag. Sci. 2007, 63, 876–881. [Google Scholar] [CrossRef]
- Kaur, R.; Subbarayalu, M.; Jagadeesan, R.; Daglish, G.J.; Nayak, M.K.; Naik, H.R.; Schlipalius, D.I. Phosphine resistance in India is characterised by a dihydrolipoamide dehydrogenase variant that is otherwise unobserved in eukaryotes. Heredity 2015, 115, 188–194. [Google Scholar] [CrossRef]
- Nayak, M.K.; Daglish, G.J.; Phillips, T.W.; Ebert, P.R. Resistance to the fumigant phosphine and its management in insect pests of stored products: A global perspective. Annu. Rev. Entomol. 2020, 65, 333–350. [Google Scholar] [CrossRef]
- Ducom, P.J.F. The return of the fumigants. In Proceedings of the Ninth International Working Conference on Stored Product Protection, Campinas, Brazil, 15–18 October 2006; Lorini, I., Bacaltchuk, B., Beckel, H., Deckers, D., Sundfeld, E., dos Santos, J.P., Biagi, J.D., Celaro, J.C., Faroni, L.R.D.A., Bortolini, L., et al., Eds.; Brazilian Post-harvest Association: Campinas, Brazil, 2006; pp. 510–516. [Google Scholar]
- Xianchang, T.; Xingwei, H.; Lizheng, C.; Jianchun, W. Research on carbonyl sulfide as a fumigant for control of stored grain insects. In Proceedings of the Seventh International Working Conference on Stored Product Protection, Beijing, China, 14–19 October 1998; Jin, Z., Liang, Q., Liang, Y., Tan, X., Guan, L., Eds.; Sichuan Publishing House of Science and Technology: Chengdu, China, 1999; pp. 567–571. [Google Scholar]
- Chaudhry, M.Q. Review A review of the mechanisms involved in the action of phosphine as an insecticide and phosphine resistance in stored-product insects. Pestic. Sci. 1997, 49, 213–228. [Google Scholar] [CrossRef]
- Hsu, C.H.; Quistad, G.B.; Casida, J.E. Phosphine-induced oxidative stress in Hepa 1c1c7 cells. Toxicol. Sci. 1998, 46, 204–210. [Google Scholar] [CrossRef]
- Haritos, V. Metabolism of carbonyl sulfide to hydrogen sulfide in insects is catalysed by carbonic anhydrase. In Proceedings of the International Conference on Controlled Atmospheres and Fumigation in Stored Products, Fresno, CA, USA, 29 October 2000; Executive Printing Services: Clovis, CA, USA, 2001; pp. 79–89. [Google Scholar]
- Liu, B.; Zhang, F.; Wang, Y. Toxicity of phosphine to Carposina niponensis (Lepidoptera: Carposinadae) at low temperature. J. Econ. Entomol. 2010, 103, 1988–1993. [Google Scholar]
- Llàcer, E.; Miret, J.A.J. Efficacy of phosphine as a fumigant against Rhynchophorus ferrugineus (Coleoptera: Curculionidae) in palms. Span. J. Agric. Res. 2010, 8, 775–779. [Google Scholar] [CrossRef]
- Kim, H.K.; Lee, S.W.; Kim, J.I.; Yang, J.O.; Koo, H.N.; Kim, G.H. Synergistic effects of oxygen on phosphine and ethyl formate for the control of Phthorimaea operculella (Lepidoptera: Gelechiidae). J. Econ. Entomol. 2015, 108, 2572–2580. [Google Scholar] [CrossRef]
| Exposure Time(h) | Species/Strain | Stages | n | Locate | Mortality | CT Value (g·h/m3) |
|---|---|---|---|---|---|---|
| O. surinamensis | Egg | 283 | Top | 100 ± 0.0a | 39.40 | |
| 284 | Middle | 100 ± 0.0a | ||||
| 286 | Bottom | 100 ± 0.0a | ||||
| 285 | Control | 4.9 ± 2.7b | ||||
| Larva | 290 | Top | 100 ± 0.0a | |||
| 268 | Middle | 100 ± 0.0a | ||||
| 291 | Bottom | 100 ± 0.0a | ||||
| 278 | Control | 3.6 ± 0.9b | ||||
| Pupa | 271 | Top | 100 ± 0.0a | |||
| 281 | Middle | 100 ± 0.0a | ||||
| 279 | Bottom | 100 ± 0.0a | ||||
| 280 | Control | 5.8 ± 4.0b | ||||
| Adult | 283 | Top | 100 ± 0.0a | |||
| 291 | Middle | 100 ± 0.0a | ||||
| 290 | Bottom | 100 ± 0.0a | ||||
| 288 | Control | 5.3 ± 0.8b | ||||
| 24 | T. castaneumd-strain | Egg | 296 | Top | 100 ± 0.0a | |
| 296 | Middle | 100 ± 0.0a | ||||
| 293 | Bottom | 100 ± 0.0a | ||||
| 294 | Control | 9.4 ± 0.8b | ||||
| Larva | 270 | Top | 100 ± 0.0a | |||
| 275 | Middle | 100 ± 0.0a | ||||
| 280 | Bottom | 100 ± 0.0a | ||||
| 270 | Control | 5.0 ± 1.5b | ||||
| Pupa | 270 | Top | 100 ± 0.0a | |||
| 270 | Middle | 100 ± 0.0a | ||||
| 270 | Bottom | 100 ± 0.0a | ||||
| 270 | Control | 7.3 ± 1.1b | ||||
| Adult | 270 | Top | 100 ± 0.0a | |||
| 270 | Middle | 100 ± 0.0a | ||||
| 270 | Bottom | 100 ± 0.0a | ||||
| 270 | Control | 3.3 ± 0.6b | ||||
| T. castaneumr-strain | Egg | 301 | Top | 3.3 ± 1.9a | ||
| 292 | Middle | 5.5 ± 1.2a | ||||
| 276 | Bottom | 7.6 ± 1.0a | ||||
| 278 | Control | 4.7 ± 1.4b | ||||
| Larva | 279 | Top | 61.2 ± 2.8a | |||
| 274 | Middle | 67.1 ± 1.9a | ||||
| 278 | Bottom | 65.8 ± 4.2a | ||||
| 281 | Control | 3.6 ± 1.6b | ||||
| Pupa | 297 | Top | 25.9 ± 1.8a | |||
| 296 | Middle | 24.0 ± 3.5a | ||||
| 291 | Bottom | 21.7 ± 3.7a | ||||
| 291 | Control | 7.2 ± 1.2b | ||||
| Adult | 273 | Top | 28.3 ± 5.1a | |||
| 284 | Middle | 27.0 ± 4.3a | ||||
| 281 | Bottom | 23.2 ± 3.9a | ||||
| 286 | Control | 5.3 ± 1.9b | ||||
| 192 | T. castaneumr-strain | Egg | 285 | Top | 100 ± 0.0a | 261.31 |
| 277 | Middle | 100 ± 0.0a | ||||
| 300 | Bottom | 100 ± 0.0a | ||||
| 279 | Control | 1.8 ± 0.8b | ||||
| Larva | 305 | Top | 100 ± 0.0a | |||
| 285 | Middle | 100 ± 0.0a | ||||
| 281 | Bottom | 100 ± 0.0a | ||||
| 273 | Control | 7.3 ± 3.1b | ||||
| Pupa | 300 | Top | 100 ± 0.0a | |||
| 300 | Middle | 100 ± 0.0a | ||||
| 300 | Bottom | 100 ± 0.0a | ||||
| 288 | Control | 7.7 ± 1.3b | ||||
| Adult | 291 | Top | 100 ± 0.0a | |||
| 284 | Middle | 100 ± 0.0a | ||||
| 279 | Bottom | 100 ± 0.0a | ||||
| 280 | Control | 8.1 ± 2.6b |
| Exposure Time(h) | Species/Strain | Stages | n | Locate | Mortality | CT Value (g·h/m3) |
|---|---|---|---|---|---|---|
| 24 | O. surinamensis | Egg | 307 | Top | 100 ± 0.0a | 1264.7 |
| 308 | Middle | 100 ± 0.0a | ||||
| 300 | Bottom | 100 ± 0.0a | ||||
| 324 | Control | 9.7 ± 3.0b | ||||
| Larva | 316 | Top | 100 ± 0.0a | |||
| 301 | Middle | 100 ± 0.0a | ||||
| 328 | Bottom | 100 ± 0.0a | ||||
| 292 | Control | 7.2 ± 2.6b | ||||
| Pupa | 280 | Top | 100 ± 0.0a | |||
| 320 | Middle | 100 ± 0.0a | ||||
| 319 | Bottom | 100 ± 0.0a | ||||
| 332 | Control | 10.2 ± 2.5b | ||||
| Adult | 317 | Top | 100 ± 0.0a | |||
| 299 | Middle | 100 ± 0.0a | ||||
| 303 | Bottom | 100 ± 0.0a | ||||
| 285 | Control | 7.1 ± 2.0b | ||||
| T. castaneumd-strain | Egg | 288 | Top | 100 ± 0.0a | ||
| 301 | Middle | 100 ± 0.0a | ||||
| 313 | Bottom | 100 ± 0.0a | ||||
| 320 | Control | 3.0 ± 1.7b | ||||
| Larva | 300 | Top | 100 ± 0.0a | |||
| 300 | Middle | 100 ± 0.0a | ||||
| 300 | Bottom | 100 ± 0.0a | ||||
| 300 | Control | 4.3 ± 1.2b | ||||
| Pupa | 270 | Top | 100 ± 0.0a | |||
| 270 | Middle | 100 ± 0.0a | ||||
| 270 | Bottom | 100 ± 0.0a | ||||
| 270 | Control | 8.5 ± 1.6b | ||||
| Adult | 282 | Top | 100 ± 0.0a | |||
| 277 | Middle | 100 ± 0.0a | ||||
| 274 | Bottom | 100 ± 0.0a | ||||
| 275 | Control | 6.5 ± 2.2b | ||||
| T. castaneumr-strain | Egg | 243 | Top | 100 ± 0.0a | ||
| 304 | Middle | 100 ± 0.0a | ||||
| 347 | Bottom | 100 ± 0.0a | ||||
| 262 | Control | 8.4 ± 2.3b | ||||
| Larva | 295 | Top | 100 ± 0.0a | |||
| 307 | Middle | 100 ± 0.0a | ||||
| 272 | Bottom | 100 ± 0.0a | ||||
| 306 | Control | 5.9 ± 1.7b | ||||
| Pupa | 289 | Top | 100 ± 0.0a | |||
| 285 | Middle | 100 ± 0.0a | ||||
| 288 | Bottom | 100 ± 0.0a | ||||
| 312 | Control | 9.1 ± 1.4b | ||||
| Adult | 343 | Top | 100 ± 0.0a | |||
| 329 | Middle | 100 ± 0.0a | ||||
| 304 | Bottom | 100 ± 0.0a | ||||
| 278 | Control | 2.5 ± 1.3b |
| Quality Criteria a | Control | PH3 | p-Value | COS | p-Value |
|---|---|---|---|---|---|
| Protein content | 5.60 ± 0.00a | 5.53 ± 0.03a | 0.12 | 5.60 ± 0.00a | - |
| Amylose content | 13.77 ± 0.0a | 14.01 ± 0.06a | 0.03 | 13.80 ± 0.03a | 0.35 |
| Head rice ratio | 76.80 ± 1.36a | 76.0 ± 1.20a | 0.68 | 76.6 ± 1.37a | 0.92 |
| Broken rice ratio | 1.37 ± 0.23a | 1.53 ± 0.35a | 0.75 | 0.90 ± 0.00a | 0.32 |
| Chalky rice ratio | 7.07 ± 0.38a | 7.67 ± 0.37a | 0.32 | 6.67 ± 0.61a | 0.91 |
| Damaged rice ratio | 14.83 ± 1.18a | 12.97 ± 0.92a | 0.39 | 15.8 ± 0.80a | 0.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.K.; Jeong, G.; Kim, H.K.; Kim, B.-S.; Yang, J.-O.; Koo, H.-N.; Kim, G.-H. Fumigation Activity against Phosphine-Resistant Tribolium castaneum (Coleoptera: Tenebrionidae) Using Carbonyl Sulfide. Insects 2020, 11, 750. https://doi.org/10.3390/insects11110750
Lee HK, Jeong G, Kim HK, Kim B-S, Yang J-O, Koo H-N, Kim G-H. Fumigation Activity against Phosphine-Resistant Tribolium castaneum (Coleoptera: Tenebrionidae) Using Carbonyl Sulfide. Insects. 2020; 11(11):750. https://doi.org/10.3390/insects11110750
Chicago/Turabian StyleLee, Han Kyung, Geunho Jeong, Hyun Kyung Kim, Bong-Su Kim, Jeong-Oh Yang, Hyun-Na Koo, and Gil-Hah Kim. 2020. "Fumigation Activity against Phosphine-Resistant Tribolium castaneum (Coleoptera: Tenebrionidae) Using Carbonyl Sulfide" Insects 11, no. 11: 750. https://doi.org/10.3390/insects11110750
APA StyleLee, H. K., Jeong, G., Kim, H. K., Kim, B.-S., Yang, J.-O., Koo, H.-N., & Kim, G.-H. (2020). Fumigation Activity against Phosphine-Resistant Tribolium castaneum (Coleoptera: Tenebrionidae) Using Carbonyl Sulfide. Insects, 11(11), 750. https://doi.org/10.3390/insects11110750





