Evaluation of Rearing Parameters of a Self-Limiting Strain of the Mediterranean Fruit Fly, Ceratitis capitata (Diptera: Tephritidae)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Adult Colony Maintenance
2.2. Egg Production
2.3. Pupal Production
2.4. Quality Control Parameters
2.4.1. Egg Production
2.4.2. Pupal Production
2.5. Statistical Analysis
3. Results
3.1. Egg Production
3.2. Pupal Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gasperi, G.; Bonizzoni, M.; Gomulski, L.; Murelli, V.; Torti, C.; Malacrida, A.; Guglielmino, C. Genetic differentiation, gene flow and the origin of infestations of the medfly, Ceratitis capitata. Genetica 2002, 116, 125–135. [Google Scholar] [CrossRef]
- Diamantidis, A.D.; Carey, J.R.; Nakas, C.T.; Papadopoulos, N.T. Ancestral populations perform better in a novel environment: Domestication of Mediterranean fruit fly populations from five global regions. Biol. J. Linn. Soc. 2011, 102, 334–345. [Google Scholar] [CrossRef] [PubMed]
- Malacrida, A.; Gomulski, L.; Bonizzoni, M.; Bertin, S.; Gasperi, G.; Guglielmino, C. Globalization and fruitfly invasion and expansion: The medfly paradigm. Genetica 2007, 131, 1. [Google Scholar] [CrossRef] [PubMed]
- Diamantidis, A.D.; Carey, J.R.; Papadopoulos, N.T. Life-history evolution of an invasive tephritid. J. Appl. Entomol. 2008, 132, 695–705. [Google Scholar] [CrossRef]
- Arouri, R.; Le Goff, G.; Hemden, H.; Navarro-Llopis, V.; M’saad, M.; Castañera, P.; Feyereisen, R.; Hernández-Crespo, P.; Ortego, F. Resistance to lambda-cyhalothrin in Spanish field populations of Ceratitis capitata and metabolic resistance mediated by P450 in a resistant strain. Pest. Manag. Sci 2015, 71, 1281–1291. [Google Scholar] [CrossRef]
- Elfekih, S.; Shannon, M.; Haran, J.; Vogler, A.P. Detection of the Acetylcholinesterase Insecticide Resistance Mutation (G328A) in Natural Populations of Ceratitis capitata. J. Econ. Entomol. 2014, 107, 1965–1968. [Google Scholar] [CrossRef]
- Kakani, E.; Mathiopoulos, K. Organophosphosphate resistance-related mutations in the acetylcholinesterase gene of Tephritidae. J. Appl. Entomol. 2008, 132, 762–771. [Google Scholar] [CrossRef]
- Magaña, C.; Hernández-Crespo, P.; Ortego, F.; Castañera, P. Resistance to Malathion in Field Populations of Ceratitis capitata. J. Econ. Entomol. 2007, 100, 1836–1843. [Google Scholar] [CrossRef]
- Vontas, J.; Hernández-Crespo, P.; Margaritopoulos, J.T.; Ortego, F.; Feng, H.-T.; Mathiopoulos, K.D.; Hsu, J.-C. Insecticide resistance in Tephritid flies. Pestic. Biochem. Physiol. 2011, 100, 199–205. [Google Scholar] [CrossRef]
- Leza, M.; Juan, A.; Capllonch, M.; Alemany, A. Female-biased mass trapping vs. bait application techniques against the Mediterranean fruit fly, Ceratitis capitata (Dipt., Tephritidae). J. Appl. Entomol. 2008, 132, 753–761. [Google Scholar] [CrossRef]
- Hendrichs, J.; Robinson, A.; Cayol, J.; Enkerlin, W. Medfly areawide sterile insect technique programmes for prevention, suppression or eradication: The importance of mating behavior studies. Fla. Entomol. 2002, 85, 1–13. [Google Scholar] [CrossRef]
- Barry, J.D.; Dowell, R.V.; Morse, J.G. Comparison of two sterile Mediterranean fruit fly (Diptera: Tephritidae) strains released in California’s preventative release program. J. Econ. Entomol. 2002, 95, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Hendrichs, J. Use of the sterile insect technique against key insect pests. Sustain. Dev. Int. 2000, 2, 75–79. [Google Scholar]
- Suckling, D.M.; Kean, J.M.; Stringer, L.D.; Cáceres-Barrios, C.; Hendrichs, J.; Reyes-Flores, J.; Dominiak, B.C. Eradication of tephritid fruit fly pest populations: Outcomes and prospects. Pest. Manag. Sci. 2014, 72, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Concha, C.; Palavesam, A.; Guerrero, F.D.; Sagel, A.; Li, F.; Osborne, J.A.; Hernandez, Y.; Pardo, T.; Quintero, G.; Vasquez, M. A transgenic male-only strain of the New World screwworm for an improved control program using the sterile insect technique. BMC Biol. 2016, 14, 72. [Google Scholar] [CrossRef]
- FAO/IAEA. Model Business Plan for a Sterile Insect Production Facility; IAEA: Viena, Austria, 2008. [Google Scholar]
- Koyama, J.; Kakinohana, H.; Miyatake, T. Eradication of the melon fly, Bactrocera cucurbitae, in Japan: Importance of behavior, ecology, genetics, and evolution. Annu. Rev. Entomol. 2004, 49, 331–349. [Google Scholar] [CrossRef]
- Hendrichs, J.; Franz, G.; Rendon, P. Increased effectiveness and applicability of the sterile insect technique through male-only releases for control of Mediterranean fruit flies during fruiting seasons. J. Appl. Entomol. 1995, 119, 371–377. [Google Scholar] [CrossRef]
- Vera, T.; Abraham, S.; Oviedo, A.; Willink, E. Demographic and quality control parameters of Anastrepha fraterculus (Diptera: Tephritidae) maintained under artificial rearing. Fla. Entomol. 2007, 90, 53–57. [Google Scholar] [CrossRef]
- Nash, W.J.; Chapman, T. Effect of Dietary Components on Larval Life History Characteristics in the Medfly Ceratitis capitata: (Diptera, Tephritidae). PLoS ONE 2014, 9, e86029. [Google Scholar] [CrossRef]
- Neto, S.; Santos, T.R.D.O.; Dias, V.S.; Joachim-Bravo, I.S.; Benevides, L.D.J.; Benevides, C.M.D.J.; Silva, M.V.L.; dos Santos, D.C.C.; Virgínio, J.; Oliveira, G.B. Mass-rearing of Mediterranean fruit fly using low-cost yeast products produced in Brazil. Sci. Agric. 2012, 69, 364–369. [Google Scholar] [CrossRef]
- Morelli, R.; Costa, K.Z.; Fagioni, K.M.; Costa, M.d.L.Z.; Nascimento, A.S.d.; Pimentel, R.M.d.A.; Walder, J.M.M. New protein sources in adults diet for mass-rearing of Anastrepha fraterculus (Diptera: Tephritidae). Braz. Arch. Biol. Technol. 2012, 55, 827–833. [Google Scholar] [CrossRef]
- Chang, C.L. Fruit fly liquid larval diet technology transfer and update. J. Appl. Entomol. 2009, 133, 164–173. [Google Scholar] [CrossRef]
- Barry, J.D.; McInnis, D.O.; Gates, D.; Morse, J.G. Effects of irradiation on Mediterranean fruit flies (Diptera: Tephritidae): Emergence, survivorship, lure attraction, and mating competition. J. Econ. Entomol. 2003, 96, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Guerfali, M.M.s.; Parker, A.; Fadhl, S.; Hemdane, H.; Raies, A.; Chevrier, C. Fitness and Reproductive Potential of Irradiated Mass-Reared Mediterranean Fruit Fly Males Ceratitis capitata (Diptera: Tephritidae): Lowering Radiation Doses. Fla. Entomol. 2011, 94, 1042–1050. [Google Scholar] [CrossRef]
- Kraaijeveld, K.; Chapman, T. Effects of male sterility on female remating in the Mediterranean fruitfly, Ceratitis capitata. Proc. Biol Sci 2004, 271 (Suppl. 4), S209–S211. [Google Scholar] [CrossRef] [PubMed]
- Lauzon, C.R.; Potter, S.E. Description of the irradiated and nonirradiated midgut of Ceratitis capitata Wiedemann (Diptera: Tephritidae) and Anastrepha ludens Loew (Diptera: Tephritidae) used for sterile insect technique. J. Pest Sci. 2012, 85, 217–226. [Google Scholar] [CrossRef]
- Lux, S.A.; Vilardi, J.C.; Liedo, P.; Gaggl, K.; Calcagno, G.E.; Munyiri, F.N.; Vera, M.T.; Manso, F. Effects of irradiation on the courtship behavior of medfly (Diptera, Tephritidae) mass reared for the sterile insect technique. Fla. Entomol. 2002, 102–112. [Google Scholar] [CrossRef]
- Rull, J.; Encarnacion, N.; Birke, A. Mass rearing history and irradiation affect mating performance of the male fruit fly, Anastrepha obliqua. J. Insect Sci 2012, 12, 45. [Google Scholar] [CrossRef]
- San Andres, V.; Ortego, F.; Castañera, P. Effects of gamma-irradiation on midgut proteolytic activity of the mediterranean fruit fly, Ceratitis capitata (Diptera: Tephritidae). Arch. Insect Biochem. Physiol. 2007, 65, 11–19. [Google Scholar] [CrossRef]
- Ant, T.; Koukidou, M.; Rempoulakis, P.; Gong, H.F.; Economopoulos, A.; Vontas, J.; Alphey, L. Control of the olive fruit fly using genetics-enhanced sterile insect technique. BMC Biol. 2012, 10, 51. [Google Scholar] [CrossRef]
- Gorman, K.; Young, J.; Pineda, L.; Márquez, R.; Sosa, N.; Bernal, D.; Torres, R.; Soto, Y.; Lacroix, R.; Naish, N. Short-term suppression of Aedes aegypti using genetic control does not facilitate Aedes albopictus. Pest. Manag. Sci. 2015, 72, 618–628. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.F.; Nimmo, D.; McKemey, A.R.; Kelly, N.; Scaife, S.; Donnelly, C.A.; Beech, C.; Petrie, W.D.; Alphey, L. Field performance of engineered male mosquitoes. Nat. Biotechnol. 2011, 29, 1034–1037. [Google Scholar] [CrossRef] [PubMed]
- Harvey-Samuel, T.; Morrison, N.I.; Walker, A.S.; Marubbi, T.; Yao, J.; Collins, H.L.; Gorman, K.; Davies, T.G.; Alphey, N.; Warner, S.; et al. Pest control and resistance management through release of insects carrying a male-selecting transgene. BMC Biol 2015, 13, 49. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, R.; McKemey, A.R.; Raduan, N.; Wee, L.K.; Ming, W.H.; Ney, T.G.; AA, S.R.; Salman, S.; Subramaniam, S.; Nordin, O. Open field release of genetically engineered sterile male Aedes aegypti in Malaysia. PLoS ONE 2012, 7, e42771. [Google Scholar] [CrossRef] [PubMed]
- Rendon, P.; McInnis, D.; Lance, D.; Stewart, J. Medfly (Diptera: Tephritidae) genetic sexing: Large-scale field comparison of males-only and bisexual sterile fly releases in Guatemala. J. Econ. Entomol. 2004, 97, 1547–1553. [Google Scholar] [CrossRef]
- Leftwich, P.T.; Koukidou, M.; Rempoulakis, P.; Gong, H.F.; Zacharopoulou, A.; Fu, G.; Chapman, T.; Economopoulos, A.; Vontas, J.; Alphey, L. Genetic elimination of field-cage populations of Mediterranean fruit flies. Proc. Biol Sci 2014, 281. [Google Scholar] [CrossRef]
- Tanaka, N.; Steiner, L.; Ohinata, K.; Okamoto, R. Low-cost larval rearing medium for mass production of oriental and Mediterranean fruit flies. J. Econ. Entomol. 1969, 62, 967–968. [Google Scholar] [CrossRef]
- FAO/IAEA/USDA. Product Quality Control for Sterile Mass-Reared and Released Tephritid Fruit Flies, Version 6.0; International Atomic Energy Agency: Vienna, Austria, 2014; p. 164. [Google Scholar]
- Enkerlin, W.; Gutiérrez-Ruelas, J.M.; Cortes, A.V.; Roldan, E.C.; Midgarden, D.; Lira, E.; López, J.L.Z.; Hendrichs, J.; Liedo, P.; Arriaga, F.J.T. Area Freedom in Mexico from Mediterranean Fruit Fly (Diptera: Tephritidae): A Review of Over 30 Years of a Successful Containment Program Using an Integrated Area-Wide SIT Approach. Florida Entomol. 2015, 98, 665–681. [Google Scholar] [CrossRef]
- Rempoulakis, P.; Taret, G.; Haq, I.U.; Wornayporn, V.; Ahmad, S.; Tomas, U.S.; Dammalage, T.; Gembinsky, K.; Franz, G.; Cáceres, C. Evaluation of Quality Production Parameters and Mating Behavior of Novel Genetic Sexing Strains of the Mediterranean Fruit Fly Ceratitis capitata (Wiedemann) (Diptera: Tephritidae). PLoS ONE 2016, 11, e0157679. [Google Scholar] [CrossRef]
- Chapman, T.; Takahisa, M.; Smith, H.K.; Partridge, L. Interactions of mating, egg production and death rates in females of the Mediterranean fruitfly, Ceratitis capitata. Proc. R. Soc. Lond. B Biol. Sci. 1998, 265, 1879–1894. [Google Scholar] [CrossRef]
- Lance, D.R.; McInnis, D.O.; Rendon, P.; Jackson, C.G. Courtship among sterile and wild Ceratitis capitata (Diptera: Tephritidae) in field cages in Hawaii and Guatemala. Ann. Entomol. Soc. Am. 2000, 93, 1179–1185. [Google Scholar] [CrossRef]
- Robinson, A.S.; Cayol, J.P.; Hendrichs, J. Recent findings on medfly sexual behavior: Implications for SIT. Fla. Entomol. 2002, 85, 171–181. [Google Scholar] [CrossRef]
- Gordillo, J.C.D. Mass rearing methods for fruit fly. In Proceedings of the South American Fruit Fly, Anastrepha Fraterculus (Wied.), Advances in Artificial Rearing, Taxonomic Status and Biological Studies, Vina del Mal, Chile, 1–2 November 1996; pp. 59–71. [Google Scholar]
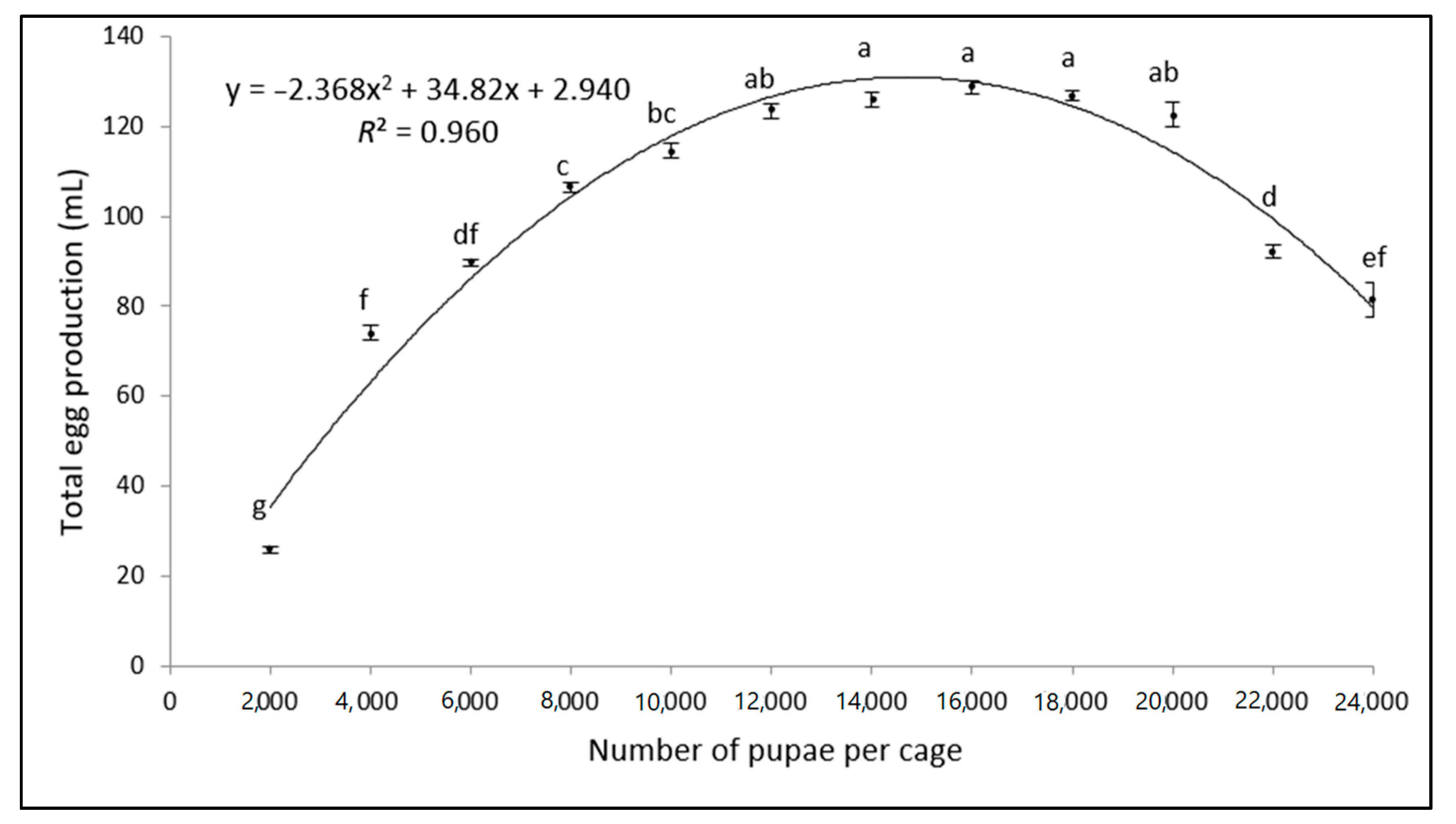
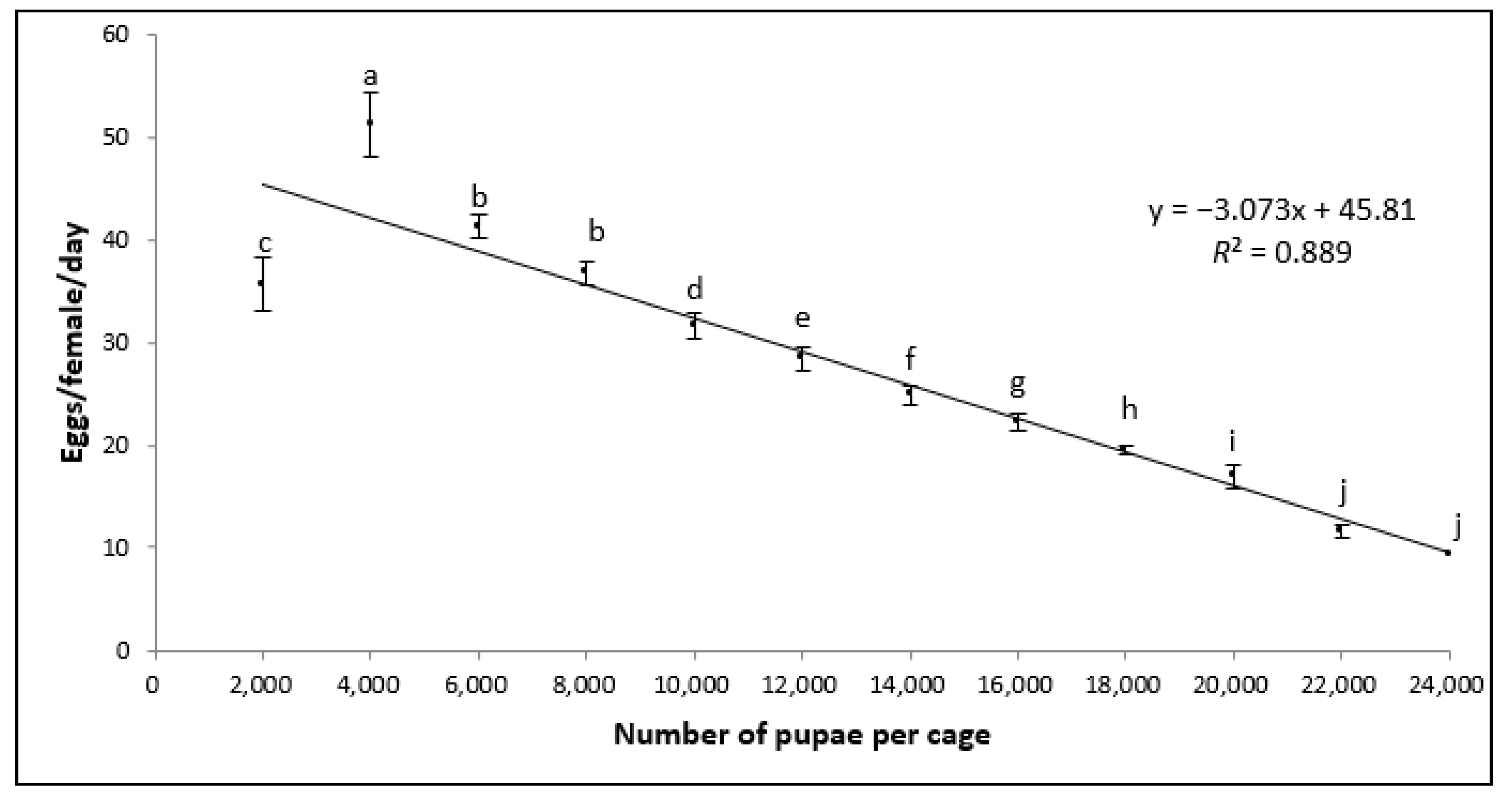
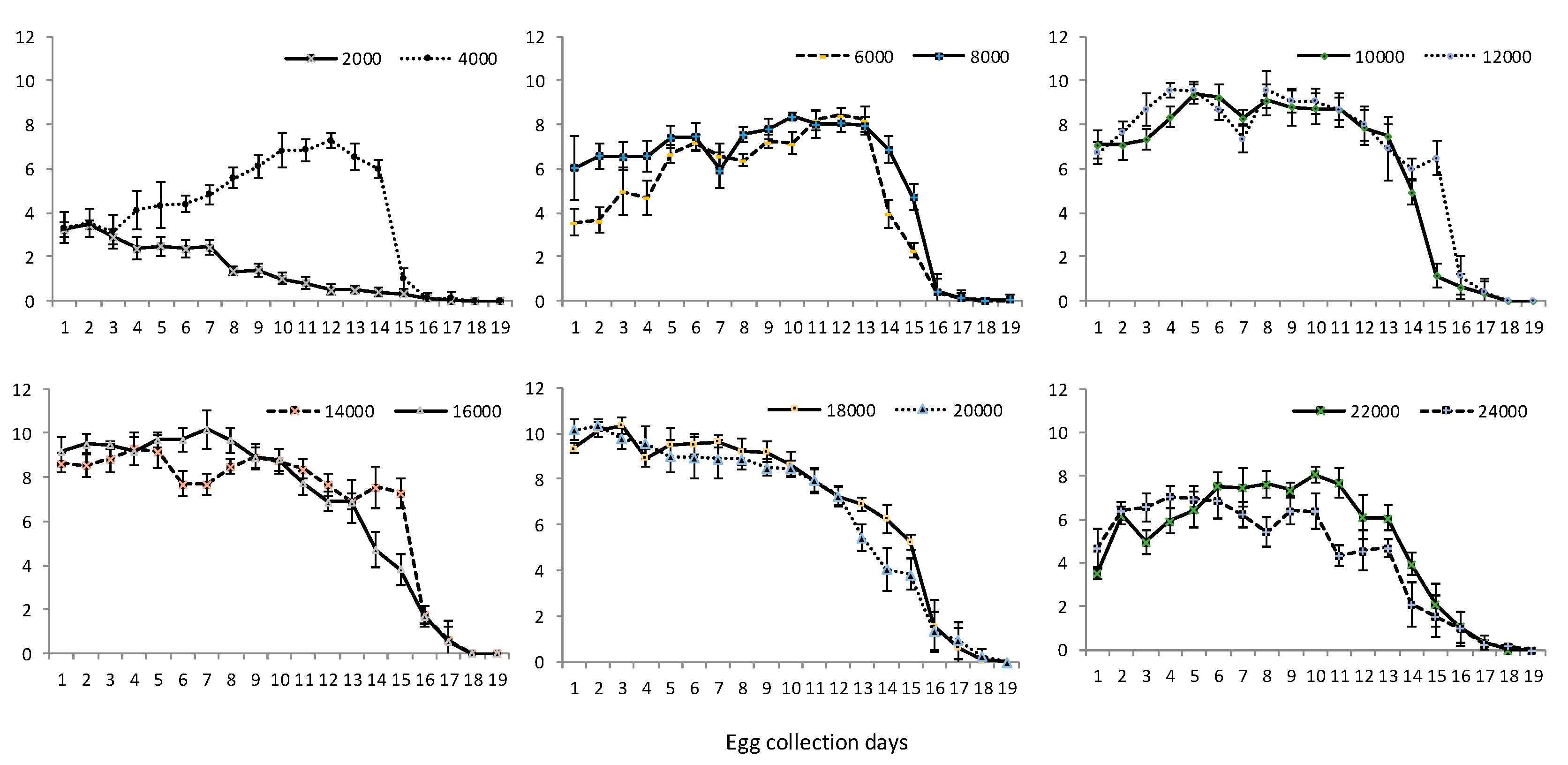
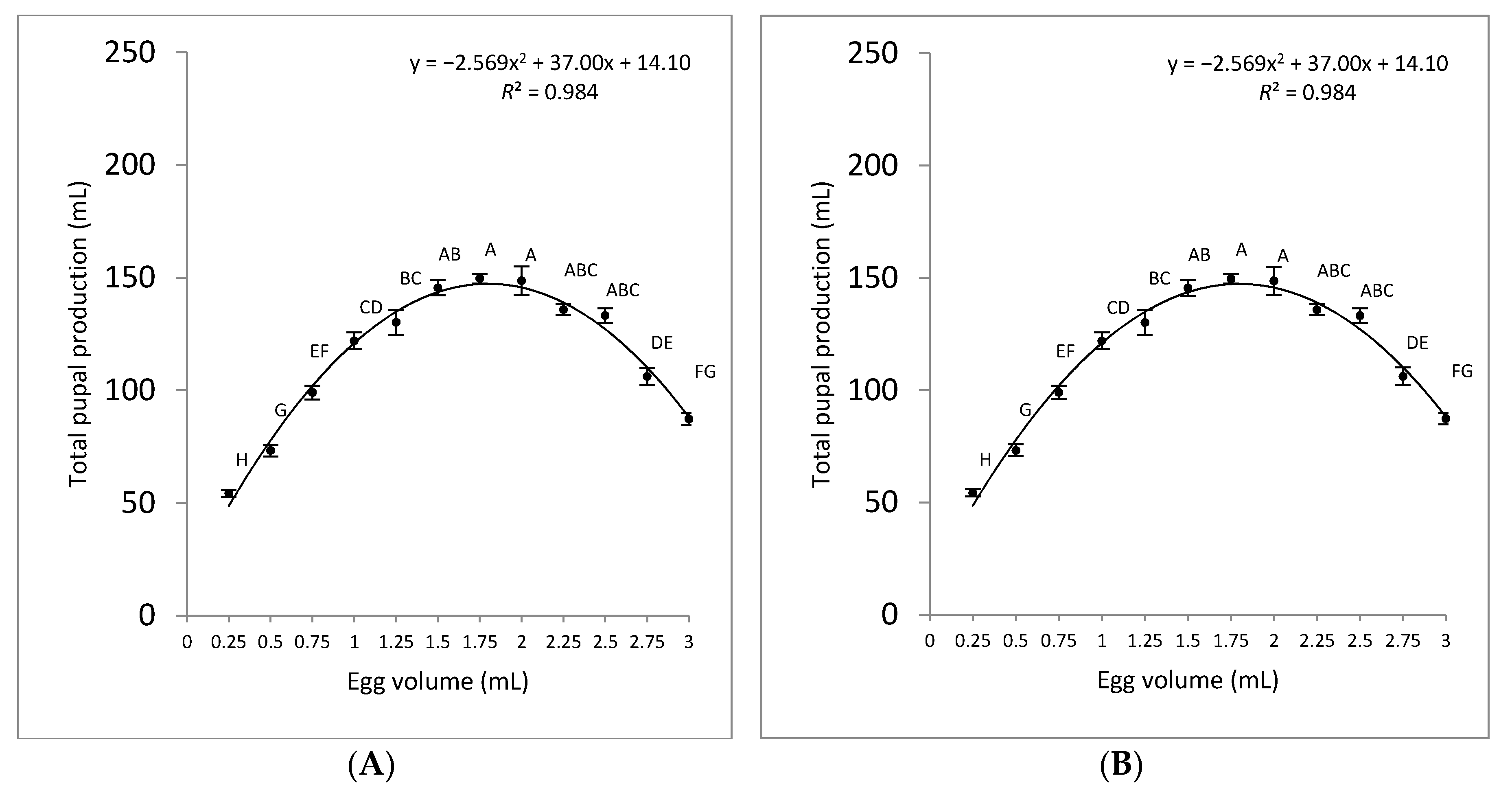
| Volume of Eggs (mL) | Total Pupal Weight (g) | Weight per 100 Pupae (g) | Number of Pupae | Pupal Recovery (%) |
|---|---|---|---|---|
| 0.25 | 39.22 ± 1.13 g | 0.90 ± 0.00 a | 4358 ± 125 g | 69.97 ± 4.78 a |
| 0.5 | 81.06 ± 1.92 de | 0.90 ± 0.00 a | 9006 ± 214 de | 77.36 ± 5.30 a |
| 0.75 | 98.12 ± 2.71 abcd | 0.90 ± 0.00 a | 10,902 ± 301 cd | 59.81 ± 1.15 b |
| 1 | 105.35 ± 3.44 abc | 0.83 ± 0.02 ab | 12,662 ± 425 bc | 51.72 ± 2.89 bc |
| 1.25 | 116.76 ± 9.47 a | 0.72 ± 0.02 c | 16,334 ± 1402 a | 59.65 ± 5.73 b |
| 1.5 | 118.80 ± 10.80 a | 0.75 ± 0.02 bc | 15,787 ± 1294 ab | 45.43 ± 3.84 cd |
| 1.75 | 117.58 ± 4.41 a | 0.75 ± 0.02 bc | 15,743 ± 770 ab | 40.66 ± 1.95 de |
| 2 | 113.12 ± 6.02 ab | 0.72 ± 0.02 c | 15,831 ± 944 ab | 36.42 ± 4.16 e |
| 2.25 | 92.42 ± 1.51 bcd | 0.80 ± 0.04 abc | 9315 ± 199 de | 18.73 ± 0.40 f |
| 2.5 | 83.50 ± 1.58 cde | 0.78 ± 0.03 bc | 8784 ± 171 def | 15.90 ± 1.31 f |
| 2.75 | 64.27 ± 0.75 ef | 0.82 ± 0.03 abc | 6762 ± 68 efg | 11.13 ± 1.11 f |
| 3 | 52.57 ± 1.64 fg | 0.75 ± 0.02 bc | 5717 ± 133 fg | 8.62 ± 0.20 f |
| Volume of Eggs (mL) | Total Pupal Weight (g) | Weight per 100 Pupae (g) | Number of Pupae | Pupal Recovery (%) |
|---|---|---|---|---|
| 0.25 | 29.68 ± 0.99 e | 0.90 ± 0.00 a | 3298 ± 110 g | 59.69 ± 1.99 a |
| 0.5 | 40.70 ± 1.42 de | 0.90 ± 0.00 a | 4522 ± 158 fg | 40.93 ± 1.43 b |
| 0.75 | 60.47 ± 4.06 bc | 0.90 ± 0.00 a | 6719 ± 451 de | 40.53 ± 2.72 bc |
| 1 | 69.07 ± 2.71 ab | 0.85 ± 0.02 ab | 8151 ± 378 cd | 36.88 ± 1.71 bcd |
| 1.25 | 72.22 ± 4.11 ab | 0.77 ± 0.02 bc | 9399 ± 374 bc | 34.02 ± 1.35 cde |
| 1.5 | 81.93 ± 2.06 a | 0.77 ± 0.02 bc | 10,725 ± 376 ab | 32.35 ± 1.13 def |
| 1.75 | 82.87 ± 2.12 a | 0.72 ± 0.02 c | 11,585 ± 356 a | 29.96 ± 0.92 ef |
| 2 | 84.13 ± 3.51 a | 0.70 ± 0.00 c | 12,018 ± 502 a | 27.19 ± 1.14 f |
| 2.25 | 80.02 ± 1.41 a | 0.78 ± 0.03 bc | 8064 ± 188 cd | 16.22 ± 0.38 g |
| 2.5 | 85.55 ± 8.30 a | 0.78 ± 0.03 bc | 8576 ± 71 c | 15.52 ± 0.13 g |
| 2.75 | 62.13 ± 2.47 bc | 0.75 ± 0.02 c | 6200 ± 343 e | 10.20 ± 0.56 gh |
| 3 | 51.32 ± 1.47 cd | 0.77 ± 0.02 bc | 5303 ± 136 ef | 8.00 ± 0.21 h |
| Criteria | tsl | OX3864A |
|---|---|---|
| Colony size (millions of adults) | 22 | 12 |
| Colony replacement (millions of pupae) | 60 | 16 |
| No. of cages for male only production | 165 | 50 |
| Egging room (m2); male only | 437 | 133 |
| No. of cages for colony maintenance | 24 | 4 |
| Total no. of cages | 189 | 54 |
| Egging room (m2); colony | 78 | 11 |
| Adult diet (sugar) (kg/day) | 24 | 9 |
| Adult diet (yeast hydrolysate) (kg/day) | 8 | 3 |
| Workers (colony + filter) | 9 | 3 |
| Total cost per million males | $510 | $207 |
| Factory and equipment | $124 | $119 |
| Consumables | $251 | $33.80 |
| Labour | $111 | $44 |
| Space | $23 | $9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elaini, R.; Asadi, R.; Naish, N.; Koukidou, M.; Ahmed, M. Evaluation of Rearing Parameters of a Self-Limiting Strain of the Mediterranean Fruit Fly, Ceratitis capitata (Diptera: Tephritidae). Insects 2020, 11, 663. https://doi.org/10.3390/insects11100663
Elaini R, Asadi R, Naish N, Koukidou M, Ahmed M. Evaluation of Rearing Parameters of a Self-Limiting Strain of the Mediterranean Fruit Fly, Ceratitis capitata (Diptera: Tephritidae). Insects. 2020; 11(10):663. https://doi.org/10.3390/insects11100663
Chicago/Turabian StyleElaini, Rachid, Romisa Asadi, Neil Naish, Martha Koukidou, and Mazih Ahmed. 2020. "Evaluation of Rearing Parameters of a Self-Limiting Strain of the Mediterranean Fruit Fly, Ceratitis capitata (Diptera: Tephritidae)" Insects 11, no. 10: 663. https://doi.org/10.3390/insects11100663
APA StyleElaini, R., Asadi, R., Naish, N., Koukidou, M., & Ahmed, M. (2020). Evaluation of Rearing Parameters of a Self-Limiting Strain of the Mediterranean Fruit Fly, Ceratitis capitata (Diptera: Tephritidae). Insects, 11(10), 663. https://doi.org/10.3390/insects11100663





