Nest Modification Protects Immature Stages of the Japanese Orchard Bee (Osmia cornifrons) from Invasion of a Cleptoparasitic Mite Pest
Abstract
:1. Introduction
2. Materials and Methods
2.1. Osmia Cornifrons Nests
2.2. Data Collection and Analysis
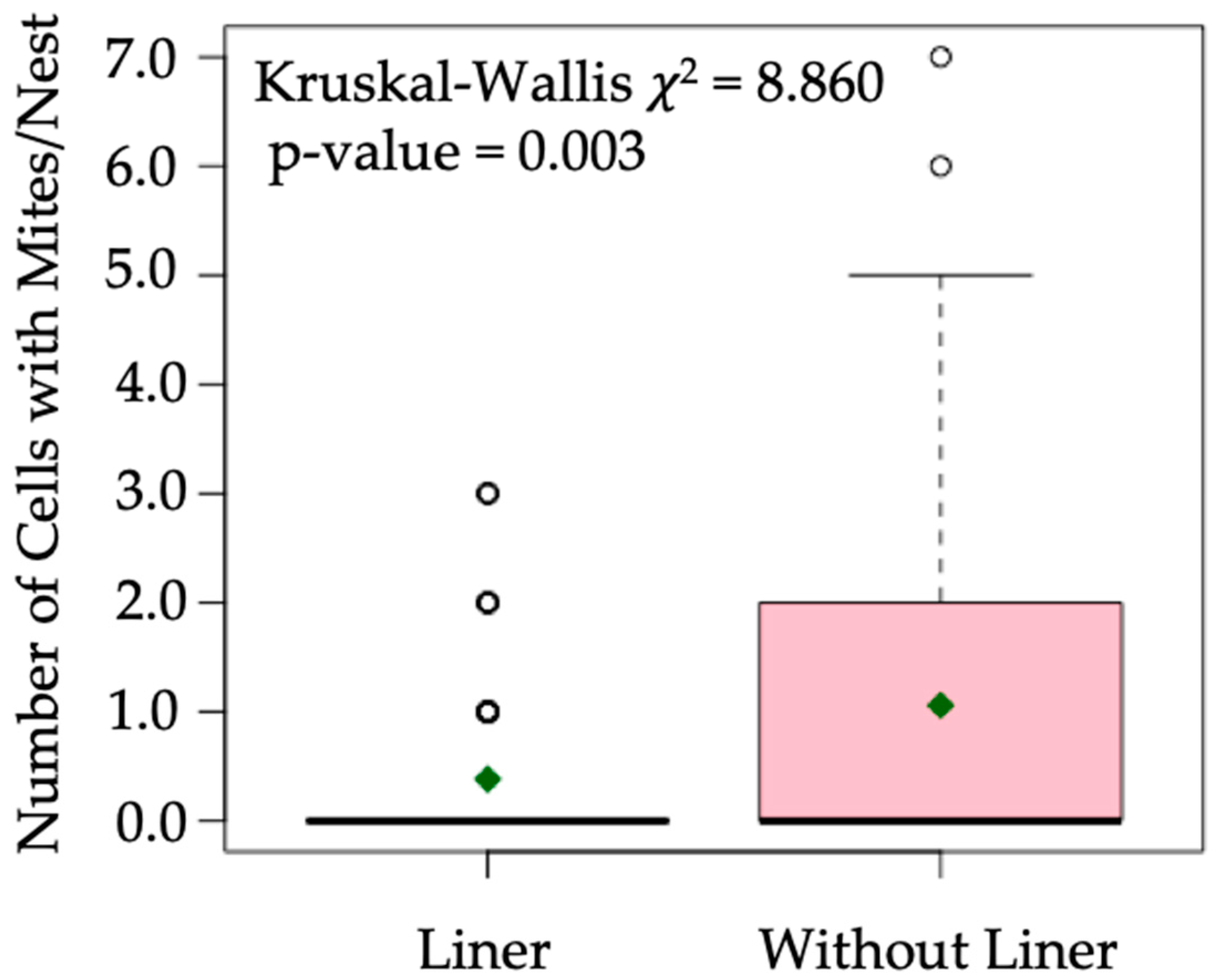
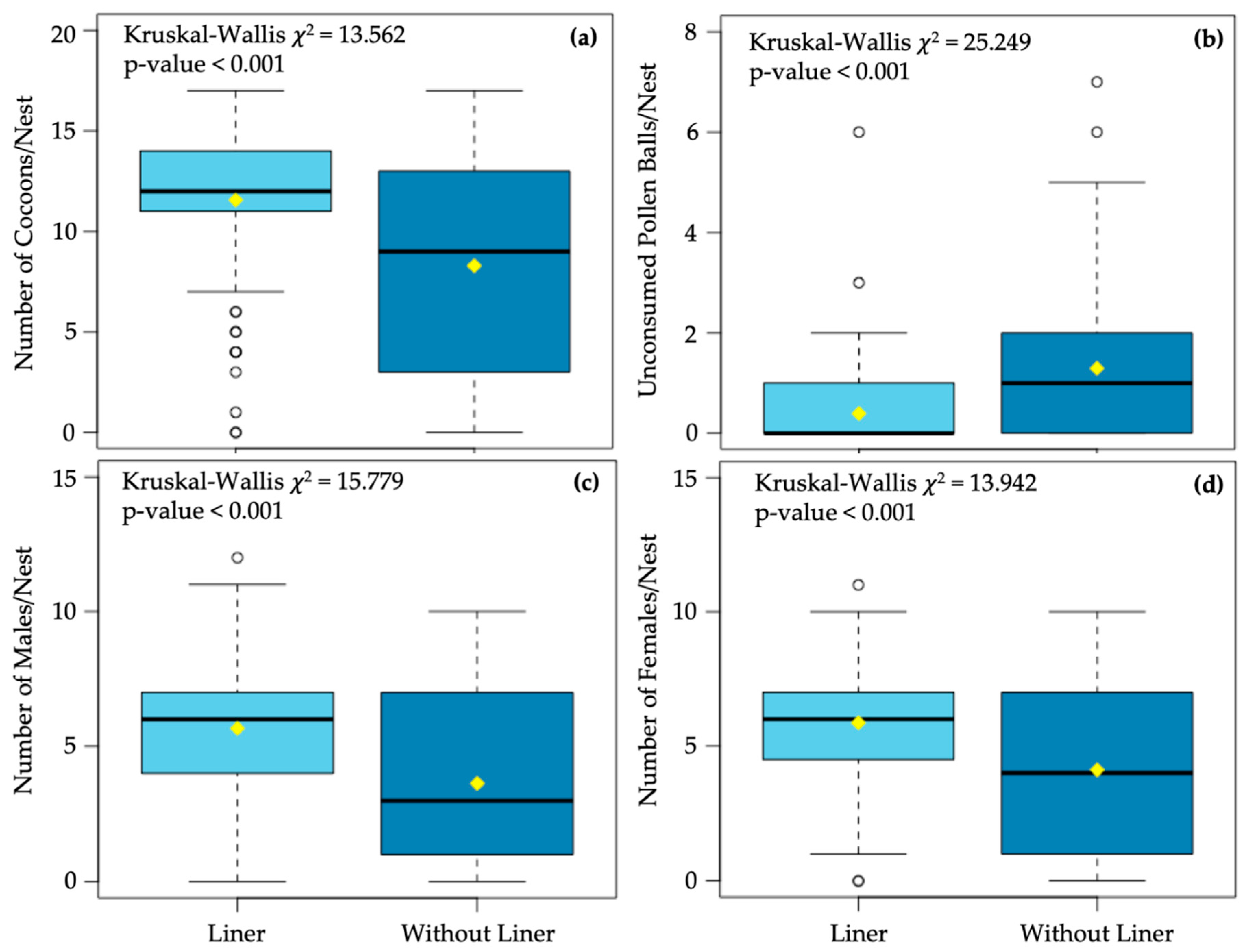
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yamada, Y.; Oyama, N.; Sekita, N.; Shirasaki, S.; Tsugawa, C. The ecology of the megachilid bee Osmia cornifrons and its utilization for apple pollination. Bull. Aomori Apple Exp. Stn. 1971, 26, 39–77. [Google Scholar]
- Xu, H.-L.; Yang, L.-I.; Kwon, Y.J. Current status on the utilization of Osmia bees as pollinators of fruit trees in China (Hymenoptera: Megachilidae). J. Apic. 1995, 10, 111–116. [Google Scholar]
- Biddinger, D.; Rajotte, E.; Joshi, N.; Ritz, A. Wild bees as alternative pollinators. Fruit Times 2011, 30, 1–4. [Google Scholar]
- Joshi, N.K.; Otieno, M.; Rajotte, E.G.; Fleischer, S.J.; Biddinger, D.J. Proximity to woodland and landscape structure drives pollinator visitation in apple orchard ecosystem. Front. Ecol. Evol. 2016, 4, 38. [Google Scholar] [CrossRef] [Green Version]
- Park, M.G.; Joshi, N.K.; Rajotte, E.J.; Biddinger, D.J.; Blitzer, E.J.; Losey, J.E.; Danforth, B.N. Apple grower pollination practices and perceptions of alternative pollinators in New York and Pennsylvania. Renew. Agric. Food Syst. 2018, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Yamada, M.; Kawashima, K.; Aizu, H. Population dynamics of the horn faced bee, Osmia cornifrons Radoszkowski, with a special reference to the population management. Bull. Aomori Apple Exp. Stn. 1984, 21, 23–92. [Google Scholar]
- Yamada, M. Control of the chaetodactylus mite, Chaetodactylus nipponicus Kurosa, an important mortality agent of hornfaced osmia bee, Osmia cornifrons Radoszkowski. Bull. Aomori Apple Exp. Stn. 1990, 26, 39–77. [Google Scholar]
- Park, Y.L.; Kondo, V.; White, J.; West, T.; McConnell, B.; McCutcheon, T. Nest-to-nest dispersal of Chaetodactylus krombeini (Acari, Chaetodactylidae) associated with Osmia cornifrons (Hym., Megachilidae). J. Appl. Entomol. 2009, 133, 174–180. [Google Scholar] [CrossRef]
- McKinney, M.I.; Park, Y.L. Distribution of Chaetodactylus krombeini (Acari: Chaetodactylidae) within Osmia cornifrons (Hymenoptera: Megachilidae) nests: Implications for population management. Exp. Appl. Acarol. 2013, 60, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Qu, D.; Maeta, Y.; Goubara, M.; Nakatsuka, K.J.; Kozo, J.; Kenji, K. Reproductive strategy in the two species of cleptoparasitic astigmatid mites, Chaetodactylus nipponicus and Tortonia sp. (Acari: Chaetodactylidae and Suidasiidae), infesting Osmia cornifrons (Hymenoptera: Megachilidae). I. Invasion/infestation patterns and partial use of the host food. Jpn. J. Entomol. New Ser. 2002, 5, 121–141. [Google Scholar]
- Qu, D.; Maeta, Y.; Nakatsuka, K.J.; Kenji, K.; Goubara, M. Reproductive strategy in the two species of cleptoparasitic astigmatid mites, Chaetodactylus nipponicus and Tortonia sp. (Acari: Chaetodactylidae and Suidasiidae), infesting Osmia cornifrons (Hymenoptera: Megachilidae) II. Life history, phoretic positions, development and reproductivity. Jpn. J. Entomol. New Ser. 2003, 6, 55–73. [Google Scholar]
- Van Asselt, L. Observations on the life cycle of Chaetodactylus osmiae (Dufour, 1839) (Acari: Chaetodactylidae) parasitic on the solitary bee, Osmia rufa (L.), 1758 (Insecta: Hymenoptera) in Belgium. Int. J. Acarol. 2000, 26, 221–228. [Google Scholar] [CrossRef]
- Sugden, E. Mitey Bees: The Blue Orchard Bee’s Mite Pest. Scarabogram 2000, 241, 2–3. [Google Scholar]
- OConnor, B.; Klimov, P. Chaetodactylus Krombeini Baker. 1962. Available online: http://insects.ummz.lsa.umich.edu/beemites/Species_Accounts/Chaetodactylus_krombeini.htm (accessed on 10 August 2018).
- Baker, E.W. Natural history of Plummers Island, Maryland. XV. Descriptions of the stages of Chaetodactylus krombeini, new species, a mite associated with the bee, Osmia lignaria Say (Acarina: Chaetodactylidae). Proc. Biol. Soc. Wash. 1962, 75, 227–236. [Google Scholar]
- Krombein, K.V. Natural history of Plummers Island, Maryland. XVI. Biological Notes on Chaetodactylus krombeini Baker, a parasitic mite of the megachilid bee, Osmia (Osmia) lignaria Say (Acarina, Chaetodactylidae). Proc. Biol. Soc. Wash. 1962, 75, 237–250. [Google Scholar]
- Eves, J.D. Biology of Monodontomerus Obscurus Westwood, a Parasite of the Alfalfa Leafcutting Bee, Megachile Rotundata (Fabricius) (Hymenoptera: Torymidae; Megachilidae). Master’s Thesis, Washington State University, Pullman, WA, USA, 1969; p. 59. [Google Scholar]
- Krunic, M.; Stanisaljevic, L.; Pinzauti, M.; Felicioli, A. The accompanying fauna of Osmia cornuta and Osmia rufa and effective measures of protection. Bull. Insectol. 2005, 58, 141–152. [Google Scholar]
- White, J.B.; Park, Y.L.; West, T.P.; Tobin, P.C. Assessment of potential fumigants to control Chaetodactylus krombeini (Acari: Chaetodactylidae) associated with Osmia cornifrons (Hymenoptera: Megachilidae). J. Econ. Entomol. 2009, 102, 2090–2095. [Google Scholar] [CrossRef] [PubMed]
- Bosch, J.; Kemp, W.P. Developing and establishing bee species as crop pollinators: The example of Osmia spp. (Hymenoptera: Megachilidae) and fruit trees. Bull. Entomol. Res. 2002, 92, 3–16. [Google Scholar] [PubMed]
- Strickler, K.; Mills, J. Loose Cell Management Systems for Solitary Bees. 2002. Available online: http://www.pollinatorparadise.com/Binderboards/Osmiabb.htm (accessed on 11 August 2018).
- Team, R.C. R: A Language and Environment for Statistical Computing; Core Team: Vienna, Austria, 2013. [Google Scholar]
- Bosch, J.; Kemp, W.P. How to Manage the Blue Orchard Bee as an Orchard Pollinator; Handbook No. 5; Sustainable Agricultural Network: Beltsville, MD, USA, 2001. [Google Scholar]
- McKinney, M.I.; Park, Y.L. Nesting activity and behavior of Osmia cornifrons (Hymenoptera: Megachilidae) elucidated using videography. Psyche J. Entomol. 2012, 2012. [Google Scholar] [CrossRef] [Green Version]
- Byers, G.W. Competitive supersedure by Monobia quadridens in nests of Osmia lignaria. J. Kans. Entomol. Soc. 1972, 45, 235–238. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joshi, N.K.; Naithani, K.; Biddinger, D.J. Nest Modification Protects Immature Stages of the Japanese Orchard Bee (Osmia cornifrons) from Invasion of a Cleptoparasitic Mite Pest. Insects 2020, 11, 65. https://doi.org/10.3390/insects11010065
Joshi NK, Naithani K, Biddinger DJ. Nest Modification Protects Immature Stages of the Japanese Orchard Bee (Osmia cornifrons) from Invasion of a Cleptoparasitic Mite Pest. Insects. 2020; 11(1):65. https://doi.org/10.3390/insects11010065
Chicago/Turabian StyleJoshi, Neelendra K., Kusum Naithani, and David J. Biddinger. 2020. "Nest Modification Protects Immature Stages of the Japanese Orchard Bee (Osmia cornifrons) from Invasion of a Cleptoparasitic Mite Pest" Insects 11, no. 1: 65. https://doi.org/10.3390/insects11010065
APA StyleJoshi, N. K., Naithani, K., & Biddinger, D. J. (2020). Nest Modification Protects Immature Stages of the Japanese Orchard Bee (Osmia cornifrons) from Invasion of a Cleptoparasitic Mite Pest. Insects, 11(1), 65. https://doi.org/10.3390/insects11010065





