A Neonicotinoid Insecticide at a Rate Found in Nectar Reduces Longevity but Not Oogenesis in Monarch Butterflies, Danaus plexippus (L.). (Lepidoptera: Nymphalidae)
Abstract
1. Introduction
2. Materials and Methods
2.1. Longevity
2.2. Oocyte Development
2.3. Data Analysis
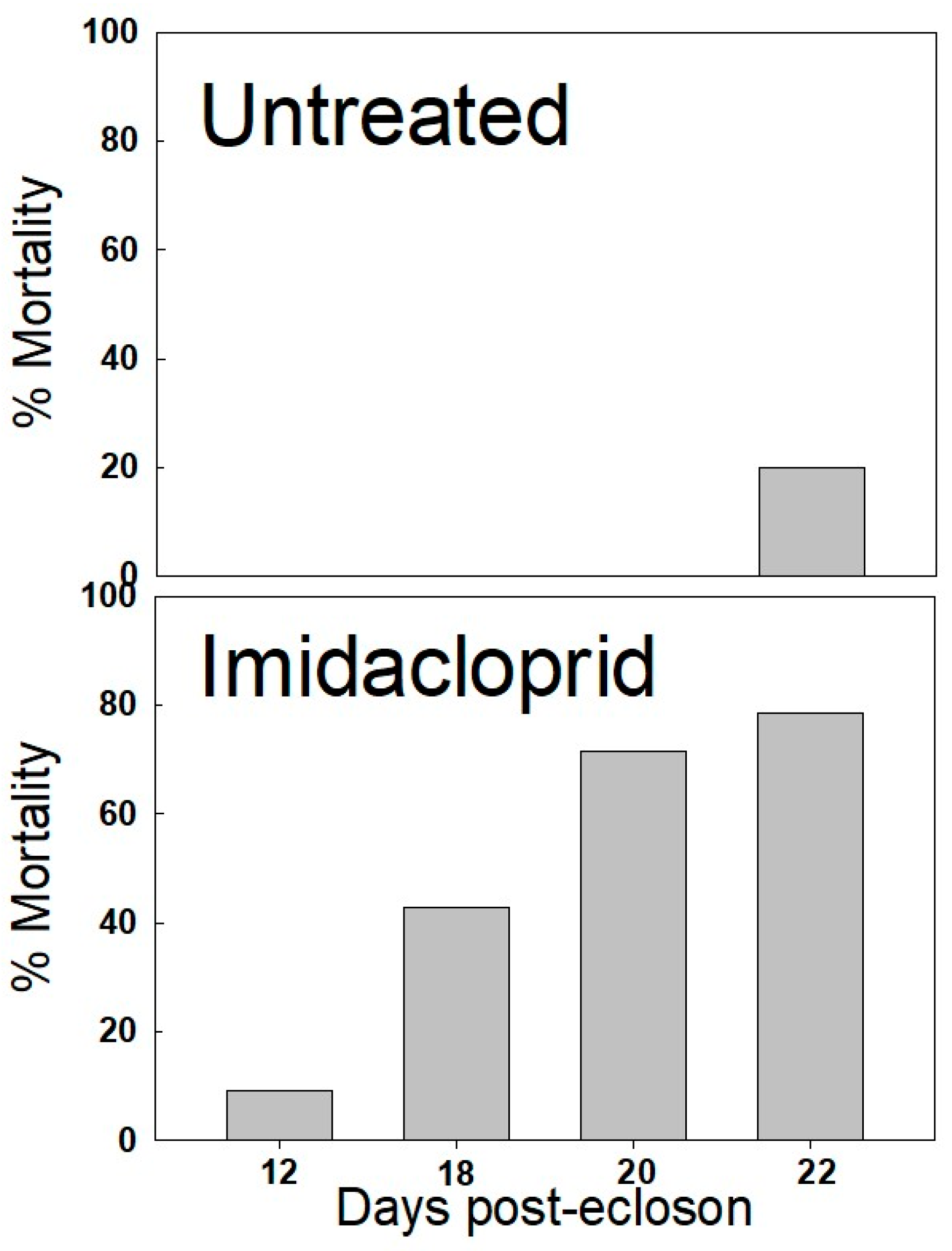
3. Results
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Brower, L.P.; Taylor, O.R.; Williams, E.H.; Slayback, D.A.; Zubieta, R.R.; Ramirez, R.R. Decline of monarch butterflies overwintering in Mexico: Is the migratory phenomenon at risk? Insect Conserv. Divers. 2012, 5, 95–100. [Google Scholar] [CrossRef]
- Semmens, B.X.; Semmens, D.J.; Thogmartin, W.E.; Wiederholt, R.; Lopez-Hoffman, L.; Diffendorfer, J.E.; Pleasants, J.M.; Oberhauser, K.M.; Yatlor, O.R. Quasi-extinction risk and population targets for the eastern migratory population of monarch butterflies (Danaus plexippus). Sci. Rep. 2016, 6, 23265. [Google Scholar] [CrossRef] [PubMed]
- Stenoien, C.; Nail, K.R.; Zalucki, J.M.; Parry, H.; Oberhauser, K.S.; Zalucki, M.P. Monarchs in decline: A collateral landscape-level effect of modern agriculture. Insect Sci. 2016. [Google Scholar] [CrossRef] [PubMed]
- Marini, L.; Zalucki, M.P. Density dependence in the declining population of the monarch butterfly. Sci. Rep. 2017, 7, 13957. [Google Scholar] [CrossRef] [PubMed]
- Pleasants, J.M.; Zalucki, M.P.; Oberhauser, K.S.; Brower, L.P.; Taylor, O.R.; Thogmartin, W.E. Interpreting surveys to estimate the size of the monarch butterfly population: Pitfalls and prospects. PLoS ONE 2017, 12, e0181245. [Google Scholar] [CrossRef] [PubMed]
- Schultz, C.B.; Brown, L.M.; Pelton, E.; Crone, E.E. Citizen science monitoring demonstrates dramatic declines of monarch butterflies in western North America. Biol. Conserv. 2017, 214, 343–345. [Google Scholar] [CrossRef]
- Thogmartin, W.E.; Lopez-Hoffman, L.; Rohweder, J.; Diffendorfer, J.; Drum, R.; Semmens, D. Restoring monarch butterfly habitat in the Midwestern US: All hands on deck. Environ. Res. Lett. 2017, 12, 074005. [Google Scholar] [CrossRef]
- Pelton, E.; Schultz, C.B.; Jepsen, S.J.; Hoffman Black, S.; Crone, E.E. Western monarch population plummets: Status, probable causes and recommended conservation actions. Front. Ecol. Evol. 2019. [Google Scholar] [CrossRef]
- Hartzler, R.G. Reduction in common milkweed (Asclepias syriaca) occurrence in Iowa cropland from 1999 to 2009. Crop Prot. 2010, 29, 1542–1544. [Google Scholar] [CrossRef]
- Pleasants, J.M.; Oberhauser, K.S. Milkweed loss in agricultural fields because of herbicide use: Effect on the monarch butterfly population. Insect Conserv. Divers. 2013, 6, 135–144. [Google Scholar] [CrossRef]
- Zaya, D.N.; Pearse, L.S.; Spyreas, G. Long term trends in Midwestern milkweed abundances and their relevance to monarch butterfly declines. Bioscience 2017, 67, 343–356. [Google Scholar] [CrossRef]
- Brower, L.P. Understanding and misunderstanding the migration of the monarch butterfly (Nymphalidae) in North America. J. Lep. Soc. 1995, 49, 304–385. [Google Scholar]
- Wassenaar, L.L.; Hobson, K.A. Natal origins of migratory monarch butterflies at wintering colonies in Mexico: New isotopic evidence. Proc. Natl. Acad. Sci. USA 1998, 95, 15436–15439. [Google Scholar] [CrossRef] [PubMed]
- James, D.G.; James, T.S.; Seymour, L.; Kappen, L.; Russell, T.; Harryman, B.; Bly, C. Citizen scientist tagging reveals destinations of migrating monarch butterflies, Danaus plexippus (L.) from the Pacific Northwest. J. Lep. Soc. 2018, 72, 127–144. [Google Scholar] [CrossRef]
- Inamine, H.; Ellner, S.P.; Springer, J.P.; Agrawal, A.A. Linking the continental migratory cycle of the monarch butterfly to understand its population decline. Oikos 2016, 125, 1081–1091. [Google Scholar] [CrossRef]
- Badgett, G.; Davis, A.K. Population trends of monarchs at a northern monitoring site: Analyses of 19 years of fall migration counts at Peninsula Point, MI. Ann. Entomol. Soc. Am. 2015, 108, 700–706. [Google Scholar] [CrossRef]
- Malcolm, S.B. Anthropogenic impacts on mortality and population viability of the monarch butterfly. Ann. Rev. Ent. 2016, 63, 277–302. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.A.; Inamine, H. Mechanisms behind the monarch’s decline. Science 2018, 360, 1294–1296. [Google Scholar] [CrossRef] [PubMed]
- Tracey, J.L.; Kantola, T.; Baum, K.A.; Coulson, R.N. Modeling fall migration pathways and spatially identifying potential migratory hazards for the eastern monarch butterfly. Landscape Ecol. 2019, 34, 443–458. [Google Scholar] [CrossRef]
- Craddock, H.A.; Huang, D.; Turner, P.C.; Quiros-Alcala, L.; Payne-Styrges, D.C. Trends in neonicotinoid pesticide residues in food and water in the United States, 1999–2015. Env. Health 2019, 18, 7. [Google Scholar] [CrossRef]
- Jeschke, P.; Nauen, R.; Schindler, M.; Elbert, A. Overview of the status and global strategy for neonicotinoids. J. Agric. Food Chem. 2011, 59, 2897–2908. [Google Scholar] [CrossRef] [PubMed]
- Pollak, P. Fine Chemicals: The Industry and the Business, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011; p. 312. [Google Scholar]
- Blake, R. EU neonicotinoid ban removes vital tools in global fight against pests. Outlooks Pest Manag. 2018, 5, 197–200. [Google Scholar] [CrossRef]
- Mizell, R.F.; Sconyers, M.C. Toxicity of imidacloprid to selected arthropod predators in the laboratory. Fla. Entomol. 1992, 75, 277–280. [Google Scholar] [CrossRef]
- James, D.G. Imidacloprid increases egg production in Amblyseius victoriensis (Acari: Phytoseiidae). Exp. Appl. Acarol. 1997, 21, 75–82. [Google Scholar] [CrossRef]
- Main, A.R.; Webb, E.B.; Goyne, K.W.; Mengel, D. Neonicotinoid insecticides negatively affect performance measures of non-target terrestrial arthropods: A meta-analysis. Ecol. Appl. 2018, 28, 1232–1244. [Google Scholar] [CrossRef] [PubMed]
- Isawa, T.; Motoyama, N.; Ambrose, J.T.; Roe, R.M. Mechanism for the differential toxicity of neonicotinoid insecticides in the honey bee, Apis mellifera. Crop Prot. 2004, 23, 371–378. [Google Scholar] [CrossRef]
- Casida, J.E. Neonicotinoids and other insect nicotinic receptor competitive modulators: Progress and prospects. Ann. Rev. Entomol. 2018, 63, 125–144. [Google Scholar] [CrossRef]
- Baron, G.L.; Raine, N.E.; Brown, M.J.F. General and species-specific impacts of a neonicotinoid insecticide on the ovary development and feeding of wild bumblebee queens. Proc. R. Soc. B Biol. Sci. 2017, 284, 20170123. [Google Scholar] [CrossRef]
- Whitehorn, P.R.; O’Connor, S.; Wackers, F.L.; Goulson, D. Neonicotinoid pesticide reduces bumble bee colony growth and queen production. Science 2012, 336, 351–352. [Google Scholar] [CrossRef]
- Feltham, H.; Park, K.; Goulson, D. Field realistic doses of pesticide imidacloprid reduce bumblebee pollen foraging efficiency. Ecotoxicology 2014, 23, 317–323. [Google Scholar] [CrossRef]
- Tan, K.; Chen, W.; Dong, S.; Liu, X.; Wang, Y.; Nish, J.C. Imidacloprid alters foraging and decreases bee avoidance of predators. PLoS ONE 2014, 9, e102725. [Google Scholar] [CrossRef] [PubMed]
- Henry, M.; Beguin, M.; Requier, F.; Rollin, O.; Odoux, J.F.; Aupinel, P. A common pesticide decreases foraging success and survival in honeybees. Science 2012, 336, 350–351. [Google Scholar] [CrossRef]
- Stanley, D.A.; Smith, K.E.; Raine, N.E. Bumblebee learning and memory are impaired by chronic exposure to a neonicotinoid pesticide. Sci. Rep. 2015, 5, 16508. [Google Scholar] [CrossRef]
- Woodcock, B.A.; Isaac, N.J.; Bullock, J.M.; Roy, D.B.; Garthwaite, D.G.; Crowe, A.; Pywell, R.F. Impacts of neonicotinoid use on long-term population changes in wild bees in England. Nat. Commun. 2016, 7, 12459. [Google Scholar] [CrossRef] [PubMed]
- Van der Sluijs, J.P.; Amaral-Rogers, V.; Belzunces, L.P.; Bijleveld van Lexmond, M.F.I.J.; Bonmatin, J.M.; Chagnon, M.; Downs, C.A.; Furlan, L.; Gibbons, D.W.; Glorio, C. Conclusions of the worldwide integrated assessment on the risks of neonicotinoids and fipronil to biodiversity and ecosystem functioning. Environ. Sci. Pollut. Res. 2015, 22, 148–154. [Google Scholar] [CrossRef]
- Parkinson, R.H.; Gray, J.R. Neural conduction, visual motion detection and insect flight behavior are disrupted by low doses of imidacloprid and its metabolites. Neurotoxicology 2019, 72, 107–113. [Google Scholar] [CrossRef]
- Eng, M.L.; Stutchbury, B.J.M.; Morrissey, C.A. Imidacloprid and chlorpyrifos insecticides impair migratory ability in a seed-eating songbird. Sci. Rep. 2017, 7, 15176. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, D.; Morrissey, C.; Mineau, P. A review of the direct and indirect effects of neonicotinoids and fipronil on vertebrate wildlife. Environ. Sci. Pollut. Res. 2015, 22, 103–118. [Google Scholar] [CrossRef]
- Berheim, E.H.; Jenks, J.A.; Lundgren, J.G.; Michel, E.S.; Grove, D.; Jensen, W.F. Effects of neonicotinoid insecticides on physiology and reproductive characteristics of captive female and fawn white-tailed deer. Sci. Rep. 2019, 9, 4534. [Google Scholar] [CrossRef] [PubMed]
- Godfray, H.C.J.; Blacquiere, T.; Field, L.M.; Halls, R.S.; Potts, S.G.; Raine, N.E.; Vanbergen, A.J.; McLean, A.R. A restatement of recent advances in the natural science evidence base concerning neonicotinoid insecticides and insect pollinators. Proc. R. Soc. B 2014, 281, 20140558. [Google Scholar] [CrossRef] [PubMed]
- Krupke, C.H.; Hunt, G.J.; Eitzer, B.D.; Andino, J.; Given, K. Multiple routes of pesticide exposure for honeybees living near agricultural fields. PLoS ONE 2012, 7, e29268. [Google Scholar] [CrossRef] [PubMed]
- Botias, C.; David, A.; Hill, E.M.; Goulson, D. Contamination of wild plants near neonicotinoid-treated crops, and implications for non-target insects. Sci. Total Environ. 2016, 566, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Limay-Rios, V.; Forero, L.G.; Xue, Y.; Smith, J.; Baute, T.; Schaafsma, A. Neonicotinoid insecticide residues in soil dust and associated parent soil in fields with a history of seed treatment use on crops in southwestern Ontario. Environ. Toxicol. Chem. 2016, 35, 303–310. [Google Scholar] [CrossRef]
- David, A.; Botias, C.; Abdul-Sada, A.; Nicholls, E.; Rotheray, E.L. Widespread contamination of wildflower and bee-collected pollen with complex mixtures of neonicotinoids and fungicides commonly applied to crops. Env. Int. 2016, 88, 169–178. [Google Scholar] [CrossRef]
- Douglas, M.R.; Rohr, J.R.; Tooker, J.F. Neonicotinoid insecticide travels through a soil food chain, disrupting biological control of non-target pests and decreasing soya bean yield. J. Appl. Ecol. 2014, 52, 250–260. [Google Scholar] [CrossRef]
- Hladik, M.L.; Kolpin, D.W.; Kuivila, K.M. Widespread occurrence of neonicotinoid insecticides in streams in a high corn and soybean producing region, USA. Environ. Pollut. 2014, 193, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Hladik, M.L.; Kolpin, D.W. First national scale reconnaissance of neonicotinoid insecticides in streams across the USA. Environ. Chem. 2015, 13, 4–11. [Google Scholar] [CrossRef]
- Morrissey, C.A.; Mineau, P.; Devries, J. Neonicotinoid contamination of global surface waters and associated risk to aquatic invertebrates: A review. Environ. Int. 2015, 74, 291–303. [Google Scholar] [CrossRef]
- Krischik, V.; Rogers, M.; Gupta, G.; Varshney, A. Soil-applied imidacloprid translocates to ornamental flowers and reduces survival of adult Coleomegilla maculata, Harmonia axyridis and Hippodamia convergens lady beetles, and larval Danaus plexippus and Vanessa cardui butterflies. PLoS ONE 2015, 10, e0119133. [Google Scholar] [CrossRef] [PubMed]
- Keim, B. Backyard Pesticide Use May Fuel Bee Die-Offs. 2012. Available online: www.wired.com/2012/04/neonicotinoids-gardens/ (accessed on 29 August 2019).
- Mule, R.; Sabella, G.; Robba, R.; Manachini, B. Systematic review of the effects of chemical insecticides on four common butterfly families. Front. Environ. Sci. 2017, 5, 32. [Google Scholar] [CrossRef]
- Braak, N.; Neve, R.; Jones, A.K.; Gibbs, M.; Breuker, C.J. The effects of insecticides on butterflies: A review. Environ. Pollut. 2018, 242, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Gilburn, A.S.; Bunnefeld, N.; McVean Wilson, J.; Botham, M.S.; Brereton, T.M.; Fox, R.; Goulson, D. Are neonicotinoid insecticides driving declines of widespread butterflies? Peer J. 2015, 3, e1402. [Google Scholar] [CrossRef]
- Forrister, M.L.; Cousens, B.; Harrison, J.G.; Anderson, K.; Thorne, J.H.; Waetjen, D.; Nice, C.C.; De Parsla, M.; Hladik, M.L.; Meese, R.; et al. Increasing neonicotinoid use and the declining butterfly fauna of lowland California. Biol. Lett. 2016, 12, 20160475. [Google Scholar] [CrossRef]
- Whitehorn, P.; Norville, G.; Gilburn, A.; Goulson, D. Larval exposure to the neonicotinoid imidacloprid impacts adult size in the farmland butterfly Pieris brassicae. Peer J. 2018, 6, e4772. [Google Scholar] [CrossRef]
- Basley, K.; Goulson, D. Effects of field-relevant concentrations of clothianidin on larval development of the butterfly Polyommatus icarus (Lepidoptera: Lycaenidae). Environ. Sci. Technol. 2018, 52, 3990–3996. [Google Scholar] [CrossRef] [PubMed]
- Pacenka, J.R.; Lundgren, J.G. Non-target effects of clothianidin on monarch butterflies. Sci. Nat. 2015, 102, 19. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, R.E.; Myers, J. Ophryocystis elektroscirrha sp. n., a neogregarine pathogen of the monarch butterfly Danaus plexippus (L.) and the Florida queen butterfly D. gilippus berenice Cramer. J. Protozool. 1970, 17, 300–305. [Google Scholar] [CrossRef]
- Altizer, S.M.; Oberhauser, K.S.; Brower, L.P. Association between host migration and the prevalence of a protozoan parasite in natural populations of monarch butterflies. Ecol. Entomol. 2000, 25, 125–139. [Google Scholar] [CrossRef]
- Suchail, S.; Guez, D.; Belzunces, L.P. Characteristics of imidacloprid toxicity in two Apis mellifera subspecies. Environ. Tox. Chem. 2000, 19, 1901–1905. [Google Scholar] [CrossRef]
- Hopwood, J.; Shepherd, M. Neonicotinoids in your garden. Wings Fall 2012, 22, 1–7. [Google Scholar]
- Kremer, A.N.; King, B.H. A neonicotinoid affects the mating behavior of Spalangia endius (Hymenoptera: Pteromalidae), a biological control agent of filth flies. Environ. Entomol. 2019, 48, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Gill, R.J.; Raine, N.E. Chronic impairment of bumblebee natural foraging behavior induced by sublethal pesticide exposure. Funct. Ecol. 2014, 28, 1459–1471. [Google Scholar] [CrossRef]
- Lamsa, J.; Kuusela, E.; Tuomi, J.; Juntunen, S.; Watts, P.C. Low dose of neonicotinoid insecticide reduces foraging motivation of bumblebees. Proc. R. Soc. B 2018, 285, 20180506. [Google Scholar] [CrossRef] [PubMed]
| Forewing (mm) | Male | Female | Wet Weight (mg) | Male | Female |
|---|---|---|---|---|---|
| Eclosion | |||||
| Untreated | 46.0 ± 1.1 a (9) | 47.2 ± 2 ± 0.7 a (9) | Untreated | 413.2 ± 18.0 a (9) | 422.8 ± 28.7 a (9) |
| Treated | 46.2 ± 0.5 a (11) | 45.1 ± 0.7 a (11) | Treated | 466.8 ± 28.7 a (11) | 389.0 ± 23.7 b (11) |
| 12 days post-eclosion | |||||
| Untreated | 48.0 ± 0.8 a (4) | 46.8 ± 0.8 a (4) | Untreated | 467.5 ± 15.2 a (4) | 423.5 ± 31.9 a (4) |
| Treated | 46.0 ± 0.4 a (4) | 46.0 ± 1.1 a (4) | Treated | 377.2 ± 21.9 a * (4) | 385.2 ± 59.2 a (4) |
| 22 days post-eclosion | |||||
| Untreated | 46.7 ± 1.2 a (3) | 47.6 ± 1.1 a (5) | Untreated | 360.0 ± 24.2 a * (3) | 457.8 ± 23.9 b (5) |
| Treated | 46.0 ± 2.0 (2) | Treated | 466.5 ± 44.5 (2) |
| Post-Eclosion Day and Treatment | No. Immature Oocytes | No. Mature Oocytes | No. Total Oocytes | t Test on Total Oocytes (Treated v Untreated) |
|---|---|---|---|---|
| 12 | ||||
| Untreated | 55.8 ± 13.1 | 59.8 ± 6.4 | 115.5 ± 19.3 (4) | |
| Treated | 42.8 ± 4.2 | 59.2 ± 9.8 | 102.0 ± 10.5 (4) | t = −0.614, 6 df, p = 0.562 |
| 22 | ||||
| Untreated | 46.0 ± 6.2 | 85.6 ± 7.0 | 131.6 ± 10.2 (5) | |
| Treated | 37.0 ± 9.0 | 68.5 ± 22.5 | 105.5 ± 31.5 (2) | t = −1.092, 5 df, p = 0.325 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
James, D.G. A Neonicotinoid Insecticide at a Rate Found in Nectar Reduces Longevity but Not Oogenesis in Monarch Butterflies, Danaus plexippus (L.). (Lepidoptera: Nymphalidae). Insects 2019, 10, 276. https://doi.org/10.3390/insects10090276
James DG. A Neonicotinoid Insecticide at a Rate Found in Nectar Reduces Longevity but Not Oogenesis in Monarch Butterflies, Danaus plexippus (L.). (Lepidoptera: Nymphalidae). Insects. 2019; 10(9):276. https://doi.org/10.3390/insects10090276
Chicago/Turabian StyleJames, David G. 2019. "A Neonicotinoid Insecticide at a Rate Found in Nectar Reduces Longevity but Not Oogenesis in Monarch Butterflies, Danaus plexippus (L.). (Lepidoptera: Nymphalidae)" Insects 10, no. 9: 276. https://doi.org/10.3390/insects10090276
APA StyleJames, D. G. (2019). A Neonicotinoid Insecticide at a Rate Found in Nectar Reduces Longevity but Not Oogenesis in Monarch Butterflies, Danaus plexippus (L.). (Lepidoptera: Nymphalidae). Insects, 10(9), 276. https://doi.org/10.3390/insects10090276





