The Thermoregulatory Behavior of Nectar Foraging Polistine Wasps (Polistes dominula and Polistes gallicus) in Different Climate Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Study Sites
2.2. Body Temperature Measurement and Environmental Conditions
2.3. Operative Temperature Model
2.4. Data Analysis
3. Results
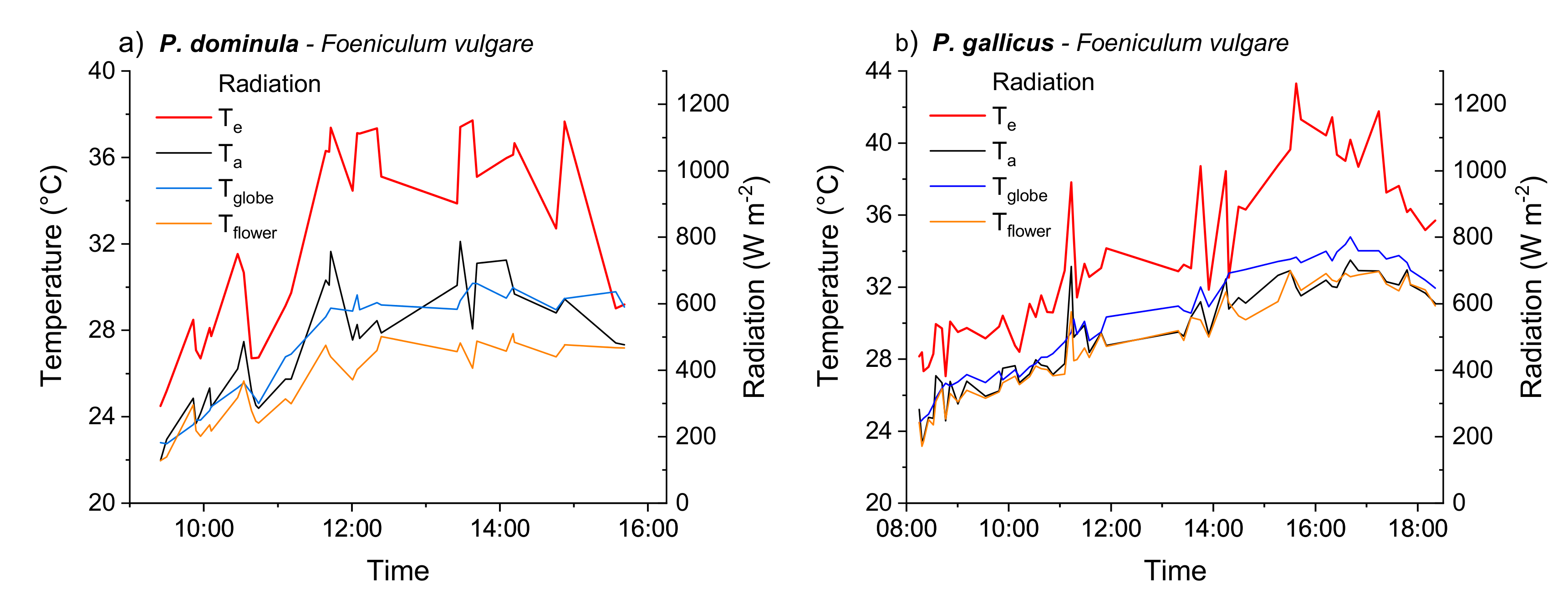
3.1. Operative Temperature
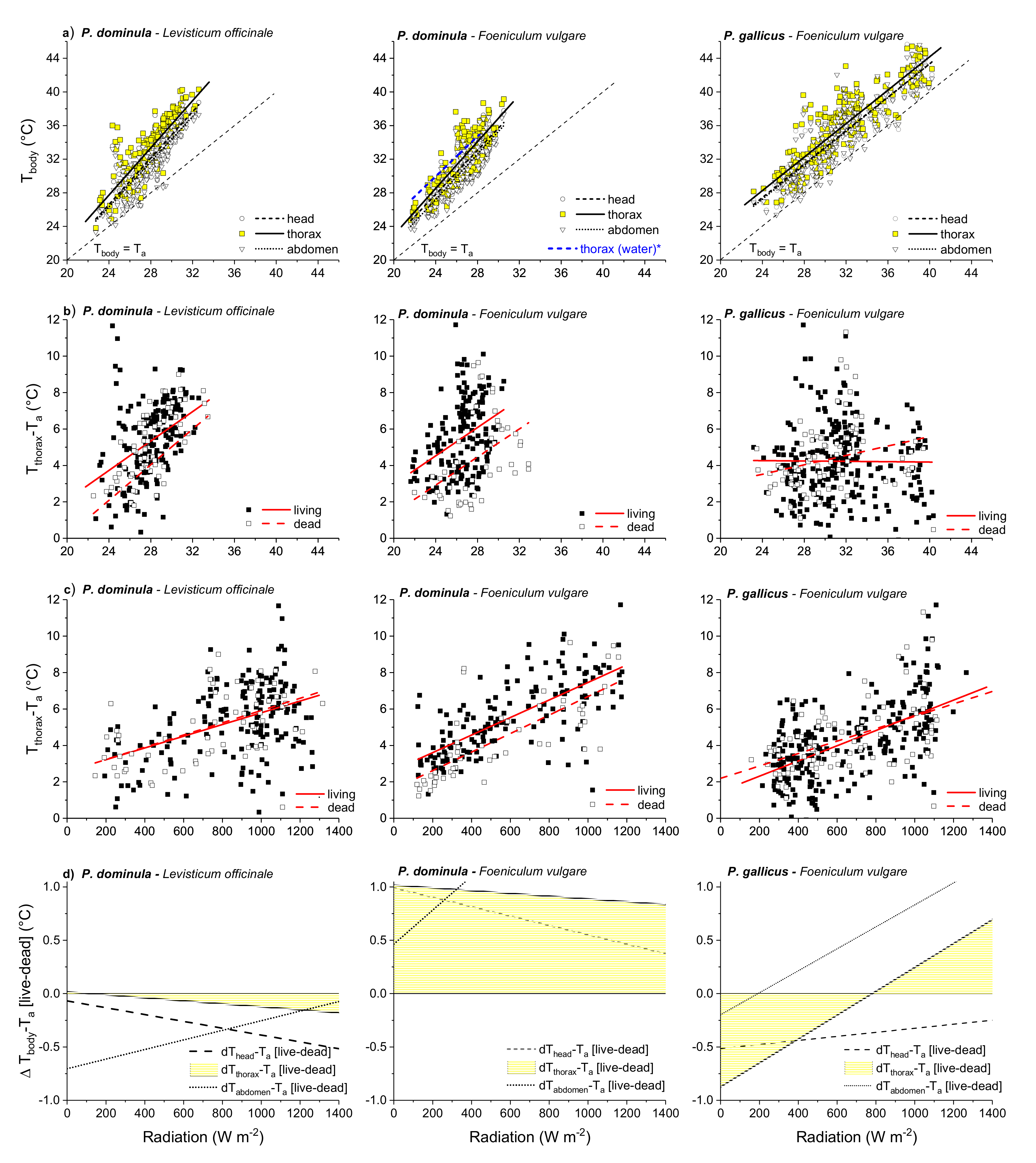
3.2. Body Temperature
3.3. Thorax Temperature Excess
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kovac, H.; Stabentheiner, A.; Schmaranzer, S. Thermoregulation of water foraging wasps (Vespula vulgaris and Polistes dominulus). J. Insect. Physiol. 2009, 55, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Kovac, H.; Stabentheiner, A. Does size matter—Thermoregulation of ‘heavyweight’ and ‘lightweight’ wasps (Vespa crabro and Vespula sp.). Biol. Open 2012, 1, 848–856. [Google Scholar] [CrossRef] [PubMed]
- Weiner, S.A.; Upton, C.T.; Noble, K.; Woods, W.A.; Starks, P.T. Thermoregulation in the primitively eusocial paper wasp, Polistes dominulus. Insect. Soc. 2010, 57, 157162. [Google Scholar] [CrossRef]
- Weiner, S.A.; Noble, K.; Upton, C.T.; Woods, W.A.; Starks, P.T. A role for thermoregulation in the Polistes dominulus invasion: A comparison of the thermoregulatory abilities of the invasive wasp P. dominulus and the native wasp P. fuscatus. Insect. Soc. 2011, 58, 185–190. [Google Scholar] [CrossRef]
- Höcherl, N.; Tautz, J. Thermoregulation of individual paper wasps (Polistes dominula) plays an important role in nest defence and dominance battles. Sci. Nat. 2015, 102, 32. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, B. Thermoregulation of African and European honeybees during foraging, attack, and hive exits and returns. J. Exp. Biol. 1979, 80, 217–229. [Google Scholar]
- Weiner, S.A.; Noble, K.; Upton, C.T.; Flynn, G.; Woods, W.A., Jr.; Starks, P.T. The cost of flight, a role in the Polistes dominulus invasion. Insect. Soc. 2012, 59, 81–86. [Google Scholar] [CrossRef]
- Coelho, J.R.; Ross, A.J. Body temperature and thermoregulation in two species of yellowjackets, Vespula germanica and V. maculifrons. J. Comp. Physiol. B 1996, 166, 68–76. [Google Scholar] [CrossRef]
- Woods, W.A.; Heinrich, B.; Stevenson, R.D. Honeybee flight metabolic rate: Does it depend upon air temperature? J. Exp. Biol. 2005, 208, 1161–1173. [Google Scholar] [CrossRef]
- Kovac, H.; Käfer, H.; Petrocelli, I.; Stabentheiner, A. Comparison of thermal traits of Polistes dominula and Polistes gallicus, two European paper wasps with strongly differing distribution ranges. J. Comp. Physiol. B 2017, 187, 277–290. [Google Scholar] [CrossRef]
- Kovac, H.; Stabentheiner, A. Effect of food quality on the body temperature of wasps (Paravespula vulgaris). J. Insect. Physiol. 1999, 45, 183–190. [Google Scholar] [CrossRef]
- Schmaranzer, S.; Stabentheiner, A. Variability of the thermal behavior of honeybees on a feeding place. J. Comp. Physiol. B 1988, 158, 135–141. [Google Scholar] [CrossRef]
- Stabentheiner, A.; Schmaranzer, S. Thermographic determination of body temperatures in honey bees and hornets: Calibration and applications. Thermology 1987, 2, 563–572. [Google Scholar]
- Stabentheiner, A.; Kovac, H.; Hetz, S.K.; Käfer, H.; Stabentheiner, G. Assessing honeybee and wasp thermoregulation and energetics—New insights by combination of flow-through respirometry with infrared thermography. Thermochim. Acta 2012, 534, 77–86. [Google Scholar] [CrossRef]
- Bishop, J.A.; Armbruster, W.S. Thermoregulatory abilities of Alaskan bees: Effects of size, phylogeny and ecology. Funct. Ecol. 1999, 13, 711–724. [Google Scholar] [CrossRef]
- Bakken, G.S.; Angilletta, M.J. How to avoid errors when quantifying thermal environments. Funct. Ecol. 2014, 28, 96–107. [Google Scholar] [CrossRef]
- Camacho, A.; Rodrigues, M.T.; Navas, C. Extreme operative temperatures are better descriptors of the thermal environment than mean temperatures. J. Therm. Biol. 2015, 49–50, 106–111. [Google Scholar] [CrossRef]
- Corbet, S.A.; Huang, S. Small bees overheat in sunlit flowers: Do they make cooling flights? Ecol. Entomol. 2016, 41, 344–350. [Google Scholar] [CrossRef]
- Kovac, H.; Stabentheiner, A.; Schmaranzer, S. Thermoregulation of water foraging honeybees—Balancing of endothermic activity with radiative heat gain and functional requirements. J. Insect. Physiol. 2010, 56, 1834–1845. [Google Scholar] [CrossRef]
- Kovac, H.; Stabentheiner, A. A comparison of body temperatures of honeybees and wasps foraging on raspberry and rhubarb. In Proceedings of the 2001 Berlin Meeting of the European Sections of the IUSSI, Berlin, Germany, 25–29 2001; Menzel, R., Rademacher, E., Eds.; p. 194, ISBN 3-901864-01-7. [Google Scholar]
- Kovac, H.; Stabentheiner, A.; Brodschneider, R. What do foraging wasps optimize in a variable environment, energy investment or body temperature? J. Comp. Physiol. A 2015, 201, 1043–1052. [Google Scholar] [CrossRef]
- Kovac, H.; Stabentheiner, A.; Brodschneider, R. Foraging strategy of wasps—Optimisation of intake rate or energetic efficiency. J. Exp. Biol. 2018, 221, jeb174169. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.R. The Effect of thorax temperature on force production during tethered flight in honeybee (Apis mellifera) drones, workers, and queens. Physiol. Zool. 1991, 64, 823–835. [Google Scholar] [CrossRef]
- Coelho, J.R.; Hoagland, J. Load-lifting capacities of three species of yellowjackets (vespula) foraging on honey-bee corpses. Funct. Ecol. 1995, 9, 171–174. [Google Scholar] [CrossRef]
- Heinrich, B. The Hot-Blooded Insects; Springer-Verlag: Berlin/Heidelberg, Germany; London, UK; Paris, France, 1993. [Google Scholar]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovac, H.; Käfer, H.; Stabentheiner, A. The Thermoregulatory Behavior of Nectar Foraging Polistine Wasps (Polistes dominula and Polistes gallicus) in Different Climate Conditions. Insects 2019, 10, 187. https://doi.org/10.3390/insects10070187
Kovac H, Käfer H, Stabentheiner A. The Thermoregulatory Behavior of Nectar Foraging Polistine Wasps (Polistes dominula and Polistes gallicus) in Different Climate Conditions. Insects. 2019; 10(7):187. https://doi.org/10.3390/insects10070187
Chicago/Turabian StyleKovac, Helmut, Helmut Käfer, and Anton Stabentheiner. 2019. "The Thermoregulatory Behavior of Nectar Foraging Polistine Wasps (Polistes dominula and Polistes gallicus) in Different Climate Conditions" Insects 10, no. 7: 187. https://doi.org/10.3390/insects10070187
APA StyleKovac, H., Käfer, H., & Stabentheiner, A. (2019). The Thermoregulatory Behavior of Nectar Foraging Polistine Wasps (Polistes dominula and Polistes gallicus) in Different Climate Conditions. Insects, 10(7), 187. https://doi.org/10.3390/insects10070187




