Foraging Behavior and Pollination Efficiency of Apis mellifera L. on the Oil Tree Peony ‘Feng Dan’ (Paeonia ostii T. Hong et J.X. Zhang)
Abstract
1. Introduction
2. Materials and Methods
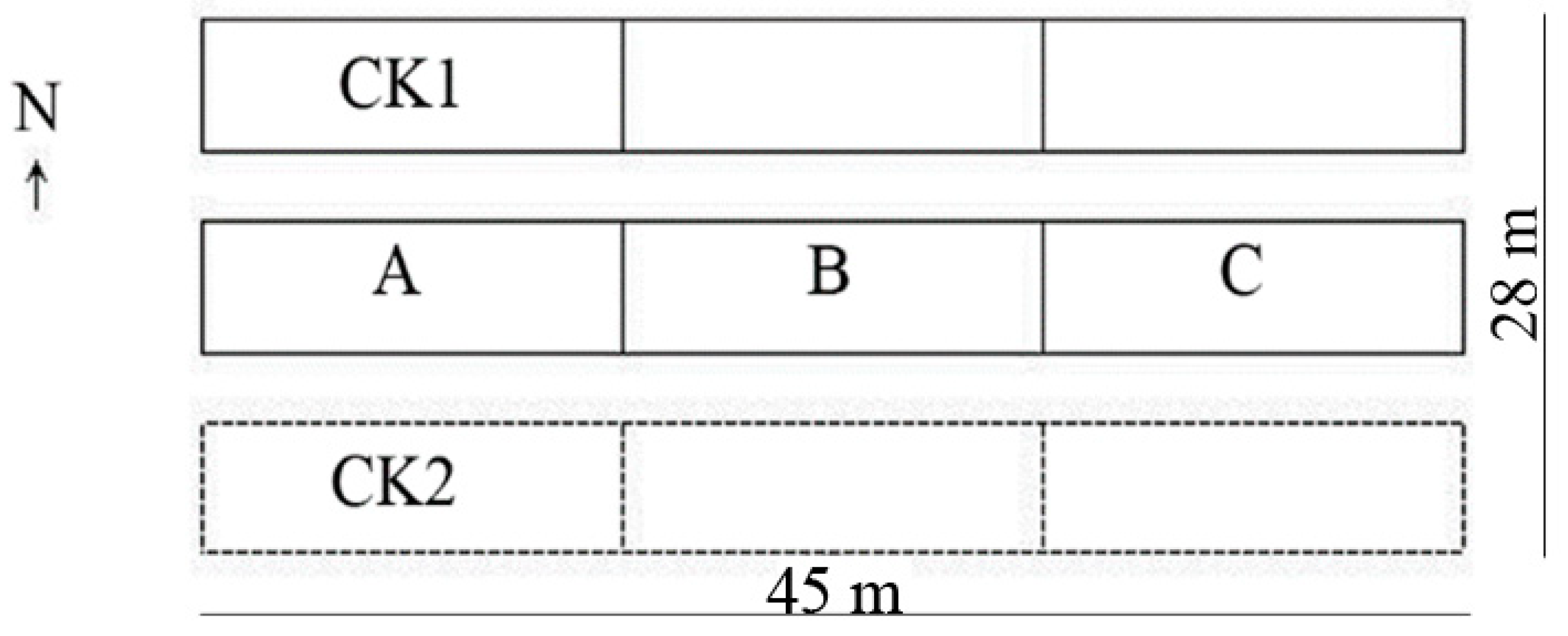
2.1. Sampling Location
2.2. Research Materials
2.3. Observation Indices
2.4. Observation of Foraging Behaviors
2.5. Observation of Pollination Efficiency
2.6. Statistical Analysis
3. Results
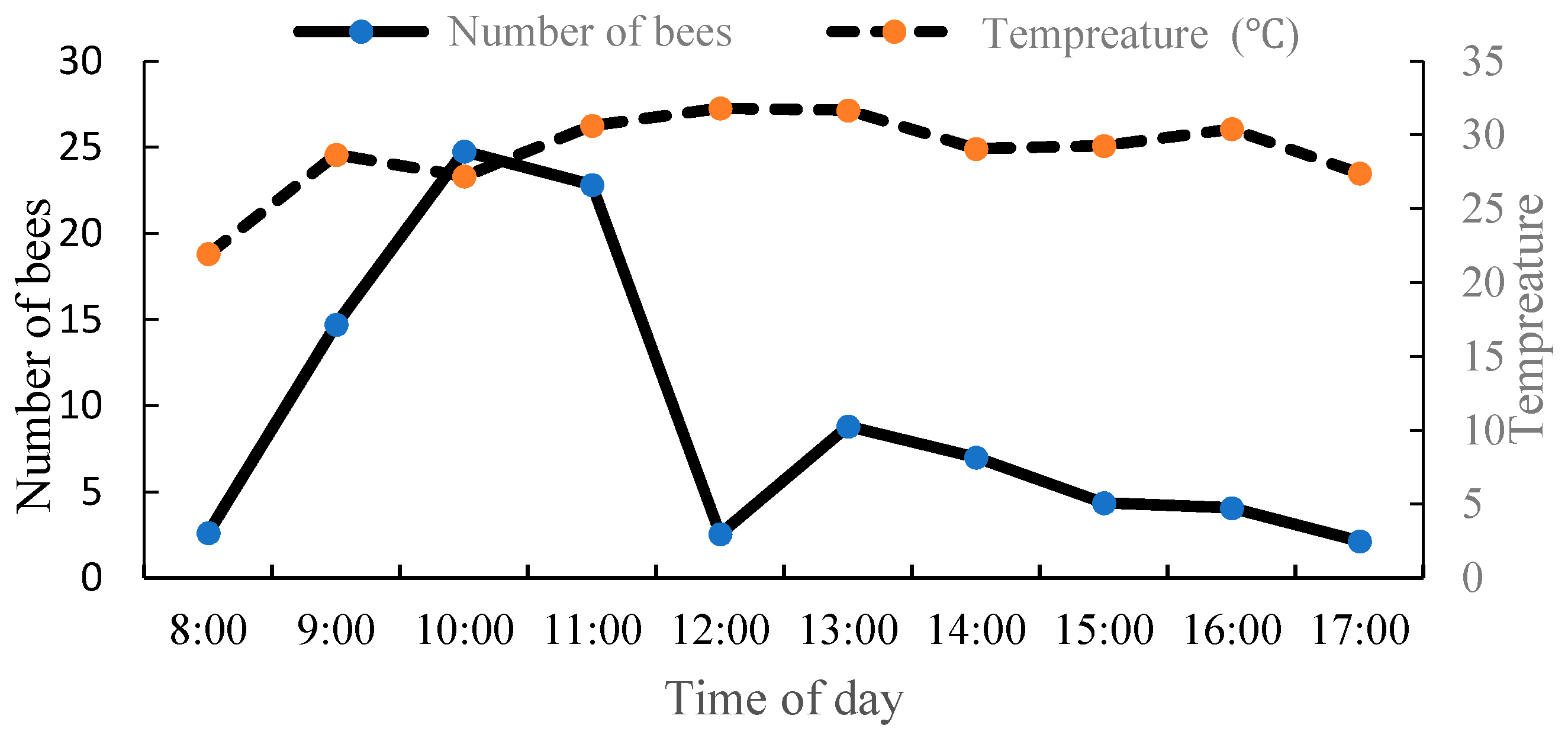
3.1. Foraging Characteristics of A. mellifera
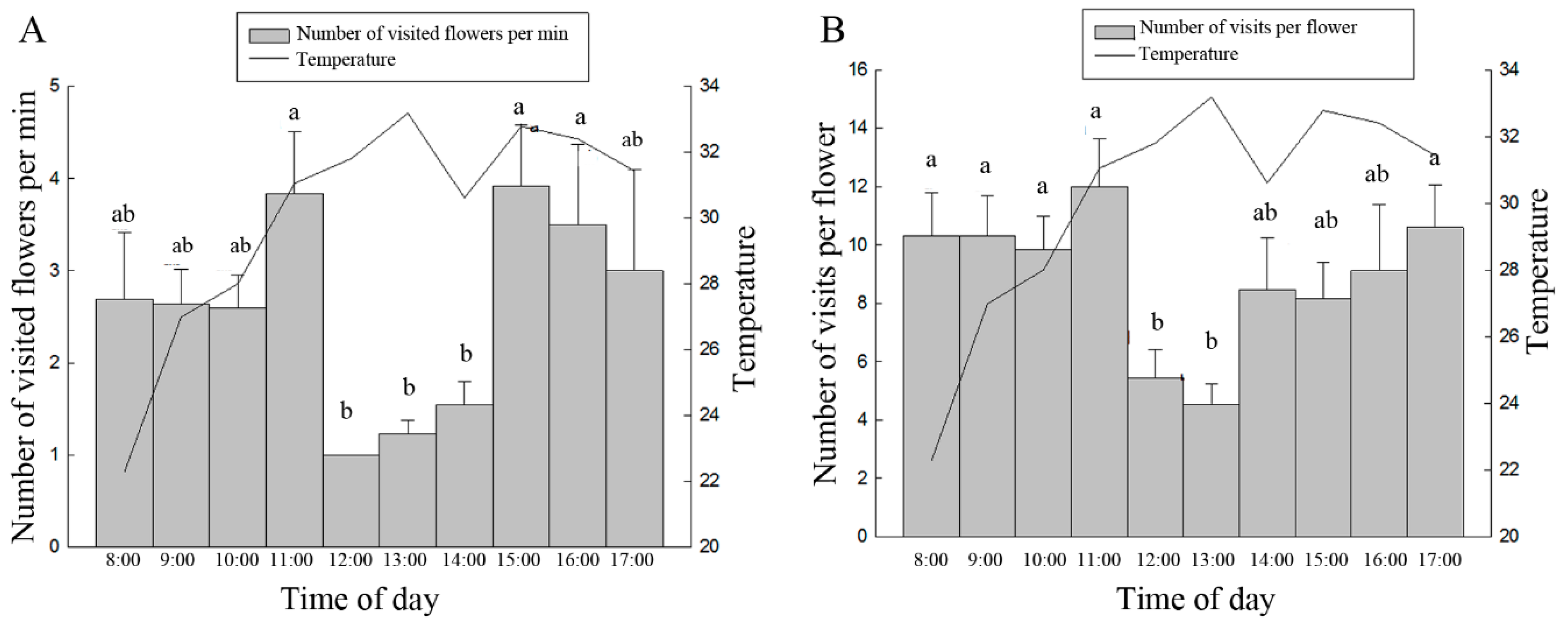
3.2. Foraging Behavior of A. mellifera on the Oil Tree Peony
3.3. Activity Patterns of A. mellifera on the Oil Tree Peony
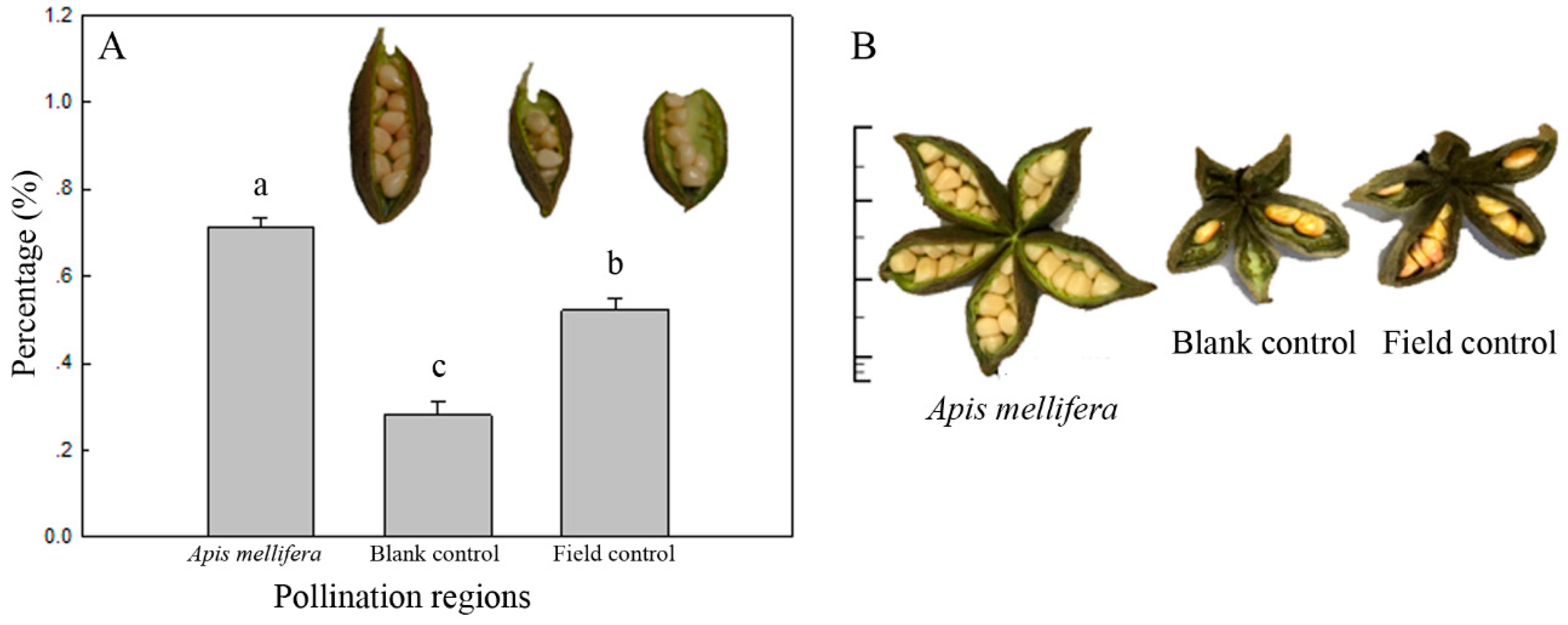
3.4. Pollination Efficiency of A. mellifera
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hong, D.Y.; Pan, K.Y. Taxonomical history and revision of Paeonia Sect. Moutan (Paeoniaceae). J. Syst. Evol. 1999, 37, 351. [Google Scholar]
- Cheng, F.Y.; Li, J.J. Exportation of Chinese tree peonies (Mudan) and their development in other countries II: Cultivated. J. Northwest Norm. Univ. (Nat. Sci.) 1998, 3, 106–111. [Google Scholar]
- Picerno, P.; Mencherini, T.; Sansone, F.; Del Gaudio, P.; Granata, I.; Porta, A.; Aquino, R.P. Screening of a polar extract of Paeonia rockii: Composition and antioxidant and antifungal activities. J. Ethnopharmacol. 2011, 138, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Chen, L.G.; Xu, Y.J.; Wang, L.J.; Wang, L.S. Identification of floral fragrances in tree peony cultivars by gas chromatography mass spectrometry. Sci. Horticul. 2012, 142, 158–165. [Google Scholar] [CrossRef]
- Peng, L.P.; Cai, C.F.; Zhong, Y.; Xu, X.X.; Xian, H.L.; Cheng, F.Y.; Mao, J.F. Genetic analyses reveal independent domestication origins of the emerging oil crop Paeonia ostii, a tree peony with a long-term cultivation history. Sci. Rep. 2017, 7, 5340. [Google Scholar] [CrossRef]
- Liu, D.J.; Jiao, X.X. Development status and countermeasures of oil tree peony industry. West. For. Sci. 2015, 44, 170–173. [Google Scholar]
- Qi, J.C.; Zhou, H.M.; Ma, J.Q.; Li, P. Analysis of the chemical constituents in peony seed oil by GC-MS. Cereals Oils 2005, 11, 23–24. [Google Scholar]
- Li, S.S.; Yuan, R.Y.; Chen, L.G.; Wang, L.S.; Hao, X.H.; Wang, L.J.; Zheng, C.X.; Du, H. Systematic qualitative and quantitative assessment of fatty acids in the seeds of 60 tree peony (Paeonia section Moutan DC.) cultivars by GC-MS. Food Chem. 2015, 173, 133–140. [Google Scholar] [CrossRef]
- Shi, G.A.; Jiao, F.X.; Jiao, Y.P.; Yang, H.A.; Han, M.W.; Wu, Y.Q.; Shi, B.R. The development prospect and countermeasures of Chinese oil peony. Cereals Oils Assoc. 2014, 29, 124–127. [Google Scholar]
- Luo, Y.B.; Pei, Y.L.; Pan, K.Y.; Hong, D.Y. A study on pollination biology of Paeonia suffruticosa A. var. spontanea R. Acta Phytotaxonomica Sinica 1998, 36, 134–144. [Google Scholar]
- Li, K.; Zheng, B.Q.; Wang, Y.; Guo, X. Flowering characteristics and breeding system of Paeonia delavayi. J. Northeast For. Uni. 2013, 41, 63–67. [Google Scholar]
- Yang, Y.; Luo, J.T.; Zhang, B.F.; Song, H.X.; Liu, G.L.; Zeng, X.L. Studies on floral characteristics and breeding system of Paeonia szechuanica F. J. Plant Resour. Environ. 2015, 24, 97–104. [Google Scholar]
- Han, X.; Cheng, F.Y.; Xiao, J.J.; Wang, Y.L.; Zhang, D.; Wang, Y.; Zhong, Y. Crosses of Paeonia ostii T.Hong et J.X.Zhang as maternal parents and an analysis on the potential in tree peony breeding. J. Beijing For. Univ. 2014, 4, 121–125. [Google Scholar]
- Abrol, D.P. The role of pollination in improving food security and livelihoods. Pollination Biol. 2011, 2011, 737–770. [Google Scholar]
- Gallai, N.; Salles, J.M.; Settele, J.; Vaissiere, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Cunningham, S.A.; Fitz Gibbon, F.; Heard, T.A. The future of pollinators for Australian agriculture. Aust. J. Agric. Res. 2002, 53, 893–900. [Google Scholar] [CrossRef]
- Rucker, R.R.; Thurman, W.N.; Burgett, M. Honey bee pollination markets and the internalization of reciprocal benefits. Am. J. Agric. Econ. 2012, 94, 956–977. [Google Scholar] [CrossRef]
- Klein, A.M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Teja Tscharntke, C.K. Importance of pollionators in changing landscapes for world crops. Proc. R. Soc. B Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef]
- Abrol, D.P.; Shankar, U. Pollination in oil crops: Recent advance sand future strategies. In Technological Innovations in Major World Oil Crops; Gupta, S.K., Ed.; Springer Science + Business Media: Berlin, Germany, 2012; Volume 2, pp. 221–265. [Google Scholar]
- Bommarco, R.; Marini, L.; Vaissière, B.E. Insect pollination enhances seed yield, quality, and market value in oilseed rape. Oecologia 2012, 169, 1025–1032. [Google Scholar] [CrossRef]
- Fishbein, M.; Venable, D.L. Diversity and temporal change in the effective pollination of Asclepias tuberosa. Ecology 1996, 77, 1061–1073. [Google Scholar] [CrossRef]
- Thomson, J.D.; Mcjenna, M.A.; Cruzan, M.B. Temporal patterns of nectar and pollen production in Aralia hispida: Implications for reproductive success. Ecology 1989, 70, 1061–1068. [Google Scholar] [CrossRef]
- Morandin, L.A.; Winston, M.L. Wild bee abundance and seed production in conventional, organic, and genetically modified canola. Ecol. Appl. 2005, 15, 871–881. [Google Scholar] [CrossRef]
- Jauker, F.; Bondarenko, B.; Becker, H.C.; Steffan-Dewenter, I. Pollination efficiency of wild bees and hoverflies provided to oilseed rape. Agric. Forest Entomol. 2012, 14, 81–87. [Google Scholar] [CrossRef]
- He, C.L.; Li, X.P.; Zhang, H.X. Food plants and foraging behaviors of Xylocopa appendiculata (Hymenoptera, Apidae) in Luoyang, Henan Province, central China. Entomologica Sinica 2012, 55, 444–456. [Google Scholar]
- Li, J.Y.; Zhang, X.F.; Zhao, X.Q. The Peony of China; Encyclopedia of China Publishing House: Beijing, China, 2011; Volume 3, pp. 30–50. [Google Scholar]
- Delaplane, K.S.; Mayer, D.E. Crop Pollination by Bees; CABI Publishing: Wallingford, CT, USA, 2000; pp. 1–20. [Google Scholar]
- Deguines, N.; Jono, C.; Baude, M.; Henry, M.; Julliard, R.; Fontaine, C. Large-scale trade-off between agricultural intensification and crop pollination services. Front. Ecol. Environ. 2014, 12, 212–217. [Google Scholar] [CrossRef]
- Blaauw, B.R.; Isaacs, R. Flower plantings increase wild bee abundance and the pollination services provided to a pollination dependent crop. J. Appl. Ecol. 2014, 51, 890–898. [Google Scholar] [CrossRef]
- Gonzalez, V.H.; Mantilla, B.; Palacios, E. Foraging activity of the solitary andean bee, Anthophora walteri (Hymenoptera: Apidae, Anthophorini). Revista Colombiana de Entomologia 2006, 32, 73–76. [Google Scholar]
- Li, J.L.; Peng, W.J.; Wu, J.; An, J.D.; Guo, Z.B.; Tong, Y.M.; Hang, J.X. Comparison of pollination behavior of Bombus lucorum and Apis mellifera for pollinating strawberry in greenhouse. Acta Entomologica Sinica 2006, 2, 342–348. [Google Scholar]
- An, J.D.; Wu, J.; Peng, W.J.; Tong, Y.M.; Guo, Z.B.; Li, J.L. Foraging behavior and pollination ecology of Bombus lucorum L. and Apis mellifera L. in greenhouse peach garden. J. Appl. Ecol. 2007, 5, 1073–1078. [Google Scholar]
- Mensah, B.A.; Kudom, A.A. Foraging dynamics and pollination efficiency of Apis mellifera and Xylocopa olivacea on Luffa aegyptiaca Mill (Cucurnitaceae) in southern Ghana. J. Pollination Ecol. 2011, 4, 34–38. [Google Scholar]
- Qin, J.D. The Relationship between Insects and Plants; Science Press: Beijing, China, 1987; pp. 188–204. [Google Scholar]
- Bosch, J.; Blas, M. Foraging behavior and pollinating efficiency of Osmia cornuat and Apis mellifera on almond (Hymenoptera, Megachilidae and Apidae). Appl. Entomol. Zool. 1994, 29, 1–9. [Google Scholar] [CrossRef]
- Thomson, J.D.; Goodell, K. Pollen removal and deposition by honeybee and bumblebee visitors to apple and almond flowers. J. Appl. Ecol. 2001, 38, 1032–1044. [Google Scholar] [CrossRef]
- Chen, S.L. The Apicultural Science in China; China Agricultural Press: Beijing, China, 2001; pp. 178–196. [Google Scholar]
- Bluthgen, N.; Klein, A.M. Functional complementarity and specialisation: The role of biodiversity in plant pollinator interactions. Basic Appl. Ecol. 2011, 12, 282–291. [Google Scholar] [CrossRef]
- Lee, K.Y.; Yim, S.H.; Kim, S.Y.; Yoon, H.J. Comparison of pollination activities between honeybee (Apis mellifera L.) and bumblebee (Bombus terrestris L.) during the flowering period of Asian pear (Pyrus pyrifolia N.) under variable weather conditions. J. Apicult. 2016, 31, 247. [Google Scholar] [CrossRef]
- Stanghellini, M.S.; Ambrose, J.T.; Schultheis, J.R. Diurnal activity, floral visitation and pollen deposition by honey bees and bumble bees on field-grown cucumber and watermelon. J. Apicult. Res. 2002, 41, 27–34. [Google Scholar] [CrossRef]
- Delaplane, K.S.; Dag, A.; Danka, R.G.; Freitas, B.M.; Garibaldi, L.A.; Goodwin, R.M.; Hormaza, J.I. Standard methods for pollination research with Apis mellifera. J. Apicult. Res. 2013, 52, 1–28. [Google Scholar] [CrossRef]
- Sabbahi, R.; Oliveira, D.; Marceau, J. Influence of honey bee (Hymenoptera: Apidae) density on the production of canola (Crucifera: Brassicacae). J. Econ. Entomol. 2005, 98, 367–372. [Google Scholar] [CrossRef]
- Peña, J.F.; Carabalí, A. Effect of honey bee (Apis mellifera L.) density on pollination and fruit set of avocado (Persea americana MILL.) cv. HASS. J. Apicult. Sci. 2018, 62, 5–14. [Google Scholar] [CrossRef]
- Si, B.; Zhang, Y.L.; Niu, L.X.; Wen, K.X.; Luo, J.R.; Xie, L.X. Selection research on pollination cultivar for Paeonia ostii T. Hong et J.X. Zhang in oil cultivation system. North. Horticult. 2016, 20, 58–61. [Google Scholar]


| Parameter | 2017 | 2018 |
|---|---|---|
| Single visitation duration (s) | 3.48 ± 0.11b | 4.33 ± 0.18a |
| Total visitation duration per flower (s) | 42.36 ± 2.62a | 46.57 ± 2.84a |
| Visitation interval (s) | 5.23 ± 0.13a | 5.06 ± 0.14a |
| Number of flowers visited per minute | 1.46 ± 0.55a | 1.50 ± 0.59a |
| Number of visits to a single flower per foraging flight | 5.15 ± 0.23a | 5.12 ± 0.30a |
| Number of bees visiting flowers | 21.23 ± 2.17a | 17.08 ± 1.33a |
| Number of stigma visits | 0.92 ± 0.14a | 0.63 ± 0.11a |
| Stigma visitation ratio (%) | 7.79a | 8.13a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, C.; Zhang, K.; Hou, X.; Han, D.; Wang, S. Foraging Behavior and Pollination Efficiency of Apis mellifera L. on the Oil Tree Peony ‘Feng Dan’ (Paeonia ostii T. Hong et J.X. Zhang). Insects 2019, 10, 116. https://doi.org/10.3390/insects10040116
He C, Zhang K, Hou X, Han D, Wang S. Foraging Behavior and Pollination Efficiency of Apis mellifera L. on the Oil Tree Peony ‘Feng Dan’ (Paeonia ostii T. Hong et J.X. Zhang). Insects. 2019; 10(4):116. https://doi.org/10.3390/insects10040116
Chicago/Turabian StyleHe, Chunling, Kaiyue Zhang, Xiaogai Hou, Dongbo Han, and Shuaibing Wang. 2019. "Foraging Behavior and Pollination Efficiency of Apis mellifera L. on the Oil Tree Peony ‘Feng Dan’ (Paeonia ostii T. Hong et J.X. Zhang)" Insects 10, no. 4: 116. https://doi.org/10.3390/insects10040116
APA StyleHe, C., Zhang, K., Hou, X., Han, D., & Wang, S. (2019). Foraging Behavior and Pollination Efficiency of Apis mellifera L. on the Oil Tree Peony ‘Feng Dan’ (Paeonia ostii T. Hong et J.X. Zhang). Insects, 10(4), 116. https://doi.org/10.3390/insects10040116




