Losing the Arms Race: Greater Wax Moths Sense but Ignore Bee Alarm Pheromones
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Chemicals
2.3. Electroantennogram Responses
2.4. Orientation Behavioral Tests
2.5. Oviposition Sites Preferences
2.6. Statistics
3. Results
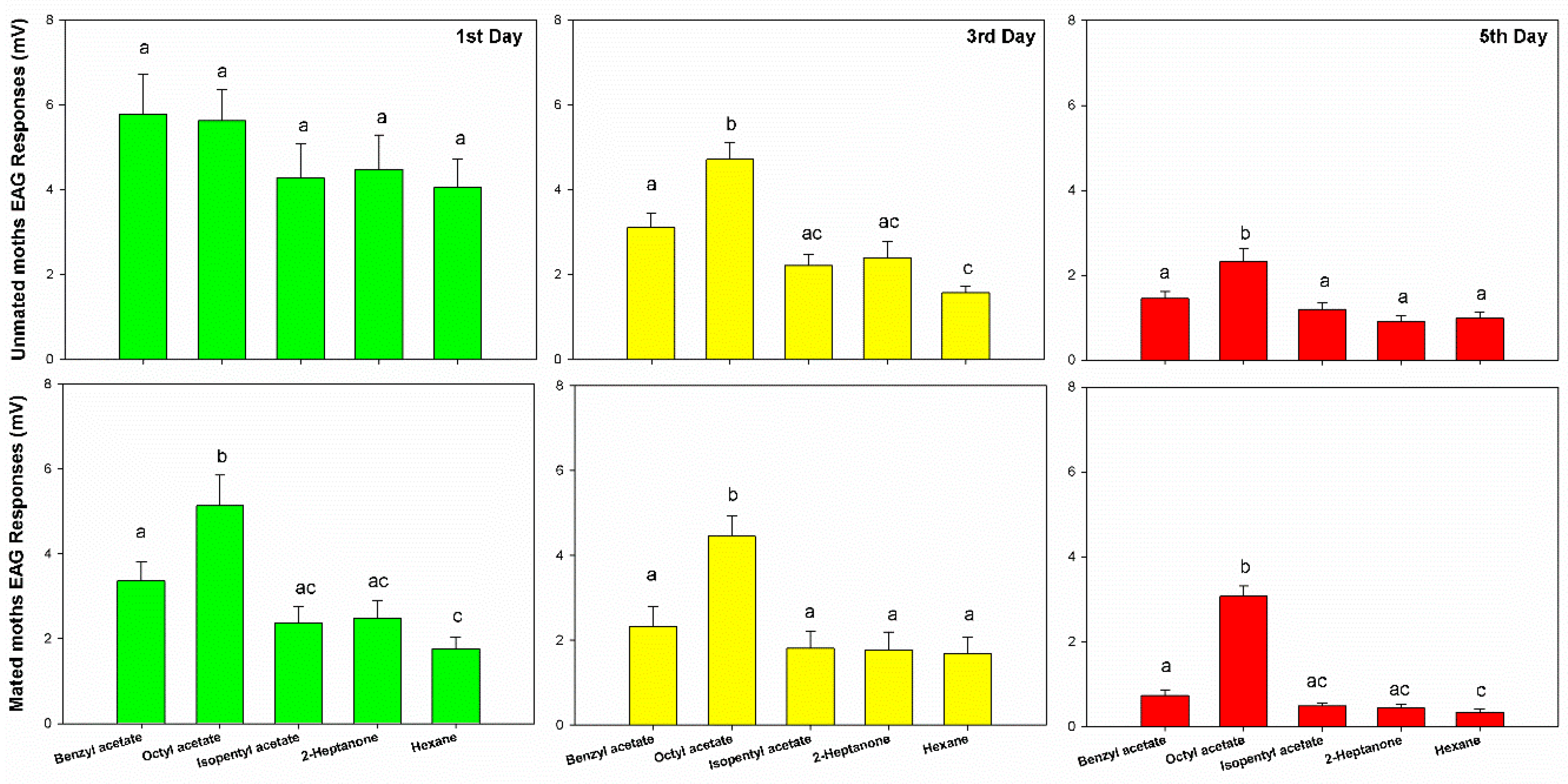
3.1. Electroantennogram Responses of Unmated/Mated Female GWM
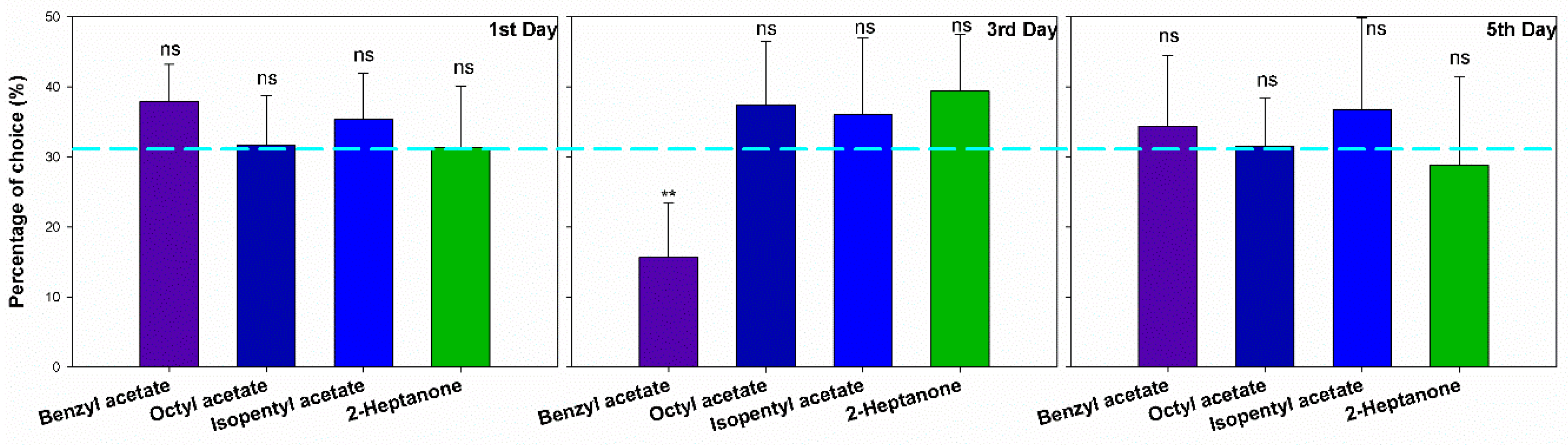
3.2. Orientation in Y Maze
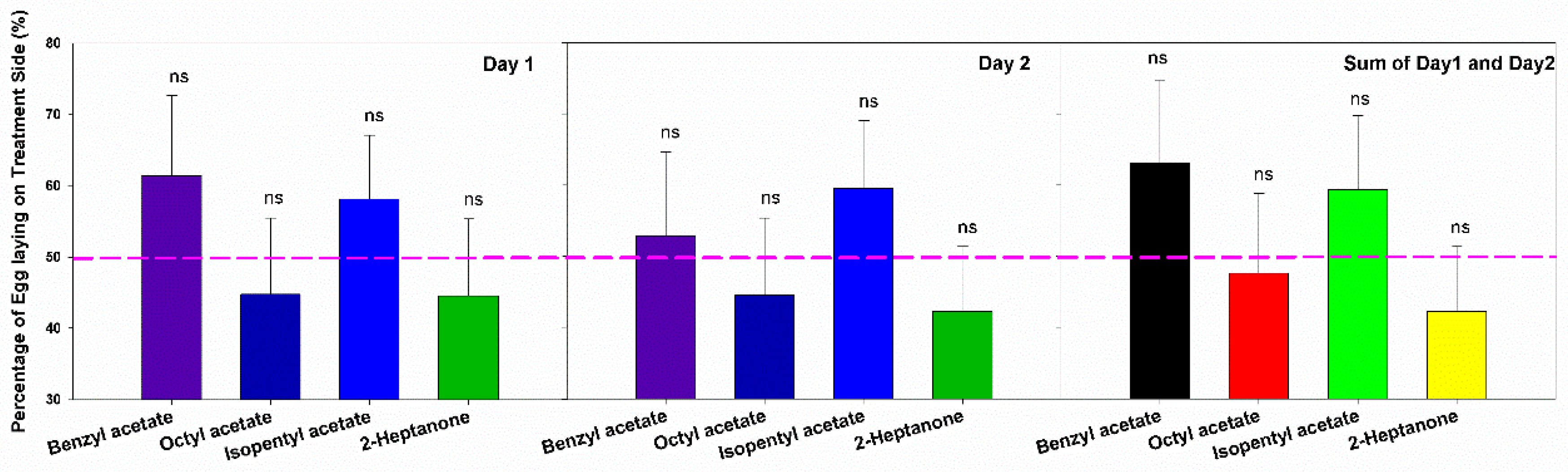
3.3. Oviposition Site Preference and Avoidance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sohail, M.; Aqueel, M.A.; Ellis, J.D.; Afzal, M.; Raza, A.M. Seasonal abundance of greater wax moths (Galleria mellonella L.) in hives of western honey bees (Apis mellifera L.) correlates with minimum and maximum ambient temperature. J. Apicult. Res. 2017, 56, 416–420. [Google Scholar] [CrossRef]
- Yang, S.; Liang, C.; Chen, S.; Song, W.; Luo, W.; Zhang, X. Different cleaning behaviors of Apis cerana and Apis mellfiera against the larvae and adults of Galleria mellonella L. J. Yunnan Agric. Univ. 2016, 31, 189–194. [Google Scholar]
- Jyothi, J.V.A. Studies on the development of Galleria mellonella Linnaeus (Lepidoptera: Pyralidae) on Apis dorsata honeycomb. J. Entomol. Res. 2003, 27, 185–188. [Google Scholar]
- Kwadha, C.A.; Ong’Amo, G.O.; Ndegwa, P.N.; Raina, S.K.; Fombong, A.T. The biology and control of the greater wax moth, Galleria mellonella. Insects 2017, 8, 61. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhang, X.; Song, W.; Miao, C.; Zhao, H.; Jiang, Y. Review of the biological and control methods on Galleria mellonella L. Apicult. China 2017, 67, 33–37. [Google Scholar]
- Nielsen, R.A.; Brister, C. Greater wax moth: Behavior of larvae. Ann. Entomol. Soc. Am. 1979, 72, 811–815. [Google Scholar] [CrossRef]
- Kecko, S.; Mihailova, A.; Kangassalo, K.; Elferts, D.; Krama, T.; Krams, R.; Luoto, S.; Rantala, M.J.; Krams, I.A. Sex-specific compensatory growth in the larvae of the greater wax moth Galleria mellonella. J. Evolut. Biol. 2017, 30, 1910–1918. [Google Scholar] [CrossRef]
- Nielsen, R.A.; Brister, D. The greater wax moth: Adult behavior. Ann. Entomol. Soc. Am. 1977, 70, 101–103. [Google Scholar] [CrossRef]
- Buyukguzel, E.; Buyukguzel, K.; Erdem, M.; Adamski, Z.; Marciniak, P.; Ziemnicki, K.; Ventrella, E.; Scrano, L.; Bufo, S.A. The influence of dietary alpha-solanine on the wax moth Galleria mellonella L. Arch. Insect Biochem. Physiol. 2013, 83, 15–24. [Google Scholar] [CrossRef]
- Coskun, M.; Kayis, T.; Sulanc, M.; Ozalp, P. Effects of different honeycomb and sucrose levels on the development of greater wax moth Galleria mellonella larvae. Int. J. Agric. Biol. 2006, 8, 855–858. [Google Scholar]
- Hanley, A.V.; Huang, Z.Y.; Pett, W.L. Effects of dietary transgenic Bt corn pollen on larvae of Apis mellifera and Galleria mellonella. J. Apicult. Res. 2003, 42, 77–81. [Google Scholar] [CrossRef]
- Burges, H.D. Control of wax moths: Physical, chemical and biological methods. Bee World 2015, 59, 129–138. [Google Scholar] [CrossRef]
- Ghimire, M.N.; Phillips, T.W. Suitability of different Lepidopteran host species for development of Bracon hebetor (Hymenoptera: Braconidae). Environ. Entomol. 2010, 39, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Dweck, H.K.M.; Svensson, G.P.; Gündüz, E.A.; Anderbrant, O. Kairomonal response of the parasitoid, Bracon hebetor Say, to the male-produced sex pheromone of its host, the greater wax moth, Galleria mellonella (L.). J. Chem. Ecol. 2010, 36, 171–178. [Google Scholar] [CrossRef]
- Leyrer, R.L.; Monroe, R.E. Isolation and identification of the scent of the moth, Galleria mellonella, and a revaluation of its sex pheromone. J. Insect Physiol. 1973, 19, 2267–2271. [Google Scholar] [CrossRef]
- Flint, H.M.; Merkle, J.R. Mating behavior, sex pheromone responses, and radiation sterilization of the greater wax moth (Lepidoptera: Pyralidae). J. Econ. Entomol. 1983, 76, 467–472. [Google Scholar] [CrossRef]
- Kwadha, C.A. Determination of Attractant Semiochemicals of the Wax Moth, Galleria mellonella L. In Honeybee Colonies; University of Nairobi: Nairobi, Kenya, 2017. [Google Scholar]
- Refsnider, J.M.; Janzen, F.J. Putting eggs in one basket: Ecological and evolutionary hypotheses for variation in oviposition-site choice. Ann. Rev. Ecol. Evolut.Syst. 2010, 41, 39–57. [Google Scholar] [CrossRef]
- And, J.A.A.R.; Chew, F.S. Oviposition behavior in Lepidoptera. Annu. Rev. Entomol. 1994, 39, 377–400. [Google Scholar]
- Li, J.; Wang, Z.; Tan, K.; Qu, Y.; Nieh, J.C. Effects of natural and synthetic alarm pheromone and individual pheromone components on foraging behavior of the giant Asian honey bee, Apis dorsata. J. Exp. Biol. 2014, 217, 3512–3518. [Google Scholar] [CrossRef]
- Wang, Z.; Wen, P.; Qu, Y.; Dong, S.; Li, J.; Tan, K.; Nieh, J.C. Bees eavesdrop upon informative and persistent signal compounds in alarm pheromones. Sci. Rep. 2016, 6, 25693. [Google Scholar] [CrossRef]
- Wen, P.; Cheng, Y.; Qu, Y.; Zhang, H.; Li, J.; Bell, H.; Tan, K.; Nieh, J. Foragers of sympatric Asian honey bee species intercept competitor signals by avoiding benzyl acetate from Apis cerana alarm pheromone. Sci. Rep. 2017, 7, 6721. [Google Scholar] [CrossRef]
- Shearer, D.; Boch, R. 2-Heptanone in the mandibular gland secretion of the honey-bee. Nature 1965, 205, 530. [Google Scholar] [CrossRef]
- Vallet, A.; Cassier, P.; Lensky, Y. Ontogeny of the fine structure of the mandibular glands of the honeybee (Apis mellifera L.) workers and the pheromonal activity of 2-heptanone. J. Insect Physiol. 1991, 37, 789–804. [Google Scholar] [CrossRef]
- Blum, M.S. Alarm pheromones. Annu. Rev. Entomol. 1969, 14, 57–80. [Google Scholar] [CrossRef]
- Koeniger, N.; Weiss, J.; Maschwitz, U. Alarm pheromones of the sting in the genus Apis. J. Insect Physiol. 1979, 25, 467–476. [Google Scholar] [CrossRef]
- Vetter, R.S.; Visscher, P.K. Influence of age on antennal response of male honey bees, Apis mellifera, to queen mandibular pheromone and alarm pheromone component. J. Chem. Ecol. 1997, 23, 1867–1880. [Google Scholar] [CrossRef]
- Yang, S.; Liu, W.; Zhao, H.; Du, Y.; Pan, J.; Wang, S.; Guo, L.; Xu, K.; Jiang, Y. Observation on antennal sensillia of Galleria mellonella L. with scanning electron microscope. Apicult. China 2017, 68, 16–19. [Google Scholar]
- Trhlin, M.; Rajchard, J. Chemical communication in the honeybee (Apis mellifera L.): A review. Vet. Med. 2011, 56, 265–273. [Google Scholar] [CrossRef]
- Van Oystaeyen, A.; Oliveira, R.C.; Holman, L.; van Zweden, J.S.; Romero, C.; Oi, C.A.; d’Ettorre, P.; Khalesi, M.; Billen, J.; Wäckers, F. Conserved class of queen pheromones stops social insect workers from reproducing. Science 2014, 343, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Ping, W.; Qi, Z.; Yuan, W.; Cheng, Y.; Tan, K.; Nieh, J.C. Olfactory eavesdropping of predator alarm pheromone by sympatric but not allopatric prey. Anim. Behav. 2018, 141, 115–125. [Google Scholar] [CrossRef]
- Finn, W.E.; Payne, T.L. Attraction of greater wax moth females to male produced pheromones. Southwest. Entomol. 1977, 2, 879–881. [Google Scholar]
- Grigor’Eva, N.Y.; Tsiklauri, P.G.; Buevich, A.V. Synthesis of Z-11-octadecanal, the sex pheromone of wax moth. Cheminform 1995, 26. [Google Scholar] [CrossRef]
- Svensson, G.P.; Gündüz, E.A.; Sjöberg, N.; Hedenström, E.; Lassance, J.-M.; Wang, H.-L.; Löfstedt, C.; Anderbrant, O. Identification, synthesis, and behavioral activity of 5, 11-dimethylpentacosane, a novel sex pheromone component of the greater wax moth, Galleria mellonella (L.). J. Chem. Ecol. 2014, 40, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Farina, W.M.; Grüter, C.; Acosta, L.; Cabe, S.M. Honeybees learn floral odors while receiving nectar from foragers within the hive. Naturwissenschaften 2007, 94, 55–60. [Google Scholar] [CrossRef]
- Dupuy, F.; Rouyar, A.; Deisig, N.; Bourgeois, T.; Limousin, D.; Wycke, M.A.; Anton, S.; Renou, M. A background of a volatile plant compound alters neural and behavioral responses to the sex pheromone blend in a moth. Front. Physiol. 2017, 8, 79. [Google Scholar] [CrossRef] [PubMed]
- Rouyar, A.; Deisig, N.; Dupuy, F.; Limousin, D.; Wycke, M.A.; Renou, M.; Anton, S. Unexpected plant odor responses in a moth pheromone system. Front. Physiol. 2015, 6, 148. [Google Scholar] [CrossRef]
- Hood, W.M.M. The small hive beetle, Aethina tumida: A review. Bee World 2004, 85, 51–59. [Google Scholar] [CrossRef]
- Kührt, U.; Samietz, J.; Dorn, S. Thermal response in adult codling moth. Physiol. Entomol. 2010, 31, 80–88. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Jiang, X.; Wang, Z.; Zhang, J.; Klett, K.; Mehmood, S.; Qu, Y.; Tan, K. Losing the Arms Race: Greater Wax Moths Sense but Ignore Bee Alarm Pheromones. Insects 2019, 10, 81. https://doi.org/10.3390/insects10030081
Li Y, Jiang X, Wang Z, Zhang J, Klett K, Mehmood S, Qu Y, Tan K. Losing the Arms Race: Greater Wax Moths Sense but Ignore Bee Alarm Pheromones. Insects. 2019; 10(3):81. https://doi.org/10.3390/insects10030081
Chicago/Turabian StyleLi, Yuan, Xingchuan Jiang, Zhengwei Wang, Junjun Zhang, Katrina Klett, Shahid Mehmood, Yufeng Qu, and Ken Tan. 2019. "Losing the Arms Race: Greater Wax Moths Sense but Ignore Bee Alarm Pheromones" Insects 10, no. 3: 81. https://doi.org/10.3390/insects10030081
APA StyleLi, Y., Jiang, X., Wang, Z., Zhang, J., Klett, K., Mehmood, S., Qu, Y., & Tan, K. (2019). Losing the Arms Race: Greater Wax Moths Sense but Ignore Bee Alarm Pheromones. Insects, 10(3), 81. https://doi.org/10.3390/insects10030081



