Establishment in an Introduced Range: Dispersal Capacity and Winter Survival of Trissolcus japonicus, an Adventive Egg Parasitoid
Abstract
1. Introduction
1.1. Parasitoid Release and Dispersal Assessment
1.2. Winter Survival as a Condition for Successful Classical Biological Control
2. Methods
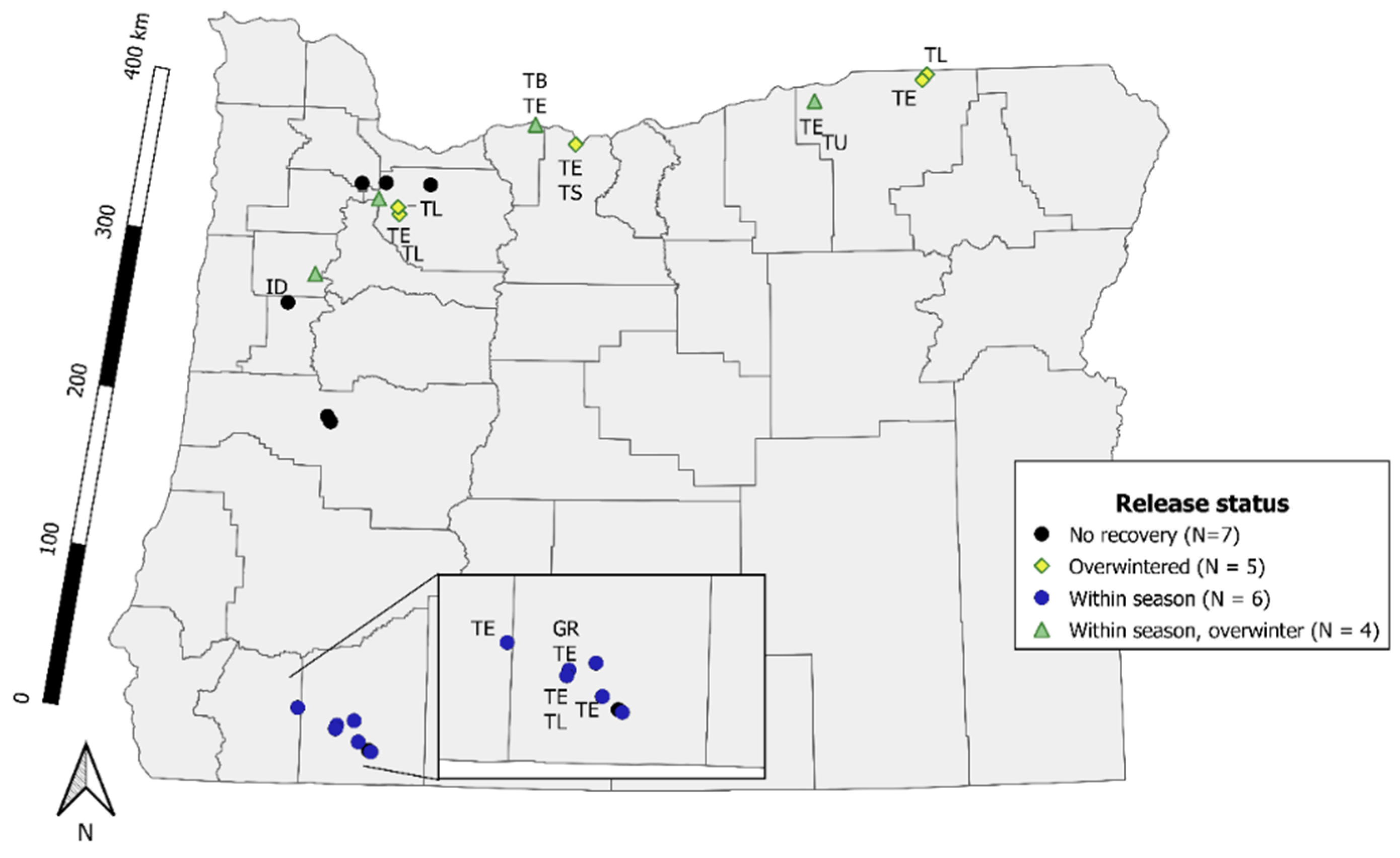
2.1. Redistribution and Short-Distance Recovery
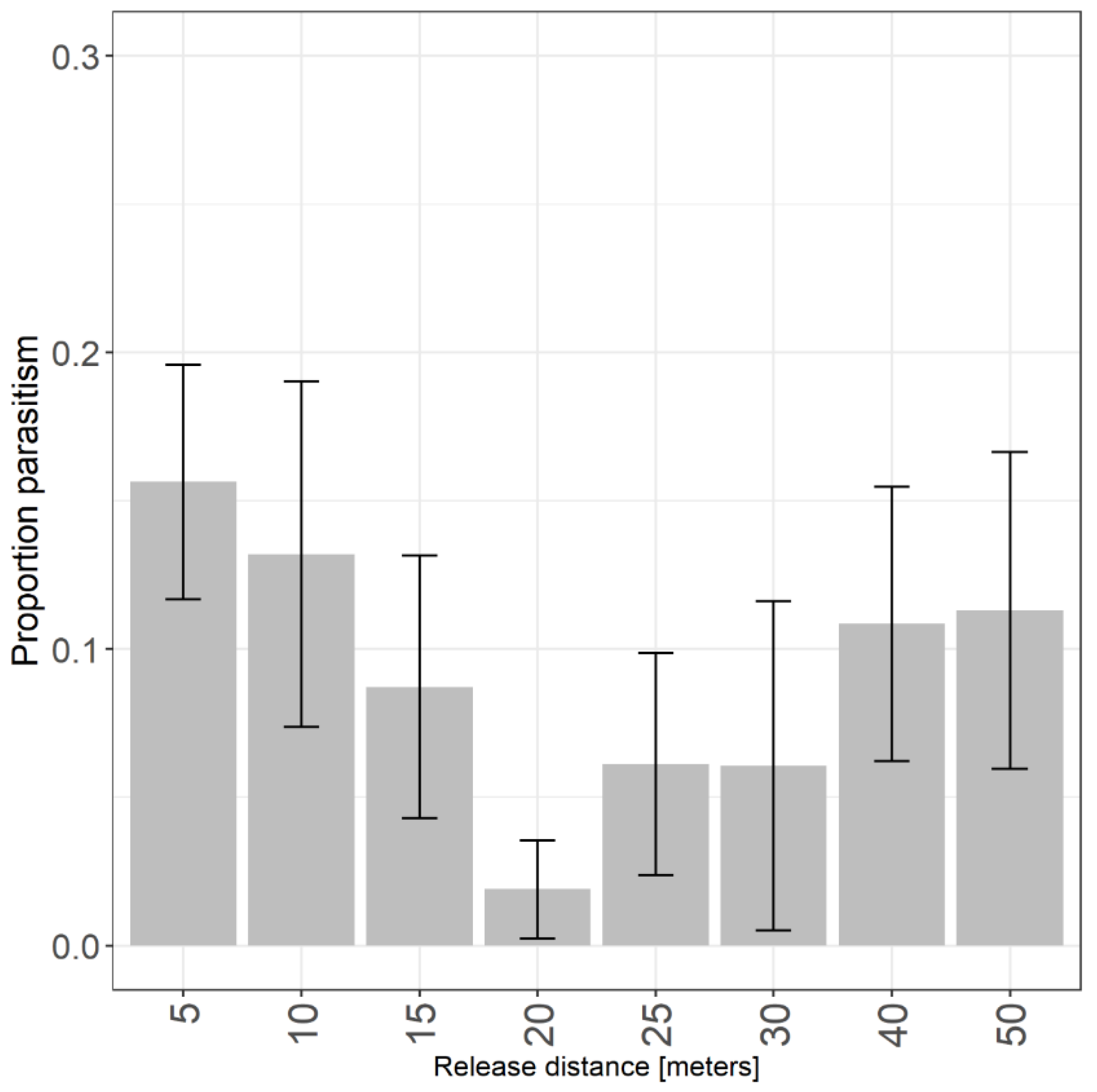
2.2. Releases and Dispersal in Crops
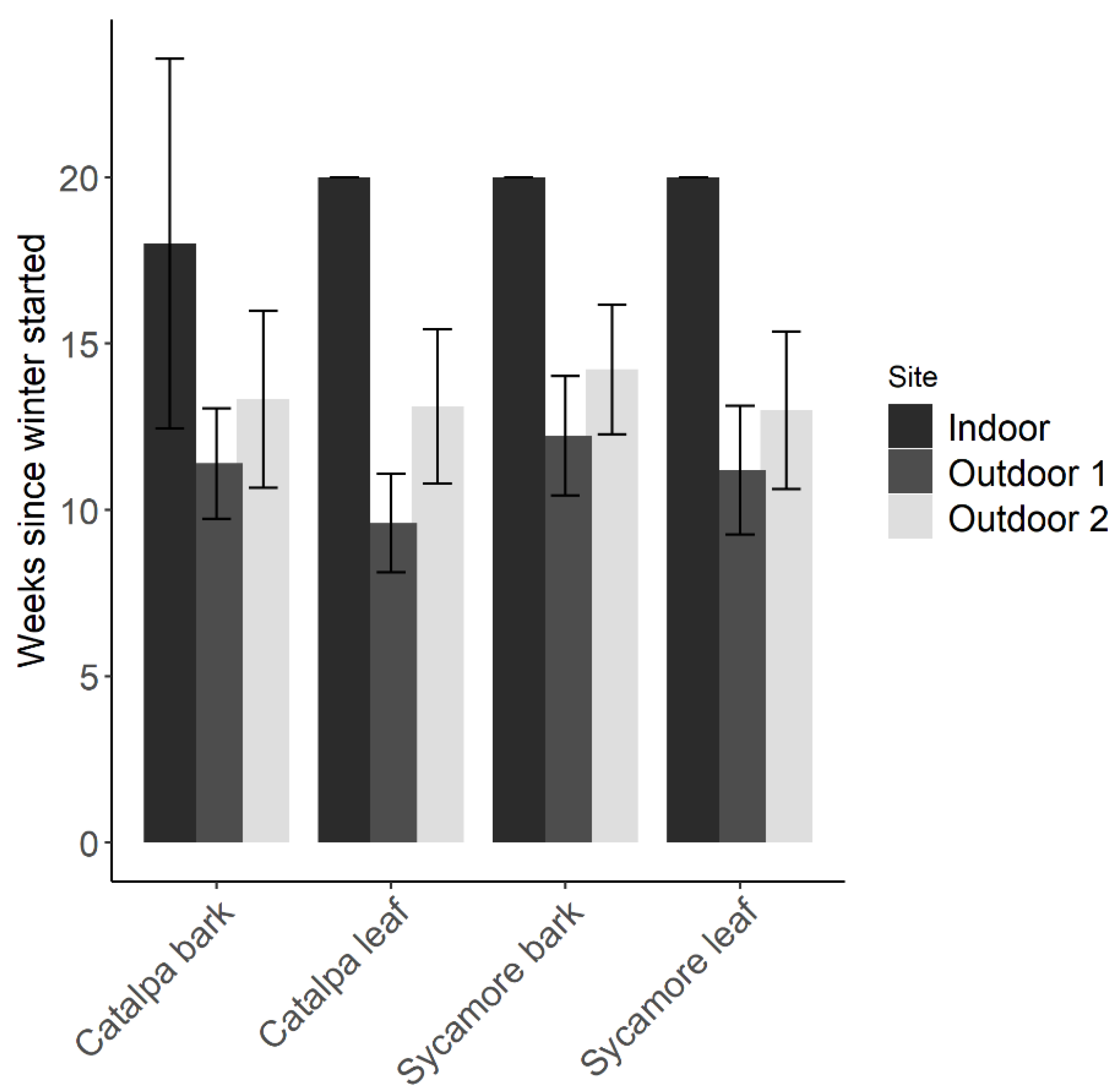
2.3. Parasitoid Longevity and Fecundity in Overwintering Habitat
2.4. Data Analysis
2.4.1. Redistribution and Short-Distance Recovery
2.4.2. Releases and Dispersal in Crops
2.4.3. Overwintering Habitat
3. Results
3.1. Redistribution and Short-Distance Recovery
3.2. Dispersal in Crops - Raspberry
3.3. Releases in Crops-Hazelnut
3.4. Parasitoid Longevity and Fecundity in Overwintering Habitat
4. Discussion
4.1. Parasitoid Release and Dispersal Assessment
4.2. Parasitoid Longevity and Fecundity in Overwintering Habitat
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hulme, P.E. Trade, transport, and trouble: Managing invasive species pathways in an era of globalization. J. Appl. Ecol. 2009, 46, 10–18. [Google Scholar] [CrossRef]
- Seebens, H.; Blackburn, T.M.; Dyer, E.E.; Genovesi, P.; Hulme, P.E.; Jeschke, J.M.; Pagad, S.; Pyšek, P.; Winter, M.; Arianoutsou, M.; et al. No saturation in the accumulation of alien species worldwide. Nat. Commun. 2017, 8, 14435. [Google Scholar] [CrossRef] [PubMed]
- Paini, D.R.; Sheppard, A.W.; Cook, D.C.; De Barro, P.J.; Worner, S.P.; Thomas, M.B. Global threat to agriculture from invasive species. Proc. Natl. Acad. Sci. USA 2016, 113, 7575–7579. [Google Scholar] [CrossRef] [PubMed]
- Holmes, T.P.; Aukema, J.E.; Van Holle, B.; Leibhold, A.; Sills, E. Economic impacts of invasive species in Forests. Ann. N. Y. Acad. Sci. 2009, 1162, 18–38. [Google Scholar] [CrossRef] [PubMed]
- Connor, E.F.; Hafernik, J.; Levy, J.; Moore, V.L.; Rickman, J.K. Insect conservation in an urban biodiversity hotspot: The San Francisco Bay area. J. Insect Conserv. 2002, 6, 247–259. [Google Scholar] [CrossRef]
- Snyder, W.E.; Evans, E.W. Ecological effects of invasive arthropod generalist predators. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 95–122. [Google Scholar] [CrossRef]
- Tillberg, C.V.; Holway, D.A.; LeBrun, E.G.; Suarez, A.V. Trophic ecology of invasive Argentine ants in their native and introduced ranges. Proc. Natl. Acad. Sci. USA 2007, 52, 20856–20861. [Google Scholar] [CrossRef]
- Gardiner, M.M.; O’Neal, M.E.; Landis, D.A. Intraguild predation and native lady beetle decline. PLoS ONE 2011, 6, e23576. [Google Scholar] [CrossRef]
- Abram, P.K.; Gariepy, T.D.; Boivin, G.; Brodeur, J. An invasive stink bug as an evolutionary trap for an indigenous parasitoid. Biol. Invasions 2014, 16, 1387–1395. [Google Scholar] [CrossRef]
- Alyokhin, A.; Sewell, G. Changes in a lady beetle community following the establishment of three alien species. Biol. Invasions 2004, 6, 463–471. [Google Scholar] [CrossRef]
- Pimentel, D.; Zuniga, R.; Morrison, D. Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol. Econ. 2005, 52, 273–288. [Google Scholar] [CrossRef]
- Colazza, S.; Giangiuliani, G.; Bin, F. Fortuitous introduction and successful establishment of Trichopoda pennipes: Parasitoid of Nezara. viridula. Biol. Control 1996, 6, 409–411. [Google Scholar] [CrossRef]
- Talamas, E.J.; Herlihy, M.V.; Dieckhoff, C.; Hoelmer, K.A.; Buffington, M.; Bon, M.; Weber, D.C. Trissolcus japonicus (Ashmead) (Hymenoptera: Scelionidae) emerges in North America. J. Hymenopt. Res. 2015, 43, 119–128. [Google Scholar] [CrossRef]
- Stahl, J.; Tortorici, F.; Pontini, M.; Bon, M.; Hoelmer, K.; Marazzi, C.; Tavella, L.; Haye, C. First discovery of adventive populations of Trissolcus japonicus in Europe. J. Pest Sci. 2019, 92, 371–379. [Google Scholar] [CrossRef]
- Sabbatini Peverieri, G.; Talamas, E.; Bon, M.; Marianelli, L.; Bernardinelli, I.; Malossini, G.; Benvenuto, L.; Roversi, P.; Hoelmer, K. Two Asian egg parasitoids of Halyomorpha halys (Hemiptera: Pentatomidae) emerge in Northern Italy: Trissolcus mitssukurii (Ashmead) and Trissolcus japonicus (Ashmead) (Hymenoptera, Scelionidae). J. Humenopt. Res. 2019, 67, 37–53. [Google Scholar] [CrossRef]
- Van Lenteren, J.C.; Bueno, V.H.P. Augmentative biological control of arthropods in Latin America. BioControl 2003, 48, 123–139. [Google Scholar] [CrossRef]
- Waage, J.K.; Ming, N.S. The reproductive strategy of a parasitic wasp. J. Anim. Ecol. 1984, 53, 401–415. [Google Scholar] [CrossRef]
- Craig, S.F.; Slobodkin, L.B.; Wray, G.A.; Biermann, C.H. The ‘paradox’ of polyembryony: A review of the cases and a hypothesis for its evolution. Evol. Ecol. 1997, 11, 127–143. [Google Scholar] [CrossRef]
- Caltagirone, L.E. Landmark examples in biological control. Annu. Rev. Entomol. 1981, 26, 213–232. [Google Scholar] [CrossRef]
- Leskey, T.C.; Nielsen, A.L. Impact of the invasive brown marmorated stink bug in North America and Europe: History, biology, ecology, and management. Annu. Rev. Entomol. 2018, 63, 599–618. [Google Scholar] [CrossRef]
- Milnes, J.M.; Wiman, N.G.; Talamas, E.J.; Brunner, J.F.; Hoelmer, K.A.; Buffington, M.L.; Beers, E.H. Discovery of an exotic egg parasitoid of the brown marmorated stink bug, Halyomorpha halys (Stål) in the Pacific Northwest. Proc. Entomol. Soc. WA 2016, 118, 466–470. [Google Scholar] [CrossRef]
- Northeastern IPM Center. Available online: Stopbmsb.org/where-is-bmsb/state-by-state/ (accessed on 21 March 2019).
- Abram, P.K.; Talamas, E.J.; Acheampong, S.; Mason, P.G.; Gariepy, T.G. First detection of the samurai wasp, Trissolcus japonicus, in Canada. J. Hymenop. Res. 2019, 68, 29–36. [Google Scholar] [CrossRef]
- Hedstrom, C.; Lowenstein, D.M.; Andrews, H.; Bai, B.; Wiman, N. Pentatomid host suitability and the discovery of introduced populations of Trissolcus japonicus in Oregon. J. Pest Sci. 2017, 90, 1169–1179. [Google Scholar] [CrossRef]
- McIntosh, H.; Lowenstein, D.M.; Wiman, N.G.; Wong, J.S.; Lee, J.C. Parasitism of frozen Halyomorpha halys eggs by Trissolcus japonicus: Applications for rearing and experimentation. Biocontrol. Sci. Technol. 2019, 29, 478–493. [Google Scholar] [CrossRef]
- Yang, Z.Q.; Yao, Y.; Qiu, L.; Li, Z. A new species of Trissolcus (Hymenoptera: Scelionidae) parasitizing eggs of Halyomorpha halys (Hemiptera: Pentatomidae) in China with comments on its biology. Ann. Entomol. Soc. Am. 2009, 102, 39–47. [Google Scholar] [CrossRef]
- Orr, D.B. Scelionid wasps as biological control agents: A review. Fla. Entomol. 1988, 71, 506–528. [Google Scholar] [CrossRef]
- Corrêa-Ferreira, B.S.; Moscardi, F. Biological control of soybean stink bug by inoculative releases of Trissolcus basalis. Entomol. Exp. Appl. 2006, 79, 1–7. [Google Scholar] [CrossRef]
- Wright, M.G.; Diez, J.M. Egg parasitism by Trissolcus basalis (Hymenoptera: Scelionidae) in architecturally varied habitats, and observations on parasitism in macademia nut orchards and other habitats following augmentative release. Proc. Hawaiian Entomol. Soc. 2011, 43, 23–31. [Google Scholar]
- Islamoglu, M. Mass rearing and release of the egg parasitoid, Trissolcus semistratus, a biological control agent of the Sunn Pest, Eurygaster integriceps, in Turkey. Egypt. J. Biol. Pest Control 2011, 21, 131–136. [Google Scholar]
- Lee, J.C.; Heimpel, G.E. Floral resources impact longevity and oviposition rate of a parasitoid in the field. J. Anim. Ecol. 2008, 77, 565–572. [Google Scholar] [CrossRef]
- Blaauw, B.R.; Isaacs, R. Wildflower plantings enhance the abundance of natural enemies and their services in adjacent blueberry fields. Biol. Control 2015, 91, 94–103. [Google Scholar] [CrossRef]
- Garcia-Salazar, C.; Landis, D.A. Marking Trichogramma brassicae with fluorescent market dust and its effect on survival and flight behavior. J. Econ. Entomol. 1997, 90, 1546–1550. [Google Scholar] [CrossRef]
- Irvin, N.; Hagler, J.; Hoddle, M. Measuring natural enemy dispersal from cover crops in a California vineyard. Biol. Control 2018, 126, 15–25. [Google Scholar] [CrossRef]
- Lee, S.C. Evaluation of Ooencyrtus Submetallicus (Howard) and Trissolcus Basalis (Wollaston) as Egg Parasites of Nezara viridula (L.). Ph.D. Dissertation, Louisiana State University, Baton Rouge, LA, USA, 1979. [Google Scholar]
- Caleca, V.; Lo Verde, G.; Rizzo, M.C.; Rizzo, R. Dispersal rate and parasitism by Closterocerus chamaeleon (Girault) after its release in Sicily to control Ophelimus maskelli (Ashmead) (Hymenoptera, Eulophidae). Biol. Control 2011, 57, 66–73. [Google Scholar] [CrossRef]
- Petit, J.N.; Hoddle, M.; Grandgirard, J.; Roderick, G.K.; Davies, N. Short-distance dispersal behavior an establishment of the parasitoid Gonatocerus ashmeadi (Hymenoptera: Mymaridae) in Tahiti: Implications for its use as a biological control agent against Homalodisca vitripennis (Hemiptera: Cicadellidae). Biol. Control 2008, 45, 344–352. [Google Scholar] [CrossRef]
- Lodos, N. Some Notes on the Sunn Pest. (Eurygaster Integriceps Put.) Problem in Iraq, Iran, Syria and Turkey; Faculty of Agriculture Press, Ege University: Izmir, Turkey, 1961; p. 115. (In Turkish) [Google Scholar]
- Qiu, L.F. Studies on Biology of the Brown Marmarated Stink Bug Halyomorpha Halys (Stål) (Hemiptera: Pentatomidae), an Important Pest for Pome Trees in China and Its Biological Control. Ph.D. Dissertation, Chinese Academy of Forestry, Beijing, China, 2007. [Google Scholar]
- Torgersen, T.R.; Ryan, R.B. Field biology of Telenomus californicus (Ashmead), an important egg parasite of douglas-fir tussock moth. Ann. Entomol. Soc. Am. 1981, 74, 185–186. [Google Scholar] [CrossRef]
- Grijpma, P. Host specificity of Telenomus nitidulus (Thomson) (Hymenoptera: Scelionidae), egg parasite of the satin moth, Leucoma salicis L. Nederlands Bosbouwtijdschrift. Rev. Appl. Entomol. 1985, 56, 201–207. [Google Scholar]
- Santacruz, E.N.; Venette, R.; Dieckhoff, C.; Hoelmer, K.; Koch, R. Cold tolerance of Trissolcus japonicus and T. cultratus, potential biological control agents of Halyomorpha halys, the brown marmorated stink bug. Biol. Control 2017, 107, 11–20. [Google Scholar] [CrossRef]
- Wiman, N. Biological control of Halyomorpha halys in Oregon 2014–2017. Unpublished data.
- Blaauw, B.R.; Polk, D.; Nielsen, A.L. IPM-CPR for peaches: Incorporating behaviorally-based methods to manage Halyomorpha halys and key pests in peach. Pest Manag. Sci. 2015, 71, 1513–1522. [Google Scholar] [CrossRef]
- Venugopal, P.D.; Dively, G.P.; Lamp, W.O. Spatiotemporal dynamics of the invasive Halyomorpha halys in and between adjacent corn and soybean fields. J. Econ. Entomol. 2015, 108, 2231–2241. [Google Scholar] [CrossRef]
- Therneau, T. A Package for Survival Analysis in S. version 2.38. 2015. Available online: https://CRAN.R-project.org/package=survival (accessed on 3 May 2019).
- Ellis, J.A.; Walter, A.D.; Tooker, J.F.; Ginzel, M.D.; Reagel, P.F.; Lacey, E.S.; Bennett, A.B.; Grossman, E.M.; Hanks, L.M. Conservation biological control in urban landscapes: Manipulating parasitoids of bagworm with flowering forbs. Biol. Control 2005, 34, 99–107. [Google Scholar] [CrossRef]
- Botias, C.; David, A.; Hill, E.M.; Goulson, D. Quantifying exposure of wild bumblebees to mixtures of agrochemicals in agricultural and urban landscapes. Environ. Pollut. 2017, 222, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Lowenstein, D.M.; Andrews, H.A.; Mugica, A.; Wiman, N.G. Sensitivity of the egg parasitoid Trissolcus japonicus to field and laboratory-applied insecticide residue. J. Econ. Entomol. 2019, 112, 2077–2084. [Google Scholar] [CrossRef] [PubMed]
- Justo, H.D.; Shepard, B.M.; Elsey, K.D. Dispersal of the egg parasitoid Trissolcus basalis (Hymenoptera: Scelionidae) in tomato. J. Agric. Entomol. 1997, 14, 139–149. [Google Scholar]
- Machtinger, E.T.; Geden, C.J.; Leppla, N.C. Linear Dispersal of the Filth Fly Parasitoid Spalangia cameroni (Hymenoptera: Pteromalidae) and Parasitism of Hosts at Increasing Distances. PLoS ONE 2015, 10, e0129105. [Google Scholar] [CrossRef]
- Weisser, W.W.; Volkl, W. Dispersal in the aphid parasitoid. Lysiphlebus cardui (Marshall). J. Appl. Entomol. 2009, 121, 23–28. [Google Scholar] [CrossRef]
- Avila, G.A.; Berndt, L.A.; Holwell, G.I. Dispersal behavior of the parasitic wasp Cotesia urabae (Hymenoptera: Braconidae): A recently introduced biocontrol agent for the control of Uraba lugens (Lepidoptera: Nolidae) in New Zealand. Biol. Control 2013, 66, 166–172. [Google Scholar] [CrossRef]
- Cronin, J.T.; Strong, D.R. Dispersal-dependent oviposition and the aggregation of parasitism. Am. Nat. 1999, 154, 23–36. [Google Scholar] [CrossRef]
- Chambers, D.L. Quality control in mass rearing. Annu. Rev. Entomol. 1977, 22, 289–308. [Google Scholar] [CrossRef]
- Jones, A.L.; Jennings, D.E.; Hooks, C.R.R.; Shrewsbury, P.M. Sentinel eggs underestimate rates of parasitism of the exotic brown marmorated stink bug. Halyomorpha. halys. Biol. Control 2014, 78, 61–66. [Google Scholar] [CrossRef]
- Tillman, P.G. Diversity of stink bug egg parasitoids in woodland and crop habitats in southwest Georgia, USA. Fl. Entomol. 2016, 99, 286–291. [Google Scholar] [CrossRef]
- Jones, V.P. Reassessment of the role of predators and Trissolcus basalis in biological control of southern green stink bug (Hemiptera: Pentatomidae) in Hawaii. Biol. Control 1995, 5, 566–572. [Google Scholar] [CrossRef]
- Quinn, N.F.; Talamas, E.J.; Acebes-Doria, A.L.; Leskey, T.C.; Bergh, J.C. Vertical sampling in tree canopies for Halyomorpha halys (Hemiptera: Pentatomidae) Life Stages and its Egg Parasitoid, Trissolcus japonicus (Hymenoptera: Scelionidae). Environ. Entomol. 2019, 48, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Voronin, K.E. Ecological aspects of the behavior of Telenominia. In Insect Behavior as a Basis for Developing Control Measures against Pests of Field Crops and Forests; Pristavko, V.P., Ed.; Amerind Publisher: New Delhi, India, 1981; pp. 36–41. [Google Scholar]
- Sakai, A. Temperature Fluctuation in Wintering Trees. Physiol. Plant. 1966, 19, 105–114. [Google Scholar] [CrossRef]
- Vermunt, B.; Cuddington, K.; Sobek-Swant, S.; Crosthwaite, J.C.; Lyons, D.B.; Sinclair, B.J. Temperatures experienced by wood-boring beetles in the under-bark microclimate. Forest. Ecol. Manag. 2012, 269, 149–157. [Google Scholar] [CrossRef]
- Olsen, J.L.; Pscheidt, J.W.; Walton, V.M. Growing Hazelnuts in the Pacific Northwest: Integrated Pest Management; Oregon State University Extension Catalog: Corvallis, OR, USA, 2017; EM9081. [Google Scholar]
- James, D.G. Fecundity, longevity, and overwintering of Trissolcus biproruli a parasitoid of Biprorulus bibax. Austral. Entomol. 1988, 27, 297–301. [Google Scholar] [CrossRef]
- Teraoka, T.; Numata, H. Winter survival an oviposition before and after overwintering of a parasitoid wasp, Ooencyrtus nezarae. Entomol. Sci. 2004, 7, 105–111. [Google Scholar] [CrossRef]
- Tarla, S.; Kornosor, S. Reproduction and survival of overwintered and F1 generation of two egg parasitoids of sunn pest, Eurygaster integriceps. Turk. J. Agric. For. 2009, 33, 257–265. [Google Scholar]
- Chen, H.; Zhang, H.; Zhu, K.Y.; Throne, J. Performance of diapausing parasitoid wasps, Habrobracon hebetor, after cold storage. Biol. Control 2013, 64, 186–194. [Google Scholar] [CrossRef]
- Ishihara, M.; Shimada, M. Trade-off in allocation of metabolic reserves: Effects of diapause on egg production and adult longevity in a multivoltine bruchid, Kyotorhinus sharpiaunus. Funct. Ecol. 1995, 9, 618–624. [Google Scholar] [CrossRef]
- Ellers, J.; Van Alphen, J.J.M. A trade-off between diapause duration and fitness in female parasitoids. Ecol. Entomol. 2002, 27, 279–284. [Google Scholar] [CrossRef]
- Foerster, L.A.; Doetzer, A.K. Cold storage of the egg parasitoids Trissolcus basalis and Telenomus podisi. Biol. Control 1996, 36, 232–237. [Google Scholar] [CrossRef]
- Colinet, H.; Boivin, G. Insect parasitoids cold storage: A comprehensive review of factors of variability and consequences. Biol. Control 2011, 58, 83–95. [Google Scholar] [CrossRef]
- Moraglio, S.T.; Tortorici, F.; Pansa, M.G.; Castelli, G.; Pontini, M.; Scovero, S.; Visentin, S.; Tavella, L. A 3 year survey of parasitism of Halyormorpha halys by egg parasitoids in Northern Italy. J. Pest Sci. 2019. [Google Scholar] [CrossRef]
- Konopka, J.K.; Poinapen, J.; Gariepy, T.; Holdsworth, D.W.; McNeil, J.M. Timing of failed parasitoid development in Halyomorpha halys eggs. Biol. Control 2020. [Google Scholar] [CrossRef]


| 2017 | 2018 | |||
|---|---|---|---|---|
| Bark | Leaves | Bark | Leaves | |
| Catalpa | 24 (10) | 24 (8) | 19 (5) | 19 (5) |
| Sycamore | 24 (8) | 24 (8) | 18 (6) | 18 (5) |
| Variable | All Sites | Outdoor | Indoor |
|---|---|---|---|
| Treatment | 13.86 ** | 23.00 ** | 10.01 * |
| Plant | 3.70 ** | 2.52 ** | 1.16 |
| Woody debris | 0.326 | 4.02 ** | 2.36 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lowenstein, D.M.; Andrews, H.; Hilton, R.J.; Kaiser, C.; Wiman, N.G. Establishment in an Introduced Range: Dispersal Capacity and Winter Survival of Trissolcus japonicus, an Adventive Egg Parasitoid. Insects 2019, 10, 443. https://doi.org/10.3390/insects10120443
Lowenstein DM, Andrews H, Hilton RJ, Kaiser C, Wiman NG. Establishment in an Introduced Range: Dispersal Capacity and Winter Survival of Trissolcus japonicus, an Adventive Egg Parasitoid. Insects. 2019; 10(12):443. https://doi.org/10.3390/insects10120443
Chicago/Turabian StyleLowenstein, David M., Heather Andrews, Richard J. Hilton, Clive Kaiser, and Nik G. Wiman. 2019. "Establishment in an Introduced Range: Dispersal Capacity and Winter Survival of Trissolcus japonicus, an Adventive Egg Parasitoid" Insects 10, no. 12: 443. https://doi.org/10.3390/insects10120443
APA StyleLowenstein, D. M., Andrews, H., Hilton, R. J., Kaiser, C., & Wiman, N. G. (2019). Establishment in an Introduced Range: Dispersal Capacity and Winter Survival of Trissolcus japonicus, an Adventive Egg Parasitoid. Insects, 10(12), 443. https://doi.org/10.3390/insects10120443




