First Report of Cattle Tick Rhipicephalus (Boophilus) annulatus in Egypt Resistant to Ivermectin
Abstract
:1. Introduction
2. Materials and Methods
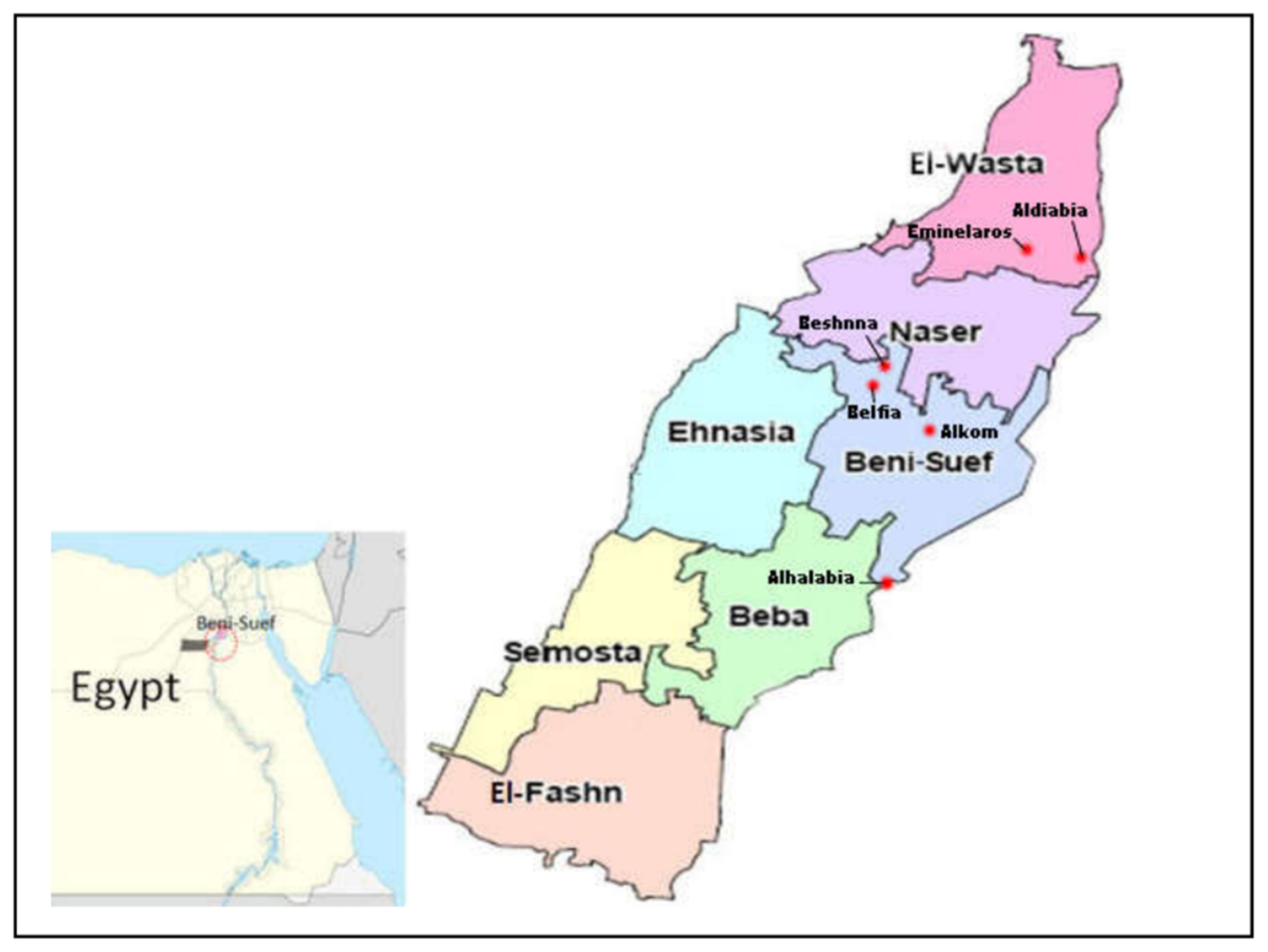
2.1. Study Area and Data Collection from Farmers
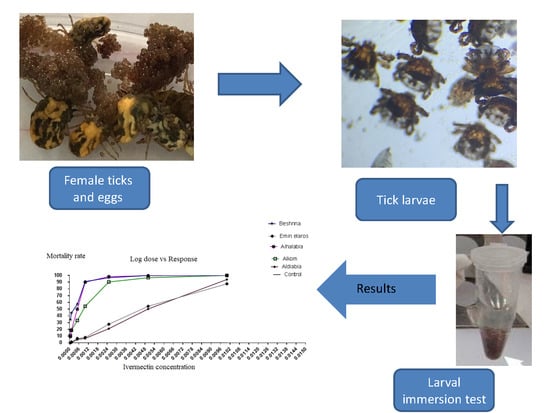
2.2. Collection of Ticks and Identification
2.3. Preparation of R. annulatus larvae
2.4. Preparation of Ivermectin for In Vitro Application
2.5. Larval Immersion Test (LIT)
2.6. Statistical Analysis
3. Results
3.1. History of Ivermectin Use at the Five Study Areas
3.2. Larval Immersion Test Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shyma, K.P.; Gupta, J.P.; Singh, V.; Patel, K.K. In Vitro Detection of Acaricidal Resistance Status of Rhipicephalus (Boophilus) microplus against Commercial Preparation of Deltamethrin, Flumethrin, and Fipronil from North Gujarat, India. J. Parasitol. Res. 2015, 2015, 506586. [Google Scholar] [CrossRef] [PubMed]
- Klafke, G.M.; Sabatini, G.A.; Thais, A.; Martins, J.R.; Kemp, D.H.; Miller, R.J.; Schumaker, T.T. Larval immersion tests with ivermectin in populations of the cattle tick Rhipicephalus (Boophilus) microplus (Acari: Ixodidae) from State of Sao Paulo, Brazil. Vet. Parasitol. 2006, 142, 386–390. [Google Scholar] [CrossRef] [PubMed]
- Aboelhadid, S.M.; Kamel, A.A.; Arafa, W.M.; Shokier, K.A. Effect of Allium sativum and Allium cepa oils on different stages of Boophilus annulatus. Parasitol. Res. 2013, 112, 1883–1890. [Google Scholar] [CrossRef] [PubMed]
- El-Ashram, S.; Al Nasr, I.; Abouhajer, F.; El-Kemary, M.; Huang, G.; Dincel, G.; Mehmood, R.; Hu, M.; Suo, X. Microbial community and ovine host response varies with early and late stages of Haemonchus contortus infection. Vet. Res. Commun. 2017, 41, 263–277. [Google Scholar] [CrossRef] [PubMed]
- El-Ashram, S.; Al Nasr, I.; El-Kemary, M.; Mehmood, R.; Hu, M.; Suo, X. Early and late gene expression profiles of the ovine mucosa in response to Haemonchus contortus infection employing Illumina RNA-seq technology. Parasitol. Int. 2017, 66, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Klafke, G.M.; Castro-Janer, E.; Mendes, M.C.; Namindome, A.; Schumaker, T.T. Applicability of in vitro bioassays for the diagnosis of ivermectin resistance in Rhipicephalus microplus (Acari: Ixodidae). Vet. Parasitol. 2012, 184, 212–220. [Google Scholar] [CrossRef]
- Martins, J.R.; Furlong, J. Avermectin resistance of the cattle tick Boophilus microplus in Brazil. Vet. Rec. 2001, 149, 64. [Google Scholar]
- Cully, D.F.; Vassilatis, D.K.; Liu, K.K.; Paress, P.S.; Van der Ploeg, L.H.; Schaeffer, J.M.; Arena, J.P. Cloning of an avermectin-sensitive glutamate-gated chloride channel from Caenorhabditis elegans. Nature 1994, 371, 707–711. [Google Scholar] [CrossRef]
- Le Gall, V.L.; Klafke, G.M.; Torres, T.T. Detoxification mechanisms involved in ivermectin resistance in the cattle tick, Rhipicephalus (Boophilus) microplus. Sci. Rep. 2018, 8, 12401. [Google Scholar] [CrossRef]
- Shaw, R.D. Culture of an organophosphorus-resistant strain of Boophilus microplus (Can.) and an assessment of its resistance spectrum. Bull. Entomol. Res. 1966, 56, 389–405. [Google Scholar] [CrossRef]
- Sabatini, G.A.; Kemp, D.H.; Hughes, S.; Nari, A.; Hansen, J. Tests to determine LC50 and discriminating doses for macrocyclic lactones against the cattle tick, Boophilus microplus. Vet. Parasitol. 2001, 95, 53–62. [Google Scholar] [CrossRef]
- Perez-Cogollo, L.C.; Rodriguez-Vivas, R.I.; Ramirez-Cruz, G.T.; Miller, R.J. First report of the cattle tick Rhipicephalus microplus resistant to ivermectin in Mexico. Vet. Parasitol. 2010, 168, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Castro-Janer, E.; Rifran, L.; Gonzalez, P.; Niell, C.; Piaggio, J.; Gil, A.; Schumaker, T.T. Determination of the susceptibility of Rhipicephalus (Boophilus) microplus (Acari: Ixodidae) to ivermectin and fipronil by Larval Immersion Test (LIT) in Uruguay. Vet. Parasitol. 2011, 178, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Aboelhadid, S.M.; Arafa, W.M.; Mahrous, L.N.; Fahmy, M.M.; Kamel, A.A. Molecular detection of Rhipicephalus (Boophilus) annulatus resistance against deltamethrin in middle Egypt. Vet. Parasitol. Reg. Stud. Rep. 2018, 13, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Salas, A.; Rodríguez-Vivas, R.I.; Alonso-Díaz, M.Á. Resistance of Rhipicephalus microplus to Amitraz and Cypermethrin in Tropical Cattle Farms in Veracruz, Mexico. J. Parasitol. 2012, 98, 1010–1015. [Google Scholar] [CrossRef]
- Hoogstraal, H.; Kaiser, M.N. The ticks (Ixodoidea) of Egypt: Abrief review and keys. J. Egypt. Public Health Assoc. 1958, 33, 51–85. [Google Scholar]
- Walker, A.R.; Bouattour, A.; Camicas, J.-L.; Estrada-Peña, A.; Horak, I.G.; Latif, A.A.; Pegram, R.G.; Preston, P.M. Ticks of Domestic Animals in Africa: A Guide to Identification of Species; Bioscience Reports: Edinburgh, UK, 2003; p. 157. [Google Scholar]
- Abou-Setta, M.M.; Sorrell, R.W.; Childers, C.C. A computer program in basic for determining probit and log-probit or logit correlation for toxicology and biology. Bull. Environ. Contam. Toxicol. 1986, 36, 242–249. [Google Scholar] [CrossRef]
- Borges, F.A.; Silva, H.C.; Buzzulini, C.; Soares, V.E.; Santos, E.; Oliveira, G.P.; Costa, A.J. Endectocide activity of a new long-action formulation containing 2.25% ivermectin+1.25% abamectin in cattle. Vet. Parasitol. 2008, 155, 299–307. [Google Scholar] [CrossRef]
- Holdsworth, P.A.; Kemp, D.; Green, P.; Peter, R.J.; De Bruin, C.; Jonsson, N.N.; Letonja, T.; Rehbein, S.; Vercruysse, J. World Association for the Advancement of Vet. Parasitol. (WAAVP) guidelines for evaluating the efficacy of acaricides against ticks (Ixodidae) on ruminants. Vet. Parasitol. 2006, 136, 29–43. [Google Scholar] [CrossRef]
- Singh, N.K.; Singh, H.; Jyoti, S.H.; Prerna, M.; Rath, S.S. First report of ivermectin resistance in field populations of Rhipicephalus (Boophilus) microplus (Acari: Ixodidae) in Punjab districts of India. Vet. Parasitol. 2015, 214, 192–194. [Google Scholar] [CrossRef]
- Chaparro, J.J.; López, A.; Puerta, J.; Villar, D. Ivermectin resistance of three Rhipicephalus microplus populations using the larval immersion test. RCCP Rev. Colomb. Cienc. Pecu. 2016, 29, 51–57. [Google Scholar]
- Cen-Aguilar, J.F.; Rodriguez-Vivas, R.I.; Dominguez-Alpizar, J.L.; Wagner, G.G. Studies on the effect of infection by Babesia sp. on oviposition of Boophilus microplus engorged females naturally infected in the Mexican tropics. Vet. Parasitol. 1998, 78, 253–257. [Google Scholar] [CrossRef]
- Lopez-Arias, A.; Villar-Argaiz, D.; Chaparro-Gutierrez, J.J.; Miller, R.J.; Perez de Leon, A.A. Reduced Efficacy of Commercial Acaricides Against Populations of Resistant Cattle Tick Rhipicephalus microplus from Two Municipalities of Antioquia, Colombia. Environ. Health Insights 2014, 8, 71–80. [Google Scholar] [CrossRef] [PubMed]
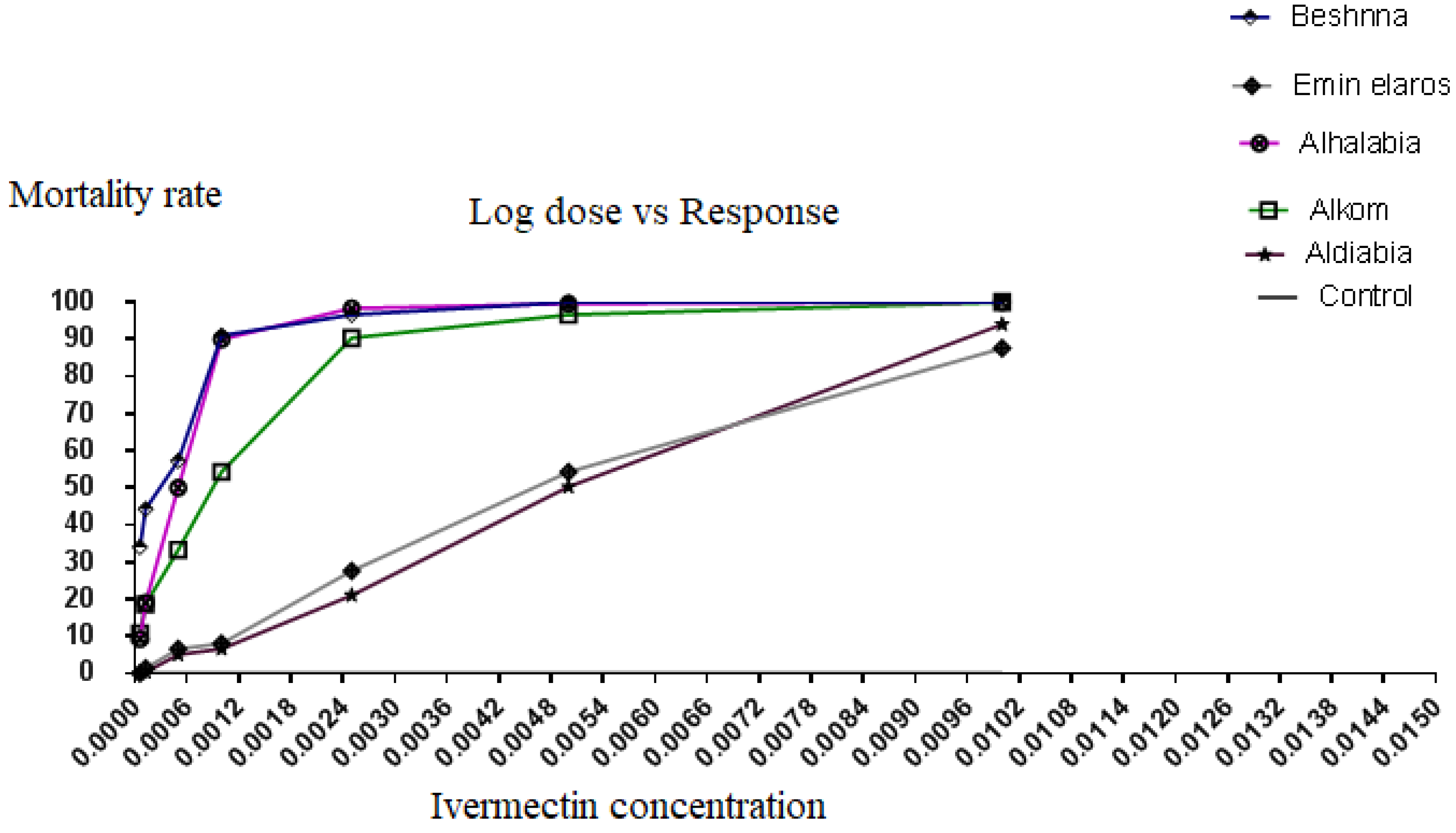
| Location/100 Larvae | Slope (SD) * | X2 ** | t for Slope *** | LC50 (95% CI) | LC90 (95% CI) |
|---|---|---|---|---|---|
| Alhalabia | 1918.292485 | 78.469074 | 2.609584 | 0.000524 (0.000175 to 0.000881) | 0.001097 (0.00056 to 0.001888) |
| Beshnna | 1113.815719 | 13.704343 | 4.840257 | 0.000286 (0.000062 to 0.000498) | 0.001272 (0.000779 to 0.00188) |
| Alkom | 705.544156 | 24.025441 | 4.823858 | 0.001091 (0.000633 to 0.001573) | 0.002648 (0.001585 to 0.003846) |
| Emin elaros | 278.371314 | 23.13296 | 6.079659 | 0.005189 (0.003788 to 0.006676) | 0.009269 (0.006482 to 0.012057) |
| Aldiabia | 330.039898 | 9.684015 | 12.838334 | 0.005093 (0.004663 to 0.005601) | 0.008422 (0.007669 to 0.009411) |
| Sample Site | |||||||
|---|---|---|---|---|---|---|---|
| Concentration | Alkom | Alhalabia | Beshnna | Aldiabia | Eminelaros | * p | |
| Ivermectin concentration % | 0.01 | 100.0 ± 0.0 | 100.0 ± 0.0 | 100.0 ± 0.0 | 94.00 ± 2.082 | 87.67 ± 1.453 | 0.0087 ** |
| 0.005 | 96.67 ± 0.8819 | 99.67 ± 0.3333 | 100.0 ± 0.0 | 50.33 ± 1.453 | 54.33 ± 2.333 | 0.0112 * | |
| 0.0025 | 90.33 ± 1.453 | 98.33 ± 0.8819 | 96.67 ± 0.8819 | 21.00 ± 2.082 | 27.67 ± 1.453 | 0.0115 * | |
| 0.001 | 54.33 ± 2.333 | 90.00 ± 0.0 | 91.00 ± 2.082 | 6.667 ± 0.8819 | 8.000 ± 1.155 | 0.0138 * | |
| 0.0005 | 33.33 ± 2.028 | 50.00 ± 0.0 | 57.33 ± 1.453 | 5.000 ± 1.155 | 6.667 ± 0.8819 | 0.0105 * | |
| 0.000125 | 18.67 ± 0.6667 | 19.00 ± 1.000 | 44.33 ± 2.333 | 0.3333 ± 0.3333 | 1.333 ± 0.8819 | 0.0139 * | |
| 0.0000625 | 10.67 ± 0.6667 | 9.333 ± 0.6667 | 34.00 ± 2.082 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0093 ** | |
| Negative control | 5 ± 2 | 6 ± 3 | 5 ± 1 | 7 ± 3 | 5 ± 2 | - | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Ashram, S.; Aboelhadid, S.M.; Kamel, A.A.; Mahrous, L.N.; Fahmy, M.M. First Report of Cattle Tick Rhipicephalus (Boophilus) annulatus in Egypt Resistant to Ivermectin. Insects 2019, 10, 404. https://doi.org/10.3390/insects10110404
El-Ashram S, Aboelhadid SM, Kamel AA, Mahrous LN, Fahmy MM. First Report of Cattle Tick Rhipicephalus (Boophilus) annulatus in Egypt Resistant to Ivermectin. Insects. 2019; 10(11):404. https://doi.org/10.3390/insects10110404
Chicago/Turabian StyleEl-Ashram, Saeed, Shawky M. Aboelhadid, Asmaa A. Kamel, Lilian N. Mahrous, and Magdy M. Fahmy. 2019. "First Report of Cattle Tick Rhipicephalus (Boophilus) annulatus in Egypt Resistant to Ivermectin" Insects 10, no. 11: 404. https://doi.org/10.3390/insects10110404
APA StyleEl-Ashram, S., Aboelhadid, S. M., Kamel, A. A., Mahrous, L. N., & Fahmy, M. M. (2019). First Report of Cattle Tick Rhipicephalus (Boophilus) annulatus in Egypt Resistant to Ivermectin. Insects, 10(11), 404. https://doi.org/10.3390/insects10110404