Cnaphalocrocis medinalis Moths Decide to Migrate when Suffering Nutrient Shortage on the First Day after Emergence
Abstract
:1. Introduction
2. Materials and Methods
2.1. Insects
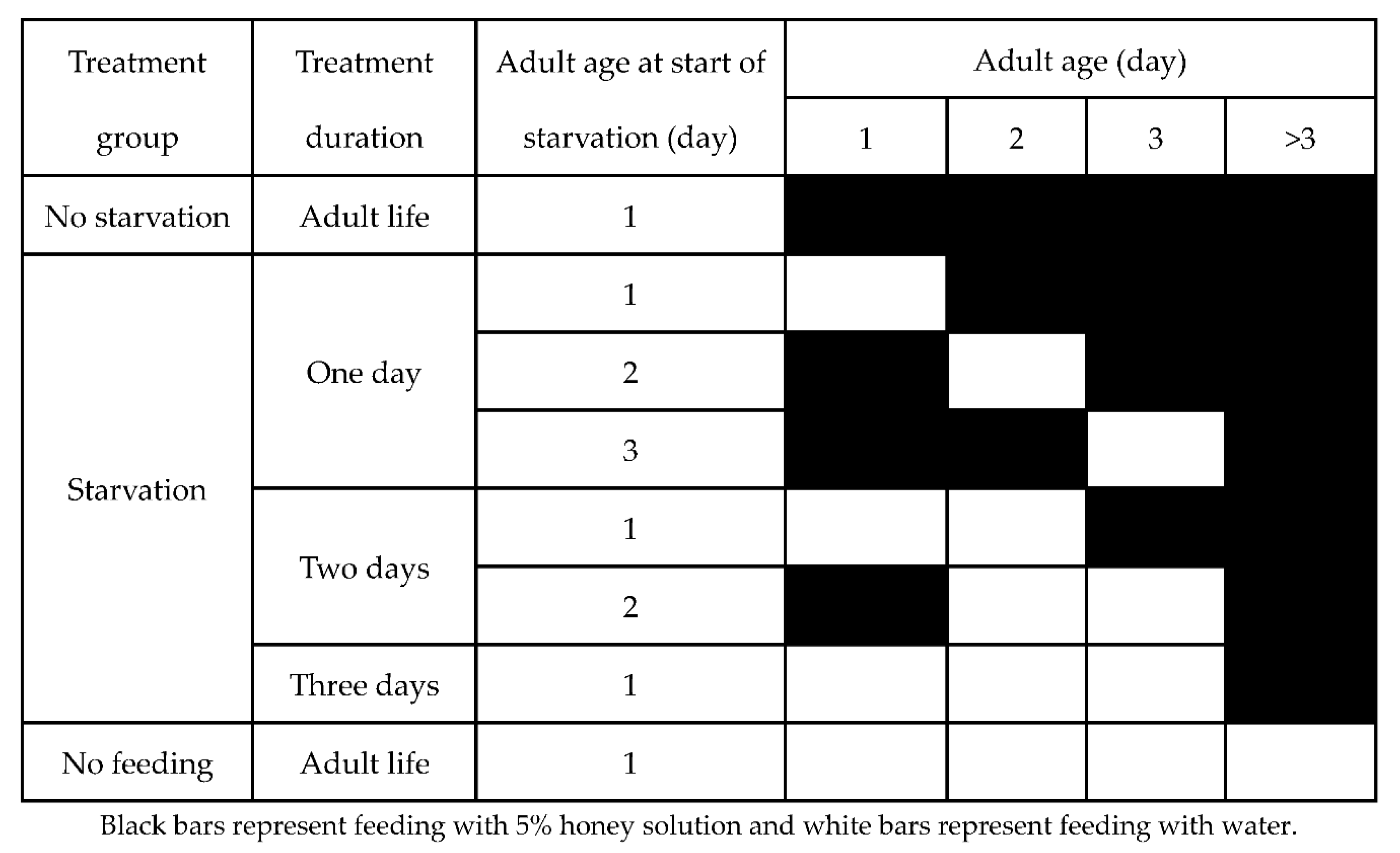
2.2. Adult Starvation
2.3. Reproductive Parameters of Female Moths
2.4. Migration Parameters
2.4.1. Percent Take-Off
2.4.2. Flight Capability Measurement
2.5. Determination of Triglyceride Content
2.6. Data Analysis
3. Results
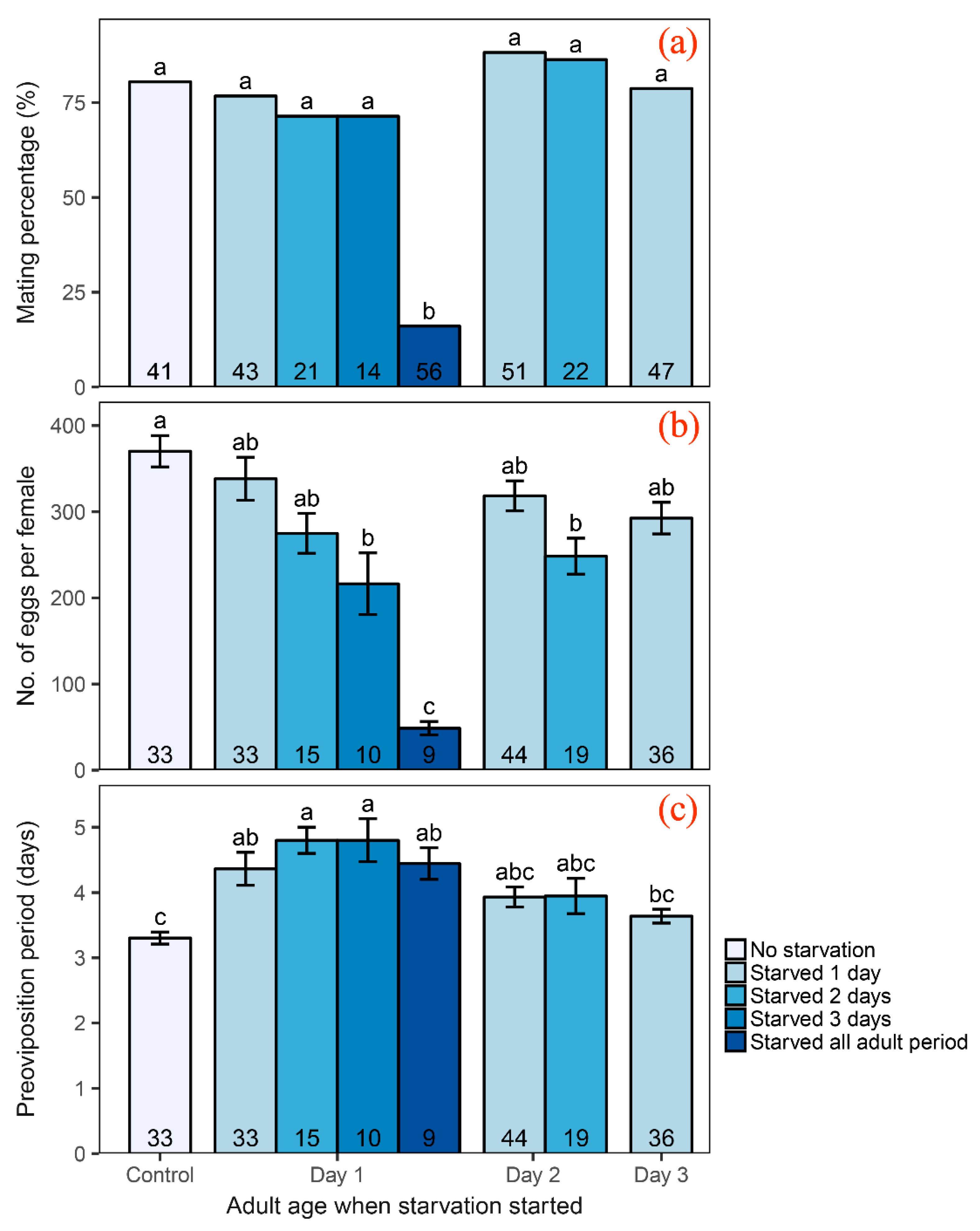
3.1. Influence of Adult Starvation on Reproduction
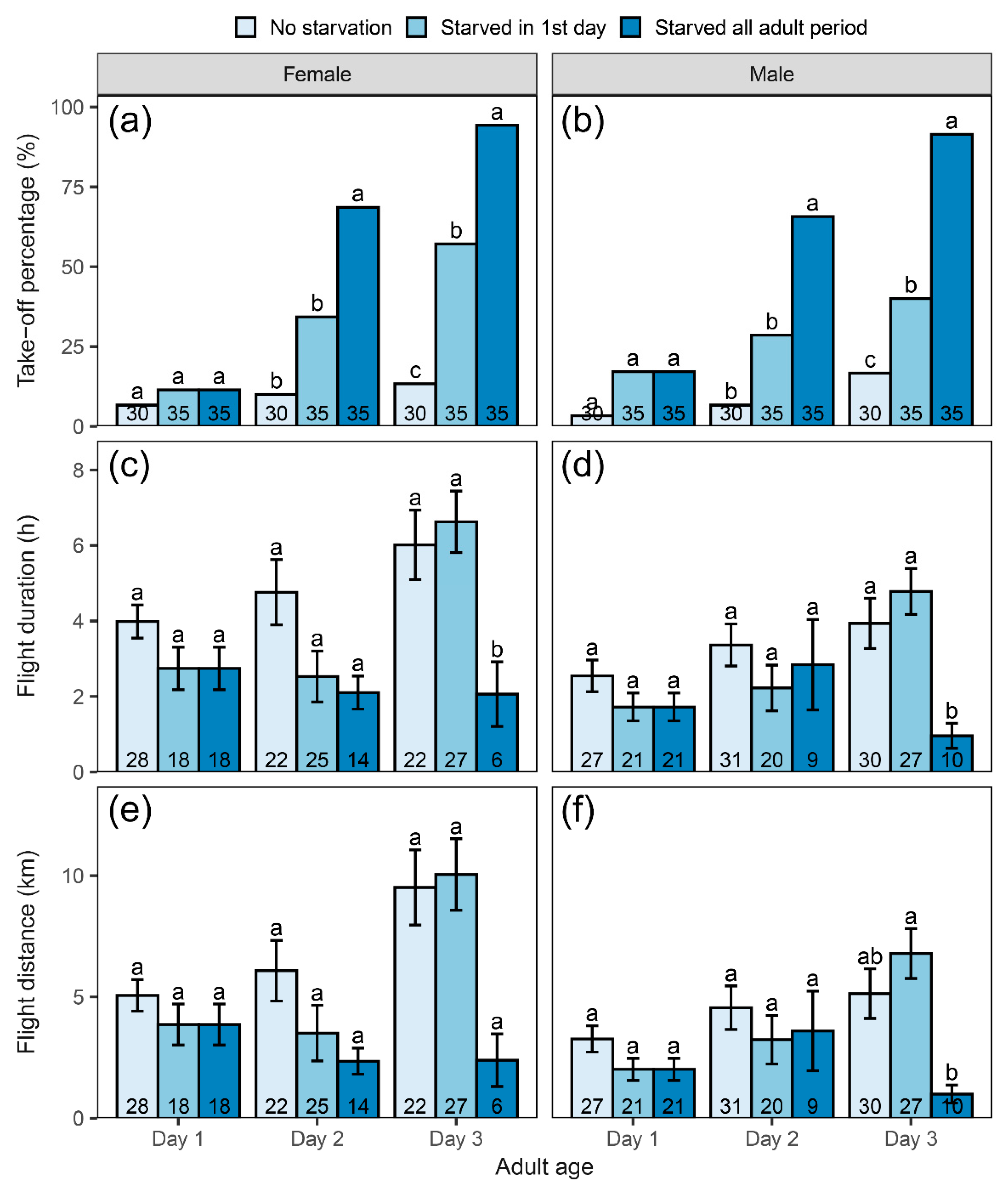
3.2. Influence of Adult Starvation on Flight Performance
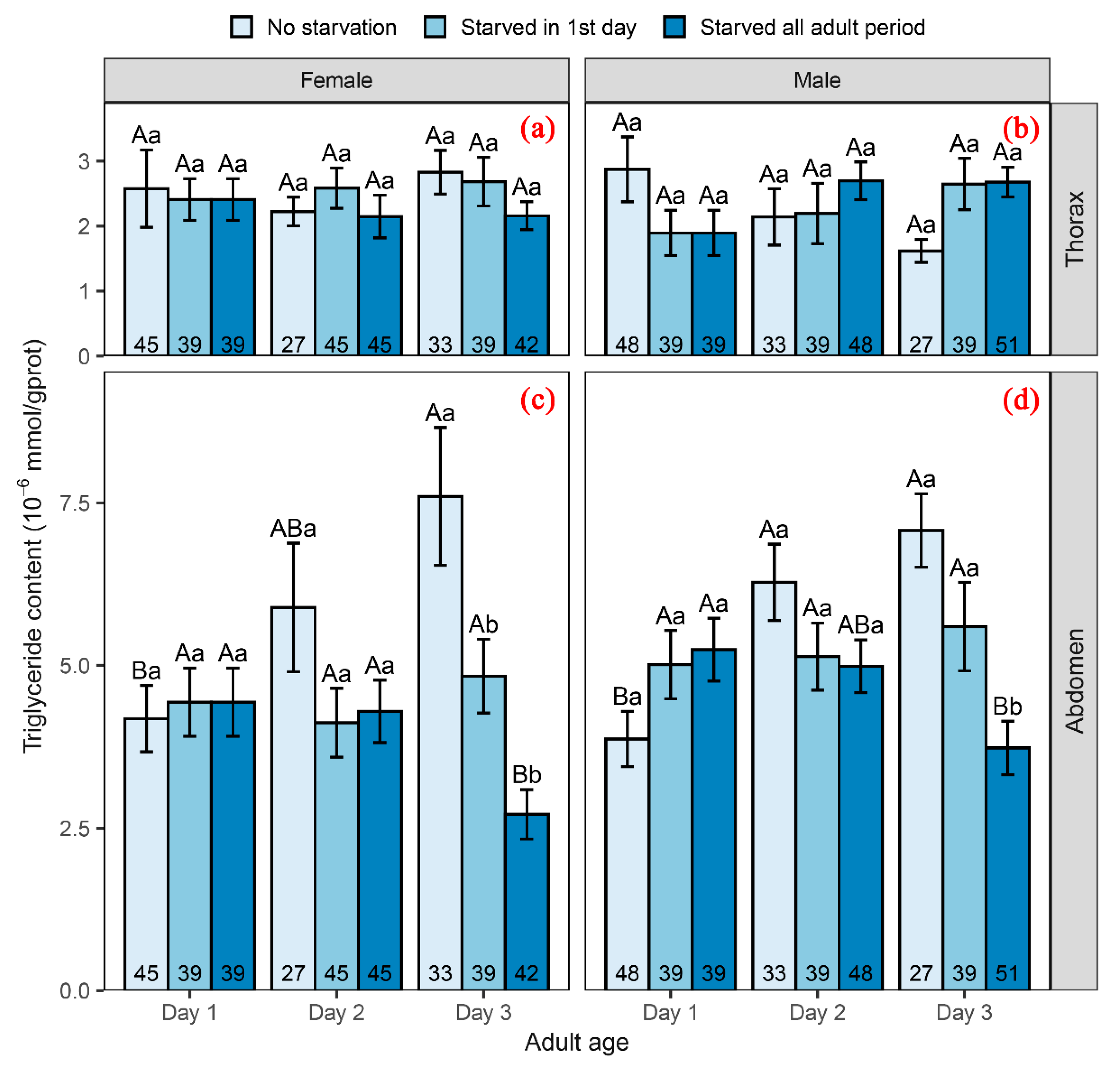
3.3. Influence of Adult Starvation on Triglyceride Content
4. Discussion
4.1. Effect of Adult Starvation on Reproductive Parameters
4.2. Effect of Adult Nutrition on Behavioral Response
4.3. Effect of Adult Nutrition on Physiological Adjustment
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chapman, J.W.; Reynolds, D.R.; Kenneth, W. Long-range seasonal migration in insects: Mechanisms, evolutionary drivers and ecological consequences. Ecol. Lett. 2015, 18, 287–302. [Google Scholar] [CrossRef]
- Dingle, H. Migration: The Biology of Life on the Move, 2nd ed.; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Kennedy, J.S. A turning point in the study of insect migration. Nature 1961, 189, 785–791. [Google Scholar] [CrossRef]
- Johnson, C.G. Migration and Dispersal of Insects by Flight; Methuen: London, UK, 1969. [Google Scholar]
- Zhang, X.X.; Wang, M.T. Study on the relationship between variation of ovary canal number and takeoff behaviour in green peach aphid. J. Plant Prot. 1991, 18, 161–166. [Google Scholar] [CrossRef]
- Liu, X.D.; Zhai, B.P.; Zhang, X.X.; Xiong, F. The relationship between flight behavior and ovary development in the cotton aphid, Aphis gossypii. Entomol. Knowl. 2003, 40, 39–42. [Google Scholar]
- Saglam, I.K.; Roff, D.A.; Fairbairn, D.J. Male sand crickets trade-off flight capability for reproductive potential. J. Evol. Biol. 2010, 21, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Zera, A.J.; Denno, R.F. Physiology and ecology of dispersal polymorphism in insects. Annu. Rev. Entomol. 1997, 42, 207–230. [Google Scholar] [CrossRef] [PubMed]
- Rankin, M.A.; Burchsted, J.C.A. The cost of migration in insects. Annu. Rev. Entomol. 1992, 37, 533–559. [Google Scholar] [CrossRef]
- Shirai, Y. Flight activity, reproduction, and adult nutrition of the beet webworm, Spoladea recurvalis (Lepidoptera: Pyralidae). Appl. Entomol. Zool. 2006, 41, 405–414. [Google Scholar] [CrossRef] [Green Version]
- Kent, J.W.; Rankin, M.A. Heritability and physiological correlates of migratory tendency in the grasshopper Melanoplus sanguinipes. Physiol. Entomol. 2010, 26, 371–380. [Google Scholar] [CrossRef]
- Zhang, X.X.; Geng, J.G.; Lu, Z.Q.; Liu, W.J. The preliminary investigation of biological and ecological characteristics in Cnaphalocrocis medinalis. Entomol. Knowl. 1980, 17, 241–244. [Google Scholar]
- Jiang, X.F.; Luo, L.Z.; Zhang, L.; Sappington, T.W.; Hu, Y. Regulation of migration in Mythimna separata (Walker) in China: A review integrating environmental, physiological, hormonal, genetic, and molecular factors. Environ. Entomol. 2011, 40, 516. [Google Scholar] [CrossRef] [PubMed]
- Boggs, C.L. Dynamics of reproductive allocation from juvenile and adult feeding: Radiotracer studies. Ecology 1997, 78, 192–202. [Google Scholar] [CrossRef]
- Fischer, K.; Boggs, D.M.; O’BRIEN, D.M.; Boggs, C.L. Allocation of larval and adult resources to reproduction in a fruit-feeding butterfly. Funct. Ecol. 2004, 18, 656–663. [Google Scholar] [CrossRef]
- Boggs, C.; Freeman, K. Larval food limitation in butterflies: Effects on adult resource allocation and fitness. Oecologia 2005, 144, 353–361. [Google Scholar] [CrossRef]
- Heimpel, G.E.; Rosenheim, J.A.; Kattari, D. Adult feeding and lifetime reproductive success in the parasitoid Aphytis melinus. Entomol. Exp. Appl. 1997, 83, 305–315. [Google Scholar] [CrossRef]
- Aluja, M.; Jacome, I.; Macias-Ordonez, R. Effect of adult nutrition on male sexual performance in four neotropical fruit fly species of the genus Anastrepha (Diptera: Tephritidae). J. Insect Behav. 2001, 14, 759–775. [Google Scholar] [CrossRef]
- Bauerfeind, S.S.; Fischer, K. Effects of adult-derived carbohydrates, amino acids and micronutrients on female reproduction in a fruit-feeding butterfly. J. Insect Physiol. 2005, 51, 545–554. [Google Scholar] [CrossRef]
- Harvey, J.A.; Cloutier, J.; Visser, B.; Ellers, J.; Wackers, F.L.; Gols, R. The effect of different dietary sugars and honey on longevity and fecundity in two hyperparasitoid wasps. J. Insect Physiol. 2012, 58, 816–823. [Google Scholar] [CrossRef]
- Kanda, K. Feeding activity of adults of the oriental armyworm, Pseudaletia separata walker. Jpn. J. Appl. Entomol. Zool. 1987, 31, 297–304. [Google Scholar] [CrossRef]
- Wang, H.S.; Xu, H.F.; Feng, C. Effect of adult foods on fecundity and ovary development of beet armyworm Spodoptera exigua (Hübner). Southwest China J. Agric. Sci. 2004, 17, 34–37. [Google Scholar]
- Murphy, D.D.; Launer, A.E.; Ehrlich, P.R. The role of adult feeding in egg production and population dynamics of the checkerspot butterfly Euphydryas editha. Oecologia 1983, 56, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.E. Spruce budworm (Lepidoptera: Tortricidae): Role of adult imbibing in reproduction. Environ. Entomol. 1987, 16, 1291–1295. [Google Scholar] [CrossRef]
- Hong, F.; Martin, T.R.; Jaskowiak, K.M.; Hatle, J.D.; Whitman, D.W.; Borst, D.W. Starvation affects vitellogenin production but not vitellogenin mRNA levels in the lubber grasshopper, Romalea microptera. J. Insect Physiol. 2005, 51, 435–443. [Google Scholar] [CrossRef]
- Rudolph, D.C.; Ely, C.A.; Schaefer, R.R.; Williamson, J.H.; Thill, R.E. Monarch (Danaus plexippus L. Nymphalidae) migration, nectar resources and fire regimes in the Ouachita Mountains of Arkansas. J. Lepid. Soc. 2006, 60, 165–170. [Google Scholar]
- Brower, L.P.; Fink, L.S.; Walford, P. Fueling the fall migration of the monarch butterfly. Integr. Comp. Biol. 2006, 46, 1123–1142. [Google Scholar] [CrossRef]
- Strong, F.E.; Bacon, O.G.; Russell, J.R. Flight habits of the alfalfa seed chalcid, Bruchophagus roddi Guss. (Hymenoptera: Eurytomidae). Hilgardia 1963, 35, 1–12. [Google Scholar] [CrossRef]
- Dingle, H. Diapause in a migrant insect, the milkweed bug Oncopeltus fasciatus (Dallas) (Hemiptera: Lygaeidae). Oecologia 1974, 17, 1–10. [Google Scholar] [CrossRef]
- Ahmad, I.; Khan, N.H. Effects of starvation on the longevity and fecundity of red cotton bug, Dysdercus cingulatus (Hemiptera: Pyrrhocoridae) in successive selected generations. Appl. Entomol. Zool. 1980, 15, 182–183. [Google Scholar] [CrossRef]
- Riley, J.R.; Reynolds, D.R.; Smith, A.D.; Edwards, A.S.; Zhang, X.X.; Cheng, X.N. Observations of the autumn migration of the rice leaf roller, Cnaphalocrocis medinalis (Lepidoptera: Pyralidae) and other moths in eastern China. Bull. Entomol. Res. 1995, 85, 397–414. [Google Scholar] [CrossRef]
- Zhang, X.X.; Geng, J.G.; Zhou, W.J. Studies on the migration of rice leaf roller, Cnaphalocrocis Medinalis Guenée. Acta Entomol. Sin. 1981, 23, 130–140. [Google Scholar]
- Wang, F.Y.; Yang, F.; Lu, M.H.; Luo, S.Y.; Zhai, B.P.; Lim, K.S.; Mcinerney, C.E.; Hu, G. Determining the migration duration of rice leaf folder (Cnaphalocrocis medinalis (Guenée)) moths using a trajectory analytical approach. Sci. Rep. 2017, 7, 39853. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Pan, P.; Sappington, T.W.; Lu, W.X.; Luo, L.Z.; Jiang, X.F. Accelerated and synchronized oviposition induced by flight of young females may intensify larval outbreaks of the rice leaf roller. PLoS ONE 2015, 10, e0121821. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.X.; Zhou, W.J.; Geng, J.G.; Su, Q.L.; Zhu, Y.C.; Tang, J.Y. Studies on the energy source of flight and migration of the rice leaf roller (Cnaphalocrocis medinalis Guenée). J. Plant Prot. 1983, 10, 153–159. [Google Scholar]
- Zou, Y.D.; Chen, J.C.; Wang, S.H. Study on the relationship between the internal comtents of energy substance and the vitality of lipase with the migration of rice leaf roller (Cnaphalocrocis medinalis Guenée) and brown planthopper (Nilaparvata lugens Stål). Acta Ecol. Sin. 1983, 3, 55–70. [Google Scholar]
- Yang, F.; Zheng, D.B.; Shi, J.J.; Gao, H.U.; Zhang, X.X.; Zhai, B.P. Observations on migratory behavior of Cnaphalocrocis medinalis: When will they take-off? Chin. J. Appl. Entomol. 2013, 50, 592–600. [Google Scholar]
- Wu, J.C. Effect of changing photoperiod, temperature and food quality on the migration of rice leaf folder Cnaphalocrocis medinalis (Guenée). Acta Entomol. Sin. 1985, 28, 398–405. [Google Scholar]
- Zhu, A.X.; Qiu, Q.; Liu, X.D. A method for rearing the rice leaf folder (Cnaphalocrocis medinalis) using wheat seedlings. Chin. J. Appl. Entomol. 2015, 52, 883–889. [Google Scholar]
- Wang, F.Y.; Zhang, X.X.; Zhai, B.P. Flight and re-migration capacity of the rice leaf folder moth, Cnaphalocrocis medinalis (Guenée) (Lepidoptera:Crambidae). Acta Entomol. Sin. 2010, 53, 1265–1272. [Google Scholar]
- Sun, B.B.; Jiang, X.F.; Zhang, L.; Stanley, D.W.; Luo, L.Z.; Long, W. Methoprene influences reproduction and flight capacity in adults of the rice leaf roller, Cnaphalocrocis medinalis (Guenée) (Lepidoptera: Pyralidae). Arch. Insect Biochem. Physiol. 2013, 82, 1–13. [Google Scholar] [CrossRef]
- Huang, X.F.; Zhang, X.X.; Zhai, B.P. Effect of copulation on flight capacity and remigration capacity of Cnaphalocrocis medinalis (Guenèe). J. Nanjing Agricult. Univ. 2010, 33, 23–28. [Google Scholar] [CrossRef]
- Himuro, C.; Fujisaki, K. Mating experience weakens starvation tolerance in the seed bug Togo hemipterus (Heteroptera: Lygaeidae). Physiol. Entomol. 2010, 35, 128–133. [Google Scholar] [CrossRef]
- O’Brien, D.M.; Boggs, C.L.; Fogel, M.L. Making eggs from nectar: The role of life history and dietary carbon turnover in butterfly reproductive resource allocation. Oikos 2004, 105, 279–291. [Google Scholar] [CrossRef]
- Kirk, K.L. Life-history responses to variable environments: Starvation and reproduction in planktonic rotifers. Ecology 1997, 78, 434–441. [Google Scholar] [CrossRef]
- Juliano, S.A. Food limitation of reproduction and survival for populations of Brachinus (Coleoptera: Carabidae). Ecology 1986, 67, 1036–1045. [Google Scholar] [CrossRef]
- Gunn, A.; Gatehouse, A.G. The effect of adult feeding on lipid and protein reserves in African armyworm, Spodoptera exempta, moths before and during reproduction. Physiol. Entomol. 1986, 11, 423–431. [Google Scholar] [CrossRef]
- Jiang, X.F.; Rui, Y.; Lei, Z.; Cheng, Y.X.; Liu, Y.Q.; Luo, L.Z. Effects of supplementary nutrition on adult reproduction and longevity of Athetis lepigone (Möschler). J. Plant Prot. 2015, 42, 1004–1008. [Google Scholar]
- Scharf, I. The multifaceted effects of starvation on arthropod behaviour. Anim. Behav. 2016, 119, 37–48. [Google Scholar] [CrossRef]
- Zhang, X.X.; Lu, Z.Q.; Geng, J.G. Application of ovarian dissection of female Cnaphalocrocis medinalis moths in prediction and forecasting system. Entomol. Knowl. 1979, 16, 97–99. [Google Scholar]
- Bertuso, A.G.; Morooka, S.; Tojo, S. Sensitive periods for wing development and precocious metamorphosis after precocene treatment of the brown planthopper, Nilaparvata lugens. J. Insect Physiol. 2002, 48, 221–229. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, X.F.; Luo, L.Z. Determination of sensitive stage for switching migrant oriental armyworms into residents. Environ. Entomol. 2008, 37, 1389–1395. [Google Scholar] [CrossRef]
- Duan, J.J.; Weber, D.C.; Dorn, S. Flight behavior of pre- and postdiapause apple blossom weevils in relation to ambient temperature. Entomol. Exp. Appl. 1998, 88, 97–99. [Google Scholar] [CrossRef]
- Sun, B.B.; Zhang, L.; Jiang, X.F.; Luo, L.Z. Effects of temperature in adult period on the reproduction behavior of the rice leaf roller, Cnaphalocrocis medinalis (Guenée). Chin. J. Appl. Entomol. 2013, 50, 622–628. [Google Scholar]
- Auerswald, L.; Gäde, G. Metabolic changes in the African fruit beetle, Pachnoda sinuata, during starvation. J. Insect Physiol. 2000, 46, 343–351. [Google Scholar] [CrossRef]
- Sappington, T.W.; Showers, W.B. Reproductive maturity, mating status, and long-duration flight behavior of Agrotis ipsilon (Lepidoptera: Noctuidae) and the conceptual misuse of the Oogenesis-Flight Syndrome by Entomologists. Environ. Entomol. 1992, 21, 677–688. [Google Scholar] [CrossRef]
- Kaufmann, C.; Collins, L.F.; Brown, M.R. Influence of age and nutritional status on flight performance of the Asian tiger mosquito Aedes albopictus (Diptera: Culicidae). Insects 2013, 4, 404–412. [Google Scholar] [CrossRef]
- Jiang, X.F.; Luo, L.Z.; Hu, Y. The effect of compensatory nutrition condition on flight ability of beet armyworm Spodoptera exigua (Hübner). J. Plant Prot. 2000, 27, 327–332. [Google Scholar]
- Li, K.B.; Gao, X.W.; Cao, Y.Z.; Luo, L.Z.; Jiang, X.F. Dynamics of energy reserves and utilization after tethered-flight in the beet armyworm, Spodoptera exigua (Hübner) (Lepidoptera: Noctuidae). J. Plant Prot. 2005, 3, 12–17. [Google Scholar]
- Harshman, L.G.; Schmid, J.L. Evolution of starvation resistance in Drosophila melanogaster: Aspects of metabolism and counter-impact selection. Evolution 1998, 52, 1679–1685. [Google Scholar] [CrossRef]
- Harshman, L.G.; Hoffmann, A.A.; Clark, A.G. Selection for starvation resistance in Drosophila melanogaster: Physiological correlates, enzyme activities and multiple stress responses. J. Evol. Biol. 1999, 12, 370–379. [Google Scholar] [CrossRef]
- Marron, M.T.; Markow, T.A.; Kain, K.J.; Gibbs, A.G. Effects of starvation and desiccation on energy metabolism in desert and mesic Drosophila. J. Insect Physiol. 2003, 49, 261–270. [Google Scholar] [CrossRef]
| Trait | Source | df | Mean-Square Value (MS) | F-Values | p |
|---|---|---|---|---|---|
| Fecundity | Starvation timing | 2 | 990 | 0.079 | 0.924 |
| Starvation duration | 4 | 230,851 | 18.46 | <0.001 | |
| Starvation timing × starvation duration | 1 | 257 | 0.02 | 0.89 | |
| Error | 191 | 12,508 | |||
| Preoviposition period | Starvation timing | 2 | 3.28 | 3.43 | 0.035 |
| Starvation duration | 4 | 8.86 | 9.26 | <0.001 | |
| Starvation timing × starvation duration | 1 | 1.03 | 1.07 | 0.30 | |
| Error | 191 | 0.96 |
| Trait | Source | df | MS | F-Values | p |
|---|---|---|---|---|---|
| Take-off percentage | Adult age | 2 | 16,891.56 | 46.52 | <0.001 |
| Sex | 1 | 154.47 | 0.43 | 0.516 | |
| Treatment | 2 | 23,182.94 | 63.844 | <0.001 | |
| Adult age × Sex | 2 | 190.81 | 0.53 | 0.593 | |
| Sex × Treatment | 2 | 95.32 | 0.26 | 0.770 | |
| Adult age × Treatment | 4 | 4294.37 | 11.83 | <0.001 | |
| Adult age × Sex × Treatment | 4 | 181.98 | 0.50 | 0.735 | |
| Error | 102 | 363.12 | |||
| Flight duration | Sex | 2 | 58.34 | 6.34 | 0.002 |
| Adult age | 1 | 85.54 | 9.30 | 0.002 | |
| Treatment | 2 | 94.98 | 10.33 | <0.001 | |
| Adult age × Sex | 2 | 11.19 | 1.22 | 0.297 | |
| Sex × Treatment | 2 | 8.54 | 0.93 | 0.396 | |
| Adult age × Treatment | 4 | 45.88 | 4.99 | <0.001 | |
| Adult age × Sex × Treatment | 4 | 1.98 | 0.22 | 0.930 | |
| Error | 358 | 9.20 | |||
| Flight distance | Sex | 2 | 163.29 | 7.00 | <0.001 |
| Adult age | 1 | 216.76 | 9.29 | 0.002 | |
| Treatment | 2 | 218.48 | 9.36 | <0.001 | |
| Adult age × sex | 2 | 47.95 | 2.06 | 0.130 | |
| Sex × Treatment | 2 | 21.22 | 0.91 | 0.404 | |
| Adult age × Treatment | 4 | 103.10 | 4.42 | 0.002 | |
| Adult age × Sex × Treatment | 4 | 6.66 | 0.29 | 0.887 | |
| Error | 358 | 23.33 |
| Trait | Source | df | MS | F-Values | p |
|---|---|---|---|---|---|
| Thorax | Sex | 1 | 0.48 | 0.26 | 0.609 |
| Adult age | 2 | 0.34 | 0.19 | 0.831 | |
| Treatment | 2 | 0.21 | 0.11 | 0.895 | |
| Adult age × Sex | 2 | 0.32 | 0.17 | 0.841 | |
| Sex × Treatment | 2 | 1.69 | 0.81 | 0.403 | |
| Adult age × Treatment | 4 | 2.19 | 1.18 | 0.319 | |
| Adult age × Sex × Treatment | 4 | 3.06 | 1.65 | 0.162 | |
| Error | 221 | 1.85 | |||
| Abdomen | Sex | 1 | 12.46 | 3.05 | 0.082 |
| Adult age | 2 | 6.71 | 1.64 | 0.196 | |
| Treatment | 2 | 39.45 | 9.67 | <0.001 | |
| Adult age × Sex | 2 | 1.36 | 0.33 | 0.718 | |
| Sex × Treatment | 2 | 5.72 | 1.40 | 0.248 | |
| Adult age × Treatment | 4 | 43.53 | 10.67 | <0.001 | |
| Adult age × Sex × Treatment | 4 | 0.62 | 0.15 | 0.962 | |
| Error | 221 | 4.08 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, J.-W.; Li, P.; Zhang, J.; Liu, X.-D.; Zhai, B.-P.; Hu, G. Cnaphalocrocis medinalis Moths Decide to Migrate when Suffering Nutrient Shortage on the First Day after Emergence. Insects 2019, 10, 364. https://doi.org/10.3390/insects10100364
Guo J-W, Li P, Zhang J, Liu X-D, Zhai B-P, Hu G. Cnaphalocrocis medinalis Moths Decide to Migrate when Suffering Nutrient Shortage on the First Day after Emergence. Insects. 2019; 10(10):364. https://doi.org/10.3390/insects10100364
Chicago/Turabian StyleGuo, Jia-Wen, Ping Li, Jie Zhang, Xiang-Dong Liu, Bao-Ping Zhai, and Gao Hu. 2019. "Cnaphalocrocis medinalis Moths Decide to Migrate when Suffering Nutrient Shortage on the First Day after Emergence" Insects 10, no. 10: 364. https://doi.org/10.3390/insects10100364






