Israeli Acute Paralysis Virus: Honey Bee Queen–Worker Interaction and Potential Virus Transmission Pathways
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experiment 1
2.3. Experiment 2
2.4. Experiment 3
2.5. Molecular Assays
2.6. Data Analysis
3. Results
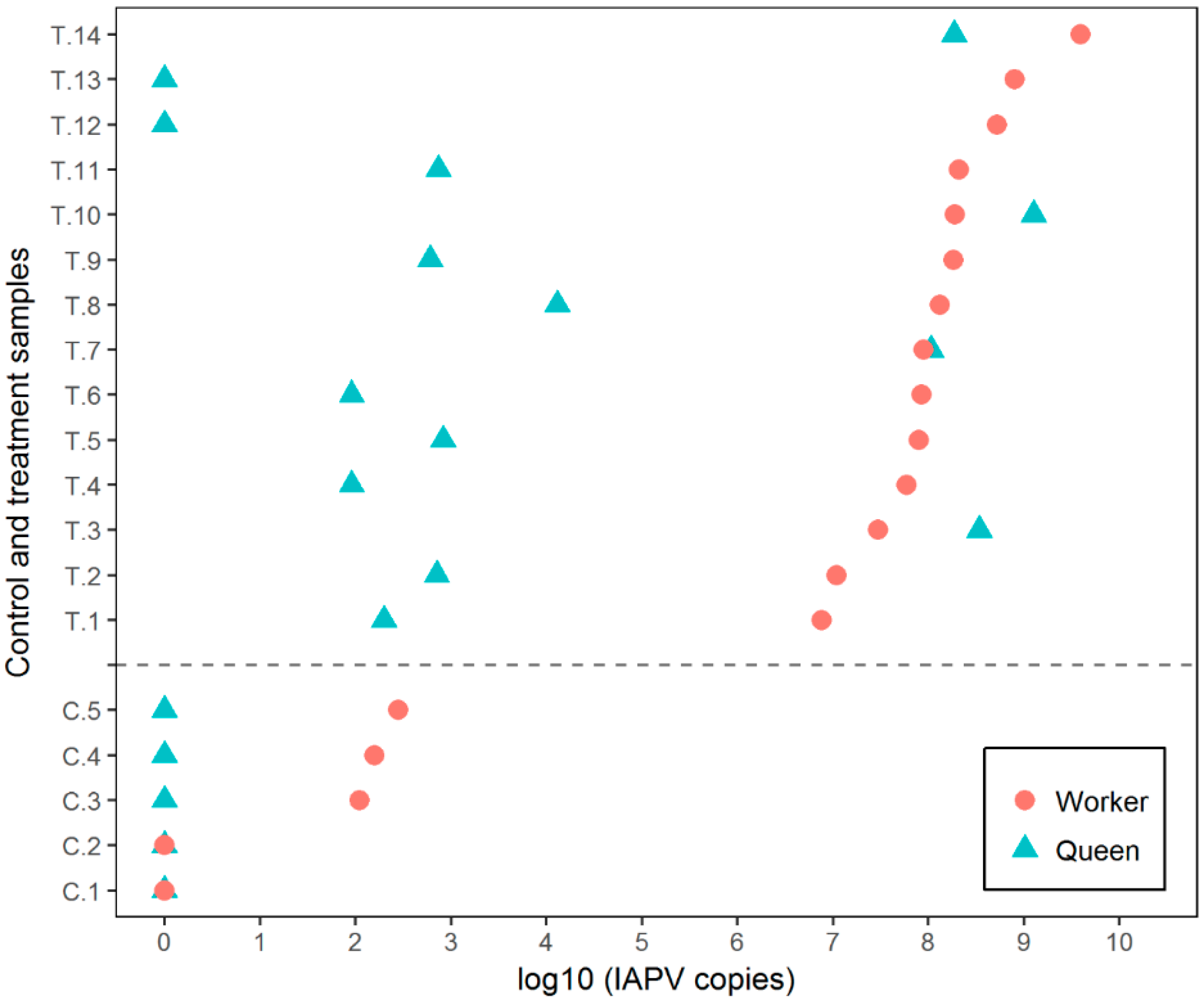
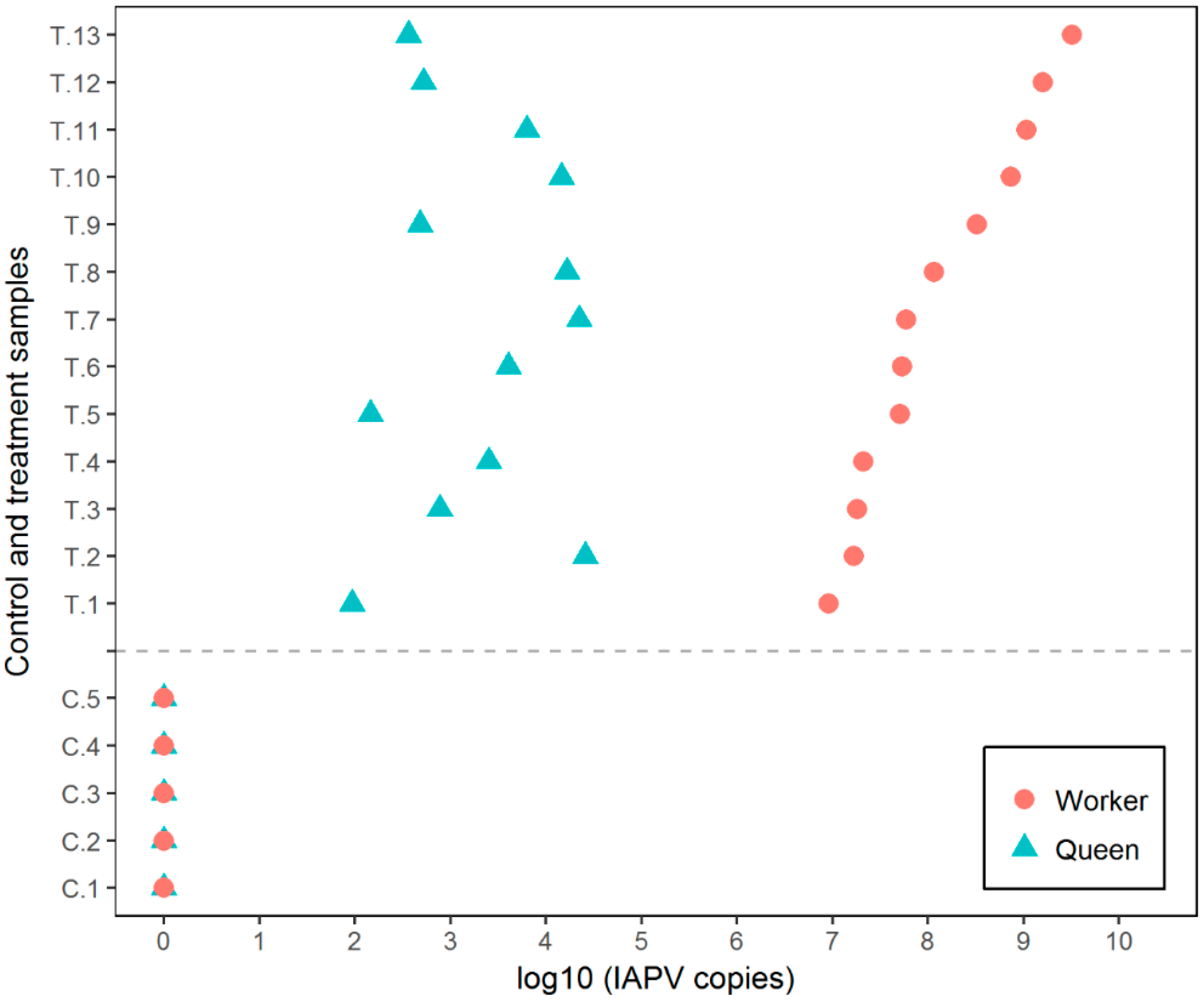
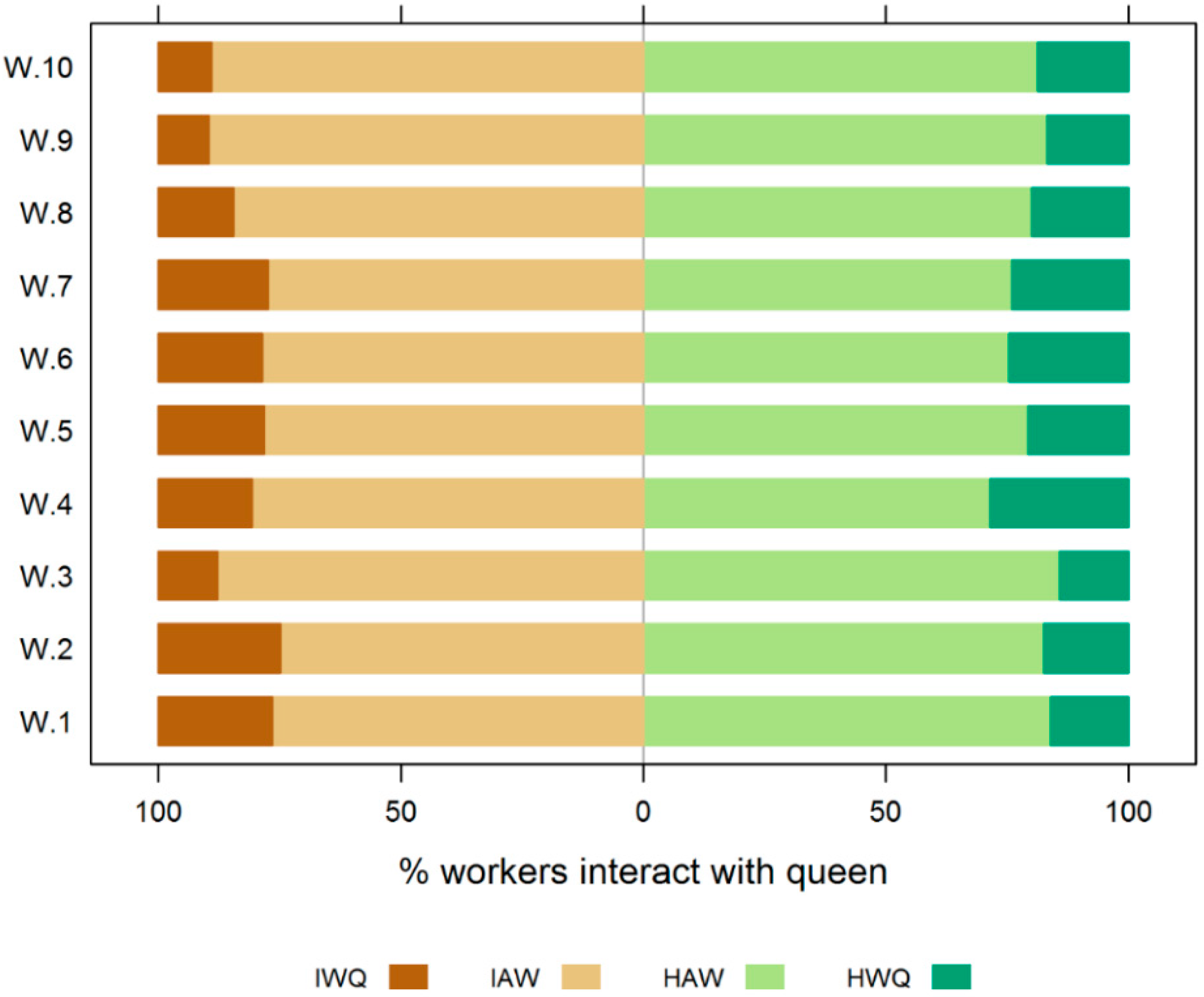
3.1. Experiment 1
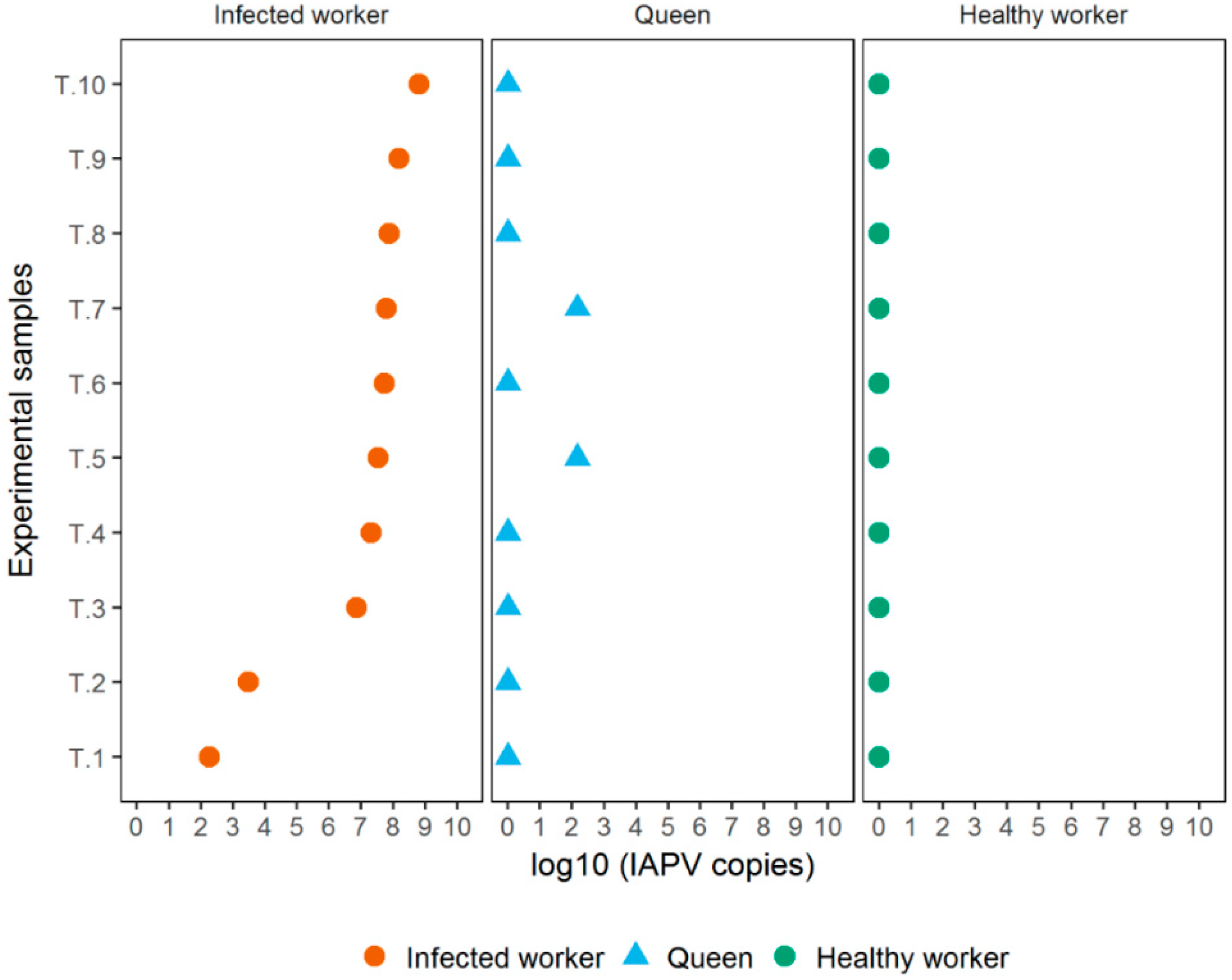
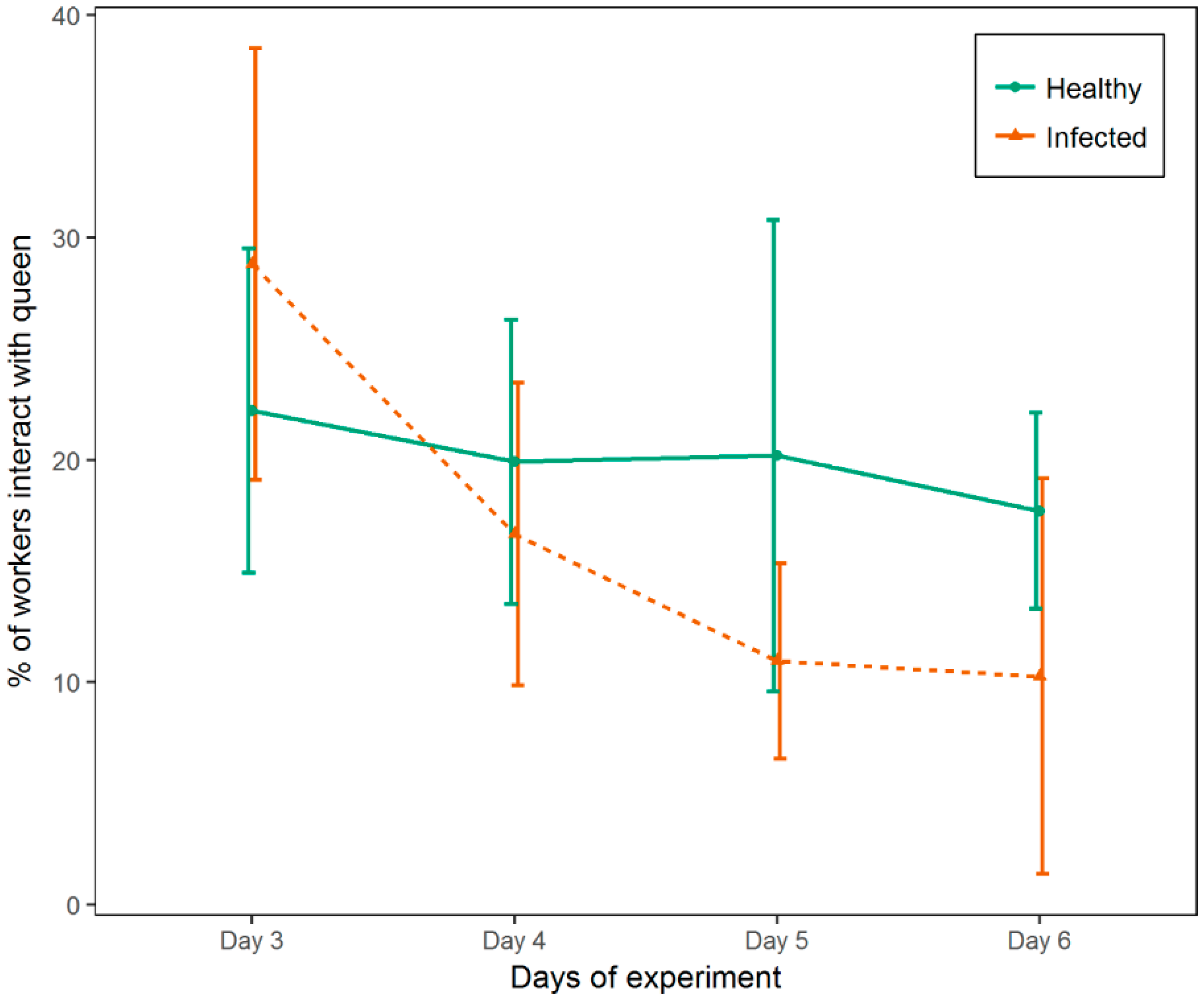
3.2. Experiment 2
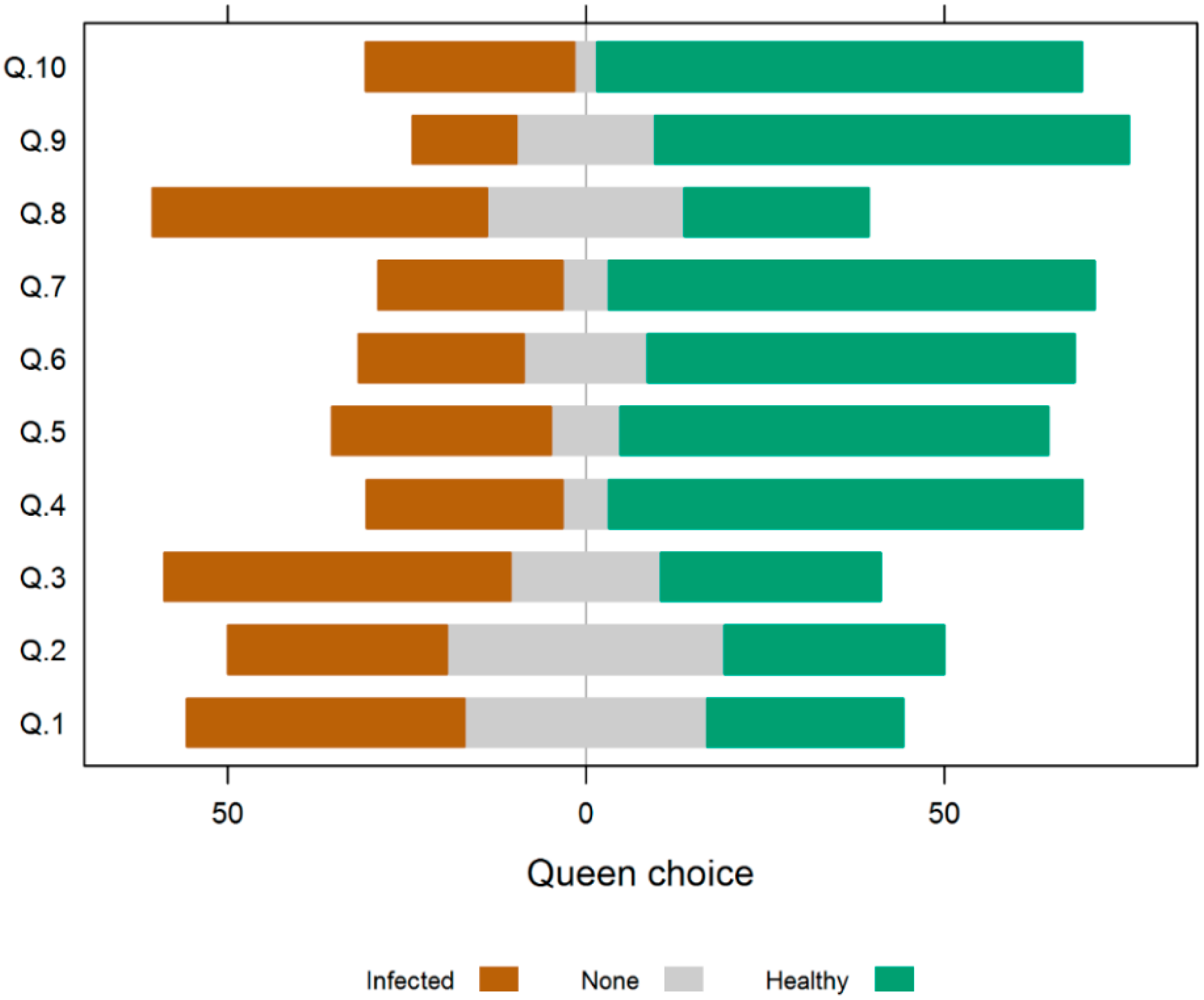
3.3. Experiment 3
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Winston, M.L. The Biology of the Honey Bee; Harvard University Press: London, UK, 1987. [Google Scholar]
- Visscher, P.K. A quantitative study of worker reproduction in honey bee colonies. Behav. Ecol. Sociobiol. 1989, 25, 247–254. [Google Scholar] [CrossRef]
- Pankiw, T.; Winston, M.L.; Slessor, K.N. Queen attendance behavior of worker honey bees (Apis mellifera L.) that are high and low responding to queen mandibular pheromone. Insectes Soc. 1995, 42, 371–378. [Google Scholar] [CrossRef]
- Free, J.B.; Ferguson, A.W.; Simpkins, J.R. The behaviour of queen honeybees and their attendants. Physiol. Entomol. 1992, 17, 43–55. [Google Scholar] [CrossRef]
- Allen, M.D. Observations on honeybees attending their queen. Anim. Behav. 1955, 3, 66–69. [Google Scholar] [CrossRef]
- Seely, T.D. Queen substance dispersal by messenger workers in honeybee colonies. Behav. Ecol. Sociobiol. 1979, 5, 391–415. [Google Scholar] [CrossRef]
- Naumann, K.; Winston, M.L.; Slessor, K.N.; Prestwich, G.D.; Webster, F.X. Production and transmission of honey bee queen (Apis mellifera L.) mandibular gland pheromone. Behav. Ecol. Sociobiol. 1991, 29, 321–332. [Google Scholar] [CrossRef]
- Rueppell, O.; Aumer, D.; Moritz, R.F.A. Ties between ageing plasticity and reproductive physiology in honey bees (Apis mellifera) reveal a positive relation between fecundity and longevity as consequence of advanced social evolution. Curr. Opin. Insect Sci. 2016, 16, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Pham-Delegue, M.-H.; Trouiller, J.; Bakchine, E.; Roger, B.; Masson, C. Age dependency of worker bee response to queen pheromone in a four-armed olfactometer. Insectes Soc. 1991, 38, 283–292. [Google Scholar] [CrossRef]
- Allen, M.D. The honeybee queen and her attendants. Anim. Behav. 1960, 8, 201–208. [Google Scholar] [CrossRef]
- Smith, M.L. The honey bee parasite Nosema ceranae: Transmissible via food exchange? PLoS ONE 2012, 7, e43319. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Evans, J.; Feldlaufer, M. Horizontal and vertical transmission of viruses in the honey bee, Apis mellifera. J. Invertebr. Pathol. 2006, 92, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Amiri, E.; Strand, M.K.; Rueppell, O.; Tarpy, D.R. Queen quality and the impact of honey bee diseases on queen health: potential for interactions between two major threats to colony health. Insects 2017, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Francis, R.M.; Nielsen, S.L.; Kryger, P. Patterns of viral infection in honey bee queens. J. Gen. Virol. 2013, 94, 668–676. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Pettis, J.S.; Feldlaufer, M.F. Detection of multiple viruses in queens of the honey bee Apis mellifera L. J. Invertebr. Pathol. 2005, 90, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Gregorc, A.; Bakonyi, T. Viral infections in queen bees (Apis mellifera carnica) from rearing apiaries. Acta Vet. Brno 2012, 81, 15–19. [Google Scholar] [CrossRef]
- Delaney, D.A.; Keller, J.J.; Caren, J.R.; Tarpy, D.R. The physical, insemination, and reproductive quality of honey bee queens (Apis mellifera L.). Apidologie 2011, 42, 1–13. [Google Scholar] [CrossRef]
- Cox-Foster, D.L.; Conlan, S.; Holmes, E.C.; Palacios, G.; Evans, J.D.; Moran, N.A.; Quan, P.-L.; Briese, T.; Hornig, M.; Geiser, D.M.; et al. A metagenomic survey of microbes in honey bee colony collapse disorder. Science 2007, 318, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Wilfert, L.; Long, G.; Leggett, H.; Schmid-Hempel, P.; Butlin, R.; Martin, S.; Boots, M. Deformed wing virus is a recent global epidemic in honeybees driven by Varroa mites. Science 2016, 351, 594–597. [Google Scholar] [CrossRef] [PubMed]
- Berthoud, H.; Imdorf, A.; Haueter, M.; Radloff, S.; Neumann, P. Virus infections and winter losses of honey bee colonies (Apis mellifera). J. Apicult. Res. 2010, 49, 60–65. [Google Scholar] [CrossRef]
- Cornman, R.S.; Tarpy, D.R.; Chen, Y.; Jeffreys, L.; Lopez, D.; Pettis, J.S.; vanEngelsdorp, D.; Evans, J.D. Pathogen webs in collapsing honey bee colonies. PLoS ONE 2012, 7, e43562. [Google Scholar] [CrossRef] [PubMed]
- Genersch, E.; Aubert, M. Emerging and re-emerging viruses of the honey bee (Apis mellifera L.). Vet. Res. 2010, 41, 54. [Google Scholar] [CrossRef] [PubMed]
- De Miranda, J.R.; Cordoni, G.; Budge, G. The Acute bee paralysis virus–Kashmir bee virus–Israeli acute paralysis virus complex. J. Invertebr. Pathol. 2010, 103, S30–S47. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Pettis, J.S.; Corona, M.; Chen, W.P.; Li, C.J.; Spivak, M.; Visscher, P.K.; DeGrandi-Hoffman, G.; Boncristiani, H.; Zhao, Y.; et al. Israeli acute paralysis virus: epidemiology, pathogenesis and implications for honey bee health. PLoS Pathog. 2014, 10, e1004261. [Google Scholar] [CrossRef] [PubMed]
- Di Prisco, G.; Pennacchio, F.; Caprio, E.; Boncristiani, H.F.; Evans, J.D.; Chen, Y. Varroa destructor is an effective vector of Israeli acute paralysis virus in the honeybee, Apis mellifera. J. Gen. Virol. 2011, 92, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Maori, E.; Lavi, S.; Mozes-Koch, R.; Gantman, Y.; Peretz, Y.; Edelbaum, O.; Tanne, E.; Sela, I. Isolation and characterization of Israeli acute paralysis virus, a dicistrovirus affecting honeybees in Israel: Evidence for diversity due to intra- and inter-species recombination. J. Gen. Virol. 2007, 88, 3428–3438. [Google Scholar] [CrossRef] [PubMed]
- Boncristiani, H.F.; Evans, J.D.; Chen, Y.; Pettis, J.; Murphy, C.; Lopez, D.L.; Simone-Finstrom, M.; Strand, M.; Tarpy, D.R.; Rueppell, O. In Vitro infection of pupae with israeli acute paralysis virus suggests disturbance of transcriptional homeostasis in honey bees (Apis mellifera). PLoS ONE 2013, 8, e73429. [Google Scholar] [CrossRef] [PubMed]
- Galbraith, D.A.; Yang, X.; Niño, E.L.; Yi, S.; Grozinger, C. Parallel epigenomic and transcriptomic responses to viral infection in honey bees (Apis mellifera). PLoS Pathog. 2015, 11, e1004713. [Google Scholar] [CrossRef] [PubMed]
- Cappelle, K.; Smagghe, G.; Dhaenens, M.; Meeus, I. Israeli acute paralysis virus infection leads to an enhanced RNA interference response and not its suppression in the bumblebee Bombus terrestris. Viruses 2016, 8, 334. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D.; Spivak, M. Socialized medicine: Individual and communal disease barriers in honey bees. J. Invertebr. Pathol. 2010, 103, S62–S72. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D.; Aronstein, K.; Chen, Y.P.; Hetru, C.; Imler, J.-L.; Jiang, H.; Kanost, M.; Thompson, G.J.; Zou, Z.; Hultmark, D. Immune pathways and defence mechanisms in honey bees Apis mellifera. Insect Mol. Biol. 2006, 15, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Cremer, S.; Armitage, S.A.O.; Schmid-Hempel, P. Social immunity. Curr. Biol. 2007, 17, R693–R702. [Google Scholar] [CrossRef] [PubMed]
- Rueppell, O.; Hayworth, M.K.; Ross, N.P. Altruistic self-removal of health-compromised honey bee workers from their hive. J. Evol. Biol. 2010, 23, 1538–1546. [Google Scholar] [CrossRef] [PubMed]
- Simone, M.; Evans, J.D.; Spivak, M. Resin collection and social immunity in honey bees. Evolution 2009, 63, 3016–3022. [Google Scholar] [CrossRef] [PubMed]
- Lecocq, A.; Jensen, A.B.; Kryger, P.; Nieh, J.C. Parasite infection accelerates age polyethism in young honey bees. Sci. Rep. 2016, 6, 22042. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-I.; Moeller, F.E. Division of labor and queen attendance behavior of nosema-infected worker honey bees. J. Econ. Entom. 1970, 63, 1540–1541. [Google Scholar]
- Biganski, S.; Kurze, C.; Müller, M.Y.; Moritz, R.F.A. Social response of healthy honeybees towards Nosema ceranae-infected workers: Care or kill? Apidologie 2018, 49, 325–334. [Google Scholar] [CrossRef]
- Traniello, J.F.A.; Rosengaus, R.B.; Savoie, K. The development of immunity in a social insect: Evidence for the group facilitation of disease resistance. Proc. Natl. Acad. Sci. USA 2002, 99, 6838–6842. [Google Scholar] [CrossRef] [PubMed]
- Maori, E.; Garbian, Y.; Kunik, V.; Mozes-Koch, R.; Malka, O.; Kalev, H.; Sabath, N.; Sela, I.; Shafir, S. A transmissible RNA pathway in honey bees. bioRxiv 2018. [Google Scholar] [CrossRef]
- Richard, F.-J.; Aubert, A.; Grozinger, C. Modulation of social interactions by immune stimulation in honey bee, Apis mellifera, workers. BMC Biol. 2008, 6, 50. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; van Oers, M.M.; van Houte, S.; Ros, V.I.D. Virus-induced behavioural changes in insects. In Host Manipulations by Parasites and Viruses; Mehlhorn, H., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 149–174. [Google Scholar]
- Amiri, E.; Meixner, M.; Büchler, R.; Kryger, P. Chronic bee paralysis virus in honeybee queens: Evaluating susceptibility and infection routes. Viruses 2014, 6, 1188–1201. [Google Scholar] [CrossRef] [PubMed]
- Laidlaw, H.H., Jr.; Page, R.E., Jr. Queen Rearing and Bee Breeding; Wicwas Press: Cheshire, UK, 1997. [Google Scholar]
- Gilley, D.C.; Tarpy, D.R. Three mechanisms of queen elimination in swarming honey bee colonies. Apidologie 2005, 36, 461–474. [Google Scholar] [CrossRef]
- De Miranda, J.R.; Bailey, L.; Ball, B.V.; Blanchard, P.; Budge, G.E.; Chejanovsky, N.; Chen, Y.; Gauthier, L.; Genersch, E.; de Graaf, D.C.; et al. Standard methods for virus research in Apis mellifera. J. Apicult. Res. 2013, 52, 1–56. [Google Scholar] [CrossRef]
- Kuster, R.D.; Boncristiani, H.F.; Rueppell, O. Immunogene and viral transcript dynamics during parasitic Varroa destructor mite infection of developing honey bee (Apis mellifera) pupae. J. Exp. Biol. 2014, 217, 1710–1718. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D. Beepath: An ordered quantitative-PCR array for exploring honey bee immunity and disease. J. Invertebr. Pathol. 2006, 93, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Francis, R.M.; Nielsen, S.L.; Kryger, P. Varroa-Virus interaction in collapsing honey bee colonies. PLoS ONE 2013, 8, e57540. [Google Scholar] [CrossRef] [PubMed]
- Ravoet, J.; De Smet, L.; Wenseleers, T.; de Graaf, D.C. Vertical transmission of honey bee viruses in a Belgian queen breeding program. BMC Vet. Res. 2015, 11, 1–6. [Google Scholar] [CrossRef] [PubMed]
- VanEngelsdorp, D.; Tarpy, D.R.; Lengerich, E.J.; Pettis, J.S. Idiopathic brood disease syndrome and queen events as precursors of colony mortality in migratory beekeeping operations in the eastern United States. Prev. Vet. Med. 2013, 108, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Bowen-Walker, P.L.; Martin, S.J.; Gunn, A. The transmission of Deformed wing virus between honeybees (Apis mellifera L.) by the ectoparasitic mite Varroa jacobsoni Oud. J. Invertebr. Pathol. 1999, 73, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Ribiere, M.; Lallemand, P.; Iscache, A.L.; Schurr, F.; Celle, O.; Blanchard, P.; Olivier, V.; Faucon, J.P. Spread of infectious Chronic bee paralysis virus by honeybee (Apis mellifera L.) feces. Appl. Environ. Microbiol. 2007, 73, 7711–7716. [Google Scholar] [CrossRef] [PubMed]
- Corona, M.; Velarde, R.A.; Remolina, S.; Moran-Lauter, A.; Wang, Y.; Hughes, K.A.; Robinson, G.E. Vitellogenin, juvenile hormone, insulin signaling, and queen honey bee longevity. Proc. Natl. Acad. Sci. USA 2007, 104, 7128–7133. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-S.; Hsu, C.-Y. Oxidative stress and anti-oxidant enzyme activities in the trophocytes and fat cells of queen honeybees (Apis mellifera). Rejuvenation Res. 2013, 16, 295–303. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amiri, E.; Seddon, G.; Zuluaga Smith, W.; Strand, M.K.; Tarpy, D.R.; Rueppell, O. Israeli Acute Paralysis Virus: Honey Bee Queen–Worker Interaction and Potential Virus Transmission Pathways. Insects 2019, 10, 9. https://doi.org/10.3390/insects10010009
Amiri E, Seddon G, Zuluaga Smith W, Strand MK, Tarpy DR, Rueppell O. Israeli Acute Paralysis Virus: Honey Bee Queen–Worker Interaction and Potential Virus Transmission Pathways. Insects. 2019; 10(1):9. https://doi.org/10.3390/insects10010009
Chicago/Turabian StyleAmiri, Esmaeil, Gregory Seddon, Wendy Zuluaga Smith, Micheline K. Strand, David R. Tarpy, and Olav Rueppell. 2019. "Israeli Acute Paralysis Virus: Honey Bee Queen–Worker Interaction and Potential Virus Transmission Pathways" Insects 10, no. 1: 9. https://doi.org/10.3390/insects10010009
APA StyleAmiri, E., Seddon, G., Zuluaga Smith, W., Strand, M. K., Tarpy, D. R., & Rueppell, O. (2019). Israeli Acute Paralysis Virus: Honey Bee Queen–Worker Interaction and Potential Virus Transmission Pathways. Insects, 10(1), 9. https://doi.org/10.3390/insects10010009






