Exposure to Herbicides Prime P450-Mediated Detoxification of Helicoverpa armigera against Insecticide and Fungal Toxin
Abstract
1. Introduction
2. Material and Methods
2.1. Insects
2.2. Chemicals
2.3. Pre-Exposure to Herbicides and Bioassays
2.4. Enzyme Activity Assay
2.5. Statistical Analysis
3. Results
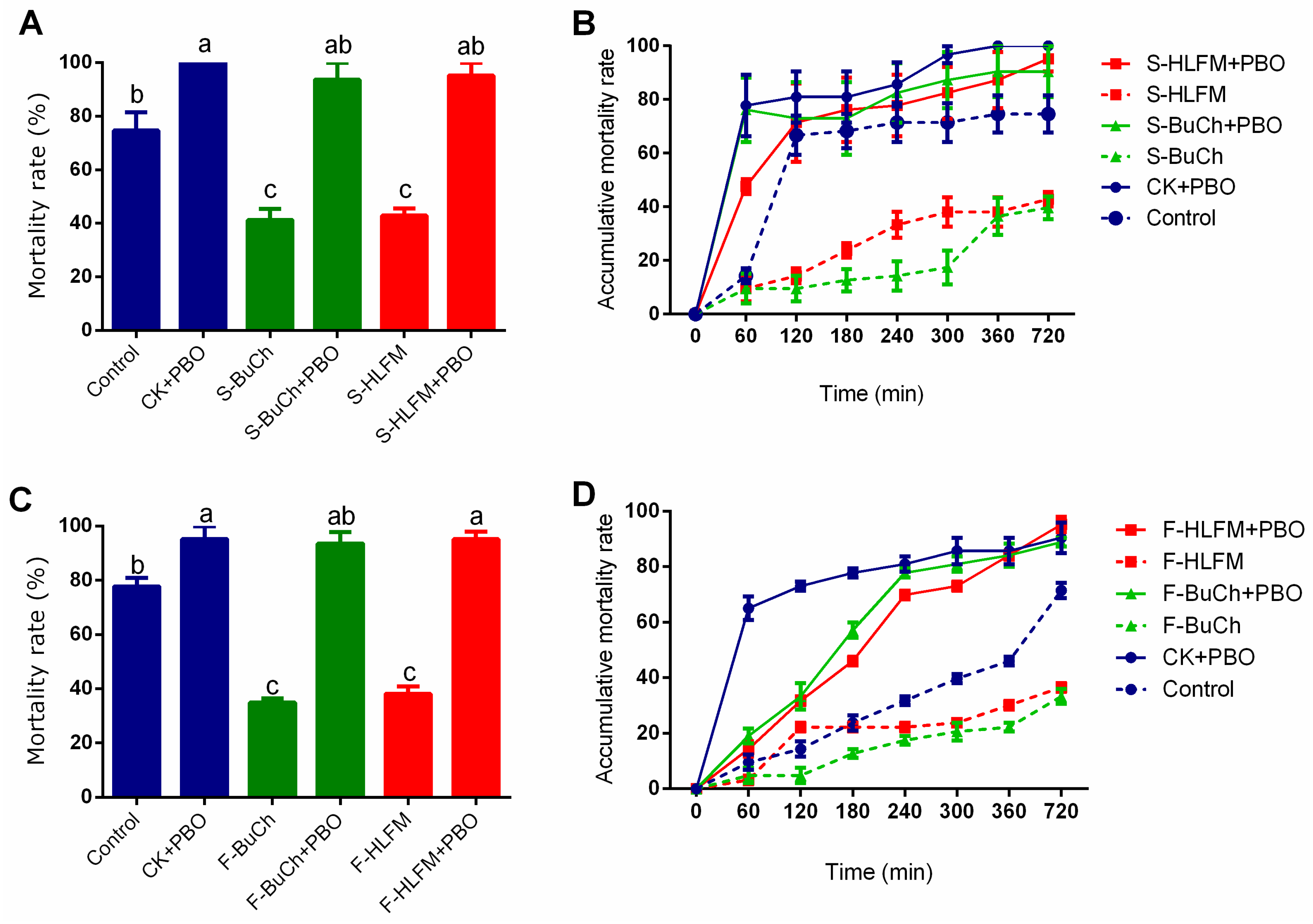
3.1. Exposure to Herbicides Prime Resistance of H. armigera against Insecticide
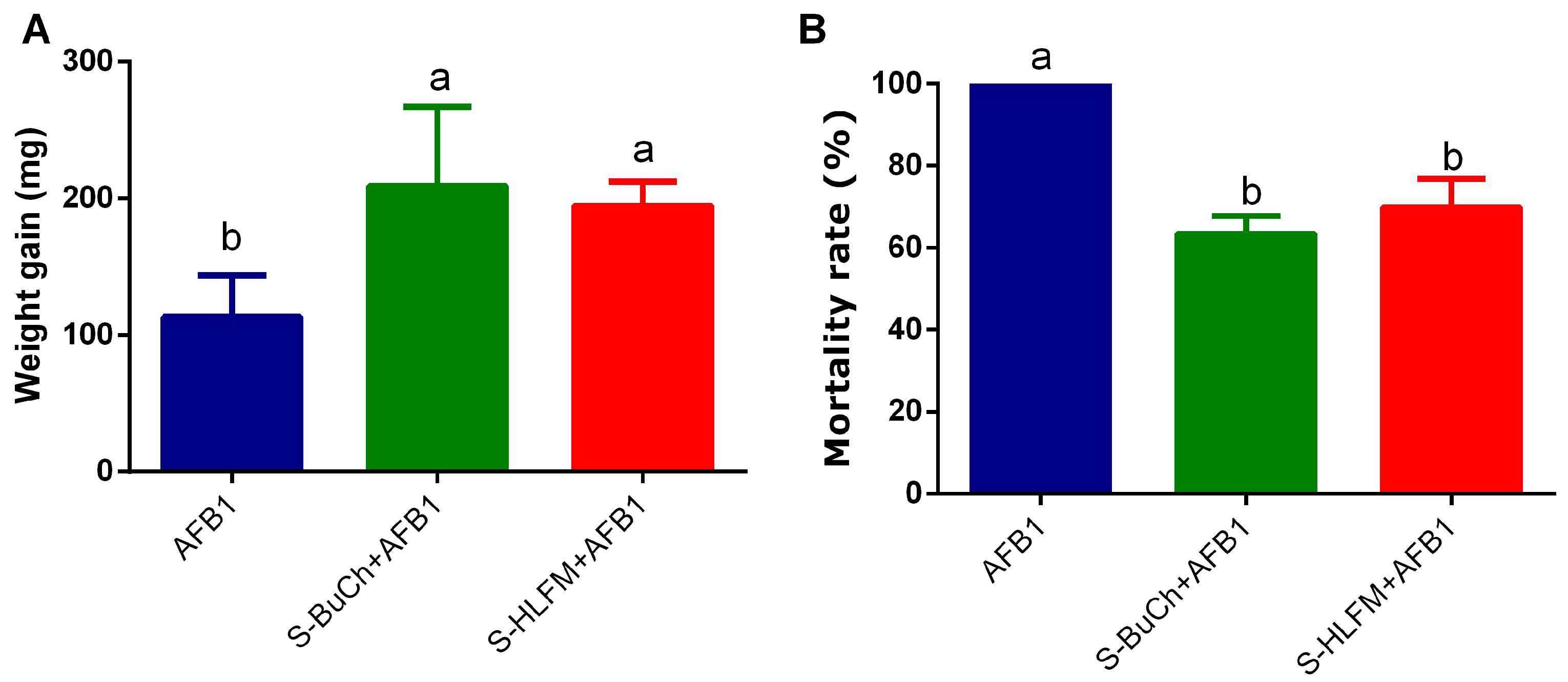
3.2. Exposure to Herbicides Prime Resistance of H. armigera against Fungal Toxin
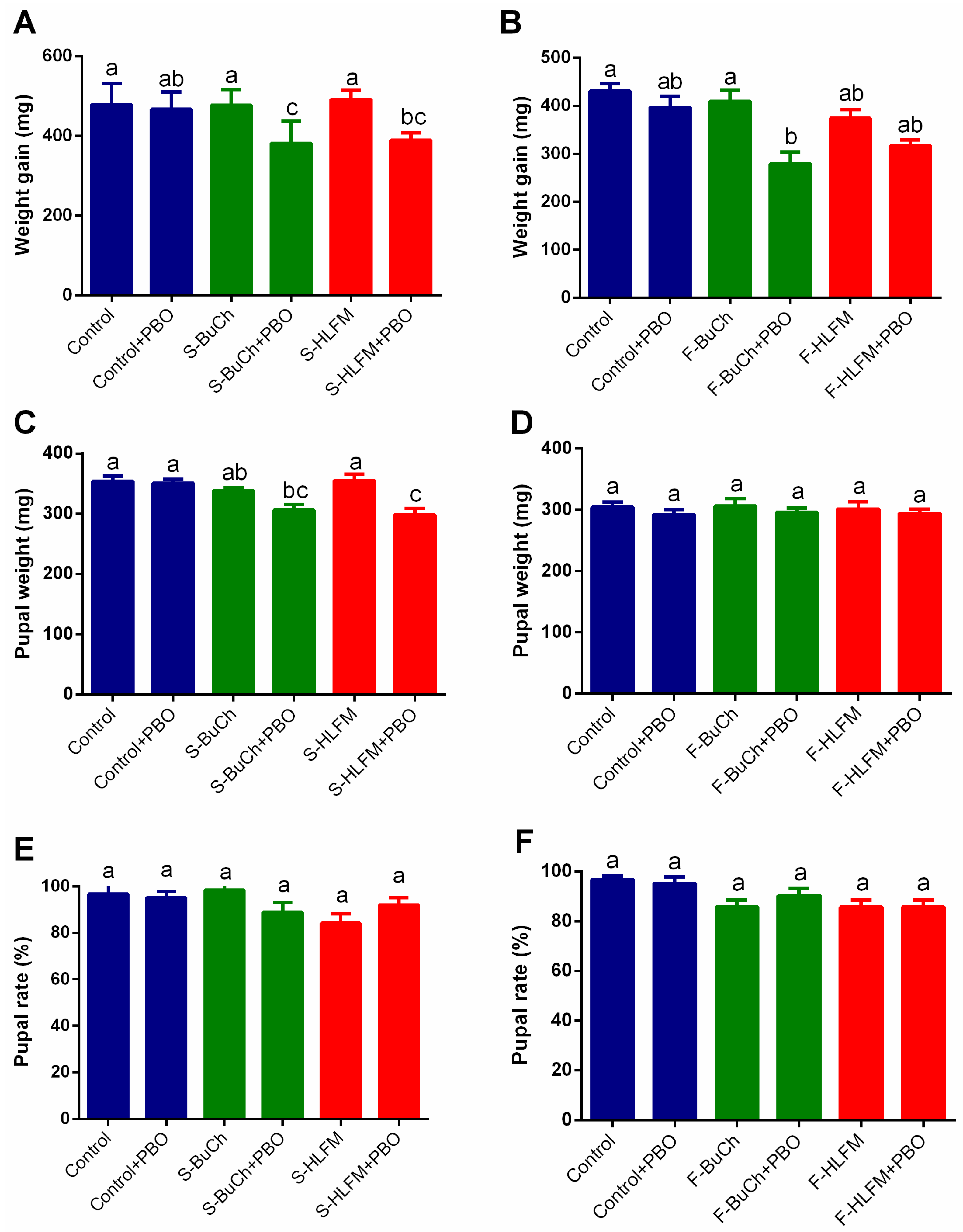
3.3. The Effects of Exposing to Herbicides on Growth and Development of H. armigera
3.4. The Effect of Exposing to Herbicides on Detoxifying Enzyme Activities in H. armigera
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wojciechowska, M.; Stepnowski, P.; Gołębiowski, M. The use of insecticides to control insect pests. Invertebr. Surviv. J. 2016, 13, 210–220. [Google Scholar]
- Parimi, S.; Meinke, L.J.; Nowatzki, T.M.; Chandler, L.D.; French, B.W.; Siegfried, B.D. Toxicity of insecticide-bait mixtures to insecticide resistant and susceptible western corn rootworms (Coleoptera: Chrysomelidae). Crop Prot. 2003, 22, 781–786. [Google Scholar] [CrossRef]
- Food and Agricultural Organization of the United Nations. Available online: http://www.fao.org/home/en/ (accessed on 18 August 2018).
- Boyer, S.; Sérandour, J.; Lempérière, G.; Raveton, M.; Ravanel, P. Do herbicide treatments reduce the sensitivity of mosquito larvae to insecticides? Chemosphere 2006, 65, 721–724. [Google Scholar] [CrossRef] [PubMed]
- Haughton, A.J.; Bell, J.R.; Wilcox, A.; Boatman, N.D. The effect of the herbicide glyphosate on non-target spiders: Part I. Direct effects on Lepthyphantes tenuis under laboratory conditions. Pest Manag. Sci. 2001, 57, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Haughton, A.J.; Bell, J.R.; Boatman, N.D.; Wilcox, A. The effect of the herbicide glyphosate on non-target spiders: Part II. Indirect effects on Lepthyphantes tenuis in field margins. Pest Manag. Sci. 2001, 57, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Hayes, T.B.; Collins, A.; Lee, M.; Mendoza, M.; Noriega, N.; Stuart, A.A.; Vonk, A. Hermaphroditic, demasculinized frogs after exposure to the herbicide atrazine at low ecologically relevant doses. Proc. Natl. Acad. Sci. USA 2002, 99, 5476–5480. [Google Scholar] [CrossRef] [PubMed]
- Le, T.H.; Lim, E.S.; Lee, S.K.; Choi, Y.W.; Kim, Y.H.; Min, J. Effects of glyphosate and methidathion on the expression of the Dhb, Vtg, Arnt, CYP4 and CYP314 in Daphnia magna. Chemosphere 2010, 79, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Poupardin, R.; Reynaud, S.; Strode, C.; Ranson, H.; Vontas, J.; David, J.P. Cross-induction of detoxification genes by environmental xenobiotics and insecticides in the mosquito. Insect Biochem. Mol. Biol. 2008, 38, 540–551. [Google Scholar] [CrossRef]
- Suwanchaichinda, C.; Brattsten, L.B. Effects of Exposure to Pesticides on Carbaryl Toxicity and Cytochrome P450 Activities in Aedes albopictus Larvae (Diptera: Culicidae). Pestic. Biochem. Physiol. 2001, 70, 63–73. [Google Scholar] [CrossRef]
- Schuler, M.A. P450s in plant-insect interactions. Biochim. Biophys. Acta 2011, 1814, 36–45. [Google Scholar] [CrossRef]
- Després, L.; David, J.-P.; Gallet, C. The evolutionary ecology of insect resistance to plant chemicals. Trends Ecol. Evol. 2007, 22, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.; Yao, J.; Li, D.; He, Y.; Zhu, Y.C.; Zhang, X.; Zhu, K.Y. Cytochrome P450 genes from the aquatic midge Chironomus tentans: Atrazine-induced up-regulation of CtCYP6EX3 enhanced the toxicity of chlorpyrifos. Chemosphere 2017, 186, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Le, G.G.; Hilliou, F.; Siegfried, B.D.; Boundy, S.; Wajnberg, E.; Sofer, L.; Audant, P.; Ffrenchconstant, R.H.; Feyereisen, R. Xenobiotic response in Drosophila melanogaster: Sex dependence of P450 and GST gene induction. Insect Biochem. Mol. Biol. 2006, 36, 674–682. [Google Scholar]
- Mao, Y.B.; Cai, W.J.; Wang, J.W.; Hong, G.J.; Tao, X.Y.; Wang, L.J.; Huang, Y.P.; Chen, X.Y. Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat. Biotechnol. 2007, 25, 1307. [Google Scholar] [CrossRef] [PubMed]
- Fukuto, T.R. Mechanism of action of organophosphorus and carbamate insecticides. Environ. Health Perspect. 1990, 87, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.C.; Sharma, H.C. Heliothis/Helicoverpa Management: Emerging Trends and Strategies for Future Research; Oxford and IBH Publishing Co.: New Delhi, India, 2005; pp. 453–463. [Google Scholar]
- Brun-Barale, A.; Héma, O.; Martin, T.; Suraporn, S.; Audant, P.; Sezutsu, H.; Feyereisen, R. Multiple P450 genes overexpressed in deltamethrin-resistant strains of Helicoverpa armigera. Pest Manag. Sci. 2010, 66, 900–909. [Google Scholar] [CrossRef] [PubMed]
- Smela, M.E.; Currier, S.S.; Bailey, E.A.; Essigmann, J.M. The chemistry and biology of aflatoxin B(1): From mutational spectrometry to carcinogenesis. Carcinogenesis 2001, 22, 535–545. [Google Scholar] [CrossRef]
- Wilson, A.G.E.; Takei, A.S. Summary of toxicology studies with butachlor. J. Pestic. Sci. 2000, 25, 75–83. [Google Scholar] [CrossRef]
- Song-Mao, L.U.; Amp, A.; Center, D. Analysis of Haloxyfop-methyl Residues in Vegetables. Agrochemicals 2014, 53, 751–752. [Google Scholar]
- Omura, T.; Sato, R. The carbon monoxide-binding pigment of liver microsomes I. Evidence for its hemoprotein nature. J. Biol. Chem. 1964, 239, 2370–2378. [Google Scholar]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-Transferases the first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [PubMed]
- Van Asperen, K. A study of housefly esterases by means of a sensitive colorimetric method. J. Insect Physiol. 1962, 8, 401–416. [Google Scholar] [CrossRef]
- Kennedy, G.G.; Storer, N.P. Life systems of polyphagous arthropod pests in temporally unstable cropping systems. Annu. Rev. Entomol. 2000, 45, 467–493. [Google Scholar] [CrossRef]
- Huang, S.; Xu, J.; Han, Z. Baseline toxicity data of insecticides against the common cutworm Spodoptera litura (Fabricius( and a comparison of resistance monitoring methods. PANS Pest Artic. News Summ. 2006, 52, 209–213. [Google Scholar]
- Xiao, P.; He, J.; Liu, Y.J.; Qiu, X.C.; Jiao, Y.Y. The relationship of resistance to lambda-cyhalothrin with detoxification enzyme activity in Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae). Acta Entomol. Sin. 2009, 52, 1097–1102. [Google Scholar]
- Jin, L.; Wang, J.; Guan, F.; Zhang, J.; Yu, S.; Liu, S.; Xue, Y.; Li, L.; Wu, S.; Wang, X. Dominant point mutation in a tetraspanin gene associated with field-evolved resistance of cotton bollworm to transgenic Bt cotton. Proc. Natl. Acad. Sci. USA 2018, 115, 11760–11765. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.J.; Oakeshott, J.G.; Tay, W.T.; Gordon, K.H.J.; Zwick, A.; Walsh, T.K. Hybridization and gene flow in the mega-pest lineage of moth, Helicoverpa. Proc. Natl. Acad. Sci. USA 2018, 115, 201718831. [Google Scholar] [CrossRef] [PubMed]
- Motasanchez, D.; Hollingworth, R.M.; Grafius, E.J.; Moyer, D.D. Resistance and cross-resistance to neonicotinoid insecticides and spinosad in the Colorado potato beetle, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae). Pest Manag. Sci. 2010, 62, 30–37. [Google Scholar] [CrossRef]
- Tao, X.Y.; Xue, X.Y.; Huang, Y.P.; Chen, X.Y.; Mao, Y.B. Gossypol-enhanced P450 gene pool contributes to cotton bollworm tolerance to a pyrethroid insecticide. Mol. Ecol. 2012, 21, 4371–4385. [Google Scholar] [CrossRef]
- Elbert, A.; Nauen, R. Resistance of Bemisia tabaci (Homoptera: Aleyrodidae) to insecticides in southern Spain with special reference to neonicotinoids. Pest Manag. Sci. 2015, 56, 60–64. [Google Scholar] [CrossRef]
- Stam, P.A.; Clower, D.F.; Graves, J.B.; Schilling, P.E. Effects of Certain Herbicides on Some Insects and Spiders Found in Louisiana Cotton Fields. J. Econ. Entomol. 1978, 71, 477–480. [Google Scholar] [CrossRef]
- Jacquet, M.; Tilquin, M.; Ravanel, P.; Boyer, S. Increase in Tolerance of Aedes aegypti Larvae (Diptera: Culicidae) to the Insecticide Temephos after Exposure to Atrazine. Afr. Entomol. 2016, 23, 110–119. [Google Scholar] [CrossRef]
- Adi, K.; Murad, G. Fitness costs associated with insecticide resistance. Pest Manag. Sci. 2012, 68, 1431–1437. [Google Scholar]
- Gassmann, A.; Carriere, Y.; Tabashnik, B. Fitness Costs of Insect Resistance to Bacillus thuringiensis. Annu. Rev. Entomol. 2009, 54, 147–163. [Google Scholar] [CrossRef] [PubMed]
- Liu, N. Insecticide resistance in mosquitoes: Impact, mechanisms, and research directions. Annu. Rev. Entomol. 2015, 60, 537–559. [Google Scholar] [CrossRef] [PubMed]
- Hilary, R.; Raphael, N.G.; Jonathan, L.; Nicolas, M.; Zinga, N.; Vincent, C. Pyrethroid resistance in African anopheline mosquitoes: What are the implications for malaria control? Trends Parasitol. 2011, 27, 91–98. [Google Scholar]
- Yang, T.; Liu, N. Permethrin resistance variation and susceptible reference line isolation in a field population of the mosquito, Culex quinquefasciatus (Diptera: Culicidae). Insect Sci. 2015, 21, 659–666. [Google Scholar] [CrossRef]
- Shi, L.; Zhang, J.; Shen, G.; Xu, Z.; Wei, P.; Zhang, Y.; Xu, Q.; He, L. Silencing NADPH-cytochrome P450 reductase results in reduced acaricide resistance in Tetranychus cinnabarinus (Boisduval). Sci. Rep. 2015, 5, 15581. [Google Scholar] [CrossRef]
- Luo, L.; Sun, Y.J.; Wu, Y.J. Abamectin resistance in Drosophila is related to increased expression of P-glycoprotein via the dEGFR and dAkt pathways. Insect Biochem. Mol. Biol. 2013, 43, 627–634. [Google Scholar] [CrossRef]
- Liang, X.; Xiao, D.; He, Y.; Yao, J.; Zhu, G.; Zhu, K.Y. Insecticide-mediated up-regulation of cytochrome P450 genes in the red flour beetle (Tribolium castaneum). Int. J. Mol. Sci. 2015, 16, 2078–2098. [Google Scholar] [CrossRef]
- Zhu, F.; Feng, J.N.; Zhang, L.; Liu, N. Characterization of two novel cytochrome P450 genes in insecticide-resistant house-flies. Insect Mol. Biol. 2010, 17, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Ranasinghe, C.; Hobbs, A.A. Isolation and characterization of two cytochrome P450 cDNA clones for CYP6B6 and CYP6B7 from Helicoverpa armigera (Hubner): Possible involvement of CYP6B7 in pyrethroid resistance. Insect Biochem. Mol. Biol. 1998, 28, 571. [Google Scholar] [CrossRef]
- Wee, C.W.; Lee, S.F.; Robin, C.; Heckel, D.G. Identification of candidate genes for fenvalerate resistance in Helicoverpa armigera using cDNA-AFLP. Insect Mol. Biol. 2010, 17, 351–360. [Google Scholar] [CrossRef] [PubMed]
| P450 | GST-DCNB | Esterase-aNA | ChE | |
|---|---|---|---|---|
| (nmole per min per mg pro) | ||||
| CK | 0.134 ± 0.009 d | 4.22 ± 0.06 b | 37.53 ± 1.33 a | 16.07 ± 2.76 a |
| S-BuCH | 0.230 ± 0.016 c | 3.19 ± 0.13 c | 30.03 ± 0.63 b | 20.14 ± 2.28 a |
| F-BuCH | 0.327 ± 0.020 bc | 3.38 ± 0.12 c | 33.03 ± 0.45 b | 19.97 ±1.23 a |
| S-HLFM | 0.383 ± 0.015 b | 5.10 ± 0.11 a | 40.98 ± 0.58 a | 22.04 ± 1.85 a |
| F-HLFM | 0.650 ± 0.034 a | 4.87 ± 0.23 ab | 17.14 ± 0.57 c | 21.48 ± 1.50 a |
| P450 | GST-DCNB | Esterase-aNA | ChE | |
|---|---|---|---|---|
| (nmole per min per mg pro) | ||||
| CK | 0.130 ± 0.034 b | 3.37 ± 0.33 b | 7.18 ± 0.35 c | 16.06 ± 0.81 a |
| S-BuCH | 0.270 ± 0.027 a | 2.66 ± 0.27 c | 11.87 ± 0.57 b | 8.19 ± 0.84 b |
| F-BuCH | 0.317 ± 0.018 a | 2.20 ± 0.18 c | 14.16 ± 0.43 b | 17.02 ± 1.01 a |
| S-HLFM | 0.343 ± 0.028 a | 3.69 ± 0.33 b | 22.24 ± 0.27 a | 13.79 ± 1.64 a |
| F-HLFM | 0.284 ± 0.054 a | 4.44 ± 0.13 a | 14.78 ± 1.37 b | 7.24 ± 0.23 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Z.; Xu, C.; Chen, S.; Shi, Q.; Wang, H.; Wang, R.; Song, Y.; Zeng, R. Exposure to Herbicides Prime P450-Mediated Detoxification of Helicoverpa armigera against Insecticide and Fungal Toxin. Insects 2019, 10, 28. https://doi.org/10.3390/insects10010028
Sun Z, Xu C, Chen S, Shi Q, Wang H, Wang R, Song Y, Zeng R. Exposure to Herbicides Prime P450-Mediated Detoxification of Helicoverpa armigera against Insecticide and Fungal Toxin. Insects. 2019; 10(1):28. https://doi.org/10.3390/insects10010028
Chicago/Turabian StyleSun, Zhongxiang, Cuicui Xu, Shi Chen, Qi Shi, Huanhuan Wang, Rumeng Wang, Yuanyuan Song, and Rensen Zeng. 2019. "Exposure to Herbicides Prime P450-Mediated Detoxification of Helicoverpa armigera against Insecticide and Fungal Toxin" Insects 10, no. 1: 28. https://doi.org/10.3390/insects10010028
APA StyleSun, Z., Xu, C., Chen, S., Shi, Q., Wang, H., Wang, R., Song, Y., & Zeng, R. (2019). Exposure to Herbicides Prime P450-Mediated Detoxification of Helicoverpa armigera against Insecticide and Fungal Toxin. Insects, 10(1), 28. https://doi.org/10.3390/insects10010028