Combined Toxicity of Insecticides and Fungicides Applied to California Almond Orchards to Honey Bee Larvae and Adults
Abstract
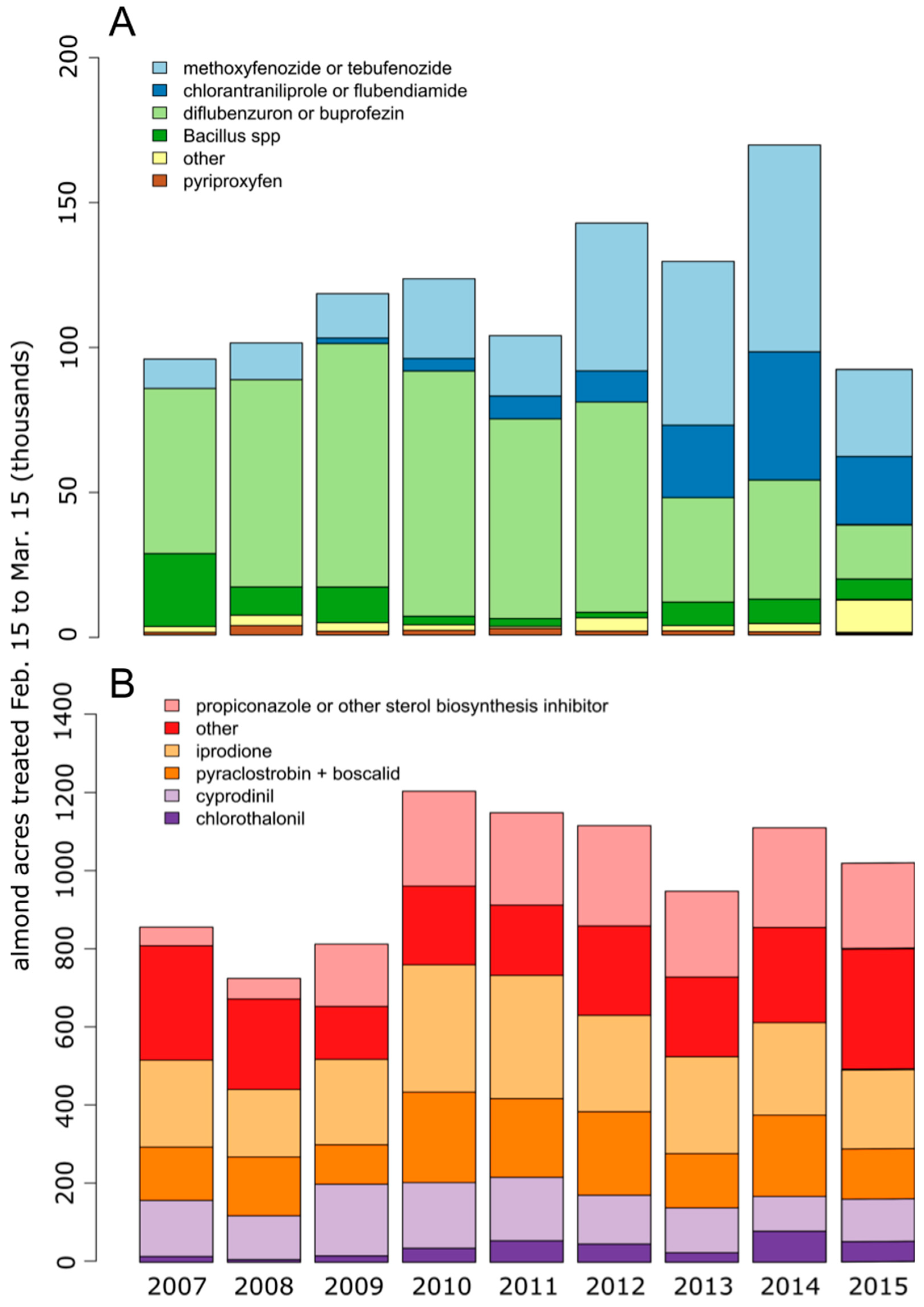
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Honey Bees
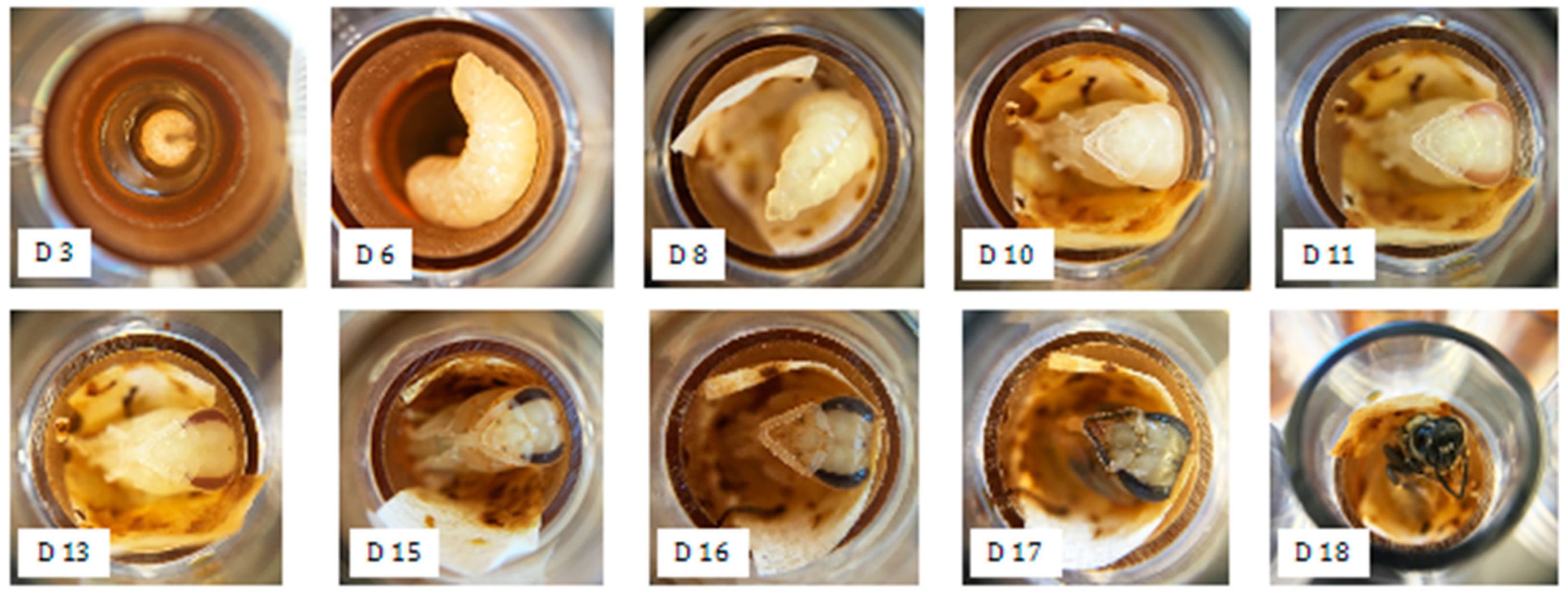
2.3. In vitro Larval Tests
2.4. Adult tests
2.5. Statistical Analysis
2.6. BeeREX Model
3. Results
3.1. In vitro Larval Tests
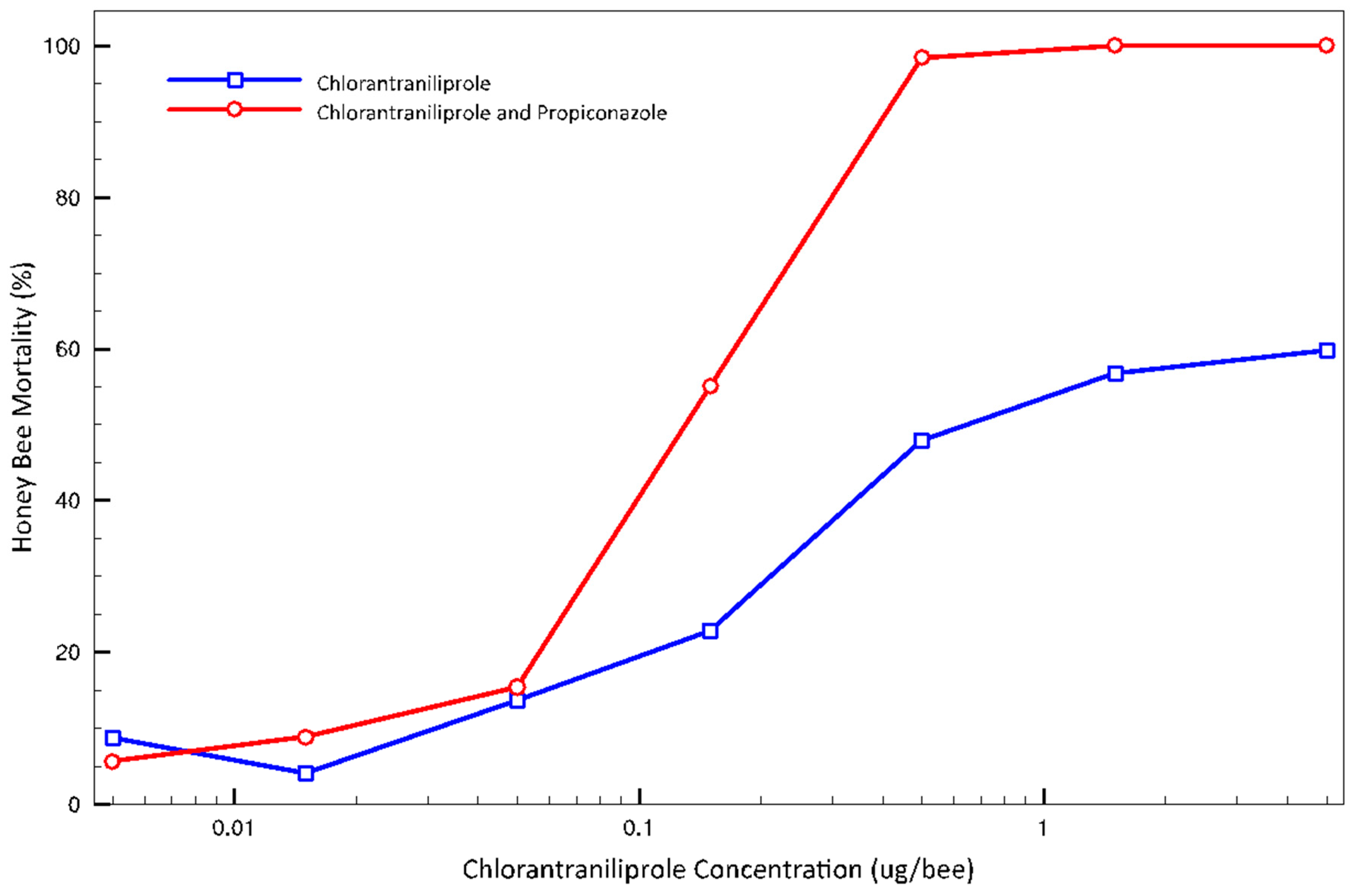
3.2. Adult Topical Tests
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Morse, R.A.; Calderone, N.W. The value of honey bee pollination the United States. Bee Cult. 2000, 128, 1–15. [Google Scholar]
- U.S. Department of Agriculture. Cost of Pollination. 2017. Available online: http://usda.mannlib.cornell.edu/usda/current/CostPoll/CostPoll-12-21-2017.pdf (accessed on 29 June 2018).
- Flottum, K. Catch the Buzz: Huge Bee Kill in Almonds. Available online: http://www.beeculture.com/catch-the-buzz-huge-bee-kill-in-almonds/ (accessed on 31 July 2017).
- California Pesticide Information Portal. Available online: http://calpip.cdpr.ca.gov/main.cfm (accessed on 17 April 2017).
- UC IPM: UC Management Guidelines for Peach Twig Borer on Almonds. Available online: http://ipm.ucanr.edu/PMG/r3300211.html (accessed on 29 June 2018).
- Casida, J.E. Golden age of RyR and GABA-R diamide and isoxazoline insecticides: Common genesis, serendipity, surprises, selectivity, and safety. Chem. Res. Toxicol. 2015, 28, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Sparks, T.C.; Nauen, R. IRAC: Mode of action classification and insecticide resistance management. Pestic. Biochem. Physiol. 2015, 121, 122–128. [Google Scholar] [CrossRef] [PubMed]
- DuPont. Altacor Product Label; E. I. du Pont de Nemours and Company: Wilmington, DE, USA, 2017. [Google Scholar]
- Dinter, A.; Brugger, K.E.; Frost, N.-M.; Woodward, M.D. Chlorantraniliprole (Rynaxypyr): A novel DuPontTM insecticide with low toxicity and low risk for honey bees (Apis mellifera) and bumble bees (Bombus terrestris) providing excellent tools for uses in integrated pest management. In Proceedings of the 10th International Symposium of the ICP-Bee Protection Group, Bucharest, Romania, 8–10 October 2008; pp. 84–96. [Google Scholar]
- U.S. Environmental Protection Agency. Flubendiamide—Notice of Intent to Cancel and Other Supporting Documents. 2016. Available online: https://www.epa.gov/ingredients-used-pesticide-products/flubendiamide-notice-intent-cancel-and-other-supporting (accessed on 29 June 2018).
- Carlson, G.R.; Dhadialla, T.S.; Hunter, R.; Jansson, R.K.; Jany, C.S.; Lidert, Z.; Slawecki, R.A. The chemical and biological properties of methoxyfenozide, a new insecticidal ecdysteroid agonist. Pest. Manag. Sci. 2001, 57, 115–119. [Google Scholar] [CrossRef]
- Dow AgroSciences. Product Safety Assessment Methoxyfenozide; Dow AgroSciences: Zionsville, IN, USA, 2014. [Google Scholar]
- Mommaerts, V.; Sterk, G.; Smagghe, G. Bumblebees can be used in combination with juvenile hormone analogues and ecdysone agonists. Ecotoxicology 2006, 15, 513–521. [Google Scholar] [CrossRef]
- Dow AgroSciences. Intrepid 2F Product Label; Dow AgroSciences: Zionsville, IN, USA, 2017. [Google Scholar]
- Fisher, A., II; Colman, C.; Hoffmann, C.; Fritz, B.; Rangel, J. The effects of the insect growth regulators methoxyfenozide and pyriproxyfen and the acaricide bifenazate on honey bee (Hymenoptera: Apidae) Forager Survival. J. Econ. Entomol. 2018, 111, 510–516. [Google Scholar] [CrossRef]
- Matsumura, F. Studies on the action mechanism of benzoylurea insecticides to inhibit the process of chitin synthesis in insects: A review on the status of research activities in the past, the present and the future prospects. Pestic. Biochem. Physiol. 2010, 97, 133–139. [Google Scholar] [CrossRef]
- Arysta. Dimilin 2L Product Label; Arysta Life Science North America: Cary, NC, USA, 2017. [Google Scholar]
- Barker, R.J.; Taber, S., III. Effects of diflubenzuron fed to caged honey bees. Environ. Entomol. 1977, 6, 167–168. [Google Scholar] [CrossRef]
- Emmett, B.J.; Archer, B.M. The toxicity of diflubenzuron to honey bee (Apis mellifera L.) colonies in apple orchards. Plant Pathol. 1980, 29, 177–183. [Google Scholar] [CrossRef]
- Tasei, J.-N. Effects of insect growth regulators on honey bees and non-Apis bees. A review. Apidologie 2001, 32, 527–545. [Google Scholar] [CrossRef]
- Chen, X.D.; Gill, T.A.; Pelz-Stelinski, K.S.; Stelinski, L.L. Risk assessment of various insecticides used for management of Asian citrus psyllid, Diaphorina citri in Florida citrus, against honey bee, Apis mellifera. Ecotoxicology 2017, 26, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Barker, R.J.; Waller, G.D. Effects of Difluhenzuron wettable powder on caged honey bee colonies. Environ. Entomol. 1978, 7, 534–535. [Google Scholar] [CrossRef]
- Gupta, P.R.; Chandel, R.S. Effects of diflubenzuron and penfluron on workers of Apis cerana indica F and Apis mellifera L. Apidologie 1995, 26, 3–10. [Google Scholar] [CrossRef]
- Abramson, C.I.; Squire, J.; Sheridan, A.; Mulder, P.G., Jr. The effect of insecticides considered harmless to honey bees (Apis mellifera): Proboscis conditioning studies by using the insect growth regulators tebufenozide and diflubenzuron. Environ. Entomol. 2004, 33, 378–388. [Google Scholar] [CrossRef]
- Chandel, R.S.; Gupta, P.R. Toxicity of diflubenzuron and penfluron to immature stages of Apis cerana indica F and Apis mellifera L. Apidologie 1992, 23, 465–473. [Google Scholar] [CrossRef]
- Thompson, H.; Wilkins, S.; Battersby, A.; Waite, R.; Wilkinson, D. The effects of four insect growth-regulating (IGR) insecticides on honeybee (Apis mellifera L.) colony development, queen rearing and drone sperm production. Ecotoxicology 2005, 14, 757–769. [Google Scholar] [CrossRef]
- Chon, K.; Lee, H.; Hwang, H.C.; Im, J.; Park, K.-H.; Paik, M.K.; Choi, Y.-S. The honey bee brood test under semi-field conditions for the assessment of positive reference chemicals in Korea. Appl. Biol. Chem. 2017, 60, 569–582. [Google Scholar] [CrossRef]
- Johnson, R.M.; Percel, E.G. Effect of a fungicide and spray adjuvant on queen-rearing success in honey bees (Hymenoptera: Apidae). J. Econ. Entomol. 2013, 106, 1952–1957. [Google Scholar] [CrossRef]
- Johnson, R.M. Honey bee toxicology. Annu. Rev. Entomol. 2015, 60, 415–434. [Google Scholar] [CrossRef]
- Mussen, E.C.; Lopez, J.E.; Peng, C.Y.S. Effects of selected fungicides on growth and development of larval honey bees, Apis mellifera L. (Hymenoptera: Apidae). Environ. Entomol. 2004, 33, 1151–1154. [Google Scholar] [CrossRef]
- Ladurner, E.; Bosch, J.; Kemp, W.P.; Maini, S. Assessing delayed and acute toxicity of five formulated fungicides to Osmia lignaria Say and Apis mellifera. Apidologie 2005, 36, 449–460. [Google Scholar] [CrossRef]
- Campbell, J.B.; Nath, R.; Gadau, J.; Fox, T.; DeGrandi-Hoffman, G.; Harrison, J.F. The fungicide Pristine® inhibits mitochondrial function in vitro but not flight metabolic rates in honey bees. J. Insect Physiol. 2016, 86, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A., II; Coleman, C.; Hoffmann, C.; Fritz, B.; Rangel, J. The synergistic effects of almond protection fungicides on honey bee (Hymenoptera: Apidae) forager survival. J. Econ. Entomol. 2017, 110, 802–808. [Google Scholar] [CrossRef]
- Colin, M.-E.; Belzunces, L.P. Evidence of synergy between prochloraz and deltamethrin in Apis mellifera L.: A convenient biological approach. Pestic. Sci. 1992, 36, 115–119. [Google Scholar] [CrossRef]
- Pilling, E.D.; Jepson, P.C. Synergism between EBI fungicides and a pyrethroid insecticide in the honeybee (Apis mellifera). Pestic. Sci. 1993, 39, 293–297. [Google Scholar] [CrossRef]
- Thompson, H.; Wilkins, S. Assessment of the synergy and repellency of pyrethroid/fungicide mixtures. Bull. Insectol. 2003, 56, 131–134. [Google Scholar]
- Iwasa, T.; Motoyama, N.; Ambrose, J.T.; Roe, R.M. Mechanism for the differential toxicity of neonicotinoid insecticides in the honey bee, Apis mellifera. Crop. Prot. 2004, 23, 371–378. [Google Scholar] [CrossRef]
- Johnson, R.M.; Dahlgren, L.; Siegfried, B.D.; Ellis, M.D. Acaricide, fungicide and drug interactions in honey bees (Apis mellifera). PLoS ONE 2013, 8, e54092. [Google Scholar] [CrossRef]
- Thompson, H.M.; Fryday, S.L.; Harkin, S.; Milner, S. Potential impacts of synergism in honeybees (Apis mellifera) of exposure to neonicotinoids and sprayed fungicides in crops. Apidologie 2014, 45, 545–553. [Google Scholar] [CrossRef]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. Disruption of quercetin metabolism by fungicide affects energy production in honey bees (Apis mellifera). Proc. Natl. Acad. Sci. USA 2017, 114, 2538–2543. [Google Scholar] [CrossRef]
- Bliss, C.I. The toxicity of poisons applied jointly. Ann. Appl. Biol. 1939, 26, 585–615. [Google Scholar] [CrossRef]
- Lepesheva, G.I.; Waterman, M.R. Sterol 14α-demethylase cytochrome P450 (CYP51), a P450 in all biological kingdoms. Biochim. Biophys. Acta (BBA) Gen. Subj. 2007, 1770, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ramamoorthy, Y.; Kilicarslan, T.; Nolte, H.; Tyndale, R.F.; Sellers, E.M. Inhibition of cytochromes P450 by antifungal imidazole derivatives. Drug Metab. Dispos. 2002, 30, 314–3318. [Google Scholar] [CrossRef] [PubMed]
- Sezutsu, H.; Le Goff, G.; Feyereisen, R. Origins of P450 diversity. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2013, 368, 20120428. [Google Scholar] [CrossRef] [PubMed]
- Berenbaum, M.R.; Johnson, R.M. Xenobiotic detoxification pathways in honey bees. Curr. Opin. Insect Sci. 2015, 10, 51–58. [Google Scholar] [CrossRef]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. CYP9Q-mediated detoxification of acaricides in the honey bee (Apis mellifera). Proc. Natl. Acad. Sci. USA 2011, 108, 12657–15662. [Google Scholar] [CrossRef] [PubMed]
- Claudianos, C.; Ranson, H.; Johnson, R.M.; Biswas, S.; Schuler, M.A.; Berenbaum, M.R.; Feyereisen, R.; Oakeshott, J.G. A deficit of detoxification enzymes: Pesticide sensitivity and environmental response in the honeybee. Insect Mol. Biol. 2006, 15, 615–636. [Google Scholar] [CrossRef] [PubMed]
- Schmehl, D.R.; Tomé, H.V.V.; Mortensen, A.N.; Martins, G.F.; Ellis, J.D. Protocol for the in vitro rearing of honey bee (Apis mellifera L.) workers. J. Apic. Res. 2016. [Google Scholar] [CrossRef]
- Office of Pesticide Programs US EPA. Ecological Effects Test Guidelines OPPTS 850.3020 Honey Bee Acute Contact Toxicity; EPA: Washington, DC, USA, 1996.
- Office of Pesticide Programs US EPA. BeeREX; United States Environmental Protection Agency: Washington, DC, USA, 2015.
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; vanEngelsdorp, D.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef] [PubMed]
- Aupinel, P.; Fortini, D. Improvement of artificial feeding in a standard in vitro method for rearing Apis mellifera larvae. Bull. Insectol. 2005, 58, 107–111. [Google Scholar]
- Robertson, J.L.; Savin, N.E.; Preisler, H.K.; Russell, R.M. Bioassays with Arthropods, Second Edition, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2007; ISBN 9780849323317. [Google Scholar]
- Office of Pesticide Programs US EPA. Guidance for Assessing Pesticide Risks to Bees; United States Environmental Protection Agency: Washington, DC, USA, 2014.

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wade, A.; Lin, C.-H.; Kurkul, C.; Regan, E.R.; Johnson, R.M. Combined Toxicity of Insecticides and Fungicides Applied to California Almond Orchards to Honey Bee Larvae and Adults. Insects 2019, 10, 20. https://doi.org/10.3390/insects10010020
Wade A, Lin C-H, Kurkul C, Regan ER, Johnson RM. Combined Toxicity of Insecticides and Fungicides Applied to California Almond Orchards to Honey Bee Larvae and Adults. Insects. 2019; 10(1):20. https://doi.org/10.3390/insects10010020
Chicago/Turabian StyleWade, Andrea, Chia-Hua Lin, Colin Kurkul, Erzsébet Ravasz Regan, and Reed M. Johnson. 2019. "Combined Toxicity of Insecticides and Fungicides Applied to California Almond Orchards to Honey Bee Larvae and Adults" Insects 10, no. 1: 20. https://doi.org/10.3390/insects10010020
APA StyleWade, A., Lin, C.-H., Kurkul, C., Regan, E. R., & Johnson, R. M. (2019). Combined Toxicity of Insecticides and Fungicides Applied to California Almond Orchards to Honey Bee Larvae and Adults. Insects, 10(1), 20. https://doi.org/10.3390/insects10010020




