Reproductive Senescence in Drones of the Honey Bee (Apis mellifera)
Abstract
1. Introduction
1.1. Pollinator Decline
1.2. The Importance of Queens
1.3. The Importance of Drones
1.4. Study Objective
2. Materials and Methods
2.1. Drone Rearing and Collection
2.2. Dissection
2.3. Semen Analyses
2.4. Digital Measurements
2.5. Statistical Analyses
3. Results
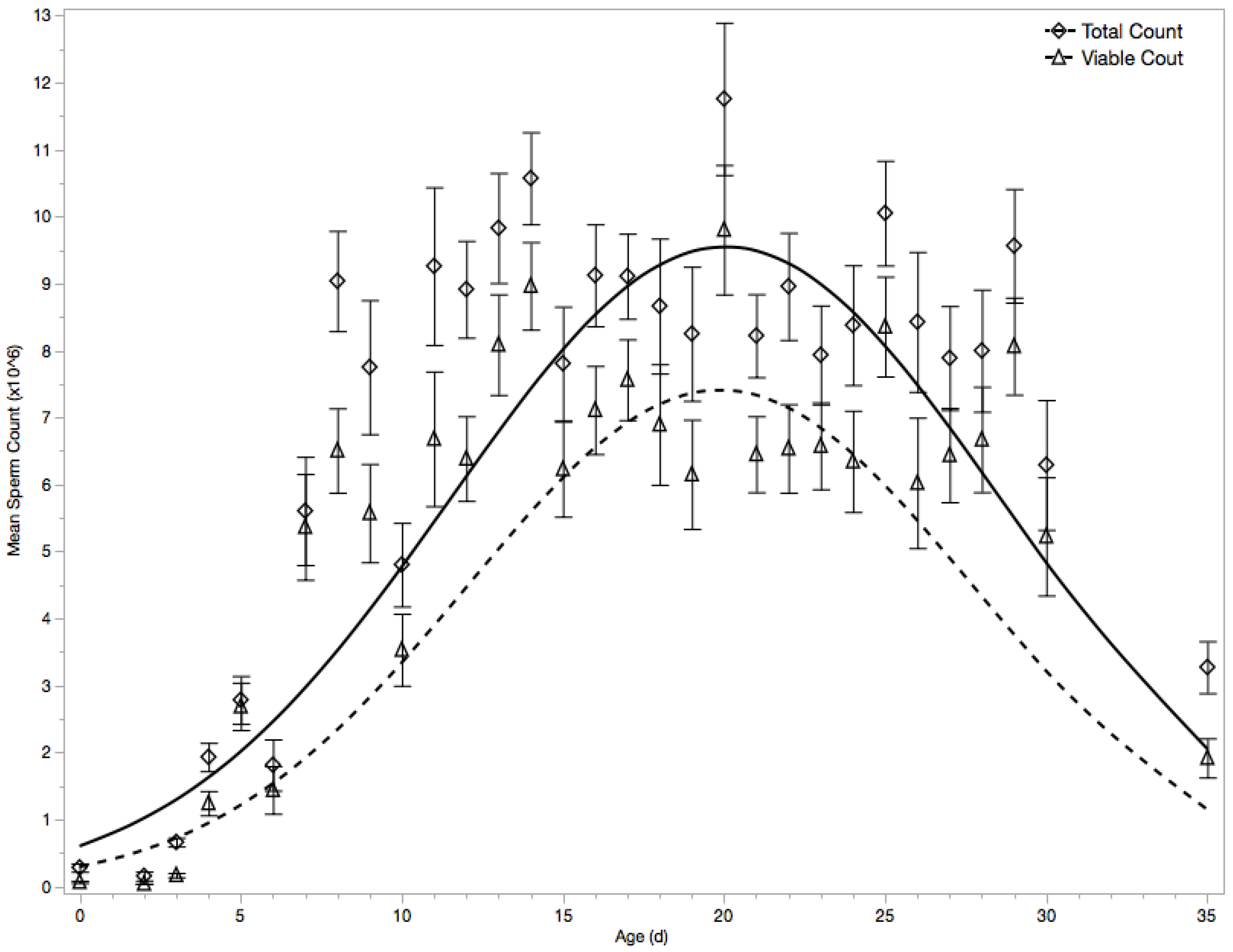
3.1. Migration of Spermatozoa to Seminal Vesicles
3.2. Viable Spermatozoa Count
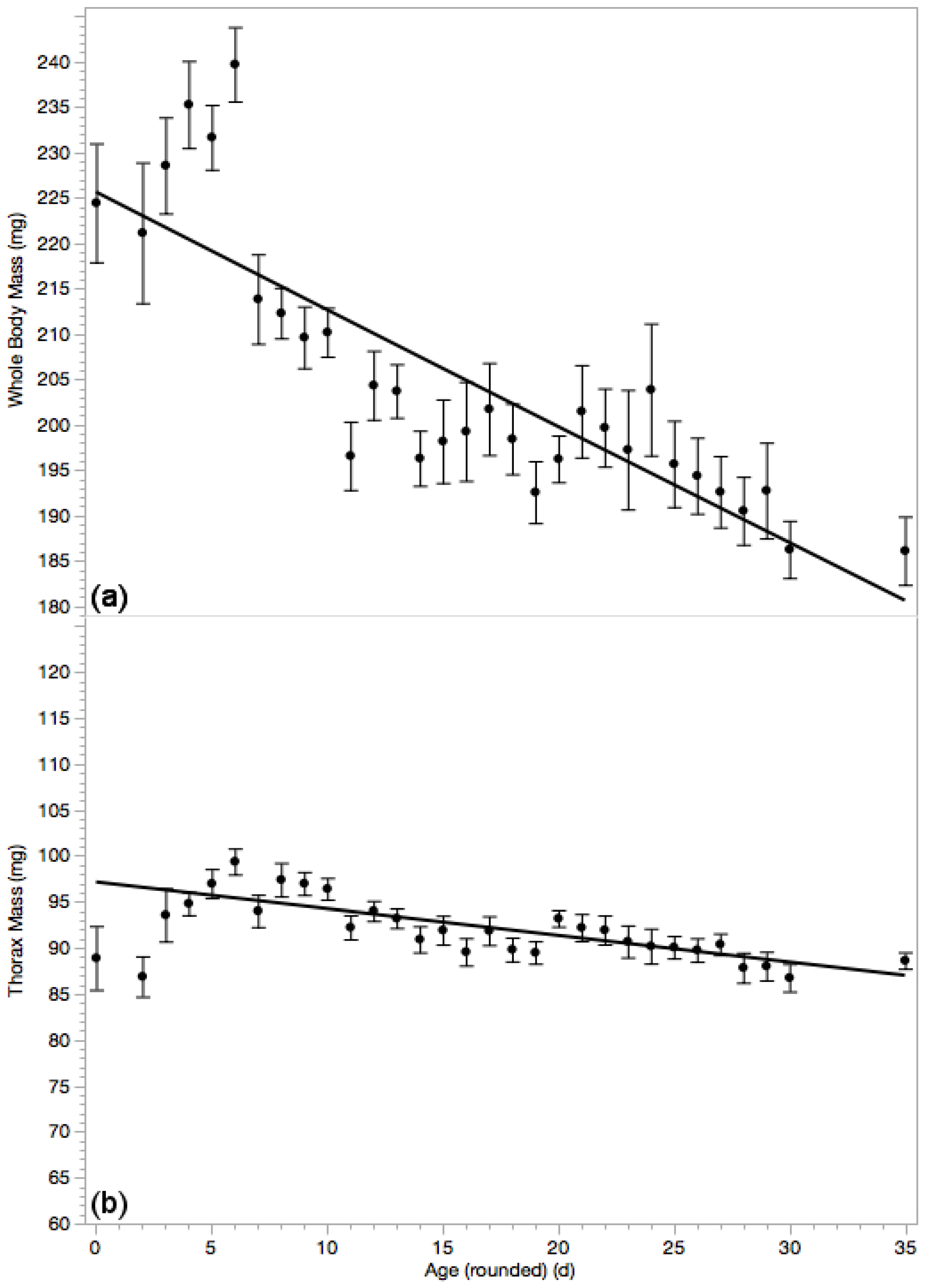
3.3. Whole Body Mass & Thorax Mass
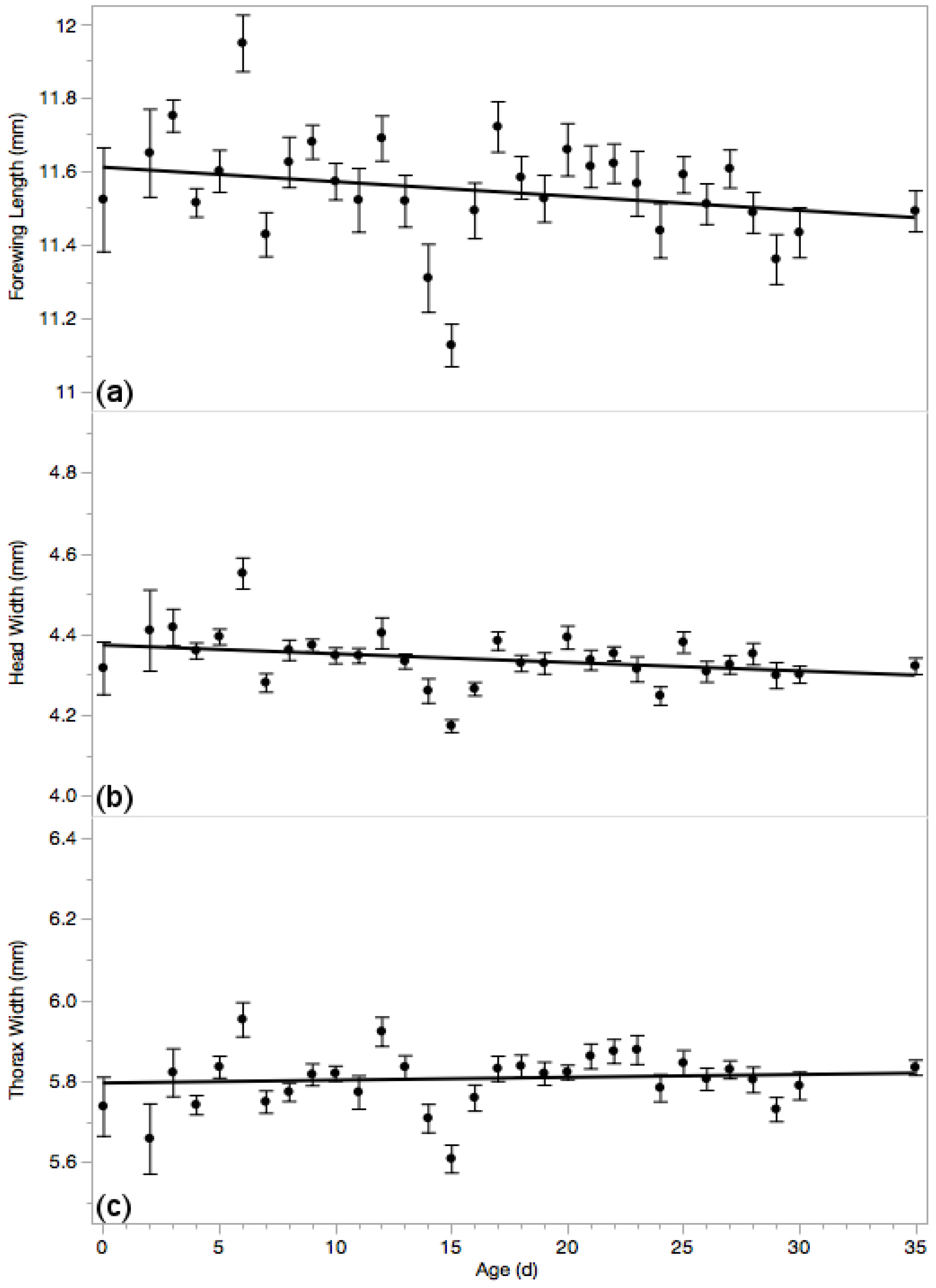
3.4. Body Measures
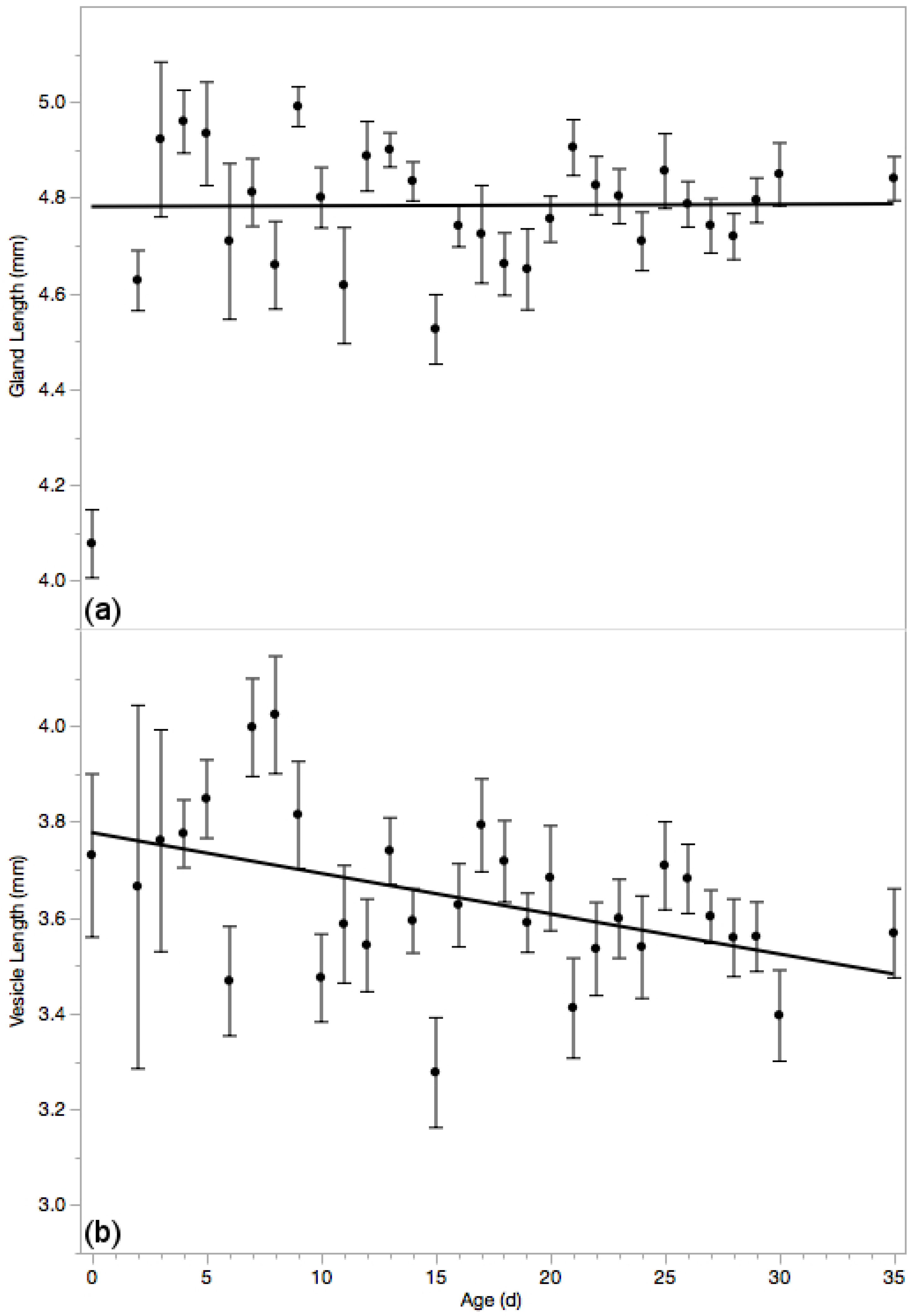
3.5. Reproductive Glands
3.6. Multivariate Analyses
4. Discussion
4.1. Spermatozoa Migration
4.2. Drone Senescence
4.3. Small Drones
4.4. Effect on Queen Mating Success
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ellis, J.D.; Evans, J.D.; Pettis, J. Colony losses, managed colony population decline, and colony collapse disorder in the United States. J. Apic. Res. 2010, 49, 134–136. [Google Scholar] [CrossRef]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schwieger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts, and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef] [PubMed]
- VanEngelsdorp, D.; Meixner, M.D. A historical review of managed honey bee populations in Europe and the United States and the factors that may affect them. J. Invertebr. Pathol. 2010, 103, S80–S95. [Google Scholar] [CrossRef] [PubMed]
- Seitz, N.; Traynor, K.S.; Steinhauer, N.; Rennich, K.; Wilson, M.E.; Ellis, J.D.; Rose, R.; Tarpy, D.R.; Sagili, R.R.; Caron, D.M.; et al. A national survey of managed honey bee 2014–2015 annual colony losses in the USA. J. Apic. Res. 2015, 54, 292–304. [Google Scholar] [CrossRef]
- VanEngelsdorp, D.; Evans, J.D.; Saegerman, C.; Mullin, C.; Haubruge, E.; Nguyen, B.K.; Frazier, M.; Frazier, J.; Cox-Foster, D.; Chen, Y.; et al. Colony collapse disorder: A descriptive study. PLoS ONE 2009, 4, e6481. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.V.; Steinhauer, N.; Rennich, K.; Wilson, M.E.; Tarpy, D.R.; Caron, D.M.; Rose, R.; Delaplane, K.S.; Baylis, K.; Lengerich, E.J.; et al. A national survey of managed honey bee 2013–2014 annual colony losses in the USA. Apidologie 2015, 46, 292–305. [Google Scholar] [CrossRef]
- Steinhauer, N.A.; Rennich, K.; Wilson, M.E.; Caron, D.M.; Lengerich, E.J.; Pettis, J.S.; Rose, R.; Skinner, J.A.; Tarpy, D.R.; Wilkes, J.T.; et al. A national survey of managed honey bee 2012–2013 annual colony losses in the USA: Results from the Bee Informed Partnership. J. Apic. Res. 2014, 53, 1–18. [Google Scholar] [CrossRef]
- Goulson, D.; Lye, G.C.; Darvill, B. Decline and conservation of bumble bees. Annu. Rev. Entomol. 2008, 53, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Koh, I.; Lonsdorf, E.V.; Williams, N.M.; Brittain, C.; Isaacs, R.; Gibbs, J.; Ricketts, T.H. Modeling the status, trends, and impacts of wild bee abundance in the United States. Proc. Natl. Acad. Sci. USA 2016, 113, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Owen, R. The Decline of Pollinators. Available online: http://www.beeculture.com/the-decline-of-pollinators/ (accessed on 8 September 2017).
- Aizen, M.A.; Garibaldi, L.A.; Cunningham, S.A.; Klein, A.M. Long-term global trends in crop yield and production reveal no current pollination shortage but increasing pollinator dependency. Curr. Biol. 2008, 18, 1572–1575. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.-M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. Lond. B Biol. Sci. 2007, 274. [Google Scholar] [CrossRef] [PubMed]
- Gallai, N.; Salles, J.-M.; Settele, J.; Vaissière, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Kulhanek, K.; Steinhauer, N.; Rennich, K.; Caron, D.M.; Sagili, R.R.; Pettis, J.S.; Ellis, J.D.; Wilson, M.E.; Wilkes, J.T.; Tarpy, D.R.; et al. A national survey of managed honey bee 2015–2016 annual colony losses in the USA. J. Apic. Res. 2017, 56, 328–340. [Google Scholar] [CrossRef]
- Traynor, K.S.; Pettis, J.S.; Tarpy, D.R.; Mullin, C.A.; Frazier, J.L.; Frazier, M.; vanEngelsdorp, D. In-hive Pesticide Exposome: Assessing risks to migratory honey bees from in-hive pesticide contamination in the eastern United States. Sci. Rep. 2016, 6, 33207. [Google Scholar] [CrossRef] [PubMed]
- Winston, M.L. The Biology of the Honey Bee; 1st Paperback; Harvard University Press: Cambridge, MA, USA, 1987; ISBN 0-674-07409-2. [Google Scholar]
- Amiri, E.; Strand, M.; Rueppell, O.; Tarpy, D. Queen Quality and the impact of honey bee diseases on queen health: Potential for interactions between two major threats to colony health. Insects 2017, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.L.; Gary, N.E. Honey productivity of honeybee colonies in relation to body weight, attractiveness and fecundity of the queen. J. Apic. Res. 1983, 22, 209–213. [Google Scholar] [CrossRef]
- Kahya, Y.; Gençer, H.V.; Woyke, J. Weight at emergence of honey bee (Apis mellifera caucasica) queens and its effect on live weights at the pre and post mating periods. J. Apic. Res. 2008, 47, 118–125. [Google Scholar] [CrossRef]
- Tarpy, D.R.; Keller, J.J.; Caren, J.R.; Delaney, D.A. Experimentally induced variation in the physical reproductive potential and mating success in honey bee queens. Insectes Soc. 2011, 58, 569–574. [Google Scholar] [CrossRef]
- Tarpy, D.R.; Keller, J.J.; Caren, J.R.; Delaney, D.A. Assessing the mating “health” of commercial honey bee queens. J. Econ. Entomol. 2012, 105, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Rangel, J.; Keller, J.J.; Tarpy, D.R. The effects of honey bee (Apis mellifera L.) queen reproductive potential on colony growth. Insectes Soc. 2013, 60, 65–73. [Google Scholar] [CrossRef]
- Rangel, J.; Böröczky, K.; Schal, C.; Tarpy, D.R. Honey bee (Apis mellifera) queen reproductive potential affects queen mandibular gland pheromone composition and worker retinue response. PLoS ONE 2016, 11, e0156027. [Google Scholar] [CrossRef] [PubMed]
- Pettis, J.S.; Rice, N.; Joselow, K.; vanEngelsdorp, D.; Chaimanee, V. Colony failure linked to low sperm viability in honey bee (Apis mellifera) queens and an exploration of potential causative factors. PLoS ONE 2016, 11, e0147220. [Google Scholar] [CrossRef]
- Delaney, D.A.; Keller, J.J.; Caren, J.R.; Tarpy, D.R. The physical, insemination, and reproductive quality of honey bee queens (Apis mellifera L.). Apidologie 2011, 42, 1–13. [Google Scholar] [CrossRef]
- Woyke, J. Multiple mating of the honeybee queen (Apis mellifica L.) in one nuptial flight. Bull. L’Acad. Pol. Sci. 1955, 3, 175–180. [Google Scholar]
- Woyke, J. Anatomo-physiological changes in queen-bees returning from mating flights, and the process of multiple mating. Bull. L’Acad. Pol. Sci. 1956, 4, 81–86. [Google Scholar]
- Tarpy, D.R.; Nielsen, R.; Nielsen, D.I. A scientific note on the revised estimates of effective paternity frequency in Apis. Insectes Soc. 2004, 51, 203–204. [Google Scholar] [CrossRef]
- Tarpy, D.R.; Delaney, D.A.; Seeley, T.D.; Crewe, R.; Solignac, M. Mating frequencies of honey bee queens (Apis mellifera L.) in a population of feral colonies in the northeastern United States. PLoS ONE 2015, 10, e0118734. [Google Scholar] [CrossRef] [PubMed]
- Mattila, H.R.; Seeley, T.D. Genetic diversity in honey bee colonies enhances productivity and fitness. Science 2007, 317, 362–364. [Google Scholar] [CrossRef] [PubMed]
- Simone-Finstrom, M.; Walz, M.; Tarpy, D.R. Genetic diversity confers colony-level benefits due to individual immunity. Biol. Lett. 2016, 12, 20151007. [Google Scholar] [CrossRef] [PubMed]
- Kairo, G.; Provost, B.; Tchamitchian, S.; Abdelkader, F. Ben; Bonnet, M.; Cousin, M.; Sénéchal, J.; Benet, P.; Kretzschmar, A.; Belzunces, L.P.; et al. Drone exposure to the systemic insecticide Fipronil indirectly impairs queen reproductive potential. Sci. Rep. 2016, 6, 31904. [Google Scholar] [CrossRef] [PubMed]
- Kairo, G.; Poquet, Y.; Haji, H.; Tchamitchian, S.; Cousin, M.; Bonnet, M.; Pelissier, M.; Kretzschmar, A.; Belzunces, L.P.; Brunet, J. Assessment of the toxic effect of pesticides on honey bee drone fertility using laboratory and semifield approaches: A case study of fipronil. Environ. Toxicol. Chem. 2017, 36, 2345–2351. [Google Scholar] [CrossRef] [PubMed]
- Koeniger, G.; Koeniger, N.; Ellis, J.; Connor, L. Mating Biology of Honey Bees (Apis mellifera), 1st ed.; Wicwas Press: Kalamazoo, MI, USA, 2014; ISBN 978-1-878075-38-3. [Google Scholar]
- Berg, S.; Koeniger, N.; Koeniger, G.; Fuchs, S. Body size and reproductive success of drones (Apis mellifera L.). Apidologie 1997, 28, 449–460. [Google Scholar] [CrossRef]
- Collins, A.M.; Pettis, J.S. Effect of Varroa infestation on semen quality. Am. Bee J. 2001, 141, 590–593. [Google Scholar]
- Peng, Y.; Baer-Imhoof, B.; Harvey Millar, A.; Baer, B. Consequences of Nosema apis infection for male honey bees and their fertility. Sci. Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Tofilski, A.; Kopel, J. The influence of Nosema apis on maturation and flight activity of honey bee drones. Pszczel. Zesz. Nauk. 1996, 40, 55–60. [Google Scholar]
- Ciereszko, A.; Wilde, J.; Dietrich, G.J.; Siuda, M.; Bąk, B.; Judycka, S.; Karol, H. Sperm parameters of honeybee drones exposed to imidacloprid. Apidologie 2017, 48, 211–222. [Google Scholar] [CrossRef]
- Kairo, G.; Biron, D.G.; Ben Abdelkader, F.; Bonnet, M.; Tchamitchian, S.; Cousin, M.; Dussaubat, C.; Benoit, B.; Kretzschmar, A.; Belzunces, L.P.; et al. Nosema ceranae, Fipronil and their combination compromise honey bee reproduction via changes in male physiology. Sci. Rep. 2017, 7, 8556. [Google Scholar] [CrossRef] [PubMed]
- Straub, L.; Villamar-Bouza, L.; Bruckner, S.; Chantawannakul, P.; Gauthier, L.; Khongphinitbunjong, K.; Retschnig, G.; Troxler, A.; Vidondo, B.; Neumann, P.; et al. Neonicotinoid insecticides can serve as inadvertent insect contraceptives. Proc. R. Soc. B 2016, 283, 20160506. [Google Scholar] [CrossRef] [PubMed]
- Rinderer, T.E.; Guzman, L.I.; Lancaster, V.A.; Delatte, G.T.; Stelzer, J.A. Varroa in the mating yard. I. The effects of Varroa jacobsoni and Apistan on drone honey bees. Am. Bee J. 1999, 139, 134–139. [Google Scholar]
- Burley, L.M. The Effects of Miticides on the Reproductive Physiology of Honey Bee (Apis mellifera L.) Queens and Drones; Virginia Polytechnic Institute and State University: Blacksburg, VA, USA, 2007. [Google Scholar]
- Johnson, R.M.; Dahlgren, L.; Siegfried, B.D.; Ellis, M.D. Effect of in-hive miticides on drone honey bee survival and sperm viability. J. Apic. Res. 2013, 52, 88–95. [Google Scholar] [CrossRef]
- Schlüns, H.; Schlüns, E.A.; van Praagh, J.; Moritz, R.F.A. Sperm numbers in drone honeybees (Apis mellifera) depend on body size. Apidologie 2003, 34, 577–584. [Google Scholar] [CrossRef]
- Gençer, H.V.; Kahya, Y. Are sperm traits of drones (Apis mellifera L.) from laying worker colonies noteworthy? J. Apic. Res. 2011, 50, 130–137. [Google Scholar] [CrossRef]
- Berg, S. Investigation on the rates of large and small drones at a drone congregation area. Apidologie 1991, 22, 437–438. [Google Scholar]
- Koeniger, G.; Koeniger, N.; Tingek, S.; Phiancharoen, M. Variance in spermatozoa number among Apis dorsata drones and among Apis mellifera drones. Apidologie 2005, 36, 279–284. [Google Scholar] [CrossRef]
- Rhodes, J.W.; Harden, S.; Spooner-Hart, R.; Anderson, D.L.; Wheen, G. Effects of age, season and genetics on semen and sperm production in Apis mellifera drones. Apidologie 2011, 42, 29–38. [Google Scholar] [CrossRef]
- Rousseau, A.; Fournier, V.; Giovenazzo, P. Apis mellifera (Hymenoptera: Apidae) drone sperm quality in relation to age, genetic line, and time of breeding. Can. Entomol. 2015, 147, 702–711. [Google Scholar] [CrossRef]
- Kraus, F.B.; Neumann, P.; Scharpenberg, H.; Van Praagh, J.; Moritz, R.F.A. Male fitness of honeybee colonies (Apis mellifera L.). J. Evol. Biol. 2003, 16, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Czekońska, K.; Chuda-Mickiewicz, B.; Samborski, J. Quality of honeybee drones reared in colonies with limited and unlimited access to pollen. Apidologie 2015, 46, 1–9. [Google Scholar] [CrossRef]
- Rousseau, A.; Giovenazzo, P. Optimizing drone fertility with spring nutritional supplements to honey bee (Hymenoptera: Apidae) colonies. J. Econ. Entomol. 2016, 109, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Boes, K.E. Honeybee colony drone production and maintenance in accordance with environmental factors: An interplay of queen and worker decisions. Insectes Soc. 2010, 57, 1–9. [Google Scholar] [CrossRef]
- Free, J.B.; Williams, I.H. Factors determining the rearing and rejection of drones by the honeybee colony. Anim. Behav. 1975, 23, 650–675. [Google Scholar] [CrossRef]
- Smith, M.L.; Ostwald, M.M.; Seeley, T.D. Honey bee sociometry: Tracking honey bee colonies and their nest contents from colony founding until death. Insectes Soc. 2016, 63, 553–563. [Google Scholar] [CrossRef]
- Smith, M.L.; Koenig, P.A.; Peters, J.M. The cues of colony size: How honey bees sense that their colony is large enough to begin to invest in reproduction. J. Exp. Biol. 2017, 220, 1597–1605. [Google Scholar] [CrossRef] [PubMed]
- Seeley, T.D.; Mikheyev, A.S. Reproductive decisions by honey bee colonies: Tuning investment in male production in relation to success in energy acquisition. Insectes Soc. 2003, 50, 134–138. [Google Scholar] [CrossRef]
- Rueppell, O.; Fondrk, M.K.; Page, R.E. Biodemographic analysis of male honey bee mortality. Aging Cell 2005, 4, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Locke, S.J.; Peng, Y.-S. The effects of drone age, semen storage and contamination on semen quality in the honey bee (Apis mellifera). Physiol. Entomol. 1993, 18, 144–148. [Google Scholar] [CrossRef]
- Mazeed, A.M.; Mohanny, K.M. Some reproductive characteristics of honeybee drones in relation to their ages. Entomol. Res. 2010, 40, 245–250. [Google Scholar] [CrossRef]
- Czekońska, K.; Chuda-Mickiewicz, B. The influence of honey bee (Apis mellifera) drone age on volume of semen and viability of spermatozoa. J. Apic. Sci. 2013, 57, 61–66. [Google Scholar] [CrossRef]
- Woyke, J. Influence of age of drones on the results of instrumental insemination of honeybee queens. Apidologie 1978, 9, 203–212. [Google Scholar] [CrossRef]
- Stürup, M.; Baer-Imhoof, B.; Nash, D.R.; Boomsma, J.J.; Baer, B. When every sperm counts: Factors affecting male fertility in the honeybee Apis mellifera. Behav. Ecol. 2013, 24, 1192–1198. [Google Scholar] [CrossRef]
- Li-Byarlay, H.; Huang, M.H.; Simone-Finstrom, M.; Strand, M.K.; Tarpy, D.R.; Rueppell, O. Honey bee (Apis mellifera) drones survive oxidative stress due to increased tolerance instead of avoidance or repair of oxidative damage. Exp. Gerontol. 2016, 83, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Gençer, H.V.; Firatli, Ç. Reproductive and morphological comparisons of drones reared in queenright and laying worker colonies. J. Apic. Res. 2005, 44, 163–167. [Google Scholar] [CrossRef]
- Jaycox, E.R. The effects of various foods and temperatures on sexual maturity of the drone honey bee (Apis mellifera). Annu. Entomol. Soc. Am. 1961, 54, 519–523. [Google Scholar] [CrossRef]
- Mackensen, O. Experiments in the technique of artificial insemination of queen bees. J. Econ. Entomol. 1955, 48, 418–421. [Google Scholar] [CrossRef]
- Page, R.E.; Erber, J. Levels of behavioral organization and the evolution of division of labor. Naturwissenschaften 2002, 89, 91–106. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9. [Google Scholar] [CrossRef]
- Ambrose, J.T.; Atkins, E.L.; Avitabile, A.; Ayers, G.S.; Blum, M.S.; Buchmann, S.L.; Caron, D.M.; Crane, E.; Dadant, C.C.; Dietz, A.; et al. The Hive and the Honey Bee, 5th ed.; Graham, J.M., Ed.; Dadant & Sons: Chelsea, MI, USA, 1992; ISBN 0-915698-09-9. [Google Scholar]
- Collins, A.M. Relationship between semen quality and performance of instrumentally inseminated honey bee queens. Apidologie 2000, 31, 421–429. [Google Scholar] [CrossRef]
- Tofilski, A. Honey Bee. Available online: http://www.honeybee.drawwing.org (accessed on 2 January 2018).
- Cortez, P.; Cerdeira, A.; Almeida, F.; Matos, T.; Reis, J. Modeling wine preferences by data mining from physicochemical properties. Decis. Support Syst. 2009, 47, 547–553. [Google Scholar] [CrossRef]
- Tozetto, S.d.O.; Bitondi, M.M.G.; Dallacqua, R.P.; Paulino Simoes, Z.L. Protein profiles of testes, seminal vesicles and accessory glands of honey bee pupae and their relation to the ecdysteroid titer. Apidologie 2007, 38, 1–11. [Google Scholar] [CrossRef]
- Kerr, W.E.; da Silveira, Z.V. A note on the formation of honeybee spermatozoa. J. Apic. Res. 1974, 13, 121–126. [Google Scholar] [CrossRef]
- Snodgrass, R.E. The Anatomy of the Honey Bee; USDA Bureau of Entomology: Washington, DC, USA, 1910; Volume 18. [Google Scholar]
- Bishop, G.H. Fertilization in the honey-bee. I. The male sexual organs: Their histological structure and physiological functioning. J. Exp. Zool. 1920, 31, 224–265. [Google Scholar] [CrossRef]
- Ruttner, F. The Instrumental Insemination of the Queen Bee, 2nd ed.; APIMONDIA International Beekeeping Technology and Economy Institute: Bucharest, Romania, 1976. [Google Scholar]
- Page, R.E., Jr.; Peng, C.Y.-S. Aging and development in social insects with emphasis on the honey bee, Apis mellifera L. Exp. Gerontol. 2001, 36, 695–711. [Google Scholar] [CrossRef]
- Heinze, J.; Schrempf, A. Experimental section aging and reproduction in social insects—A mini-review. Gerontology 2008, 54, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Visscher, K.P. A quantitative study of worker reproduction in honeybee colonies. Behav. Ecol. Sociobiol. 1989, 25, 247–254. [Google Scholar] [CrossRef]
- Couvillon, M.J.; Hughes, W.O.H.; Perez-Sato, J.A.; Martin, S.J.; Roy, G.G.F.; Ratnieks, F.L.W. Sexual selection in honey bees: Colony variation and the importance of size in male mating success. Behav. Ecol. 2010, 21, 520–525. [Google Scholar] [CrossRef]
- Jarolimek, J.; Otis, G. A comparison of fitness components in large and small honeybee drones. Am. Bee J. 2001, 12, 891–892. [Google Scholar]

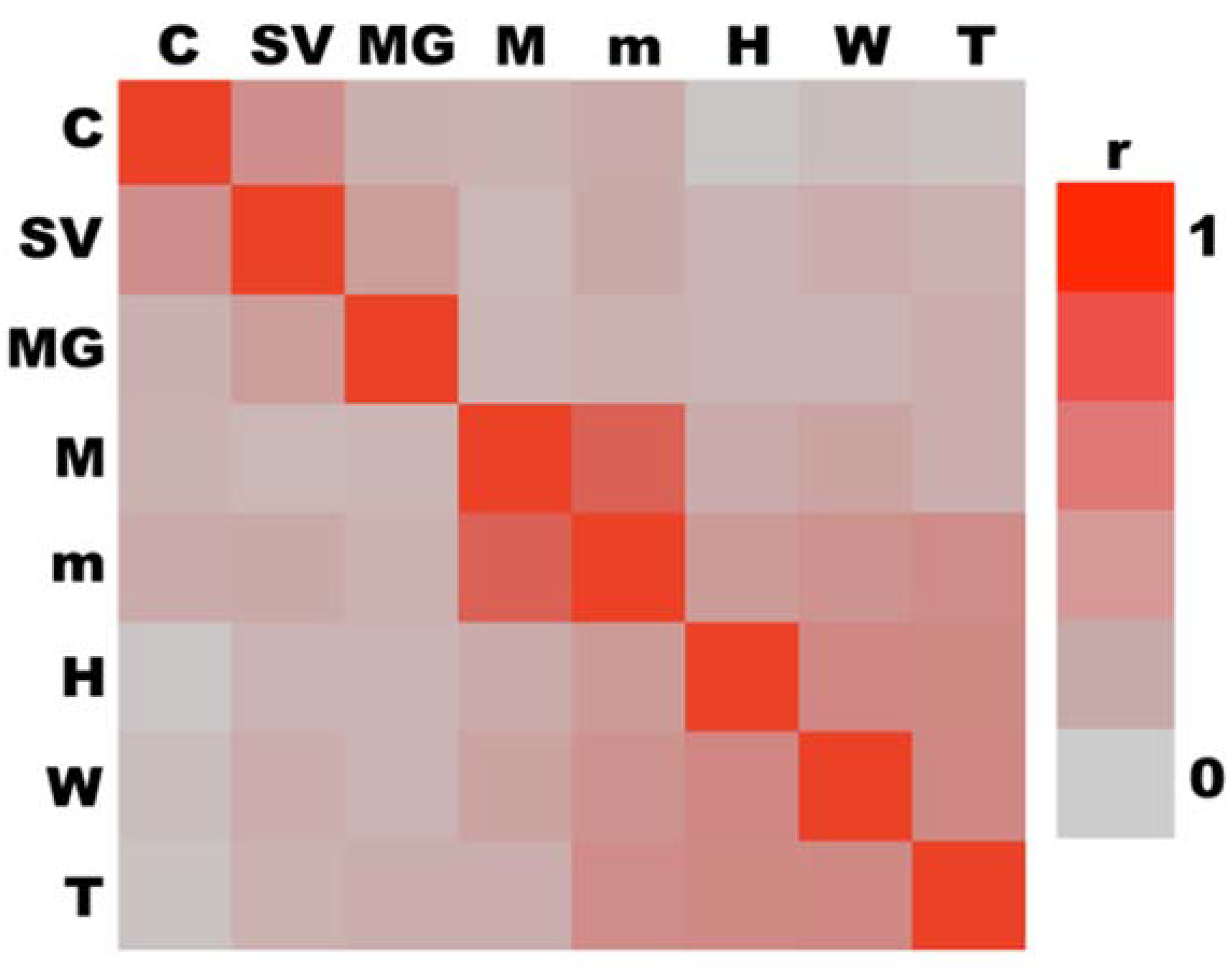
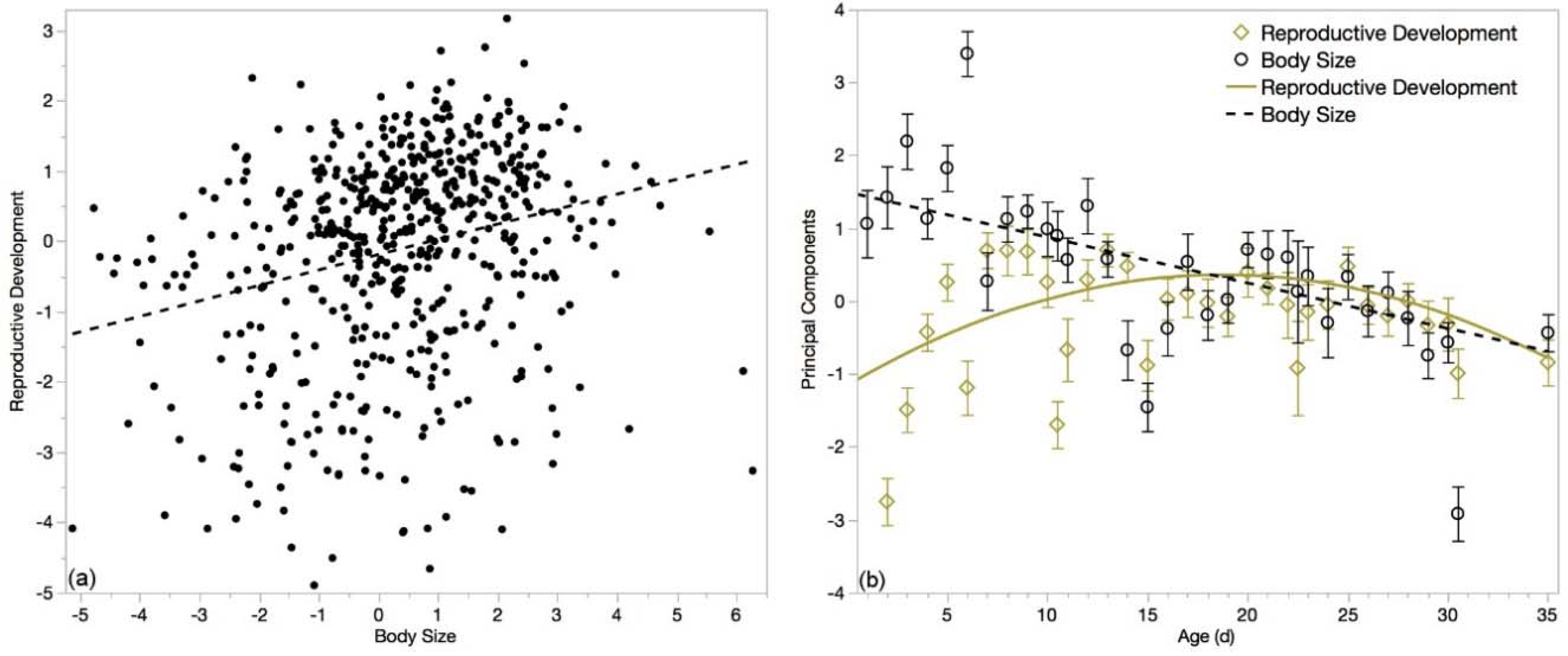
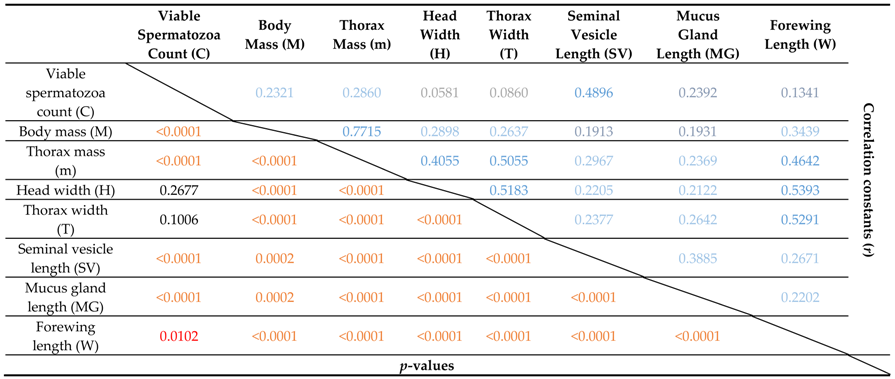
| Loading Variable | Eigenvector |
|---|---|
| Viable spermatozoa count (C) | 0.576 |
| Seminal Vesicle length (SV) | 0.638 |
| Mucus Gland length (MG) | 0.511 |
| Loading Variable | Eigenvector |
|---|---|
| Body mass (M) | 0.418 |
| Thorax mass (m) | 0.498 |
| Head width (H) | 0.426 |
| Thorax width (T) | 0.441 |
| Forewing length (W) | 0.450 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Metz, B.N.; Tarpy, D.R. Reproductive Senescence in Drones of the Honey Bee (Apis mellifera). Insects 2019, 10, 11. https://doi.org/10.3390/insects10010011
Metz BN, Tarpy DR. Reproductive Senescence in Drones of the Honey Bee (Apis mellifera). Insects. 2019; 10(1):11. https://doi.org/10.3390/insects10010011
Chicago/Turabian StyleMetz, Bradley N., and David R. Tarpy. 2019. "Reproductive Senescence in Drones of the Honey Bee (Apis mellifera)" Insects 10, no. 1: 11. https://doi.org/10.3390/insects10010011
APA StyleMetz, B. N., & Tarpy, D. R. (2019). Reproductive Senescence in Drones of the Honey Bee (Apis mellifera). Insects, 10(1), 11. https://doi.org/10.3390/insects10010011




