Clinical Effect of the C-Reactive Protein to Serum Albumin Ratio in Patients with Metastatic Gastric or Gastroesophageal Junction Cancer Treated with Trifluridine/Tipiracil
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Patients
2.3. Assessment of Treatment Response and Adverse Events after FTD/TPI Therapy
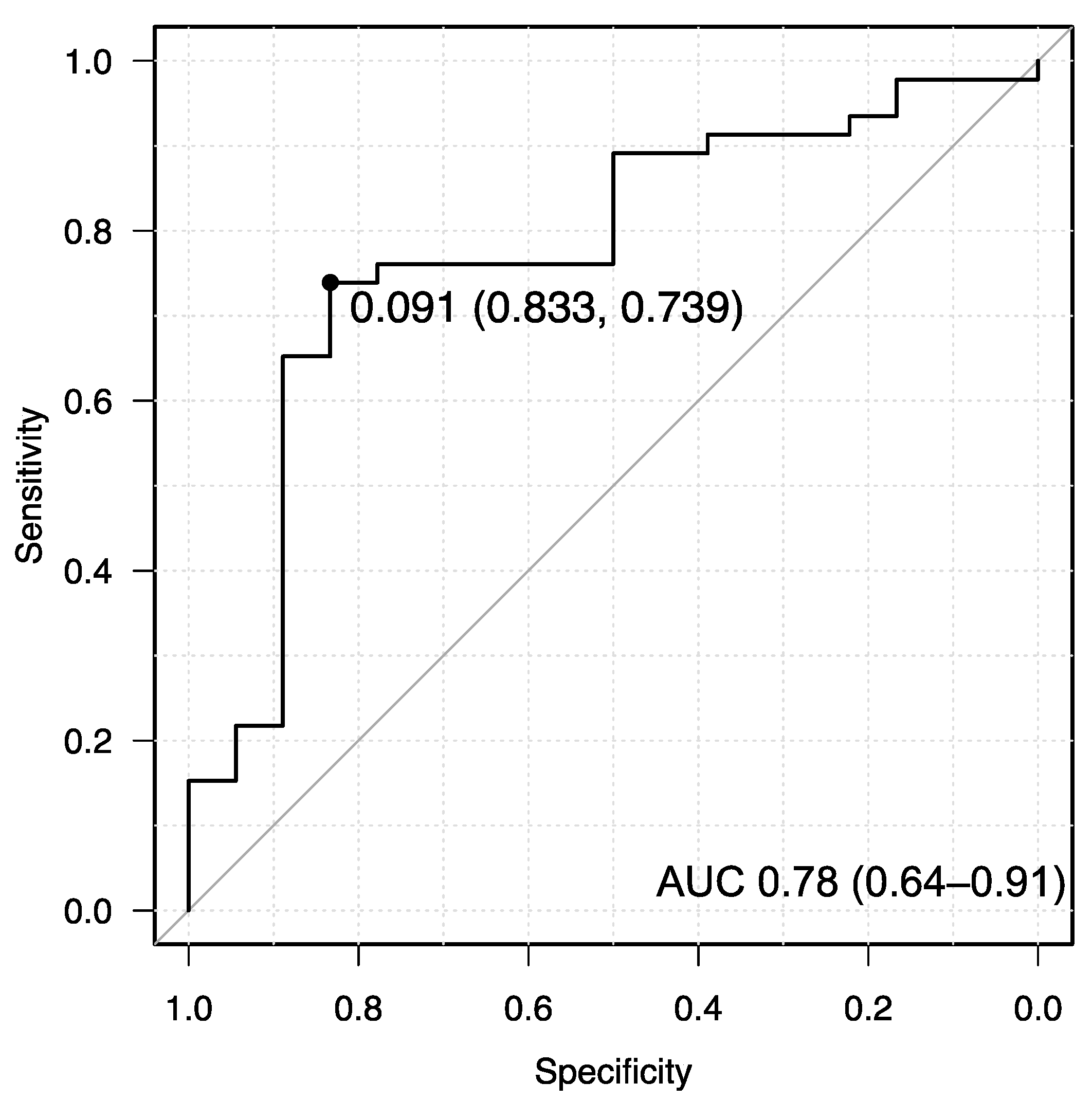
2.4. Definition of CAR
2.5. Statistical Analyses
3. Results
3.1. Patients
3.2. Relationship between CAR and Clinicopathological Factors
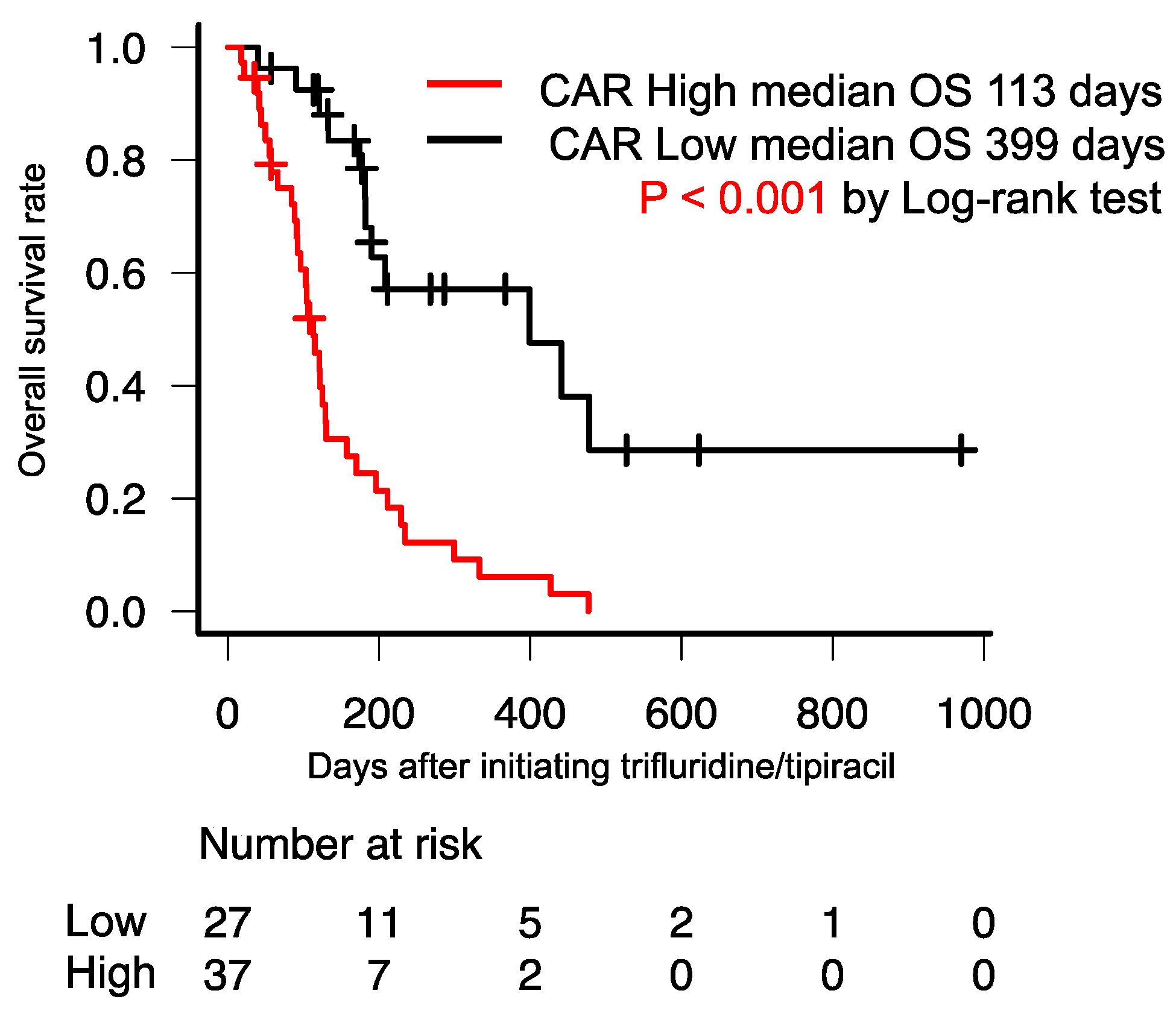
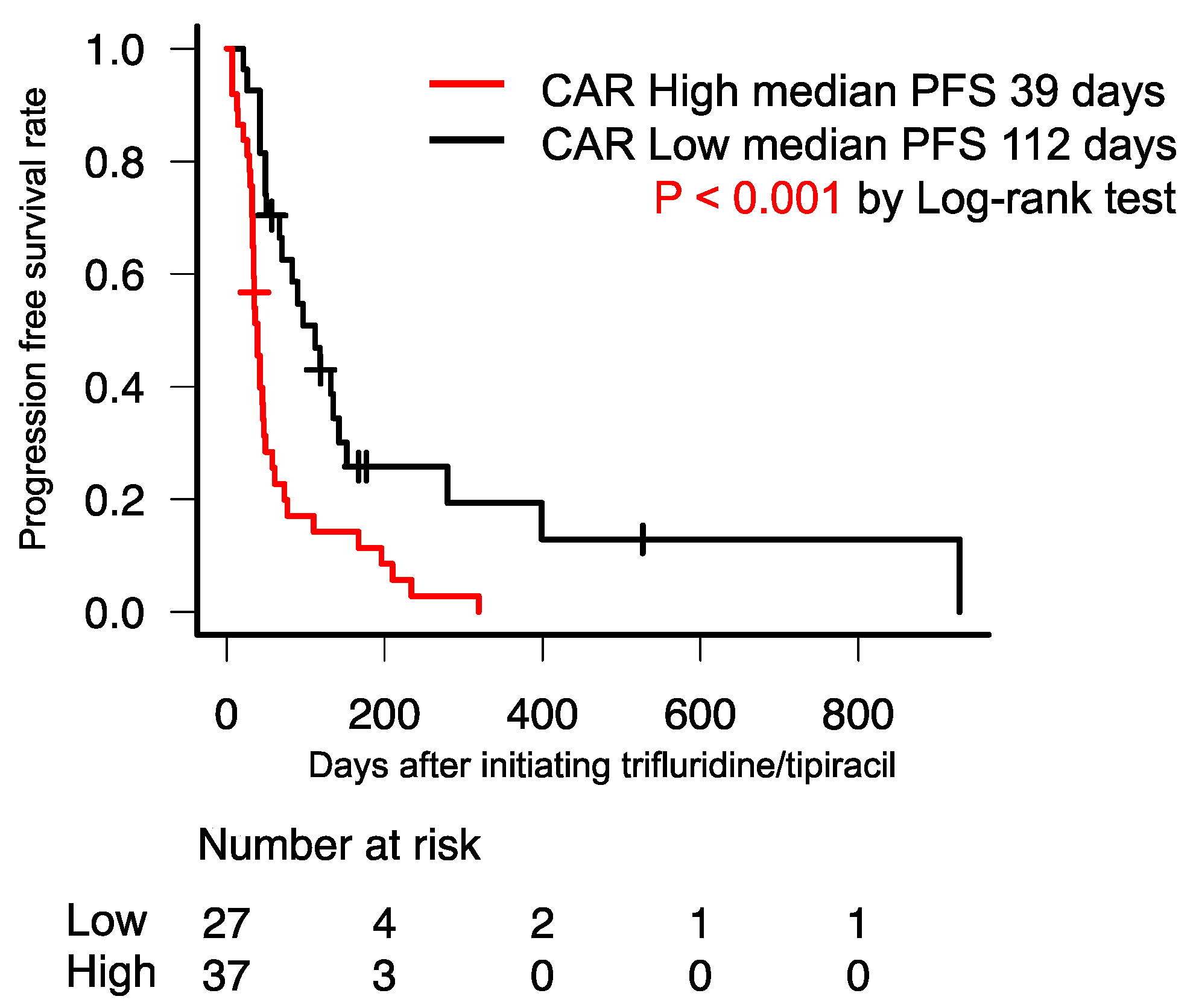
3.3. Median OS and PFS Based on the CAR in Patients with mGC/GEJC Treated with FTD/TPI
3.4. Univariate and Multivariate Analyses of OS and PFS
3.5. Relationship between CAR and Treatment Response to FTD/TPI Therapy
3.6. Adverse Events of FTD/TPI Therapy in the High- and Low-CAR Groups
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Smyth, E.C.; Nilsson, M.; Grabsch, H.I.; van Grieken, N.C.; Lordick, F. Gastric cancer. Lancet 2020, 396, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.-K.; Boku, N.; Satoh, T.; Ryu, M.-H.; Chao, Y.; Kato, K.; Chung, H.C.; Chen, J.-S.; Muro, K.; Kang, W.K.; et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): A randomised, double-blind, placebo-controlled, Phase 3 trial. Lancet 2017, 390, 2461–2471. [Google Scholar] [CrossRef] [PubMed]
- Janjigian, Y.Y.; Shitara, K.; Moehler, M.; Garrido, M.; Salman, P.; Shen, L.; Wyrwicz, L.; Yamaguchi, K.; Skoczylas, T.; Campos Bragagnoli, A.; et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): A randomised, open-label, Phase 3 trial. Lancet 2021, 398, 27–40. [Google Scholar] [CrossRef]
- Shitara, K.; Özgüroğlu, M.; Bang, Y.J.; Di Bartolomeo, M.; Mandalà, M.; Ryu, M.H.; Fornaro, L.; Olesiński, T.; Caglevic, C.; Chung, H.C.; et al. Pembrolizumab versus paclitaxel for previously treated, advanced castric or gastro-oesophageal junction cancer (KEYNOTE-061): A randomised, open-label, controlled, Phase 3 Trial. Lancet 2018, 392, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Shitara, K.; Bang, Y.J.; Iwasa, S.; Sugimoto, N.; Ryu, M.H.; Sakai, D.; Chung, H.C.; Kawakami, H.; Yabusaki, H.; Lee, J.; et al. Trastuzumab deruxtecan in previously treated HER2-positive gastric cancer. N. Engl. J. Med. 2020, 382, 2419–2430. [Google Scholar] [CrossRef]
- Murakami, Y.; Kazuno, H.; Emura, T.; Tsujimoto, H.; Suzuki, N.; Fukushima, M. Different mechanisms of acquired resistance to fluorinated pyrimidines in human colorectal cancer cells. Int. J. Oncol. 2000, 17, 277–283. [Google Scholar] [CrossRef]
- Fukushima, M.; Suzuki, N.; Emura, T.; Yano, S.; Kazuno, H.; Tada, Y.; Yamada, Y.; Asao, T. Structure and activity of specific inhibitors of thymidine phosphorylase to potentiate the function of antitumor 2′-deoxyribonucleosides. Biochem. Pharmacol. 2000, 59, 1227–1236. [Google Scholar] [CrossRef]
- Emura, T.; Nakagawa, F.; Fujioka, A.; Ohshimo, H.; Yokogawa, T.; Okabe, H.; Kitazato, K. An optimal dosing schedule for a novel combination antimetabolite, TAS-102, based on its intracellular metabolism and its incorporation into DNA. Int. J. Mol. Med. 2004, 13, 249–255. [Google Scholar] [CrossRef]
- Tanaka, N.; Sakamoto, K.; Okabe, H.; Fujioka, A.; Yamamura, K.; Nakagawa, F.; Nagase, H.; Yokogawa, T.; Oguchi, K.; Ishida, K.; et al. Repeated oral dosing of TAS-102 confers high trifluridine incorporation into DNA and sustained antitumor activity in mouse models. Oncol. Rep. 2014, 32, 2319–2326. [Google Scholar] [CrossRef]
- Shitara, K.; Doi, T.; Dvorkin, M.; Mansoor, W.; Arkenau, H.T.; Prokharau, A.; Alsina, M.; Ghidini, M.; Faustino, C.; Gorbunova, V.; et al. Trifluridine/Tipiracil versus placebo in patients with heavily pretreated metastatic gastric cancer (TAGS): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2018, 19, 1437–1448. [Google Scholar] [CrossRef]
- Japanese Gastric Cancer Association. Japanese Gastric Cancer Treatment Guidelines 2021 (6th edition). Gastric Cancer 2023, 26, 1–25. [Google Scholar] [CrossRef]
- Ilson, D.H.; Tabernero, J.; Prokharau, A.; Arkenau, H.T.; Ghidini, M.; Fujitani, K.; Van Cutsem, E.; Thuss-Patience, P.; Beretta, G.D.; Mansoor, W.; et al. Efficacy and safety of trifluridine/Tipiracil treatment in patients with metastatic gastric cancer who had undergone gastrectomy: Subgroup analyses of a randomized clinical trial. JAMA Oncol. 2020, 6, e193531. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, W.; Arkenau, H.T.; Alsina, M.; Shitara, K.; Thuss-Patience, P.; Cuffe, S.; Dvorkin, M.; Park, D.; Ando, T.; Van Den Eynde, M.; et al. Trifluridine/Tipiracil in patients with metastatic gastroesophageal junction cancer: A subgroup analysis from the Phase 3 TAGS study. Gastric Cancer 2021, 24, 970–977. [Google Scholar] [CrossRef]
- Shitara, K.; Doi, T.; Hosaka, H.; Thuss-Patience, P.; Santoro, A.; Longo, F.; Ozyilkan, O.; Cicin, I.; Park, D.; Zaanan, A.; et al. Efficacy and safety of trifluridine/Tipiracil in older and younger patients with metastatic gastric or gastroesophageal junction cancer: Subgroup analysis of a randomized Phase 3 study (TAGS). Gastric Cancer 2022, 25, 586–597. [Google Scholar] [CrossRef]
- Muscaritoli, M.; Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN practical guideline: Clinical nutrition in cancer. Clin. Nutr. 2021, 40, 2898–2913. [Google Scholar] [CrossRef] [PubMed]
- Maiorino, L.; Daßler-Plenker, J.; Sun, L.; Egeblad, M. Innate immunity and cancer pathophysiology. Annu. Rev. Pathol. 2022, 17, 425–457. [Google Scholar] [CrossRef]
- Namikawa, T.; Shimizu, S.; Yokota, K.; Tanioka, N.; Munekage, M.; Uemura, S.; Maeda, H.; Kitagawa, H.; Kobayashi, M.; Hanazaki, K. Neutrophil-to-lymphocyte ratio and C-reactive protein-to-albumin ratio as prognostic factors for unresectable advanced or recurrent gastric cancer. Langenbecks Arch. Surg. 2022, 407, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Ikoma, T.; Matsumoto, T.; Kurioka, Y.; Takatani, M.; Nagai, H.; Matsumoto, Y.; Satake, H.; Yasui, H. Improvement of body weight and nutritional status in gastric cancer patients enhances the benefit of nivolumab therapy. J. Clin. Med. 2022, 11, 6100. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Kimura, M.; Iwai, M.; Usami, E.; Teramachi, H.; Yoshimura, T. Prognostic factors in patients with advanced and recurrent colorectal cancer receiving last-line chemotherapy. Pharmazie 2018, 73, 115–119. [Google Scholar] [CrossRef]
- Skuja, E.; Gerina-Berzina, A.; Hegmane, A.; Zvirbule, Z.; Vecvagare, E.; Purkalne, G. Duration of previous treatment as a prognostic factor in metastatic colorectal cancer treated with trifluridine/Tipiracil. Mol. Clin. Oncol. 2018, 8, 699–702. [Google Scholar] [CrossRef]
- Hamauchi, S.; Yamazaki, K.; Masuishi, T.; Kito, Y.; Komori, A.; Tsushima, T.; Narita, Y.; Todaka, A.; Ishihara, M.; Yokota, T.; et al. Neutropenia as a predictive factor in metastatic colorectal cancer treated with TAS-102. Clin. Color. Cancer 2017, 16, 51–57. [Google Scholar] [CrossRef]
- Kimura, M.; Usami, E.; Iwai, M.; Teramachi, H.; Yoshimura, T. Severe neutropenia: A prognosticator in patients with advanced/recurrent colorectal cancer under oral trifluridine-Tipiracil (TAS-102) chemotherapy. Pharmazie 2017, 72, 49–52. [Google Scholar] [CrossRef]
- Nose, Y.; Kagawa, Y.; Hata, T.; Mori, R.; Kawai, K.; Naito, A.; Sakamoto, T.; Murakami, K.; Katsura, Y.; Ohmura, Y.; et al. Neutropenia is an indicator of outcomes in metastatic colorectal cancer patients treated with FTD/TPI plus bevacizumab: A retrospective study. Cancer Chemother. Pharm. 2020, 86, 427–433. [Google Scholar] [CrossRef]
- Makihara, K.; Fukui, R.; Uchiyama, H.; Shigeoka, Y.; Toyokawa, A. Decreased percentage of neutrophil is a predict factor for the efficacy of trifluridine and Tipiracil hydrochloride for pretreated metastatic colorectal cancer. J. Gastrointest. Oncol. 2019, 10, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, A.; Yamada, T.; Matsumoto, S.; Sakurazawa, N.; Kawano, Y.; Shinozuka, E.; Sekiguchi, K.; Suzuki, H.; Yoshida, H. Pretreatment neutrophil-to-lymphocyte ratio predicts survival after TAS-102 treatment of patients with metastatic colorectal cancer. Anticancer Res. 2019, 39, 4343–4350. [Google Scholar] [CrossRef] [PubMed]
- Stavraka, C.; Pouptsis, A.; Synowiec, A.; Angelis, V.; Satterthwaite, L.; Khan, S.; Chauhan, M.; Holden, C.; Young, S.; Karampera, C.; et al. Trifluridine/Tipiracil in metastatic colorectal cancer: A UK multicenter real-world analysis on efficacy, safety, predictive and prognostic factors. Clin. Color. Cancer 2021, 20, 342–349. [Google Scholar] [CrossRef]
- Shibutani, M.; Nagahara, H.; Fukuoka, T.; Iseki, Y.; Matsutani, S.; Wang, E.N.; Maeda, K.; Hirakawa, K.; Ohira, M. Prognostic significance of the C-reactive protein-to-albumin ratio in patients with metastatic colorectal cancer treated with trifluridine/thymidine phosphorylase inhibitor as later-line chemotherapy. Anticancer Res. 2019, 39, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Bedford, M.; Begum, R.; Mitchell, H.; Hodson, J.; Whiting, J.; Griffiths, E. The value of inflammation based prognostic scores in patients undergoing surgical resection for oesophageal and gastric carcinoma. J. Surg. Oncol. 2018, 117, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Golder, A.M.; McMillan, D.C.; Park, J.H.; Mansouri, D.; Horgan, P.G.; Roxburgh, C.S. The prognostic value of combined measures of the systemic inflammatory response in patients with colon cancer: An analysis of 1700 patients. Br. J. Cancer 2021, 124, 1828–1835. [Google Scholar] [CrossRef]
- Potempa, L.A.; Rajab, I.M.; Olson, M.E.; Hart, P.C. C-reactive protein and cancer: Interpreting the differential bioactivities of Its pentameric and monomeric, modified isoforms. Front. Immunol. 2021, 12, 744129. [Google Scholar] [CrossRef] [PubMed]
- Marcason, W. Should albumin and prealbumin be used as indicators for malnutrition? J. Acad. Nutr. Diet. 2017, 117, 1144. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Oshima, Y.; Matsumoto, Y.; Seto, Y.; Yamashita, H.; Hayano, K.; Kano, M.; Ono, H.A.; Mitsumori, N.; Fujisaki, M.; et al. The new prognostic score for unresectable or recurrent gastric cancer treated with nivolumab: A multi-institutional cohort study. Ann. Gastroenterol. Surg. 2021, 5, 794–803. [Google Scholar] [CrossRef]
- Furukawa, T.; Tabata, S.; Yamamoto, M.; Kawahara, K.; Shinsato, Y.; Minami, K.; Shimokawa, M.; Akiyama, S.-I. Thymidine phosphorylase in cancer aggressiveness and chemoresistance. Pharmacol. Res. 2018, 132, 15–20. [Google Scholar] [CrossRef]
- Warfield, B.M.; Reigan, P. Multifunctional role of thymidine phosphorylase in cancer. Trends Cancer 2022, 8, 482–493. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.S.; Lee, W.S.; Lee, J.S.; Kim, H.W.; Ko, G.H.; Ha, W.S. The expression of thymidine phosphorylase in cancer-infiltrating inflammatory cells in stomach cancer. J. Korean Med. Sci. 2007, 22, S109–S114. [Google Scholar] [CrossRef]
- Huang, L.; Liu, S.; Lei, Y.; Wang, K.; Xu, M.; Chen, Y.; Liu, B.; Chen, Y.; Fu, Q.; Zhang, P.; et al. Systemic immune-inflammation index, thymidine phosphorylase and survival of localized gastric cancer patients after curative resection. Oncotarget 2016, 7, 44185–44193. [Google Scholar] [CrossRef]
- Diakos, C.I.; Charles, K.A.; McMillan, D.C.; Clarke, S.J. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014, 15, e493–e503. [Google Scholar] [CrossRef]
- Shinko, D.; Diakos, C.I.; Clarke, S.J.; Charles, K.A. Cancer-related systemic inflammation: The challenges and therapeutic opportunities for personalized medicine. Clin. Pharmacol. Ther. 2017, 102, 599–610. [Google Scholar] [CrossRef]
- Bower, J.E. Cancer-related fatigue—Mechanisms, risk factors, and treatments. Nat. Rev. Clin. Oncol. 2014, 11, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Groenvold, M.; Petersen, M.A.; Idler, E.; Bjorner, J.B.; Fayers, P.M.; Mouridsen, H.T. Psychological distress and fatigue predicted recurrence and survival in primary breast cancer patients. Breast Cancer Res. Treat. 2007, 105, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Visacri, M.B.; Duarte, N.C.; Lima, T.M.; de Souza, R.N.; Cobaxo, T.S.; Teixeira, J.C.; Barbosa, C.R.; Dias, L.P.; Tavares, M.G.; Pincinato, E.C.; et al. Adverse reactions and adherence to capecitabine: A prospective study in patients with gastrointestinal cancer. J. Oncol. Pharm. Pract. 2022, 28, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Thong, M.S.Y.; van Noorden, C.J.F.; Steindorf, K.; Arndt, V. Cancer-related fatigue: Causes and current treatment options. Curr. Treat. Options Oncol. 2020, 21, 17. [Google Scholar] [CrossRef]
- Alexander, S.; Minton, O.; Andrews, P.; Stone, P. A comparison of the characteristics of disease-free breast cancer survivors with or without cancer-related fatigue syndrome. Eur. J. Cancer 2009, 45, 384–392. [Google Scholar] [CrossRef]
- Bower, J.E.; Ganz, P.A.; Tao, M.L.; Hu, W.; Belin, T.R.; Sepah, S.; Cole, S.; Aziz, N. Inflammatory biomarkers and fatigue during radiation therapy for breast and prostate cancer. Clin. Cancer Res. 2009, 15, 5534–5540. [Google Scholar] [CrossRef] [PubMed]
- Pertl, M.M.; Hevey, D.; Boyle, N.T.; Hughes, M.M.; Collier, S.; O’Dwyer, A.M.; Harkin, A.; Kennedy, M.J.; Connor, T.J. C-reactive protein predicts fatigue independently of depression in breast cancer patients prior to chemotherapy. Brain Behav. Immun. 2013, 34, 108–119. [Google Scholar] [CrossRef]
| Variables | All Patients (n = 64) | CAR | p-Value | |
|---|---|---|---|---|
| Low (n = 27) | High (n = 37) | |||
| Age | <65 | 10 (37.0) | 16 (43.2) | 0.797 |
| ≧65 | 17 (63.0) | 21 (56.8) | ||
| Sex | Male | 11 (40.7) | 12 (32.4) | 0.600 |
| Female | 16 (59.3) | 25 (67.6) | ||
| ECOG PS | 0 | 20 (74.1) | 15 (40.5) | 0.022 |
| 1 | 6 (22.2) | 19 (51.4) | ||
| 2 | 1 (3.7) | 3 (8.1) | ||
| Histological type | Intestinal | 12 (44.4) | 19 (51.4) | 0.621 |
| Diffuse | 15 (55.6) | 18 (48.6) | ||
| HER2 status | Negative | 24 (88.9) | 33 (89.2) | 1.000 |
| Positive | 3 (11.1) | 4 (10.8) | ||
| Macroscopic classification | Non-type 4 | 25 (92.6) | 27 (73.0) | 0.058 |
| Type 4 | 2 (7.4) | 10 (27.0) | ||
| Primary tumor site | GC | 21 (77.8) | 29 (78.4) | 1.000 |
| EGJC | 6 (22.2) | 8 (21.6) | ||
| Disease status | Unresectable | 12 (44.4) | 25 (67.6) | 0.078 |
| Recurrence | 15 (55.6) | 12 (32.4) | ||
| Peritoneal metastasis | − | 16 (59.3) | 14 (37.8) | 0.129 |
| + | 11 (40.7) | 23 (62.2) | ||
| Hematogenous metastasis | − | 12 (44.4) | 20 (54.1) | 0.613 |
| + | 15 (55.6) | 17 (45.9) | ||
| Lymphatic metastasis | − | 15 (55.6) | 22 (59.5) | 0.802 |
| + | 12 (44.4) | 15 (40.5) | ||
| Number of metastatic sites | 1 | 16 (59.3) | 18 (48.6) | 0.454 |
| ≧2 | 11 (40.7) | 19 (51.4) | ||
| Number of previous regimens | 2 | 20 (74.1) | 30 (81.1) | 0.551 |
| ≧3 | 7 (25.9) | 7 (18.9) | ||
| Previous ICI administered | − | 10 (37.0) | 14 (37.8) | 1.000 |
| + | 17 (63.0) | 23 (62.2) | ||
| Number of FTD/TPI | 1 | 5 (18.5) | 18 (48.6) | 0.018 |
| ≧2 | 22 (81.5) | 19 (51.4) | ||
| Post-FTD/TPI therapy | − | 12 (44.4) | 30 (81.1) | 0.004 |
| + | 13 (48.1) | 7 (18.9) | ||
| FTD/TPI ongoing | 2 (7.4) | 0 (0.0) | ||
| Factors | Univariate | p-Value | Multivariate | p-Value | |||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | HR | 95% CI | ||||
| Age | <65 | 1 | |||||
| ≧65 | 0.74 | 0.41–1.32 | 0.30 | ||||
| Sex | Male | 1 | |||||
| Female | 0.62 | 0.34–1.13 | 0.12 | ||||
| ECOG PS | 0 | 1 | 1 | ||||
| 1, 2 | 1.83 | 1.02–3.29 | 0.04 | 1.32 | 0.72–2.43 | 0.37 | |
| HER2 status | Negative | 1 | |||||
| Positive | 0.76 | 0.27–2.13 | 0.60 | ||||
| Disease status | Recurrence | 1 | |||||
| Unresectable | 0.67 | 0.37–1.23 | 0.20 | ||||
| Peritoneal metastasis | - | 1 | 1 | ||||
| + | 2.57 | 1.37–4.82 | 0.003 | 1.51 | 0.76–2.98 | 0.24 | |
| No. of previous regimens | 3 | 1 | |||||
| ≧4 | 0.90 | 0.43–1.87 | 0.77 | ||||
| CAR | Low | 1 | 1 | ||||
| High | 4.48 | 2.24–8.97 | <0.001 | 3.56 | 1.67–7.55 | <0.001 | |
| Factors | Univariate | p-Value | Multivariate | p-Value | |||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | HR | 95% CI | ||||
| Age | <65 | 1 | |||||
| ≧65 | 0.68 | 0.40–1.16 | 0.16 | ||||
| Sex | Male | 1 | |||||
| Female | 0.66 | 0.39–1.13 | 0.13 | ||||
| ECOG PS | 0 | 1 | |||||
| 1, 2 | 1.56 | 0.92–2.65 | 0.10 | ||||
| HER2 status | Negative | 1 | |||||
| Positive | 1.37 | 0.61–3.07 | 0.44 | ||||
| Disease status | Recurrence | 1 | |||||
| Unresectable | 0.78 | 0.45–1.33 | 0.36 | ||||
| Peritoneal metastasis | - | 1 | 1 | ||||
| + | 1.78 | 1.04–3.05 | 0.04 | 1.28 | 0.72–2.28 | 0.41 | |
| No. of previous regimens | 3 | 1 | |||||
| ≧4 | 0.80 | 0.42–1.51 | 0.49 | ||||
| CAR | Low | 1 | 1 | ||||
| High | 2.67 | 1.53–4.66 | <0.001 | 2.42 | 1.33–4.41 | 0.004 | |
| Tumor Response Data | Low-CAR (n = 27) | High-CAR (n = 37) | p-Value |
|---|---|---|---|
| Best response | |||
| Complete response (CR) | 2 (7.4) | 0 (0.0) | 0.18 |
| Partial response (PR) | 4 (14.8) | 2 (5.4) | |
| Stable disease (SD) | 8 (29.6) | 8 (21.6) | |
| Progressive disease (PD) | 12 (44.4) | 25 (67.6) | |
| Not evaluated (NE) | 1 (3.7) | 2 (5.4) | |
| Overall response rate (ORR) | 6 (22.2) | 2 (5.4) | 0.07 |
| Disease control rate (DCR) | 14 (51.9) | 10 (27.0) | 0.06 |
| Low-CAR (n = 27) | High-CAR (n = 37) | p-Value | |
|---|---|---|---|
| Total adverse events | 22 (81.5) | 29 (78.4) | 1.000 |
| Anorexia | 9 (33.3) | 17 (45.9) | 0.440 |
| Decreased neutrophil count | 14 (51.9) | 7 (18.9) | 0.008 |
| Fatigue | 0 (0.0) | 9 (24.3) | 0.008 |
| Diarrhea | 1 (3.7) | 3 (8.1) | 0.632 |
| Decreased platelet count | 1 (3.7) | 1 (2.7) | 1.000 |
| Anemia | 0 (0.0) | 1 (2.7) | 1.000 |
| Biliary tract infection | 0 (0.0) | 1 (2.7) | 1.000 |
| Febrile neutropenia | 1 (3.7) | 0 (0.0) | 0.422 |
| Hyponatremia | 1 (3.7) | 0 (0.0) | 0.422 |
| Myalgia | 0 (0.0) | 1 (2.7) | 1.000 |
| Peripheral sensory neuropathy | 0 (0.0) | 1 (2.7) | 1.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hashimoto, I.; Kano, K.; Onuma, S.; Suematsu, H.; Nagasawa, S.; Kanematsu, K.; Furusawa, K.; Hamaguchi, T.; Watanabe, M.; Hayashi, K.; et al. Clinical Effect of the C-Reactive Protein to Serum Albumin Ratio in Patients with Metastatic Gastric or Gastroesophageal Junction Cancer Treated with Trifluridine/Tipiracil. J. Pers. Med. 2023, 13, 923. https://doi.org/10.3390/jpm13060923
Hashimoto I, Kano K, Onuma S, Suematsu H, Nagasawa S, Kanematsu K, Furusawa K, Hamaguchi T, Watanabe M, Hayashi K, et al. Clinical Effect of the C-Reactive Protein to Serum Albumin Ratio in Patients with Metastatic Gastric or Gastroesophageal Junction Cancer Treated with Trifluridine/Tipiracil. Journal of Personalized Medicine. 2023; 13(6):923. https://doi.org/10.3390/jpm13060923
Chicago/Turabian StyleHashimoto, Itaru, Kazuki Kano, Shizune Onuma, Hideaki Suematsu, Shinsuke Nagasawa, Kyohei Kanematsu, Kyoko Furusawa, Tomomi Hamaguchi, Mamoru Watanabe, Kei Hayashi, and et al. 2023. "Clinical Effect of the C-Reactive Protein to Serum Albumin Ratio in Patients with Metastatic Gastric or Gastroesophageal Junction Cancer Treated with Trifluridine/Tipiracil" Journal of Personalized Medicine 13, no. 6: 923. https://doi.org/10.3390/jpm13060923
APA StyleHashimoto, I., Kano, K., Onuma, S., Suematsu, H., Nagasawa, S., Kanematsu, K., Furusawa, K., Hamaguchi, T., Watanabe, M., Hayashi, K., Furuta, M., Inokuchi, Y., Machida, N., Aoyama, T., Yamada, T., Rino, Y., Ogata, T., & Oshima, T. (2023). Clinical Effect of the C-Reactive Protein to Serum Albumin Ratio in Patients with Metastatic Gastric or Gastroesophageal Junction Cancer Treated with Trifluridine/Tipiracil. Journal of Personalized Medicine, 13(6), 923. https://doi.org/10.3390/jpm13060923






