A Promising Biomarker and Therapeutic Target in Patients with Advanced PDAC: The Stromal Protein βig-h3
Abstract
:1. Introduction
2. Material and Methods
2.1. Patients and Data Collection
2.2. Evaluation Criteria
2.3. Statistical Methods
2.4. Biological Analysis Methods
3. Results
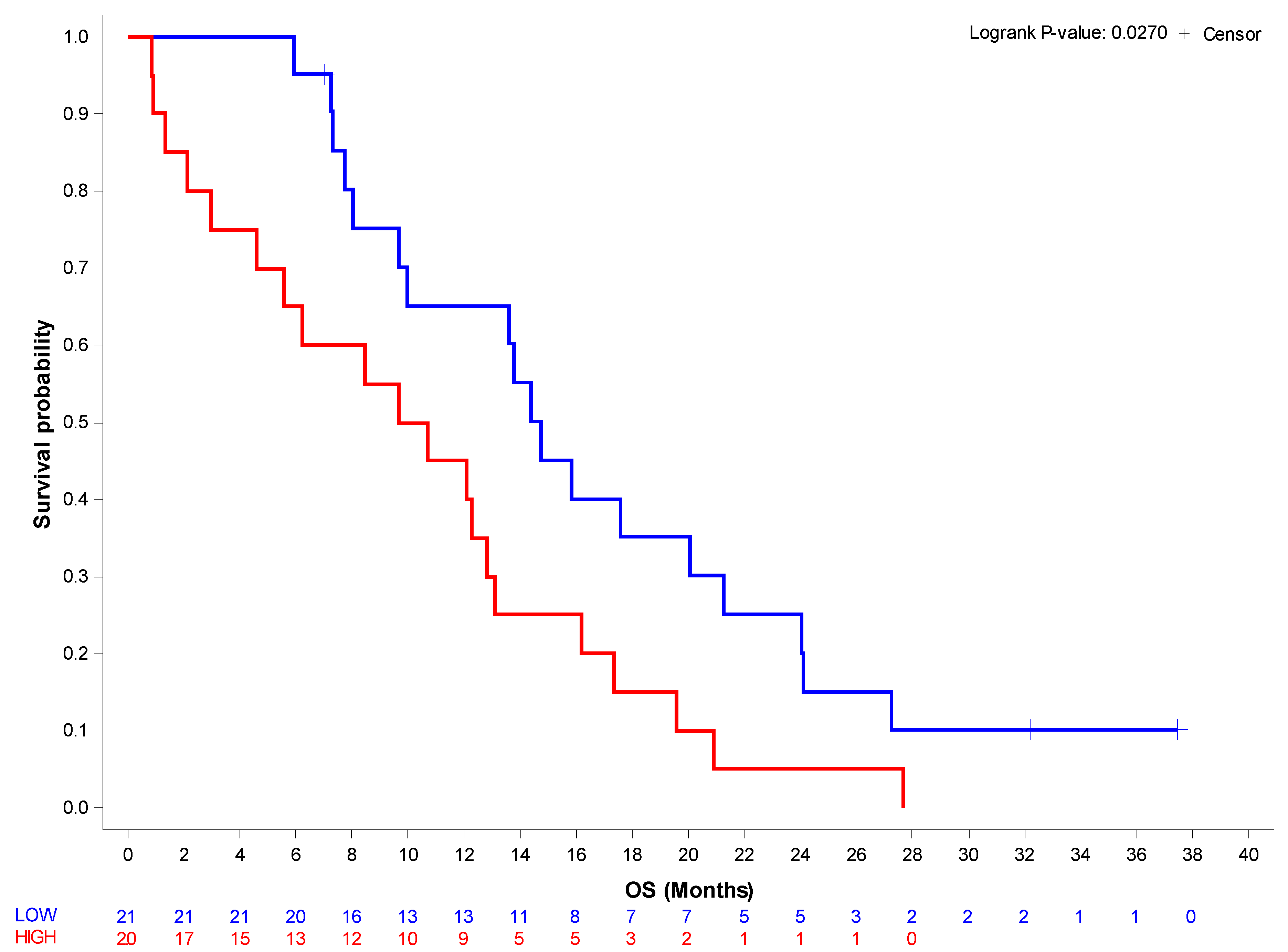
3.1. βig-h3 Serum Levels Significantly Correlate with Overall Survival in Stage IV PDAC
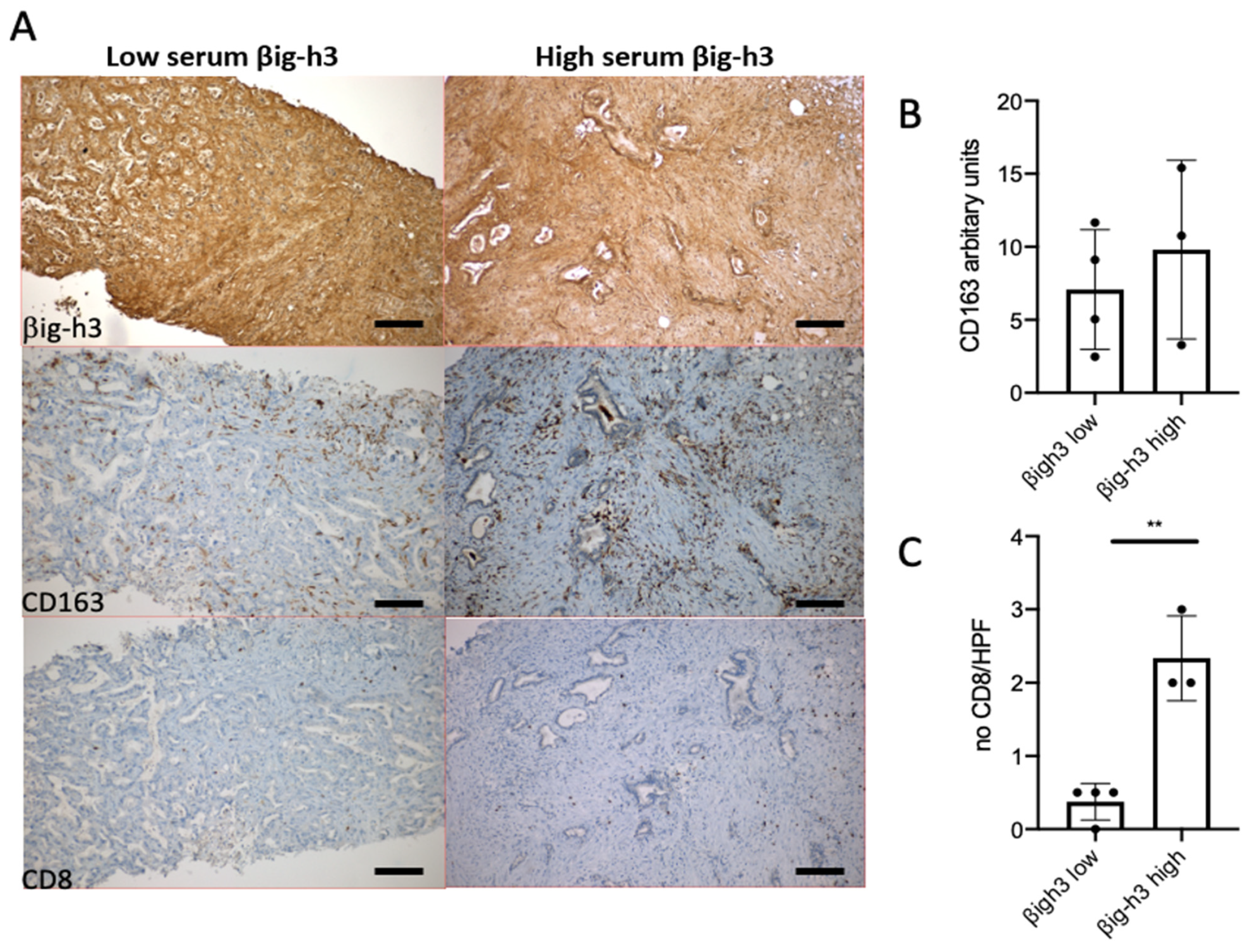
3.2. βig-h3, CD8, and CD163 Staining in Tumor Biopsies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khalaf, N.; El-Serag, H.B.; Abrams, H.R.; Thrift, A.P. Burden of Pancreatic Cancer: From Epidemiology to Practice. Clin. Gastroenterol. Hepatol. 2021, 19, 876–884. [Google Scholar] [CrossRef]
- Mizrahi, J.D.; Surana, R.; Valle, J.W.; Shroff, R.T. Pancreatic Cancer. Lancet 2020, 395, 2008–2020. [Google Scholar] [CrossRef]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.-L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef] [Green Version]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased Survival in Pancreatic Cancer with Nab-Paclitaxel plus Gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Golan, T.; Hammel, P.; Reni, M.; Van Cutsem, E.; Macarulla, T.; Hall, M.J.; Park, J.-O.; Hochhauser, D.; Arnold, D.; Oh, D.-Y.; et al. Maintenance Olaparib for Germline BRCA-Mutated Metastatic Pancreatic Cancer. N. Engl. J. Med. 2019, 381, 317–327. [Google Scholar] [CrossRef]
- Marabelle, A.; Le, D.T.; Ascierto, P.A.; Di Giacomo, A.M.; De Jesus-Acosta, A.; Delord, J.-P.; Geva, R.; Gottfried, M.; Penel, N.; Hansen, A.R.; et al. Efficacy of Pembrolizumab in Patients With Noncolorectal High Microsatellite Instability/Mismatch Repair-Deficient Cancer: Results from the Phase II KEYNOTE-158 Study. J. Clin. Oncol. 2019, 38, JCO1902105. [Google Scholar] [CrossRef] [PubMed]
- Marabelle, A.; Fakih, M.; Lopez, J.; Shah, M.; Shapira-Frommer, R.; Nakagawa, K.; Chung, H.C.; Kindler, H.L.; Lopez-Martin, J.A.; Miller, W.H.; et al. Association of Tumour Mutational Burden with Outcomes in Patients with Advanced Solid Tumours Treated with Pembrolizumab: Prospective Biomarker Analysis of the Multicohort, Open-Label, Phase 2 KEYNOTE-158 Study. Lancet Oncol. 2020, 21, 1353–1365. [Google Scholar] [CrossRef]
- Gamradt, P.; De La Fouchardière, C.; Hennino, A. Stromal Protein-Mediated Immune Regulation in Digestive Cancers. Cancers 2021, 13, 146. [Google Scholar] [CrossRef] [PubMed]
- Patry, M.; Teinturier, R.; Goehrig, D.; Zetu, C.; Ripoche, D.; Kim, I.-S.; Bertolino, P.; Hennino, A. Βig-H3 Represses T-Cell Activation in Type 1 Diabetes. Diabetes 2015, 64, 4212–4219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goehrig, D.; Nigri, J.; Samain, R.; Wu, Z.; Cappello, P.; Gabiane, G.; Zhang, X.; Zhao, Y.; Kim, I.-S.; Chanal, M.; et al. Stromal Protein Βig-H3 Reprogrammes Tumour Microenvironment in Pancreatic Cancer. Gut 2019, 68, 693–707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lecker, L.S.M.; Berlato, C.; Maniati, E.; Delaine-Smith, R.; Pearce, O.M.T.; Heath, O.; Nichols, S.J.; Trevisan, C.; Novak, M.; McDermott, J.; et al. TGFBI Production by Macrophages Contributes to an Immunosuppressive Microenvironment in Ovarian Cancer. Cancer Res. 2021, 81, 5706–5719. [Google Scholar] [CrossRef] [PubMed]
- Bachy, S.; Wu, Z.; Gamradt, P.; Thierry, K.; Milani, P.; Chlasta, J.; Hennino, A. Βig-H3-Structured Collagen Alters Macrophage Phenotype and Function in Pancreatic Cancer. iScience 2022, 25, 103758. [Google Scholar] [CrossRef]
- Palmquist, C.; Dehlendorff, C.; Calatayud, D.; Hansen, C.P.; Hasselby, J.P.; Johansen, J.S. Prediction of Unresectability and Prognosis in Patients Undergoing Surgery on Suspicion of Pancreatic Cancer Using Carbohydrate Antigen 19-9, Interleukin 6, and YKL-40. Pancreas 2020, 49, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Giannis, D.; Moris, D.; Barbas, A.S. Diagnostic, Predictive and Prognostic Molecular Biomarkers in Pancreatic Cancer: An Overview for Clinicians. Cancers 2021, 13, 1071. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chen, P.; Dai, F.; Wang, J. Up-regulation of TGFBI and TGFB2 in the plasma of gestational diabetes mellitus patients and its clinical significance. Ir. J. Med. Sci. 2021. [Google Scholar] [CrossRef] [PubMed]
- Helms, E.; Onate, M.K.; Sherman, M.H. Fibroblast Heterogeneity in the Pancreatic Tumor Microenvironment. Cancer Discov. 2020, 10, 648–656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bauer, C.A. Immunosuppressive Βig-H3 Links Tumour Stroma and Dysfunctional T Cells in Pancreatic Cancer. Gut 2019, 68, 581. [Google Scholar] [CrossRef] [PubMed]
| All n = 41 | βig-h3 | Test | ||
|---|---|---|---|---|
| Low (n = 21) | High (n = 20) | |||
| Age | Wilcoxon | |||
| Median (min; max) | 65.0 (41; 78) | 61.0 (41; 76) | 67.0 (45; 78) | p = 0.196 |
| Gender | Fisher Exact p = 0.043 | |||
| Female | 13 (31.7%) 28 (68.3%) | 10 (47.6%) | 3 (15.0%) | |
| Male | 11 (52.4%) | 17 (85.0%) | ||
| Performance Status (ECOG) | Fisher Exact p = 1.000 | |||
| 0 | 3 (7.3%) | 1 (4.8%) | 2 (10.0%) | |
| 1 | 33 (80.5) | 17 (81.0%) | 16 (80.0%) | |
| 2 | 5 (12.2%) | 3 (14.3%) | 2 (10.0%) | |
| Tobacco | Fisher Exact p = 0.758 | |||
| Never | 22 (53.7%) 19 (46.3%) | 12 (57.1%) | 10 (50.0%) | |
| Current/former | 9 (42.9%) | 10 (50.0%) | ||
| Diabetes | Fisher Exact p = 0.697 | |||
| No | 34 (82.9%) 7 (17.1%) | 18 (85.7%) | 16 (80.0%) | |
| Yes | 3 (14.3%) | 4 (20.0%) | ||
| Primary tumor localization | Fisher Exact p = 0.111 | |||
| Head | 14 (34.1%) | 7 (33.3%) | 7 (35.0%) | |
| Body | 15 (36.6%) | 5 (23.8%) | 10 (50.0%) | |
| Tail | 12 (29.3%) | 9 (42.9%) | 3 (15.0%) | |
| Metastatic site (at diagnosis) | Fisher Exact p = 0.484 | |||
| Liver | ||||
| No | 11 (26.8%) | 7 (33.3%) | 4 (20.0%) | |
| Yes | 30 (73.2%) | 14 (66.7%) | 16 (80.0%) | |
| Lung | Fisher Exact p = 1.000 | |||
| No | 34 (82.9%) 7 (17.1%) | 17 (81.0%) | 17 (85.0%) | |
| Yes | 4 (19.0%) | 3 (15.0%) | ||
| Peritoneum | Fisher Exact p = 0.277 | |||
| No | 31 (75.6%) 10 (24.4) | 14 (66.7%) | 17 (85.0%) | |
| Yes | 7 (33.3%) | 3 (15.0%) | ||
| Lymph nodes | Fisher Exact p = 0.208 | |||
| No | 25 (61.0%) 16 (39.0%) | 15 (71,4%) | 10 (50%) | |
| Yes | 6 (28.6%) | 10 (50%) | ||
| Bone | Fisher Exact p = 1.000 | |||
| No | 37 (90.2%) 4 (9.8%) | 19 (90.5%) | 18 (90.0%) | |
| Yes | 2 (9.5%) | 2 (10.0%) | ||
| Number of metastatic sites | Wilcoxon p = 0.316 | |||
| 1 | 20 (48.8%) | 13 (61.9%) | 7 (35.0%) | |
| 2 | 12 (29.3%) | 3 (14.3%) | 9 (45.0%) | |
| 3 | 7 (17.1%) | 3 (14.3%) | 4 (20.0%) | |
| 4 | 2 (4.9%) | 2 (9.5%) | 0 (0.0%) | |
| Median (min; max) | 2.0 (1; 4) | 1.0 (1; 4) | 2.0 (1; 3) | |
| Differentiation grade | Fisher Exact p = 0.060 | |||
| Unknown | 2 | 0 | 2 | |
| Low | 8 (20.5%) | 7 (33.3%) | 1 (5.6%) | |
| Intermediate | 24 (61.5%) | 12 (57.1%) | 12 (66.7%) | |
| High | 7 (17.9%) | 2 (9.5%) | 5 (27.8%) | |
| Ca 19-9 at diagnosis (UI/l) | Wilcoxon p = 0.735 | |||
| Median (min; max) | 2497.0 | 2497.0 | 2027.0 | |
| (9; 128600) | (10; 128600) | (9; 84720) | ||
| CEA at diagnosis (ng/mL) | Wilcoxon p = 0.666 | |||
| Median (min; max) | 8.0 (2; 810) | 8.0 (2; 258) | 8.5 (2; 810) | |
| Chemotherapy lines number | Fisher Exact p = 0.396 | |||
| 1 | 8 (20.0%) | 2 (9.5%) | 6 (31.6%) | |
| 2 | 15 (37.5%) | 8 (38.1%) | 7 (36.8%) | |
| 3 | 14 (35.0%) | 9 (42.9%) | 5 (26.3%) | |
| 4 | 3 (7.5%) | 2 (9.5%) | 1 (5.3%) | |
| Chemotherapy regimen (L1) | Fisher Exact p = 1.000 | |||
| FOLFIRINOX | 29 (72.5%) | 15 (71.4%) | 14 (73.7%) | |
| Gemcitabine/nab-paclitaxel | 1 (2.5%) | 1 (4.8%) | 0 (0.0%) | |
| Gemcitabine | 3 (7.5%) | 2 (9.5%) | 1 (5.3%) | |
| Other | 7 (17.5%) | 3 (14.3%) | 4 (21.1%) | |
| Event/Total | Median (95% CI) KM | Hazard Ratio (95% CI) Cox | Survival Estimates (95% CI) KM | p-Value | |
|---|---|---|---|---|---|
| βig-h3_cut-off | 0.0270 * | ||||
| Low | 18/21 | 14.8 (9.7–21.3) | Reference | 6 months: 0.95 (0.71–0.99) 12 months: 0.65 (0.40–0.82) 24 months: 0.25 (0.09–0.45) | |
| High | 20/20 | 10.2 (3.0–13.1) | 2.05 (1.07–3.93) | 6 months: 0.65 (0.40–0.82) 12 months: 0.45 (0.23–0.65) 24 months: 0.05 (0.00–0.21) |
| Univariate Cox Model | Multivariate Cox Model | ||||||
|---|---|---|---|---|---|---|---|
| HR | CI95% | p Value | HR | CI95% | p Value | ||
| βig-h3 (cut-off median) | Low | 0.0301 | 0.0156 | ||||
| High | 2.053 | [1.07–3.93] | 2.332 | [1.174–4.633] | |||
| ECOG-PS | 0–1 | 0.1591 | 0.0413 | ||||
| 2,3,4 | 1.997 | [0.76–5.23] | 2.964 | [1.044–8.418] | |||
| Age | <65 | 0.6911 | |||||
| ≥65 | 1.139 | [0.60–2.16] | |||||
| Liver metastases | No | 0.0561 | ns | ||||
| Yes | 2.093 | [0.98–4.46] | |||||
| CA19-9 (cut-off median) | Low | 0.3118 | |||||
| High | 1.397 | [0.73–2.67] | |||||
| Neutrophil-to-lymphocyte ratio | ≤5 | 0.0016 | 0.0024 | ||||
| >5 | 5.068 | [1.85–13.9] | 4.962 | [1.764–13.964] | |||
| Gender | F | 0.0593 | ns | ||||
| M | 2.038 | [0.97–4.27] | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de la Fouchardière, C.; Gamradt, P.; Chabaud, S.; Raddaz, M.; Blanc, E.; Msika, O.; Treilleux, I.; Bachy, S.; Cattey-Javouhey, A.; Guibert, P.; et al. A Promising Biomarker and Therapeutic Target in Patients with Advanced PDAC: The Stromal Protein βig-h3. J. Pers. Med. 2022, 12, 623. https://doi.org/10.3390/jpm12040623
de la Fouchardière C, Gamradt P, Chabaud S, Raddaz M, Blanc E, Msika O, Treilleux I, Bachy S, Cattey-Javouhey A, Guibert P, et al. A Promising Biomarker and Therapeutic Target in Patients with Advanced PDAC: The Stromal Protein βig-h3. Journal of Personalized Medicine. 2022; 12(4):623. https://doi.org/10.3390/jpm12040623
Chicago/Turabian Stylede la Fouchardière, Christelle, Pia Gamradt, Sylvie Chabaud, Maxime Raddaz, Ellen Blanc, Olivier Msika, Isabelle Treilleux, Sophie Bachy, Anne Cattey-Javouhey, Pierre Guibert, and et al. 2022. "A Promising Biomarker and Therapeutic Target in Patients with Advanced PDAC: The Stromal Protein βig-h3" Journal of Personalized Medicine 12, no. 4: 623. https://doi.org/10.3390/jpm12040623
APA Stylede la Fouchardière, C., Gamradt, P., Chabaud, S., Raddaz, M., Blanc, E., Msika, O., Treilleux, I., Bachy, S., Cattey-Javouhey, A., Guibert, P., Sarabi, M., Rochefort, P., Funk-Debleds, P., Coutzac, C., Ray-Coquard, I., Peyrat, P., Meeus, P., Rivoire, M., Dupré, A., & Hennino, A. (2022). A Promising Biomarker and Therapeutic Target in Patients with Advanced PDAC: The Stromal Protein βig-h3. Journal of Personalized Medicine, 12(4), 623. https://doi.org/10.3390/jpm12040623









