Risk of Burning Mouth Syndrome in Patients with Migraine: A Nationwide Cohort Study
Abstract
:1. Introduction
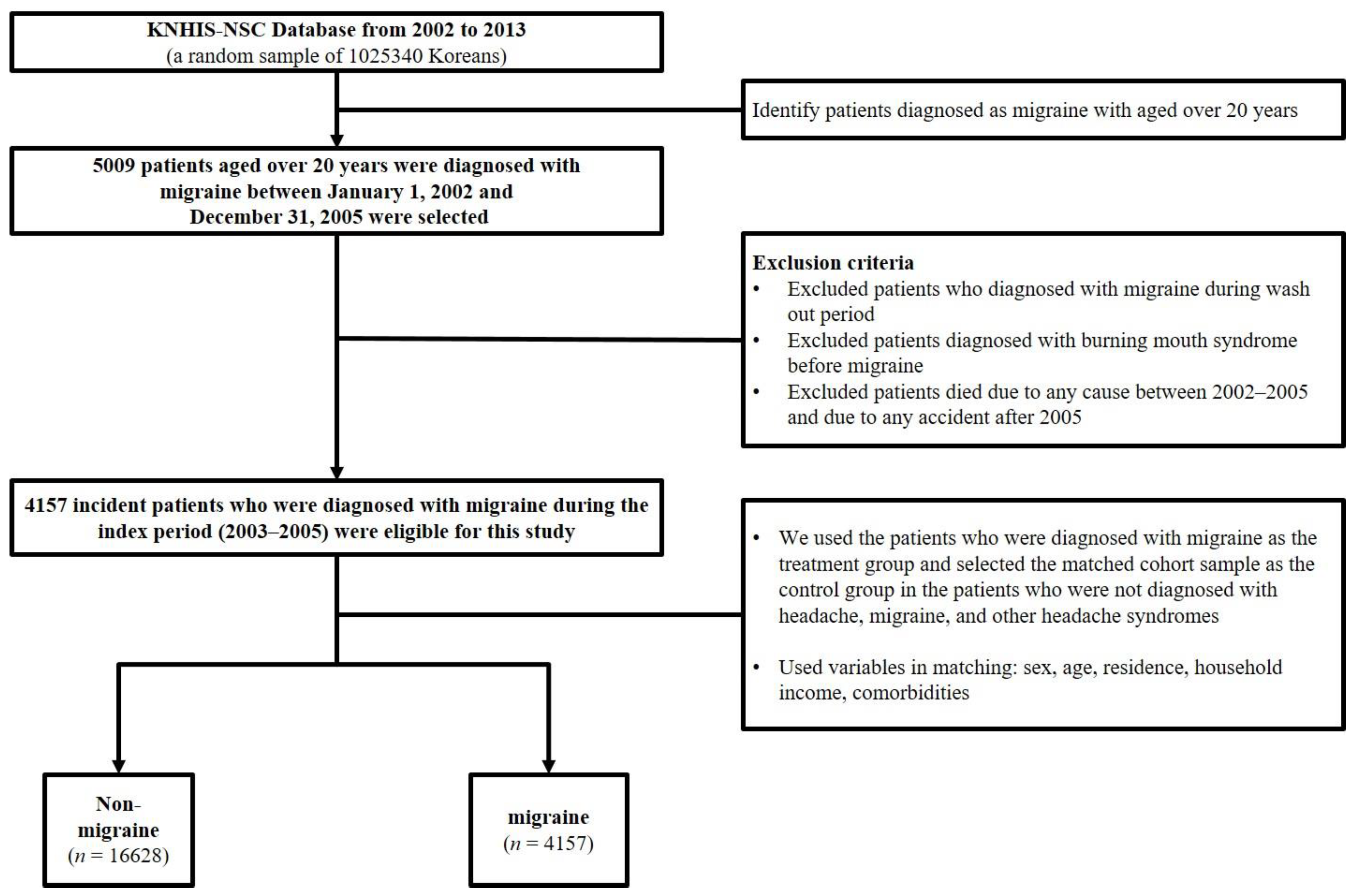
2. Materials and Methods
2.1. Study Population
2.2. Outcome Variables
2.3. Statistical Analyses
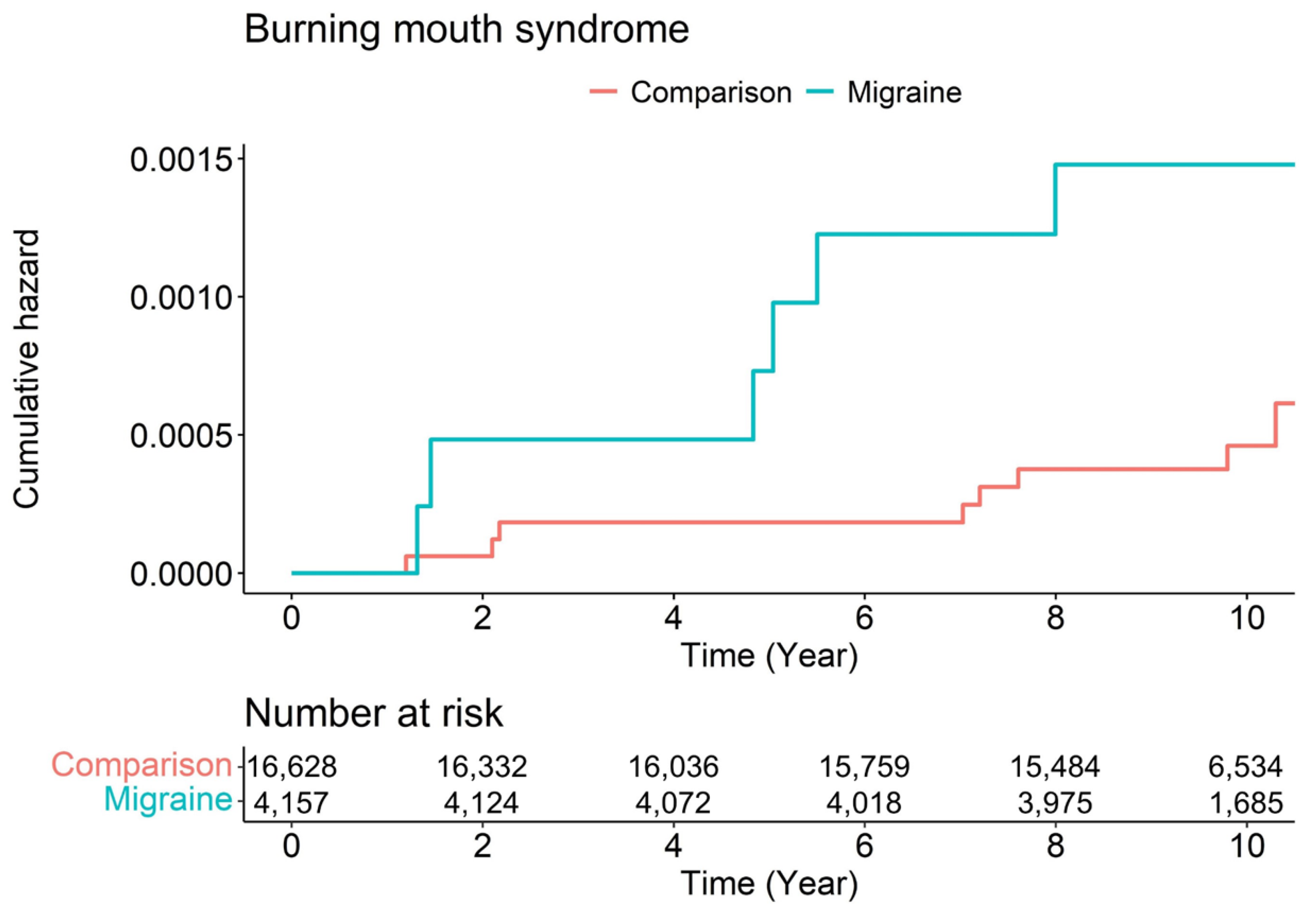
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| The Number of Burning Mouth Syndrome Event | |
|---|---|
| Event | 14 |
| Comparison | 8 |
| Migraine | 6 |
| Total censored (No event) | 20,771 |
| Comparison | 16,620 |
| Migraine | 4151 |
| Termination of study | 19,111 |
| Comparison | 15,185 |
| Migraine | 3926 |
| Loss to follow up/Drop-out | 1660 |
| Comparison | 1435 |
| Migraine | 225 |
| Sex | Male | Female | ||
|---|---|---|---|---|
| Comparison | Migraine | Comparison | Migraine | |
| Burning mouth syndrome | ||||
| Unadjusted HR (95% CI) | 1.00 (ref) | 3.98 (0.25–63.61) | 1.00 (ref) | 2.97 (0.94–9.38) |
| Adjusted HR (95% CI) | 1.00 (ref) | 3.70 (0.23–59.89) | 1.00 (ref) | 2.87 (0.91–9.07) |
| Ages | <45 | 45–64 | >64 | |||
|---|---|---|---|---|---|---|
| Comparison | Migraine | Comparison | Migraine | Comparison | Migraine | |
| Burning mouth syndrome | ||||||
| Unadjusted HR (95% CI) | 1.00 (ref) | 0.00 (0-Inf) | 1.00 (ref) | 3.13 (0.70–14.03) | 1.00 (ref) | 3.80 (0.77–18.86) |
| Adjusted HR (95% CI) | 1.00 (ref) | 0.00 (0-Inf) | 1.00 (ref) | 3.06 (0.68–13.71) | 1.00 (ref) | 3.92 (0.79–19.54) |
| CCI | 0 | 1 | ≥2 | |||
|---|---|---|---|---|---|---|
| Comparison | Migraine | Comparison | Migraine | Comparison | Migraine | |
| Unadjusted HR (95% CI) | 1.00 (ref) | 3.97 (0.56–28.19) | 1.00 (ref) | 1.05 (0.17–9.38) | 1.00 (ref) | 6.11 (1.02–36.76) |
| Adjusted HR (95% CI) | 1.00 (ref) | 4.00 (0.56–28.37) | 1.00 (ref) | 1.02 (0.11–9.17) | 1.00 (ref) | 5.76 (0.96–34.70) |
References
- Stovner, L.J.; Hagen, K.; Jensen, R.; Katsarava, Z.; Lipton, R.; Scher, A.; Steiner, T.; Zwart, J.A. The Global Burden of Headache: A Documentation of Headache Prevalence and Disability Worldwide. Cephalalgia 2007, 27, 193–210. [Google Scholar] [CrossRef] [PubMed]
- Lipton, R.B.; Schwedt, T.J.; Friedman, B.W. GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, Regional, and National Incidence, Prevalence, and Years Lived with Disability for 310 Diseases and Injuries, 1990–2015: A Systematic Analysis for the Global Prevalence Burden of Disease Study 2015. Lancet 2015, 388, 1545–1602. [Google Scholar]
- GBD, Disease and Injury Incidence and Prevalence Collaborators. Global, Regional, and National Incidence, Prevalence, and Years Lived with Disability for 328 Diseases and Injuries for 195 Countries, 1990–2016: A Systematic Analysis for the Global Burden of Disease Study 2016. Lancet 2016, 390, 1211–1259. [Google Scholar]
- Tajti, J.; Szok, D.; Majláth, Z.; Tuka, B.; Csáti, A.; Vécsei, L. Migraine and Neuropeptides. Neuropeptides 2015, 52, 19–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nassini, R.; Materazzi, S.; Benemei, S.; Geppetti, P. The TRPA1 Channel in Inflammatory and Neuropathic Pain and Migraine. In Reviews of Physiology, Biochemistry and Pharmacology; Springer: Berlin/Heidelberg, Germany, 2014; Volume 167, pp. 1–43. [Google Scholar] [CrossRef]
- Charles, A. The Pathophysiology of Migraine: Implications for Clinical Management. Lancet Neurol. 2018, 17, 174–182. [Google Scholar] [CrossRef]
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd Edition. Cephalalgia 2018, 38, 1–211. [Google Scholar] [CrossRef] [PubMed]
- Momin, S. Burning Mouth Syndrome-A Frustrating Problem. JAMA Otolaryngol. Head Neck Surg. 2021, 147, 580. [Google Scholar] [CrossRef]
- Silvestre, F.J.; Silvestre-Rangil, J.; López-Jornet, P. Burning Mouth Syndrome: A Review and Update. Rev. Neurol. 2015, 60, 457–463. [Google Scholar]
- Galli, F.; Pravettoni, G. Burning Mouth Syndrome-Opening the Door to a Psychosomatic Approach in the Era of Patient-Centered Medicine. JAMA Otolaryngol. Head Neck Surg. 2020, 146, 569–570. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, Y.S.; Ko, I.; Kim, D.K. Association Between Burning Mouth Syndrome and the Development of Depression, Anxiety, Dementia, and Parkinson Disease. JAMA Otolaryngol. Head Neck Surg. 2020, 146, 561–569. [Google Scholar] [CrossRef]
- Yoo, H.S.; Jin, S.H.; Lee, Y.J.; Song, C.M.; Ji, Y.B.; Tae, K. The Role of Psychological Factors in the Development of Burning Mouth Syndrome. Int. J. Oral Maxillofac. Surg. 2018, 47, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Lauria, G.; Majorana, A.; Borgna, M.; Lombardi, R.; Penza, P.; Padovani, A.; Sapelli, P. Trigeminal Small-Fiber Sensory Neuropathy Causes Burning Mouth Syndrome. Pain 2005, 115, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Carreño-Hernández, I.; Cassol-Spanemberg, J.; Rodríguez de Rivera-Campillo, E.; Estrugo-Devesa, A.; López-López, J. Is Burning Mouth Syndrome a Neuropathic Pain Disorder? A Systematic Review. J. Oral Facial Pain Headache 2021, 35, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Croft, P.; Blyth, F.M.; van der Windt, D. (Eds.) The global occurrence of chronic pain: An introduction. In Chronic Pain Epidemiology: From Aetiology to Public Health; Oxford University Press: Oxford, UK, 2010; pp. 9–18. [Google Scholar]
- Crocetto, F.; Coppola, N.; Barone, B.; Leuci, S.; Imbimbo, C.; Mignogna, M.D. The association between burning mouth syndrome and urologic chronic pelvic pain syndrome: A case-control study. J. Oral Pathol. Med. 2020, 49, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Rabenstein, A.; Catarino, C.B.; Rampeltshammer, V.; Schindler, D.; Gallenmüller, C.; Priglinger, C.; Pogarell, O.; Rüther, T.; Klopstock, T. Smoking and Alcohol, Health-Related Quality of Life and Psychiatric Comorbidities in Leber’s Hereditary Optic Neuropathy Mutation Carriers: A Prospective Cohort Study. Orphanet J. Rare Dis. 2021, 16, 127. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Dong, Y.; Jin, X.; Zhang, C.; Zhang, T.; Zhao, J.; Shi, J.; Li, J. The Novel and Potent Anti-Depressive Action of Triptolide and Its Influences on Hippocampal Neuroinflammation in a Rat Model of Depression Comorbidity of Chronic Pain. Brain Behav. Immun. 2017, 64, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.K.; Kavelaars, A.; Heijnen, C.J.; Dantzer, R. Neuroinflammation and Comorbidity of Pain and Depression. Pharmacol. Rev. 2014, 66, 80–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peres, M.F.P.; Mercante, J.P.P.; Tobo, P.R.; Kamei, H.; Bigal, M.E. Anxiety and Depression Symptoms and Migraine: A Symptom-Based Approach Research. J. Headache Pain 2017, 18, 37. [Google Scholar] [CrossRef] [Green Version]
- Louter, M.A.; Pijpers, J.A.; Wardenaar, K.J.; van Zwet, E.W.; van Hemert, A.M.; Zitman, F.G.; Ferrari, M.D.; Penninx, B.W.; Terwindt, G.M. Symptom Dimensions of Affective Disorders in Migraine Patients. J. Psychosom. Res. 2015, 79, 458–463. [Google Scholar] [CrossRef]
- Minen, M.T.; Begasse De Dhaem, O.; Kroon Van Diest, A.; Powers, S.; Schwedt, T.J.; Lipton, R.; Silbersweig, D. Migraine and Its Psychiatric Comorbidities. J. Neurol. Neurosurg. Psychiatry 2016, 87, 741–749. [Google Scholar] [CrossRef]
- Sikora, M.; Verzak, Ž.; Matijević, M.; Včev, A.; Siber, S.; Musić, L.; Carek, A. Anxiety and Depression Scores in Patients with Burning Mouth Syndrome. Psychiatr. Danub. 2018, 30, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Malta, C.E.N.; Costa, F.W.G.; Dias, C.C.; Carlos, A.C.A.M.; Sousa, F.B.; Silva, P.G.B.; Teófilo, C.R. Association of Anxiety, Depression, and Stress with Burning Mouth Syndrome: A Case-Control Study. Gen. Dent. 2021, 69, 46–52. [Google Scholar] [PubMed]
- Schiavone, V.; Adamo, D.; Ventrella, G.; Morlino, M.; De Notaris, E.B.; Ravel, M.G.; Kusmann, F.; Piantadosi, M.; Pollio, A.; Fortuna, G.; et al. Anxiety, Depression, and Pain in Burning Mouth Syndrome: First Chicken or Egg? Headache 2012, 52, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Bigal, M.E.; Ferrari, M.; Silberstein, S.D.; Lipton, R.B.; Goadsby, P.J. Migraine in the Triptan Era: Lessons from Epidemiology, Pathophysiology, and Clinical Science. Headache 2009, 49 (Suppl. 1), S21–S33. [Google Scholar] [CrossRef]
- Noseda, R.; Burstein, R. Migraine Pathophysiology: Anatomy of the Trigeminovascular Pathway and Associated Neurological Symptoms, CSD, Sensitization and Modulation of Pain. Pain 2013, 154 (Suppl. 1), S44–S53. [Google Scholar] [CrossRef] [Green Version]
- Teruel, A.; Patel, S. Burning Mouth Syndrome: A Review of Etiology, Diagnosis, and Management. Gen. Dent. 2019, 67, 24–29. [Google Scholar]
- Minguez-Sanz, M.P.; Salort-Llorca, C.; Silvestre-Donat, F.J. Etiology of Burning Mouth Syndrome: A Review and Update. Med. Oral Patol. Oral Cir. Bucal. 2011, 16, e144–e148. [Google Scholar] [CrossRef] [Green Version]
- De Tommaso, M.; Lavolpe, V.; Di Venere, D.; Corsalini, M.; Vecchio, E.; Favia, G.; Sardaro, M.; Livrea, P.; Nolano, M. A case of unilateral burning mouth syndrome of neuropathic origin. Headache 2011, 51, 441–443. [Google Scholar] [CrossRef]
- Yilmaz, Z.; Renton, T.; Yiangou, Y.; Zakrzewska, J.; Chessell, I.P.; Bountra, C.; Anand, P. Burning mouth syndrome as a trigeminal small fibre neuropathy: Increased heat and capsaicin receptor TRPV1 in nerve fibres correlates with pain score. J. Clin. Neurosci. 2007, 14, 864–871. [Google Scholar] [CrossRef]
- Braud, A.; Touré, B.; Agbo-Godeau, S.; Descroix, V.; Boucher, Y. Characteristics of Pain Assessed with Visual Analog Scale and Questionnaire in Burning Mouth Syndrome Patients: A Pilot Study. J. Orofac. Pain 2013, 27, 235–242. [Google Scholar] [CrossRef]
- Lopez-Jornet, P.; Lucero-Berdugo, M.; Castillo-Felipe, C.; Zamora Lavella, C.; Ferrandez-Pujante, A.; Pons-Fuster, A. Assessment of Self-Reported Sleep Disturbance and Psychological Status in Patients with Burning Mouth Syndrome. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Lee, B.C.; Park, K.J.; Son, G.M. Effect of Obstructive Sleep Apnea on Immunity in Cases of Chronic Rhinosinusitis with Nasal Polyps. Clin. Exp. Otorhinolaryngol. 2021, 14, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.S.; Lee, J.J.; Lee, S.H.; Kim, C.; Yu, H.; Sohn, J.H.; Kim, D.K. Risk of Dementia in Patients Who Underwent Surgery Under Neuraxial Anesthesia: A Nationwide Cohort Study. J. Pers. Med. 2021, 11, 1386. [Google Scholar] [CrossRef]
- Lee, H.J.; Yu, H.; Gil Myeong, S.; Park, K.; Kim, D.K. Mid- and Late-Life Migraine Is Associated with an Increased Risk of All-Cause Dementia and Alzheimer’s Disease, but Not Vascular Dementia: A Nationwide Retrospective Cohort Study. J. Pers. Med. 2021, 11, 990. [Google Scholar] [CrossRef] [PubMed]
- Sohn, J.H.; Lee, J.J.; Lee, S.H.; Kim, C.; Yu, H.; Kwon, Y.S.; Kim, D.K. Longitudinal Study of the Association Between General Anesthesia and Increased Risk of Developing Dementia. J. Pers. Med. 2021, 11, 1215. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Lee, S.; Cha, J.; Son, G.; Kim, D.K. Chronic Kidney Disease Is Associated with Increased Risk of Sudden Sensorineural Hearing Loss and Meniere’s Disease: A Nationwide Cohort Study. Sci. Rep. 2021, 11, 20194. [Google Scholar] [CrossRef]
- Lee, S.; Cha, J.; Kim, J.Y.; Son, G.M.; Kim, D.K. Detection of Unknown Ototoxic Adverse Drug Reactions: An Electronic Healthcare Record-Based Longitudinal Nationwide Cohort Analysis. Sci. Rep. 2021, 11, 14045. [Google Scholar] [CrossRef]
- Seong, S.C.; Kim, Y.Y.; Park, S.K.; Khang, Y.H.; Kim, H.C.; Park, J.H.; Kang, H.J.; Do, C.H.; Song, J.S.; Lee, E.J.; et al. Cohort Profile: The National Health Insurance Service-National Health Screening Cohort (NHIS-HEALS) in Korea. BMJ Open 2017, 7, e016640. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Lee, J.S.; Park, S.H.; Shin, S.A.; Kim, K. Cohort Profile: The National Health Insurance Service-National Sample Cohort (NHIS-NSC), South Korea. Int. J. Epidemiol. 2017, 46, e15. [Google Scholar] [CrossRef]
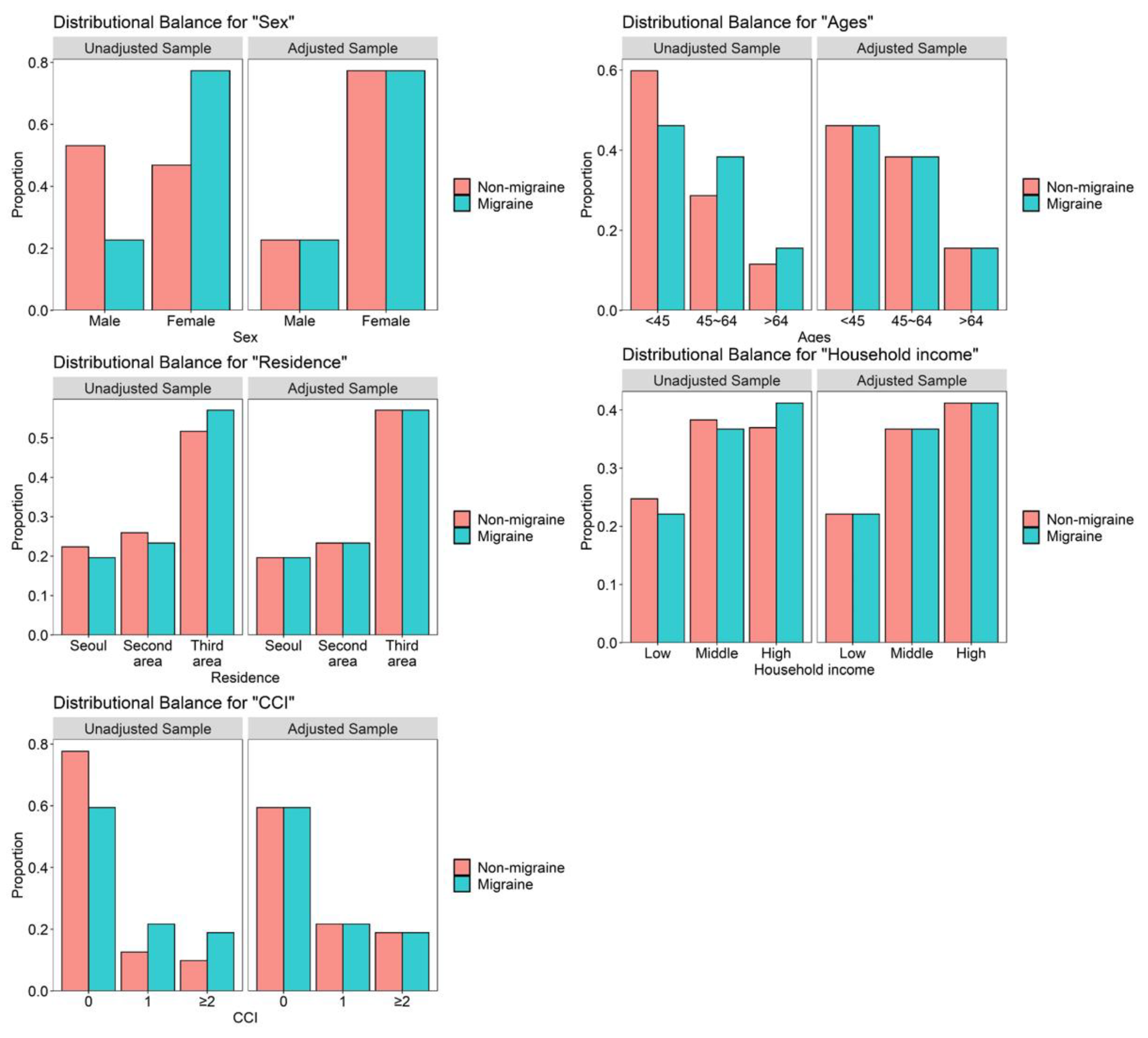
| Variables | Comparison (n = 16,628) | Migraine (n = 4157) | p Value |
|---|---|---|---|
| Sex | 1000 | ||
| Men | 3772 (22.7%) | 943 (22.7%) | |
| Women | 12,856 (77.3%) | 3214 (77.3%) | |
| Ages (years) | 1000 | ||
| <45 | 7672 (46.1%) | 1918 (46.1%) | |
| 45–64 | 6376 (38.3%) | 1594 (38.3%) | |
| >64 | 2580 (15.5%) | 645 (15.5%) | |
| Residence | 1000 | ||
| Seoul | 3264 (19.6%) | 816 (19.6%) | |
| Second area | 3880 (23.3%) | 970 (23.3%) | |
| Third area | 9484 (57.0%) | 2371 (57.0%) | |
| Household income | 1000 | ||
| Low (0–30%) | 3676 (22.1%) | 919 (22.1%) | |
| Middle (30–70%) | 6104 (36.7%) | 1526 (36.7%) | |
| High (70–100%) | 6848 (41.2%) | 1712 (41.2%) | |
| CCI | 1000 | ||
| 0 | 9880 (59.4%) | 2470 (59.4%) | |
| 1 | 3604 (21.7%) | 901 (21.7%) | |
| ≥2 | 3144 (18.9%) | 786 (18.9%) |
| Variables | N | Case | Person-Year | Incidence | Unadjusted HR (95% CI) | Adjusted HR (95% CI) | p Value |
|---|---|---|---|---|---|---|---|
| Group | |||||||
| Comparison | 16,628 | 8 | 162,034.51 | 0.05 | 1.00 (ref) | 1.00 (ref) | |
| Migraine | 4157 | 6 | 39,371.21 | 0.15 | 3.10 (1.08–8.97) | 2.96 (1.02–8.56) | 0.045 |
| Sex | |||||||
| Men | 4715 | 2 | 44,313.74 | 0.05 | 1.00 (ref) | 1.00 (ref) | |
| Women | 16,070 | 12 | 157,091.98 | 0.08 | 1.68 (0.38–7.52) | 1.65 (0.36–7.44) | 0.516 |
| Age (years) | |||||||
| <45 | 9590 | 1 | 96,319.54 | 0.01 | 1.00 (ref) | 1.00 (ref) | |
| 45–64 | 7970 | 7 | 78,831.25 | 0.09 | 8.55 (1.05–69.46) | 7.39 (0.90–60.59) | 0.062 |
| >64 | 3225 | 6 | 26,254.93 | 0.23 | 22.47 (2.70–186.89) | 15.04 (1.75–128.92) | 0.013 |
| Residence | |||||||
| Seoul | 4080 | 3 | 40,086.03 | 0.07 | 1.00 (ref) | 1.00 (ref) | |
| Second area | 4850 | 1 | 47,235.26 | 0.02 | 0.28 (0.03–2.73) | 0.28 (0.03–2.68) | 0.268 |
| Third area | 11,855 | 10 | 114,084.43 | 0.09 | 1.17 (0.32–4.26) | 0.94 (0.26–3.44) | 0.928 |
| Household income | |||||||
| Low | 4595 | 6 | 43,909.28 | 0.14 | 1.00 (ref) | 1.00 (ref) | |
| Middle | 7630 | 4 | 74,159.59 | 0.05 | 0.39 (0.11–1.40) | 0.50 (0.14–1.79) | 0.288 |
| High | 8560 | 4 | 83,336.85 | 0.05 | 0.35 (0.10–1.24) | 0.39 (0.11–1.38) | 0.143 |
| CCI | |||||||
| 0 | 12,350 | 4 | 121,791.34 | 0.03 | 1.00 (ref) | 1.00 (ref) | |
| 1 | 4505 | 5 | 43,961.34 | 0.11 | 3.46 (0.93–12.90) | 2.44 (0.64–9.23) | 0.190 |
| ≥2 | 3930 | 5 | 35,653.04 | 0.14 | 4.28 (1.15–15.92) | 2.98 (0.79–11.27) | 0.108 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, D.-K.; Lee, H.-J.; Lee, I.H.; Lee, J.-J. Risk of Burning Mouth Syndrome in Patients with Migraine: A Nationwide Cohort Study. J. Pers. Med. 2022, 12, 620. https://doi.org/10.3390/jpm12040620
Kim D-K, Lee H-J, Lee IH, Lee J-J. Risk of Burning Mouth Syndrome in Patients with Migraine: A Nationwide Cohort Study. Journal of Personalized Medicine. 2022; 12(4):620. https://doi.org/10.3390/jpm12040620
Chicago/Turabian StyleKim, Dong-Kyu, Hyun-Joo Lee, Il Hwan Lee, and Jae-Jun Lee. 2022. "Risk of Burning Mouth Syndrome in Patients with Migraine: A Nationwide Cohort Study" Journal of Personalized Medicine 12, no. 4: 620. https://doi.org/10.3390/jpm12040620
APA StyleKim, D.-K., Lee, H.-J., Lee, I. H., & Lee, J.-J. (2022). Risk of Burning Mouth Syndrome in Patients with Migraine: A Nationwide Cohort Study. Journal of Personalized Medicine, 12(4), 620. https://doi.org/10.3390/jpm12040620






