Network Analysis-Based Disentanglement of the Symptom Heterogeneity in Asian Patients with Schizophrenia: Findings from the Research on Asian Psychotropic Prescription Patterns for Antipsychotics
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Overview and Participants
2.2. Brief Psychiatric Rating Scale
2.3. Operational Classification of Psychotropic Drugs
2.4. Statistical Analyses
3. Results
3.1. General Description of the Study Participants
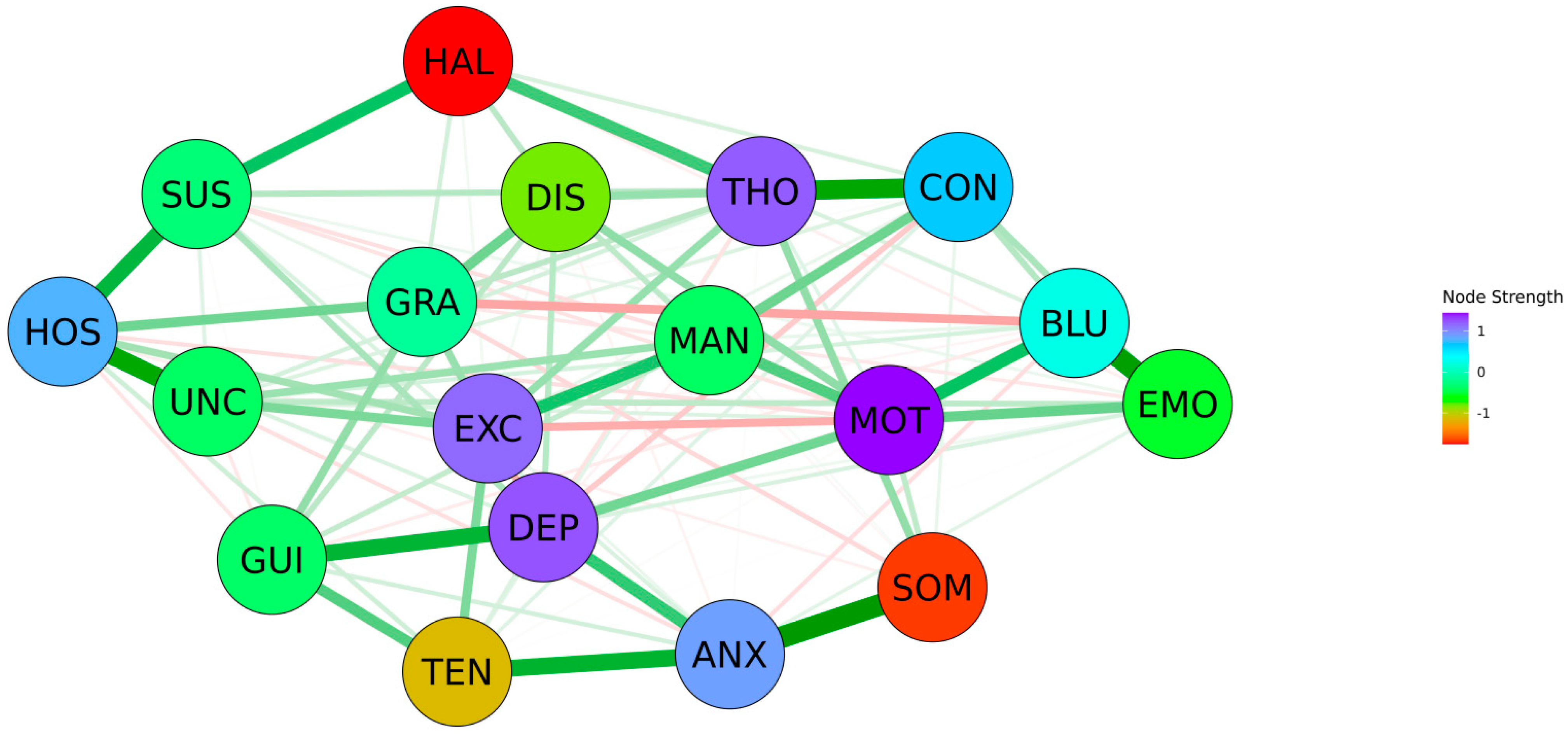
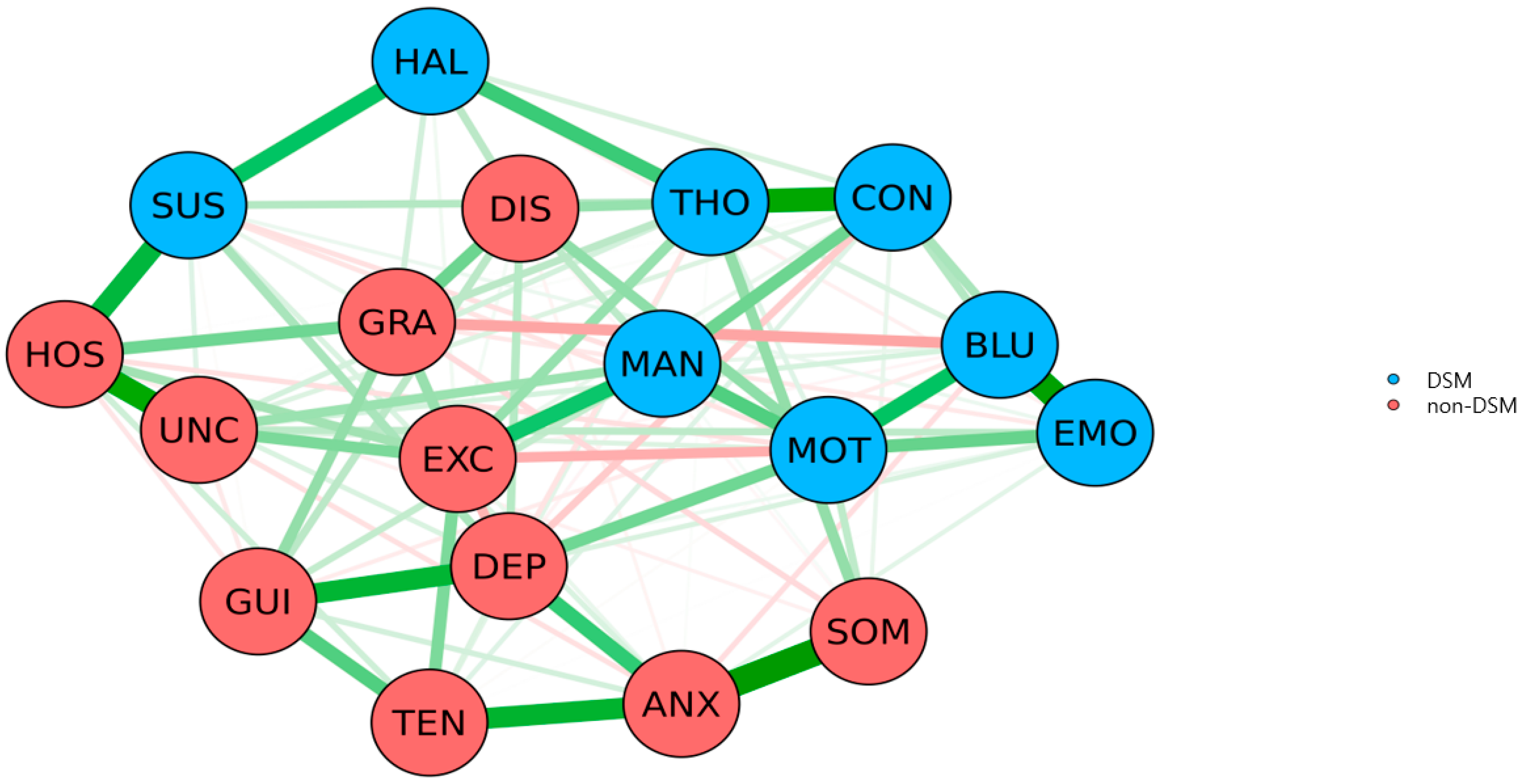
3.2. Edge Statistics
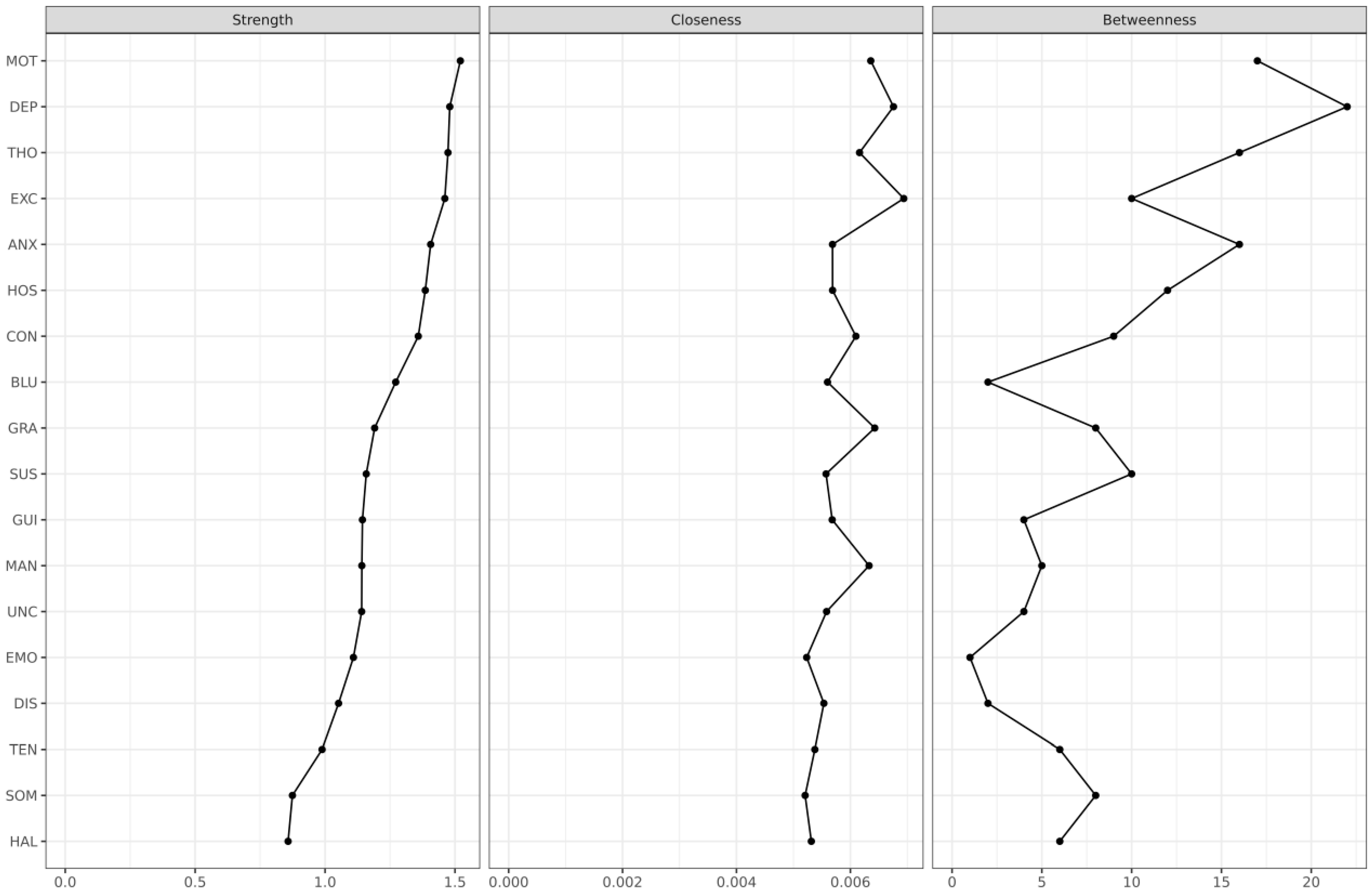
3.3. Node Statistics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tandon, R.; Keshavan, M.S.; Nasrallah, H.A. Schizophrenia, “just the facts”: What we know in 2008. Part 1: Overview. Schizophr. Res. 2008, 100, 4–19. [Google Scholar] [CrossRef]
- Tandon, R.; Keshavan, M.S.; Nasrallah, H.A. Schizophrenia, “just the facts”: What we know in 2008. 2. Epidemiology and etiology. Schizophr. Res. 2008, 102, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Tandon, R.; Nasrallah, H.A.; Keshavan, M.S. Schizophrenia, “just the facts.” 4. Clinical features and conceptualization. Schizophr. Res. 2009, 110, 1–23. [Google Scholar] [CrossRef]
- Kraepelin, E. Dementia Praecox and Hebephrenia, 1919; Robertson, G., Ed.; Krieger: New York, NY, USA, 1971. [Google Scholar]
- Kraepelin, E. Psychiatry—A Textbook for Students and Doctors, 1899; Acanet: Seoul, Korea, 2021. [Google Scholar]
- Bleuler, E. Dementia Praecox or the Groups of Schizophrenia, 1911; International University Press: New York, NY, USA, 1950. [Google Scholar]
- Schneider, K. Clinical Psychopathology, 1946; Grune and Stratton: New York, NY, USA, 1959. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 2nd ed.; (DSM-II); American Psychiatric Publishing, Inc.: Washington, DC, USA, 1968. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed.; (DSM-III); American Psychiatric Publishing, Inc.: Washington, DC, USA, 1980. [Google Scholar]
- Parnas, I. Philosophical Issues in Psychiatry II: Nosology; Oxford University Press: Oxford, UK, 2012. [Google Scholar]
- Tammiga, C.A.; Sirovatka, P.J.; Regier, D.A.; van Os, J. Deconstructing Psychosis:Refining the Research Agenda for DSM-V; American Psychiatric Publishing, Inc.: Washington, DC, USA, 2009. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; (DSM-5); American Psychiatric Publishing, Inc.: Washington, DC, USA, 2013. [Google Scholar]
- Kincaid, H.; Sullivan, J.A. Classifying Psychopathology: Mental Kinds and Natural Kinds; The MIT Press: New York, NY, USA, 2014. [Google Scholar]
- Jeong, S.H. “Does schizophrenia exist?”: A number of related questions in psychiatric nosology. Korean J. Schizophr. Res. 2008, 11, 78–88. [Google Scholar]
- Wittgenstein, L. Philosophical Investigations. (German Text, with a Revised English Translation, Trans GEM Anscombe), 3rd ed.; Blackwell: Oxford, UK, 2001. [Google Scholar]
- Rosenman, S.; Nasti, J. Psychiatric diagnoses are not mental processes: Wittgenstein on conceptual confusion. Aust. N. Z. J. Psychiatry 2012, 46, 1046–1052. [Google Scholar] [CrossRef]
- Insel, T.R. Next-generation treatments for mental disorders. Sci. Transl. Med. 2012, 4, 155psc19. [Google Scholar] [CrossRef]
- Insel, T.R.; Cuthbert, B.N. Endophenotypes: Bridging genomic complexity and disorder heterogeneity. Biol. Psychiatry 2009, 66, 988–989. [Google Scholar] [CrossRef]
- Posner, J. The role of precision medicine in child psychiatry: What can we expect and when? J. Am. Acad. Child Adolesc. Psychiatry 2018, 57, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Tognin, S.; van Hell, H.H.; Merritt, K.; Winter-van Rossum, I.; Bossong, M.G.; Kempton, M.J.; Modinos, G.; Fusar-Poli, P.; Mechelli, A.; Dazzan, P.; et al. Towards precision medicine in psychosis: Benefits and challenges of multimodal multicenter studies–PSYSCAN: Translating neuroimaging findings from research into clinical practice. Schizophr. Bull. 2020, 46, 432–441. [Google Scholar] [CrossRef]
- Roca, P.; Diez, G.G.; Castellanos, N.; Vazquez, C. Does mindfulness change mind? A novel psychonectome perspective based on network analysis. PLoS ONE 2019, 14, e0219793. [Google Scholar] [CrossRef] [PubMed]
- Saxe, G.N. Network psychiatry: Computational methods to understand the complexity of psychiatric disorders. J. Am. Acad. Child. Adolesc. Psychiatry 2017, 56, 639–641. [Google Scholar] [CrossRef]
- Barabasi, A.L. Network medicine form obesity to the “diseasome”. N. Engl. J. Med. 2017, 357, 404–407. [Google Scholar] [CrossRef]
- Armour, C.; Fried, E.I.; Deserno, M.K.; Tsai, J.; Pietrzak, R.H. A network analysis of DSM-5 posttraumatic stress disorder symptoms and correlates in U.S. military veterans. J. Affect. Disord. 2017, 45, 49–59. [Google Scholar] [CrossRef]
- Van Borkulo, C.; Boschloo, L.; Borsboom, D.; Penninx, B.W.J.H.; Waldorp, L.J.; Schoevers, R.A. Association of symptom network structure with the course of depression. JAMA Psychiatry 2015, 72, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- McNally, R.J.; Robinaugh, D.J.; Wu, G.W.; Wang, L.; Deserno, M.K.; Borsboom, D. Mental disorders as causal systems a network approach to posttraumatic stress disorder. Clin. Psychol. Sci. 2015, 23, 836–849. [Google Scholar] [CrossRef]
- Park, Y.C.; Lee, M.S.; Si, T.M.; Chiu, H.F.K.; Kanba, S.; Chong, M.-Y.; Tripathi, A.; Udomratn, P.; Chee, K.Y.; Tanra, A.J.; et al. Psychotropic Drug-Prescribing Correlates of Disorganized Speech in Asians with Schizophrenia: The REAP-AP Study. Saudi Pharm. J. 2019, 27, 246–253. [Google Scholar] [CrossRef]
- Park, Y.C.; Kanba, S.; Chong, M.-Y.; Tripathi, A.; Kallivayalil, R.A.; Avasthi, A.; Grover, S.; Chee, K.Y.; Tanra, A.J.; Maramis, M.M.; et al. To Use the Brief Psychiatric Rating Scale to Detect Disorganized Speech in Schizophrenia: Findings from the REAP-AP Study. Kaohsiung J. Med. Sci. 2018, 34, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Overall, J.E.; Gorham, D.R. The brief psychiatric rating scale. Psychol. Rep. 1962, 10, 779–812. [Google Scholar] [CrossRef]
- World Health Organization. Anatomical Therapeutic Chemical (ATC) Classification System. Available online: https://www.whocc.no/atc/structure_and_principles/ (accessed on 18 January 2021).
- Leucht, S.; Kane, J.M.; Kissling, W.; Hamann, J.; Etschel, E.; Engel, R. Clinical implications of brief psychiatric rating scale score. Br. J. Psychiatry 2005, 187, 366–371. [Google Scholar] [CrossRef]
- Hedlund, J.L.; View, B.W. The brief psychiatric rating scale: A comprehensive review. J. Oper. Psychiatr. 1980, 11, 48–65. [Google Scholar]
- Sim, K.; Su, A.; Leong, I.Y.; Yip, K.; Chong, M.Y.; Fujii, S.; Yang, S.; Ungvari, G.S.; Si, T.; Chung, E.K.; et al. High dose antipsychotic use in schizophrenia: Findings of the REAP (Research on East Asian Psychotropic Prescriptions) study. Pharmacopsychiatry 2004, 37, 175–179. [Google Scholar] [CrossRef]
- Tihonen, J.; Mittendorfer-Rutz, E.; Tornianen, M.; Alexanderson, K.; Tanskanen, A. Mortality and cumulative exposure to antipsychotics, antidepressants, and benzodiazepine in patients with schizophrenia: An observational follow-up study. Am. J. Psychiatry 2016, 173, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Epskamp, S.; Cramer, A.O.J.; Waldorp, L.J.; Schmittman, V.D.; Borsboom, D. Qgraph: Network visualizations of relationships in psychometric data. J. Stat. Softw. 2012, 48, 1–18. [Google Scholar] [CrossRef]
- Tibshirani, R. Regression shrinkage and selection via the lasso. J. R. Stat. Soc. Ser. B 1996, 58, 267–288. [Google Scholar] [CrossRef]
- Chen, J.; Chen, Z. Extended Bayesian information criteria for model selection with large model speaces. Biometrika 2008, 95, 759–771. [Google Scholar] [CrossRef]
- Reichardt, J.; Bornholdt, S. Statistical mechanics of community detection. Phys. Rev. E 2006, 74, 016110. [Google Scholar] [CrossRef]
- Csardi, G.; Nepusz, T. The igraph software package for complex network research. Inter J. Complex Syst. 2006, 1695, 1–9. [Google Scholar]
- Opsahl, T.; Agneessens, F.; Skvoretz, J. Node centrality in weighted networks: Generalizing degrees and shortest paths. Soc. Netw. 2010, 32, 245–251. [Google Scholar] [CrossRef]
- Fried, E.I.; Epskamp, S.; Nesse, R.M.; Tuerlinckx, F.; Borsboom, D. What are ‘good’ depression symptoms? Comparing the centrality of DSM and non-DSM symptoms of depression in a network analysis. J. Affect. Disord. 2016, 189, 314–329. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences (Revised Edition); Academic Press: New York, NY, USA, 1977. [Google Scholar]
- Epskamp, S.; Borsboom, D.; Fried, E.I. Estimating psychological networks and their accuracy: A tutorial paper. Behav. Res. Methods 2018, 50, 195–212. [Google Scholar] [CrossRef] [PubMed]
- Northoff, G.; Hirjak, D.; Wolf, R.C.; Magioncalda, P.; Martino, M. All roads lead to the motor cortex: Psychomotor mechanisms and their biochemical modulation in psychiatric disorders. Mol. Psychiatry 2021, 26, 92–102. [Google Scholar] [CrossRef]
- Hirjak, D.; Meyer-Lindenberg, A.; Sambataro, F.; Fritze, S.; Kukovic, J.; Kubera, K.K.; Wolf, R.C. Progress in sensorimotor neuroscience of schizophrenia spectrum disorders: Lessens learned and future directions. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 111, 110370. [Google Scholar] [CrossRef] [PubMed]
- Kent, J.S.; Disner, S.G.; van Voorhis, A.C.; Urosevic, S.; Caligiuri, M.P.; Sponheim, S.R. Exploring the relationship of transdiagnostic mood and psychosis symptom domains with mood dysfunction. Neuropsychobiology 2020, 79, 301–312. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Choi, J.; Park, S.-C. A novel bio-psychosocial-behavioral treatment model in schizophrenia. Int. J. Mol. Sci. 2017, 18, 734. [Google Scholar] [CrossRef]
- Keshavan, M.S.; Lawler, A.N.; Nasrallah, H.A.; Tandon, R. New drug developments in psychosis: Challenges, opportunities and strategies. Prog. Neurobiol. 2017, 152, 3–30. [Google Scholar] [CrossRef]
- Zhou, N.; Masterson, S.P.; Damron, J.K.; Guido, W.; Bickford, M. The mouse pulvinar nucleus links the lateral extrastriate cortex, striatum, and amygdala. J. Neurosci. 2018, 38, 347–362. [Google Scholar] [CrossRef]
- Chou, X.-L.; Fang, Q.; Yan, L.; Zhong, W.; Peng, B.; Li, H.; Wei, J.; Tao, H.W.; Zhang, L.I. Contextual and cross-modality modulation of auditory cortical processing through pulvinar mediated suppression. Elife 2020, 9, e54157. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; Chou, X.-L.; Peng, B.; Zhong, W.; Zhang, L.I.; Tao, H.W. A differential circuit via retiono-colliculo-pulvinar pathway enhances feature selectively in visual cortex through surround suppression. Neuron 2020, 105, 355–369. [Google Scholar] [CrossRef]
- Ibrahim, L.A.; Mesik, L.; Ji, X.-Y.; Fang, Q.; Li, H.-F.; Li, Y.-T.; Zingg, B.; Zhang, L.I.; Tao, H.W. Cross-modality sharpening of visual cortical processing through layer-1-mediated inhibition and disinhibition. Neuron 2016, 89, 1031–1045. [Google Scholar] [CrossRef] [PubMed]
- Andrews, J.; Wang, L.; Csernansky, J.G.; Gado, M.H.; Barch, D.M. Abnormalities of thalamic activation and cognition in schizophrenia. Am. J. Psychiatry 2006, 163, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Hagele, C.; Schlagenhauf, F.; Rapp, M.; Sterzer, P.; Beck, A.; Bermpohl, F.; Stoy, M.; Strohle, A.; Wittchen, H.U.; Dolan, R.J.; et al. Dimensional psychiatry: Reward dysfunction and depressive mood across psychiatric disorders. Psychopharmacology 2015, 232, 331–341. [Google Scholar] [CrossRef]
- Ortiz, B.B.; Higuch, C.H.; Noto, C.; Joyce, D.W.; Correll, C.U.; Bressan, R.A.; Gadelha, A. A symptom combination predicting treatment-resistant schizophrenia—A strategy for real-world clinical practice. Schizophr. Res. 2020, 218, 195–200. [Google Scholar] [CrossRef] [PubMed]
- McCarthy-Jones, S.; Smailes, D.; Corvin, A.; Gill, M.; Morris, D.W.; Dinan, T.G.; Murphy, K.C.; Neil, F.A.O.; Waddington, J.L.; Austrailian Schizophrenia Research Bank; et al. Occurrence and co-occurrence of hallucinations by modality in schizophrenia-spectrum disorders. Psychiatry Res. 2017, 252, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Dugre, J.R.; West, M.L. Disentangling compliance with command hallucinations in schizophrenia spectrum disorders. Schizophr. Bull. 2019, 212, 33–39. [Google Scholar]
- Sinkeviciute, I.; Hugdahl, K.; Bartz-Johannessen, C.; Kroken, R.A.; Loberg, E.-M.; Kjelby, E.; Rettenbacher, M.A.; Joa, I.; Reitan, S.K.; Alisauskiene, R.; et al. Differential effectiveness of atypical antipsychotics on hallucinations: A pragmatic randomized controlled trial. J. Clin. Psychopharmacol. 2021, 41, 389–396. [Google Scholar] [CrossRef]
- Sawamur, J.; Morishita, S.; Ishigooka, J. A modification of the relative weightings of symptoms utilizing a logistic function to enhance the linearity of the Brief Psychiatric Rating Scale: A retrospective analysis. J. Behav. Brain Sci. 2012, 2, 225–238. [Google Scholar] [CrossRef][Green Version]
| Continuous Variable | Mean | SD |
|---|---|---|
| Age (years) | 39.9 | 12.5 |
| Chlorpromazine equivalent dose (mg/day) | 501.5 | 396.5 |
| Discrete variable | n | % |
| Sex | ||
| Male | 830 | 57.7 |
| Female | 608 | 42.3 |
| Country | ||
| India | 400 | 27.8 |
| Indonesia | 261 | 18.2 |
| Japan | 98 | 6.8 |
| Malaysia | 299 | 20.8 |
| Taiwan | 380 | 26.4 |
| Duration of illness | ||
| <3 months | 52 | 3.6 |
| 3–6 months | 38 | 2.6 |
| 6–12 months | 46 | 3.2 |
| 1–5 years | 274 | 19.1 |
| 5–10 years | 240 | 16.7 |
| 10–20 years | 429 | 29.8 |
| >20 years | 359 | 25.0 |
| Duration of untreated psychosis | ||
| <3 months | 524 | 36.4 |
| 3–12 months | 492 | 34.2 |
| 1–5 years | 270 | 18.8 |
| >5 years | 152 | 10.6 |
| Inpatient | 664 | 46.2 |
| Unemployed | 237 | 16.5 |
| Antipsychotic polypharmacy | 536 | 37.3 |
| Adjunctive mood stabilizer | 142 | 9.9 |
| Adjunctive antidepressant | 129 | 9.0 |
| Adjunctive antiparkinsonian | 560 | 38.9 |
| High dose antipsychotic † | 161 | 11.2 |
| Long-acting injectable antipsychotic | 305 | 21.2 |
| Clozapine | 293 | 20.4 |
| Electroconvulsive therapy | 42 | 2.9 |
| Cannabis use (lifetime) | 119 | 8.3 |
| Items | Abbreviation | Mean (SD) | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|---|---|
| n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | |||
| Somatic concern | SOM | 1.8 (1.2) | 840 (58.4) | 258 (17.9) | 199 (13.8) | 87 (6.1) | 37 (2.6) | 13 (0.9) | 4 (0.3) |
| Anxiety | ANX | 2.1 (1.2) | 596 (41.4) | 348 (24.2) | 290 (20.2) | 143 (9.9) | 44 (3.1) | 16 (1.1) | 1 (0.1) |
| Emotional withdrawal | EMO | 2.5 (1.5) | 511 (35.5) | 274 (19.1) | 294 (20.4) | 221 (15.4) | 85 (5.9) | 42 (2.9) | 11 (0.8) |
| Conceptual disorganization | CON | 2.4 (1.5) | 599 (41.7) | 251 (17.5) | 265 (18.4) | 176 (12.2) | 102 (7.1) | 32 (2.2) | 13 (0.9) |
| Guilt feelings | GUI | 1.5 (0.9) | 1016 (70.7) | 237 (16.5) | 140 (9.7) | 32 (2.2) | 7 (0.5) | 4 (0.3) | 2 (0.1) |
| Tension | TEN | 2.0 (1.1) | 656 (45.6) | 336 (23.4) | 286 (19.9) | 121 (8.4) | 31 (2.2) | 8 (0.6) | 0 (0.0) |
| Mannerism and posturing | MAN | 1.5 (1.0) | 1090 (75.8) | 148 (10.3) | 117 (8.1) | 56 (3.9) | 18 (1.3) | 9 (0.6) | 0 (0.0) |
| Grandiosity | GRA | 1.5 (1.0) | 1107 (77.0) | 140 (9.7) | 97 (6.7) | 63 (4.4) | 14 (1.0) | 12 (0.8) | 5 (0.3) |
| Depressive mood | DEP | 1.8 (1.1) | 804 (55.9) | 301 (20.9) | 233 (16.2) | 74 (5.1) | 14 (1.0) | 9 (0.6) | 3 (0.2) |
| Hostility | HOS | 1.9 (1.3) | 807 (56.1) | 256 (17.8) | 169 (11.8) | 136 (9.5) | 47 (3.3) | 15 (1.0) | 8 (0.6) |
| Suspiciousness | SUS | 2.4 (1.5) | 587 (40.8) | 248 (17.2) | 269 (18.7) | 201 (14.0) | 81 (5.6) | 41 (2.9) | 11 (0.8) |
| Hallucinatory behavior | HAL | 2.6 (1.6) | 530 (36.9) | 234 (16.3) | 259 (18.0) | 196 (13.6) | 118 (8.2) | 80 (5.6) | 21 (1.5) |
| Motor retardation | MOT | 1.7 (1.2) | 898 (62.4) | 244 (17.0) | 158 (11.0) | 82 (5.7) | 37 (2.6) | 15 (1.0) | 4 (0.3) |
| Uncooperativeness | UNC | 1.9 (1.2) | 814 (56.6) | 273 (19.0) | 182 (12.7) | 103 (7.2) | 38 (2.6) | 22 (1.5) | 6 (0.4) |
| Unusual thought content | THO | 2.5 (1.6) | 578 (40.2) | 239 (17.5) | 251 (17.5) | 181 (12.6) | 118 (8.2) | 54 (3.8) | 17 (1.2) |
| Blunted affect | BLU | 2.3 (1.4) | 572 (39.8) | 285 (19.8) | 285 (19.8) | 162 (11.3) | 84 (5.8) | 43 (3.0) | 7 (0.5) |
| Excitement | EXC | 1.6 (1.2) | 1001 (69.6) | 184 (12.8) | 113 (7.9) | 77 (5.4) | 49 (3.4) | 13 (0.9) | 1 (0.1) |
| Disorientation | DIS | 1.4 (0.8) | 1144 (79.6) | 162 (11.3) | 87 (6.1) | 30 (2.1) | 7 (0.5) | 3 (0.2) | 5 (0.3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.; Yoon, H.-J.; Park, J.H.; Nakagami, Y.; Kubota, C.; Inada, T.; Kato, T.A.; Yang, S.-Y.; Lin, S.-K.; Chong, M.-Y.; et al. Network Analysis-Based Disentanglement of the Symptom Heterogeneity in Asian Patients with Schizophrenia: Findings from the Research on Asian Psychotropic Prescription Patterns for Antipsychotics. J. Pers. Med. 2022, 12, 33. https://doi.org/10.3390/jpm12010033
Choi J, Yoon H-J, Park JH, Nakagami Y, Kubota C, Inada T, Kato TA, Yang S-Y, Lin S-K, Chong M-Y, et al. Network Analysis-Based Disentanglement of the Symptom Heterogeneity in Asian Patients with Schizophrenia: Findings from the Research on Asian Psychotropic Prescription Patterns for Antipsychotics. Journal of Personalized Medicine. 2022; 12(1):33. https://doi.org/10.3390/jpm12010033
Chicago/Turabian StyleChoi, Joonho, Hyung-Jun Yoon, Jae Hong Park, Yukako Nakagami, Chika Kubota, Toshiya Inada, Takahiro A. Kato, Shu-Yu Yang, Sih-Ku Lin, Mian-Yoon Chong, and et al. 2022. "Network Analysis-Based Disentanglement of the Symptom Heterogeneity in Asian Patients with Schizophrenia: Findings from the Research on Asian Psychotropic Prescription Patterns for Antipsychotics" Journal of Personalized Medicine 12, no. 1: 33. https://doi.org/10.3390/jpm12010033
APA StyleChoi, J., Yoon, H.-J., Park, J. H., Nakagami, Y., Kubota, C., Inada, T., Kato, T. A., Yang, S.-Y., Lin, S.-K., Chong, M.-Y., Avasthi, A., Grover, S., Kallivayalil, R. A., Tanra, A. J., Chee, K. Y., Xiang, Y.-T., Sim, K., Javed, A., Tan, C. H., ... Park, S.-C. (2022). Network Analysis-Based Disentanglement of the Symptom Heterogeneity in Asian Patients with Schizophrenia: Findings from the Research on Asian Psychotropic Prescription Patterns for Antipsychotics. Journal of Personalized Medicine, 12(1), 33. https://doi.org/10.3390/jpm12010033









