Long-Term Clinical Outcomes of Fractional Flow Reserve-Guided Coronary Artery Revascularization in Chronic Kidney Disease
Abstract
:1. Introduction
2. Materials and Methods
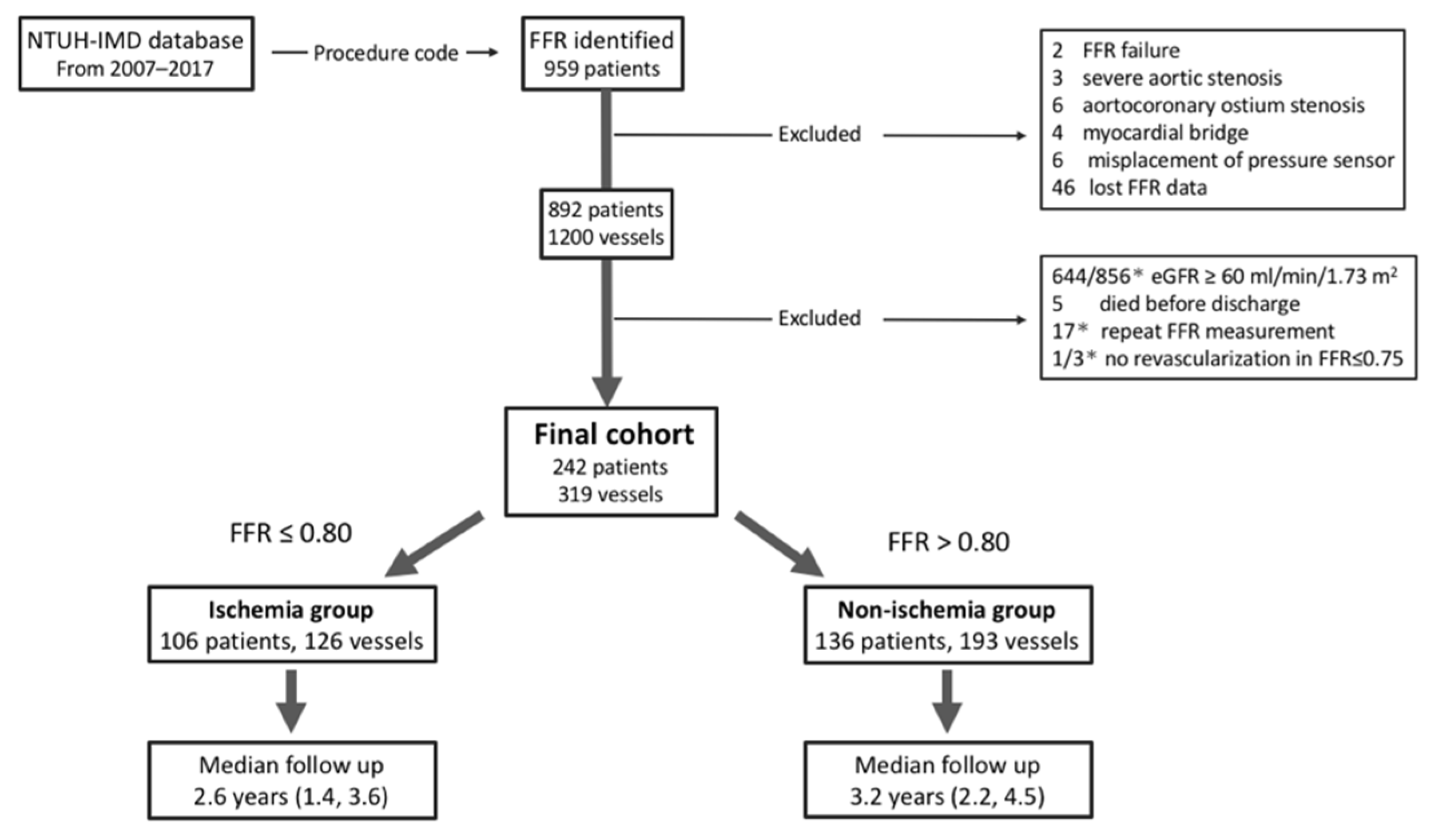
2.1. Study Design and Subjects
2.2. Data Collection and Ethical Approval
2.3. Outcomes and Follow-Up
2.4. Statistical Methods
3. Results
3.1. Patient and Vessel Characteristics
3.2. Clinical Outcomes
3.3. The Predicted Value and Best Cutoff of FFR to Predict Clinical Outcome
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD Chronic Kidney Disease Collaboration. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [CrossRef] [Green Version]
- Wen, C.P.; Cheng, T.Y.D.; Tsai, M.K.; Chang, Y.C.; Chan, H.T.; Tsai, S.P.; Chiang, P.H.; Hsu, C.C.; Sung, P.K.; Hsu, Y.H.; et al. All-cause mortality attributable to chronic kidney disease: A prospective cohort study based on 462,293 adults in Taiwan. Lancet 2008, 371, 2173–2182. [Google Scholar] [CrossRef]
- Chou, M.-T.; Wang, J.-J.; Sun, Y.-M.; Sheu, M.-J.; Chu, C.-C.; Weng, S.-F.; Chio, C.-C.; Kan, W.-C.; Chien, C.-C. Epidemiology and mortality among dialysis patients with acute coronary syndrome: Taiwan National Cohort Study. Int. J. Cardiol. 2013, 167, 2719–2723. [Google Scholar] [CrossRef] [PubMed]
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C.-Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Eng. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef]
- Bangalore, S.; Maron, D.J.; O’Brien, S.M.; Fleg, J.L.; Kretov, E.I.; Briguori, C.; Kaul, U.; Reynolds, H.R.; Mazurek, T.; Sidhu, M.S.; et al. Management of coronary disease in patients with advanced kidney disease. N. Engl. J. Med. 2020, 382, 1608–1618. [Google Scholar] [CrossRef] [PubMed]
- Bangalore, S. Stress testing in patients with chronic kidney disease: The need for ancillary markers for effective risk stratification and prognosis. J. Nucl. Cardiol. 2016, 23, 570–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pijls, N.H.; van Son, J.A.; Kirkeeide, R.L.; De Bruyne, B.; Gould, K.L. Experimental basis of determining maximum coronary, myocardial, and collateral blood flow by pressure measurements for assessing functional stenosis severity before and after percutaneous transluminal coronary angioplasty. Circulation 1993, 87, 1354–1367. [Google Scholar] [CrossRef] [Green Version]
- Neumann, F.J.; Sousa-Uva, M.; Ahlsson, A.; Alfonso, F.; Banning, A.P.; Benedetto, U.; Byrne, R.A.; Collet, J.P.; Falk, V.; Head, S.J.; et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur. Heart J. 2019, 40, 87–165. [Google Scholar] [CrossRef]
- Pijls, N.H.; Fearon, W.F.; Tonino, P.A.; Siebert, U.; Ikeno, F.; Bornschein, B.; van’t Veer, M.; Klauss, V.; Manoharan, G.; Engstrøm, T.; et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention in patients with multivessel coronary artery disease: 2-year follow-up of the FAME (Fractional Flow Reserve Versus Angiography for Multivessel Evaluation) study. J. Am. Coll. Cardiol. 2010, 56, 177–184. [Google Scholar] [CrossRef] [Green Version]
- Xaplanteris, P.; Fournier, S.; Pijls, N.H.; Fearon, W.F.; Barbato, E.; Tonino, P.A.; Engstrøm, T.; Kääb, S.; Dambrink, J.-H.; Rioufol, G.; et al. Five-year outcomes with PCI guided by fractional flow reserve. N. Engl. J. Med. 2018, 379, 250–259. [Google Scholar] [CrossRef]
- Charytan, D.M.; Skali, H.; Shah, N.R.; Veeranna, V.; Cheezum, M.K.; Taqueti, V.R.; Kato, T.; Bibbo, C.R.; Hainer, J.; Dorbala, S.; et al. Coronary flow reserve is predictive of the risk of cardiovascular death regardless of chronic kidney disease stage. Kidney Int. 2018, 93, 501–509. [Google Scholar] [CrossRef]
- Jong, C.-B.; Lu, T.-S.; Liu, P.Y.-T.; Hsieh, M.-Y.; Meng, S.-W.; Huang, C.-C.; Kao, H.-L.; Wu, C.-C. High dose escalation of intracoronary adenosine in the assessment of fractional flow reserve: A retrospective cohort study. PLoS ONE 2020, 15, e0240699. [Google Scholar] [CrossRef]
- Lu, T.-H.; Lee, M.-C.; Chou, M.-C. Accuracy of cause-of-death coding in Taiwan: Types of miscoding and effects on mortality statistics. Int. J. Epidemiol. 2000, 29, 336–343. [Google Scholar] [CrossRef]
- Davies, J.E.; Sen, S.; Dehbi, H.-M.; Al-Lamee, R.; Petraco, R.; Nijjer, S.S.; Bhindi, R.; Lehman, S.J.; Walters, D.; Sapontis, J.; et al. Use of the instantaneous wave-free ratio or fractional flow reserve in PCI. N. Engl. J. Med. 2017, 376, 1824–1834. [Google Scholar] [CrossRef] [Green Version]
- Uno, H.; Cai, T.; Tian, L.; Wei, L.J. Evaluating prediction rules for t-year survivors with censored regression models. J. Am. Stat. Assoc. 2007, 102, 527–537. [Google Scholar] [CrossRef]
- Lee, J.M.; Hwang, D.; Choi, K.H.; Rhee, T.-M.; Park, J.; Kim, H.Y.; Jung, H.W.; Hwang, J.-W.; Lee, H.-J.; Jang, H.-J.; et al. Prognostic implications of relative increase and final fractional flow reserve in patients with stent implantation. JACC Cardiovasc. Interv. 2018, 11, 2099–2109. [Google Scholar] [CrossRef]
- Agarwal, S.K.; Kasula, S.; Hacioglu, Y.; Ahmed, Z.; Uretsky, B.F.; Hakeem, A. Utilizing post-intervention fractional flow reserve to optimize acute results and the relationship to long-term outcomes. JACC Cardiovasc. Interv. 2016, 9, 1022–1031. [Google Scholar] [CrossRef]
- Tebaldi, M.; Biscaglia, S.; Fineschi, M.; Manari, A.; Menozzi, M.; Secco, G.G.; Di Lorenzo, E.; D’Ascenzo, F.; Fabbian, F.; Tumscitz, C.; et al. Fractional flow reserve evaluation and chronic kidney disease: Analysis from a multicenter italian registry (the FREAK Study). Catheter. Cardiovasc. Interv. 2016, 88, 555–562. [Google Scholar] [CrossRef]
- Valdivielso, J.M.; Rodríguez-Puyol, D.; Pascual, J.; Barrios, C.; Bermúdez-López, M.; Sánchez-Niño, M.D.; Pérez-Fernández, M.; Ortiz, A. Atherosclerosis in chronic kidney disease: More, less, or just different? Arterioscler. Thromb. Vasc. Biol. 2019, 39, 1938–1966. [Google Scholar] [CrossRef]
- Kirigaya, H.; Matsushita, K.; Okada, K.; Iwahashi, N.; Maejima, N.; Ebina, T.; Tamura, K.; Hibi, K.; Kimura, K. TCT-152 Clinical predictors of discordance between instantaneous wave-free ratio and fractional flow reserve. J. Am. Coll. Cardiol. 2018, 72 (Suppl. 13), B65. [Google Scholar] [CrossRef]
- Jong, C.-B.; Lu, T.-S.; Liu, P.Y.-T.; Wu, C.-C.; Kao, H.-L.; Chen, J.-W.; Hung, K.-Y.; Huang, C.-C. Impact of renal insufficiency on the relationship between the adenosine-free physiologic index and fractional flow reserve. Acta Cardiol. Sin. 2021. under revision. [Google Scholar]
- Johnson, N.P.; Tóth, G.G.; Lai, D.; Zhu, H.; Açar, G.; Agostoni, P.; Appelman, Y.; Arslan, F.; Barbato, E.; Chen, S.L.; et al. Prognostic value of fractional flow reserve: Linking physiologic severity to clinical outcomes. J. Am. Coll. Cardiol. 2014, 64, 1641–1654. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Demographic | Revascularization (n = 106 Patients) | Non-Revascularization (n = 136 Patients) | p-Value |
|---|---|---|---|
| Age | 69.3 ± 11.4 | 72.3 ± 10.5 | 0.050 |
| Sex (female), n (%) | 29 (27.4) | 55 (40.0) | 0.034 |
| Body mass index | 26.0 ± 3.9 | 25.7 ± 4.1 | 0.475 |
| Current smoker, n (%) | 19 (17.9) | 18 (13.3) | 0.326 |
| Comorbidity, n (%) | |||
| Hypertension | 90 (84.9) | 119 (87.5) | 0.560 |
| Diabetes mellitus | 64 (60.4) | 71 (52.2) | 0.204 |
| Heart failure | 34 (32.1) | 27 (19.9) | 0.030 |
| LVEF ≤ 50% | 21 (20.6) | 19 (14.7) | 0.243 |
| Previous myocardial infarction | 5 (4.7) | 3 (2.2) | 0.278 |
| History of coronary artery bypass grafting | 6 (5.7) | 9 (6.6) | 0.759 |
| CKD status | |||
| Mild CKD | 43 (40.6) | 62 (45.6) | 0.434 |
| Moderate to advanced CKD | 23 (21.7) | 48 (35.3) | 0.021 |
| Dialysis-dependent CKD | 40 (37.7) | 26 (19.1) | 0.001 |
| Distribution of arterial disease, n (%) | |||
| Left main disease | 15 (14.2) | 7 (5.1) | 0.004 |
| CAD single-vessel disease | 12 (11.3) | 52 (38.2) | <0.0001 |
| CAD double-vessel disease | 41 (38.7) | 34 (25.0) | 0.015 |
| CAD triple-vessel disease | 53 (50.0) | 50 (36.8) | 0.055 |
| Peripheral arterial disease | 16 (15.1) | 15 (11.0) | 0.348 |
| History of stroke | 13 (12.3) | 13 (9.6) | 0.500 |
| Clinical presentation *, n (%) | |||
| Acute coronary syndrome | 19 (15.1) | 19 (9.8) | 0.165 |
| Stable CAD | 106 (84.1) | 174 (90.2) | |
| Heart failure | 1 (0.8) | 0 (0) | |
| Lab data | |||
| Estimated glomerular filtration rate, ml/min/1.73 m2 | 46.7 ± 10.7 | 45.0 ± 11.9 | 0.536 |
| Hemoglobin, g/dL | 12.0 ± 2.2 | 12.6 ± 2.1 | 0.041 |
| HbA1c, mmol/mol | 7.0 ± 1.7 | 7.1 ± 1.8 | 0.858 |
| LDL, mg/dL | 96.6 ± 37.1 | 94.0 ± 32.9 | 0.840 |
| Target vessel *, n(%) | |||
| Left main or ostium/proximal part of left anterior descending artery | 48 (38.1) | 35 (18.1) | 0.0004 |
| Other coronary arteries | 78 (61.9) | 158 (81.9) | |
| Extent of atherosclerosis *, n (%) | |||
| Diffuse † | 23 (18.3) | 5 (2.6) | <0.0001 |
| Tandem lesion ‡ | 39 (31.0) | 26 (13.5) | 0.0002 |
| Lesion stenosis *, n (%) | |||
| 30–49 | 1 (0.8) | 11 (5.7) | <0.0001 |
| 50–70 | 115 (91.3) | 178 (92.2) | |
| 71–90 | 10 (7.9) | 4 (2.1) | |
| Invasive physiologic index * | |||
| Median FFR value | 0.75 (0.70, 0.78) | 0.88 (0.84, 0.91) | <0.0001 |
| Median NTG-Pd/Pa | 0.80 (0.74, 0.84) | 0.92 (0.88, 0.95) | <0.0001 |
| Treatment strategy *, n (%) | |||
| Revascularization | 114 (90.5) | 1 (0.5) | NA |
| Medical therapy alone | 12 (9.5) | 192 (99.5) | |
| Median time to first event (yr) | 2.59 (1.44, 3.60) | 3.22 (2.15, 4.46) | 0.002 |
| Demographic | Revascularization (n = 106 Patients) | Non-Revascularization (n = 136 Patients) | p-Value |
|---|---|---|---|
| Medication at discharge, n (%) | |||
| Aspirin or P2Y12 inhibitor | 97 (91.5) | 117 (86.0) | 0.186 |
| Statin | 56 (52.8) | 75 (55.1) | 0.720 |
| Beta-blocker | 67 (63.2) | 81 (59.6) | 0.563 |
| Revascularization strategy *, n (%) | |||
| Coronary artery bypass grafting | 2 (1.6) | 0(0) | - |
| Percutaneous coronary intervention | 112 (88.9) | 1 † (0.5) | |
| Drug-eluting stent or bioresorbable vascular scaffold | 71 (63.4) | 1 † (100) | |
| Bare metal stent | 33 (29.5) | 0 (0) | |
| Drug-coated balloon or plain old balloon angioplasty only | 8 (7.1) | 0 (0) | |
| Revascularization | Non-Revascularization | Crude HR (95% CI) | p-Value | Adjusted HR * (95% CI) | p-Value | |||
|---|---|---|---|---|---|---|---|---|
| Total Number of Events (%) | Incidence Rate (Per 100 Person-Years) | Total Number of Events (%) | Incidence Rate (Per 100 Person-Years) | |||||
| Composite outcome | 27 (25.5) | 9.03 | 18 (13.2) | 3.84 | 2.32 (1.28–4.24) | 0.006 | 2.06 (1.07–3.97) | 0.030 |
| Death from any cause | 31 (29.3) | 10.4 | 33 (24.3) | 7.04 | 1.54 (0.94–2.52) | 0.086 | 1.67 (0.95–2.92) | 0.073 |
| Cardiac death | 9 (8.5) | 3.01 | 8 (5.9) | 1.71 | 1.78 (0.69–4.64) | 0.236 | 1.97 (0.69–5.60) | 0.203 |
| Non-fatal MI | 3 (2.8) | 1.00 | 2 (1.5) | 0.43 | 2.32 (0.38–14.0) | 0.359 | 2.61 (0.26–26.0) | 0.413 |
| Cardiac death + non-fatal MI | 12 (11.3) | 4.02 | 10 (7.4) | 2.13 | 1.89 (0.81–4.39) | 0.138 | 2.20 (0.89–5.43) | 0.089 |
| TVF * | 22 (17.5) | 6.34 | 16 (8.3) | 2.46 | 2.53 (1.33–4.82) | 0.005 | 2.19 (1.10–4.37) | 0.026 |
| Revascularization | Non-Revascularization | HR (95% CI) | ||||
|---|---|---|---|---|---|---|
| Total Number of Events (%) | Incidence Rate (Per 100 Person-Years) | Total Number of Events (%) | Incidence Rate (Per 100 Person-Years) | Interaction p-Value | ||
| Mild CKD | 9 (20.9) | 0.14 | 6 (9.7) | 0.038 | 2.49 (0.88–7.03) | 0.540 |
| Moderate to advanced CKD | 4 (17.4) | 0.31 | 5 (10.4) | 0.066 | 2.45 (0.65–9.24) | |
| Dialysis dependent CKD | 14 (35) | 0.40 | 7 (26.9) | 0.47 | 1.13 (0.45–2.83) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jong, C.-B.; Lu, T.-S.; Liu, P.Y.-T.; Chen, J.-W.; Huang, C.-C.; Kao, H.-L. Long-Term Clinical Outcomes of Fractional Flow Reserve-Guided Coronary Artery Revascularization in Chronic Kidney Disease. J. Pers. Med. 2022, 12, 21. https://doi.org/10.3390/jpm12010021
Jong C-B, Lu T-S, Liu PY-T, Chen J-W, Huang C-C, Kao H-L. Long-Term Clinical Outcomes of Fractional Flow Reserve-Guided Coronary Artery Revascularization in Chronic Kidney Disease. Journal of Personalized Medicine. 2022; 12(1):21. https://doi.org/10.3390/jpm12010021
Chicago/Turabian StyleJong, Chien-Boon, Tsui-Shan Lu, Patrick Yan-Tyng Liu, Jeng-Wei Chen, Ching-Chang Huang, and Hsien-Li Kao. 2022. "Long-Term Clinical Outcomes of Fractional Flow Reserve-Guided Coronary Artery Revascularization in Chronic Kidney Disease" Journal of Personalized Medicine 12, no. 1: 21. https://doi.org/10.3390/jpm12010021
APA StyleJong, C.-B., Lu, T.-S., Liu, P. Y.-T., Chen, J.-W., Huang, C.-C., & Kao, H.-L. (2022). Long-Term Clinical Outcomes of Fractional Flow Reserve-Guided Coronary Artery Revascularization in Chronic Kidney Disease. Journal of Personalized Medicine, 12(1), 21. https://doi.org/10.3390/jpm12010021






