Opinion Piece: Patient-Specific Implants May Be the Next Big Thing in Spinal Surgery
Abstract
1. Introduction
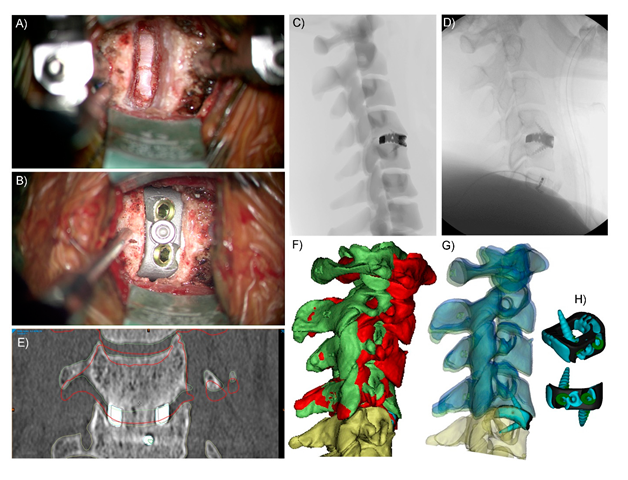
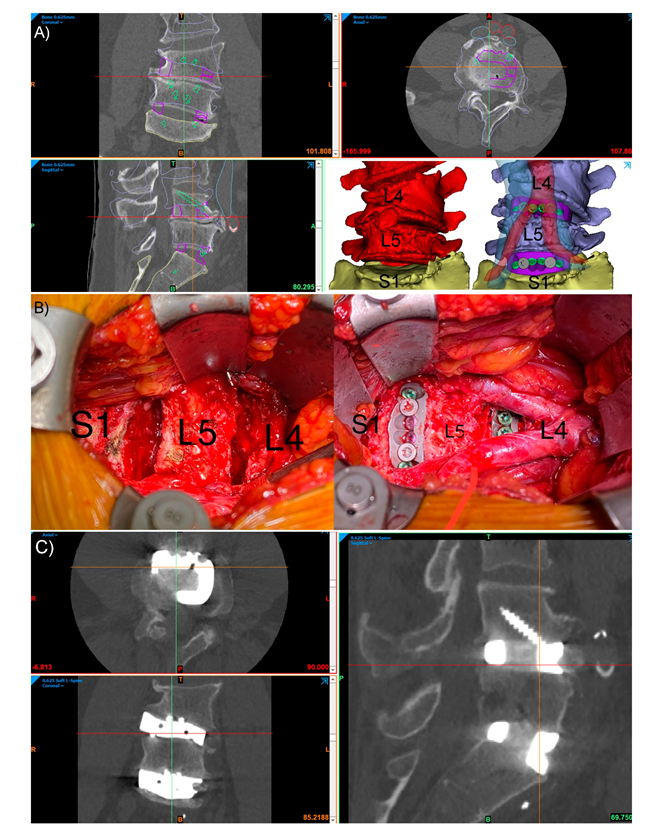
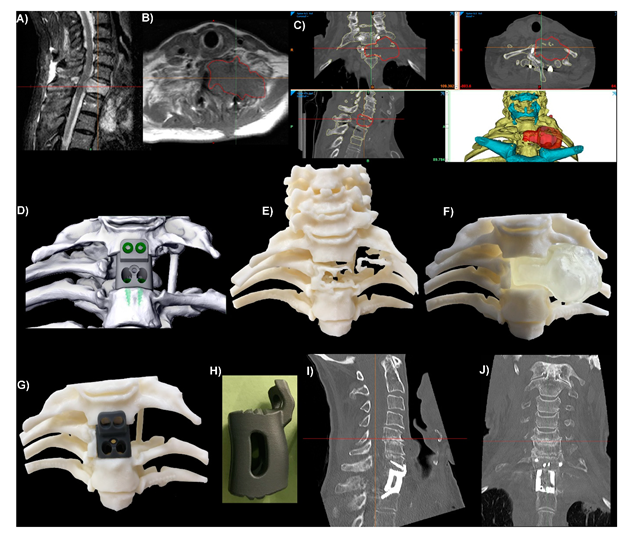
2. Current State of PSI Use in Spinal Surgery
3. Why Should Spinal Surgeons Use PSIs?
4. What Are the Issues?
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Sheha, E.D.; Gandhi, S.D.; Colman, M.W. 3D printing in spine surgery. Ann. Transl. Med. 2019, 7, S164. [Google Scholar] [CrossRef]
- Maniar, R.N.; Singhi, T. Patient specific implants: Scope for the future. Curr. Rev. Musculoskelet. Med. 2014, 7, 125–130. [Google Scholar] [CrossRef]
- Wong, K.C. 3D-printed patient-specific applications in orthopedics. Orthop. Res. Rev. 2016, 8, 57–66. [Google Scholar] [CrossRef] [PubMed]
- D’Urso, P.S.; Askin, G.; Earwaker, J.S.; Merry, G.S.; Thompson, R.G.; Barker, T.M.; Effeney, D.J. Spinal biomodeling. Spine 1999, 24, 1247–1251. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, B.; Mobbs, R.J.; Wu, A.-M.; Phan, K. Systematic review of 3D printing in spinal surgery: The current state of play. J. Spine Surg. 2017, 3, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Girolami, M.; Boriani, S.; Bandiera, S.; Barbanti-Bródano, G.; Ghermandi, R.; Terzi, S.; Tedesco, G.; Evangelisti, G.; Pipola, V.; Gasbarrini, A. Biomimetic 3D-printed custom-made prosthesis for anterior column reconstruction in the thoracolumbar spine: A tailored option following en bloc resection for spinal tumors: Preliminary results on a case-series of 13 patients. Eur. Spine J. 2018, 27, 3073–3083. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.; Li, Z.; Liu, Z.; Liu, X.; Jiang, L.; Yu, M.; Xu, N.; Wu, F.; Dang, L.; Zhou, H.; et al. Upper cervical spine reconstruction using customized 3D-printed vertebral body in 9 patients with primary tumors involving C2. Ann. Transl. Med. 2020, 8, 332. [Google Scholar] [CrossRef]
- Parr, W.C.H.; Burnard, J.L.; Singh, T.; McEvoy, A.; Walsh, W.R.; Mobbs, R.J. C3-C5 Chordoma Resection and Reconstruction with a Three-Dimensional Printed Titanium Patient-Specific Implant. World Neurosurg. 2020, 136, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Willemsen, K.; Nizak, R.; Noordmans, H.J.; Castelein, R.M.; Weinans, H.; Kruyt, M.C. Challenges in the design and regulatory approval of 3D-printed surgical implants: A two-case series. Lancet Digit. Health 2019, 1, e163–e171. [Google Scholar] [CrossRef]
- Yang, X.; Wan, W.; Gong, H.; Xiao, J. Application of Individualized 3D-Printed Artificial Vertebral Body for Cervicothoracic Reconstruction in a Six-Level Recurrent Chordoma. Turk. Neurosurg. 2020, 30, 149–155. [Google Scholar] [CrossRef]
- Chung, K.S.; Shin, D.A.; Kim, K.N.; Ha, Y.; Yoon, D.H.; Yi, S. Vertebral Reconstruction with Customized 3-Dimensional−Printed Spine Implant Replacing Large Vertebral Defect with 3-Year Follow-up. World Neurosurg. 2019, 126, 90–95. [Google Scholar] [CrossRef]
- Chin, B.Z.; Ji, T.; Tang, X.; Yang, R.; Guo, W. Three-Level Lumbar En Bloc Spondylectomy with Three-Dimensional-Printed Vertebrae Reconstruction for Recurrent Giant Cell Tumor. World Neurosurg. 2019, 129, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Choy, W.J.; Parr, W.C.H.; Phan, K.; Walsh, W.R.; Mobbs, R.J. 3-dimensional printing for anterior cervical surgery: A review. J. Spine Surg. 2018, 4, 757–769. [Google Scholar] [CrossRef] [PubMed]
- Burnard, J.L.; Parr, W.C.H.; Choy, W.J.; Walsh, W.R.; Mobbs, R.J. 3D-printed spine surgery implants: A systematic review of the efficacy and clinical safety profile of patient-specific and off-the-shelf devices. Eur. Spine J. 2019. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Wei, F.; Liu, X.; Jiang, L.; Cai, H.; Li, Z.; Yu, M.; Wu, F.; Liu, Z. Reconstruction of the Upper Cervical Spine Using a Personalized 3D-Printed Vertebral Body in an Adolescent With Ewing Sarcoma. Spine 2016, 41, E50–E54. [Google Scholar] [CrossRef]
- Phan, K.; Sgro, A.; Maharaj, M.M.; D’Urso, P.; Mobbs, R.J. Application of a 3D custom printed patient specific spinal implant for C1/2 arthrodesis. J. Spine Surg. 2016, 2, 314–318. [Google Scholar] [CrossRef]
- Wei, R.; Guo, W.; Ji, T.; Zhang, Y.; Liang, H. One-step reconstruction with a 3D-printed, custom-made prosthesis after total en bloc sacrectomy: A technical note. Eur. Spine J. 2017, 26, 1902–1909. [Google Scholar] [CrossRef]
- Siu, T.L.; Rogers, J.M.; Lin, K.; Thompson, R.; Owbridge, M. Custom-Made Titanium 3-Dimensional Printed Interbody Cages for Treatment of Osteoporotic Fracture-Related Spinal Deformity. World Neurosurg. 2018, 111, 1–5. [Google Scholar] [CrossRef]
- Choy, W.J.; Mobbs, R.J.; Wilcox, B.; Phan, S.; Phan, K.; Sutterlin, C.E., 3rd. Reconstruction of Thoracic Spine Using a Personalized 3D-Printed Vertebral Body in Adolescent with T9 Primary Bone Tumor. World Neurosurg. 2017, 1032, e1013–e1017. [Google Scholar] [CrossRef]
- Parr, W.C.H.; Burnard, J.L.; Wilson, P.J.; Mobbs, R.J. 3D printed anatomical (bio)models in spine surgery: Clinical benefits and value to health care providers. J. Spine Surg. 2019. [Google Scholar] [CrossRef]
- Mobbs, R.J.; Parr, W.C.H.; Choy, W.J.; McEvoy, A.; Walsh, W.R.; Phan, K. Anterior Lumbar Interbody Fusion Using a Personalized Approach: Is Custom the Future of Implants for Anterior Lumbar Interbody Fusion Surgery? World Neurosurg. 2019. [Google Scholar] [CrossRef]
- Hartman, J. Anatomy and clinical significance of the uncinate process and uncovertebral joint: A comprehensive review. Clin. Anat. 2014, 27, 431–440. [Google Scholar] [CrossRef]
- Devereaux, M.W. Anatomy and examination of the spine. Neurol. Clin. 2007, 25, 331–351. [Google Scholar] [CrossRef] [PubMed]
- Saker, E.; Graham, R.A.; Nicholas, R.; D’Antoni, A.V.; Loukas, M.; Oskouian, R.J.; Tubbs, R.S. Ligaments of the Costovertebral Joints including Biomechanics, Innervations, and Clinical Applications: A Comprehensive Review with Application to Approaches to the Thoracic Spine. Cureus 2016, 8, e874. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.-C.; Chou, P.-H.; Lin, H.-H.; Wang, S.-T.; Liu, C.-L.; Chang, M.-C. Risk Factors of Cage Subsidence in Patients Received Minimally Invasive Transforaminal Lumbar Interbody Fusion. Spine 2020, 45, E1279–E1285. [Google Scholar] [CrossRef] [PubMed]
- Amin, T.; Lin, H.; Parr, W.C.H.; Lim, P.; Mobbs, R.J. Revision of a Failed C5-7 Corpectomy Complicated by Esophageal Fistula Using a 3-Dimensional−Printed Zero-Profile Patient-Specific Implant: A Technical Case Report. World Neurosurg. 2021, 151, 29–38. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Zhao, Y.; Liu, J.; Xiao, S.; Mao, K. Multilevel 3D Printing Implant for Reconstructing Cervical Spine With Metastatic Papillary Thyroid Carcinoma. Spine 2017, 42, E1326–E1330. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, L.; Ma, Y.; Zhang, K.; Li, G.; Lu, B.; Lu, C.; Chen, C.; Wang, L.; Wang, H.; et al. 3D-Printed Titanium Cage with PVA-Vancomycin Coating Prevents Surgical Site Infections (SSIs). Macromol. Biosci. 2020, 20, e1900394. [Google Scholar] [CrossRef]
- Dong, J.; Zhang, S.; Liu, H.; Li, X.; Liu, Y.; Du, Y. Novel alternative therapy for spinal tuberculosis during surgery: Reconstructing with anti-tuberculosis bioactivity implants. Expert Opin. Drug Deliv. 2014, 11, 299–305. [Google Scholar] [CrossRef]
- Ogura, T.; Le, K.; Merkely, G.; Bryant, T.; Minas, T. A high level of satisfaction after bicompartmental individualized knee arthroplasty with patient-specific implants and instruments. Knee Surg. Sports Traumatol. Arthrosc. 2019, 27, 1487–1496. [Google Scholar] [CrossRef] [PubMed]
- Arbab, D.; Reimann, P.; Brucker, M.; Bouillon, B.; Lüring, C. Alignment in total knee arthroplasty—A comparison of patient-specific implants with the conventional technique. Knee 2018, 25, 882–887. [Google Scholar] [CrossRef]
- Schroeder, L.; Martin, G. In Vivo Tibial Fit and Rotational Analysis of a Customized, Patient-Specific TKA versus Off-the-Shelf TKA. J Knee Surg. 2019, 32, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Haglin, J.M.; Eltorai, A.E.M.; Gil, J.A.; Marcaccio, S.E.; Botero-Hincapie, J.; Daniels, A.H. Patient-Specific Orthopaedic Implants. Orthop. Surg. 2016, 8, 417–424. [Google Scholar] [CrossRef]
- Schwarzkopf, R.; Brodsky, M.; Garcia, G.A.; Gomoll, A.H. Surgical and Functional Outcomes in Patients Undergoing Total Knee Replacement With Patient-Specific Implants Compared With “Off-the-Shelf” Implants. Orthop. J. Sports Med. 2015, 3. [Google Scholar] [CrossRef]
- Rojanasopondist, P.; Galea, V.P.; Connelly, J.W.; Matuszak, S.J.; Rolfson, O.; Bragdon, C.R.; Malchau, H. What Preoperative Factors are Associated With Not Achieving a Minimum Clinically Important Difference After THA? Findings from an International Multicenter Study. Clin. Orthop. Relat. Res. 2019, 477, 1301–1312. [Google Scholar] [CrossRef]
- Vogenberg, F.R.; Isaacson Barash, C.; Pursel, M. Personalized medicine: Part 1: Evolution and development into theranostics. Pharm. Ther. 2010, 35, 560–576. [Google Scholar]
- Mobbs, R.J.; Ho, D.; Choy, W.J.; Betteridge, C.; Lin, H. COVID-19 is shifting the adoption of wearable monitoring and telemedicine (WearTel) in the delivery of healthcare: Opinion piece. Ann. Transl. Med. 2020, 8, 1285. [Google Scholar] [CrossRef] [PubMed]
- Barberan-Garcia, A.; Ubré, M.; Roca, J.; Lacy, A.M.; Burgos, F.; Risco, R.; Momblán, D.; Balust, J.; Blanco, I.; Martínez-Pallí, G. Personalised Prehabilitation in High-risk Patients Undergoing Elective Major Abdominal Surgery: A Randomized Blinded Controlled Trial. Ann. Surg. 2018, 267, 50–56. [Google Scholar] [CrossRef]
- van der Meij, E.; Anema, J.R.; Leclercq, W.K.G.; Bongers, M.Y.; Consten, E.C.J.; Schraffordt Koops, S.E.; van de Ven, P.M.; Terwee, C.B.; van Dongen, J.M.; Schaafsma, F.G.; et al. Personalised perioperative care by e-health after intermediate-grade abdominal surgery: A multicentre, single-blind, randomised, placebo-controlled trial. Lancet 2018, 392, 51–59. [Google Scholar] [CrossRef]
- Nicolaidis, S. Personalized medicine in neurosurgery. Metabolism 2013, 62 (Suppl. 1), S45–S48. [Google Scholar] [CrossRef]
- Tarpada, S.P.; Morris, M.T.; Burton, D.A. Spinal fusion surgery: A historical perspective. J. Orthop. 2016, 14, 134–136. [Google Scholar] [CrossRef] [PubMed]
- McClelland, S., 3rd; Goldstein, J.A. Minimally Invasive versus Open Spine Surgery: What Does the Best Evidence Tell Us? J. Neurosci. Rural Pract. 2017, 8, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Meheux, C.J.; Park, K.J.; Clyburn, T.A. A Retrospective Study Comparing a Patient-specific Design Total Knee Arthroplasty With an Off-the-Shelf Design: Unexpected Catastrophic Failure Seen in the Early Patient-specific Design. J. Am. Acad. Orthop. Surg. Glob. Res. Rev. 2019, 3. [Google Scholar] [CrossRef]
- Tong, Y.; Kaplan, D.J.; Spivak, J.M.; Bendo, J.A. Three-dimensional printing in spine surgery: A review of current applications. Spine J. 2020, 20, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Mobbs, R.J.; Phan, K.; Malham, G.; Seex, K.; Rao, P.J. Lumbar interbody fusion: Techniques, indications and comparison of interbody fusion options including PLIF, TLIF, MI-TLIF, OLIF/ATP, LLIF and ALIF. J. Spine Surg. 2015, 1, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Martelli, N.; Serrano, C.; van den Brink, H.; Pineau, J.; Prognon, P.; Borget, I.; El Batti, S. Advantages and disadvantages of 3-dimensional printing in surgery: A systematic review. Surgery 2016, 159, 1485–1500. [Google Scholar] [CrossRef]
| Advantages | Disadvantages |
|---|---|
| Easier Implantation | Lack of Quality Data |
| Minimal Endplate Preparation | Research Challenges |
| Improved Device–Bone Load Distribution | Skilled Labour and Equipment Requirements |
| Improved Primary Stabilisation | Increased Preoperative Planning |
| Range of Customisable Features | Reduced Intraoperative Flexibility |
| Enhanced Osseointegration | Multiple Implants Need to be Produced Per Case |
| Minimised Operative Time | Off the Shelf Devices Often Also Kept on Hand |
| Tailor to Specific Operative Challenges and Clinical Scenarios | Challenging Regulatory Environment |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amin, T.; Parr, W.C.H.; Mobbs, R.J. Opinion Piece: Patient-Specific Implants May Be the Next Big Thing in Spinal Surgery. J. Pers. Med. 2021, 11, 498. https://doi.org/10.3390/jpm11060498
Amin T, Parr WCH, Mobbs RJ. Opinion Piece: Patient-Specific Implants May Be the Next Big Thing in Spinal Surgery. Journal of Personalized Medicine. 2021; 11(6):498. https://doi.org/10.3390/jpm11060498
Chicago/Turabian StyleAmin, Tajrian, William C.H. Parr, and Ralph J. Mobbs. 2021. "Opinion Piece: Patient-Specific Implants May Be the Next Big Thing in Spinal Surgery" Journal of Personalized Medicine 11, no. 6: 498. https://doi.org/10.3390/jpm11060498
APA StyleAmin, T., Parr, W. C. H., & Mobbs, R. J. (2021). Opinion Piece: Patient-Specific Implants May Be the Next Big Thing in Spinal Surgery. Journal of Personalized Medicine, 11(6), 498. https://doi.org/10.3390/jpm11060498







