Effects of Respiratory Muscle Training on Baroreflex Sensitivity, Respiratory Function, and Serum Oxidative Stress in Acute Cervical Spinal Cord Injury
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Ethical Approval and Consent to Participate
2.3. Assessment of Respiratory Muscle and Lung Function
2.4. Respiratory Muscle Training Protocol
2.5. Assessment of Autonomic Function
2.6. Blood Sampling and Assessment of Oxidative Stress
2.7. Clinical Manifestations and Quality-of-Life Assessment
2.8. Statistics
3. Results
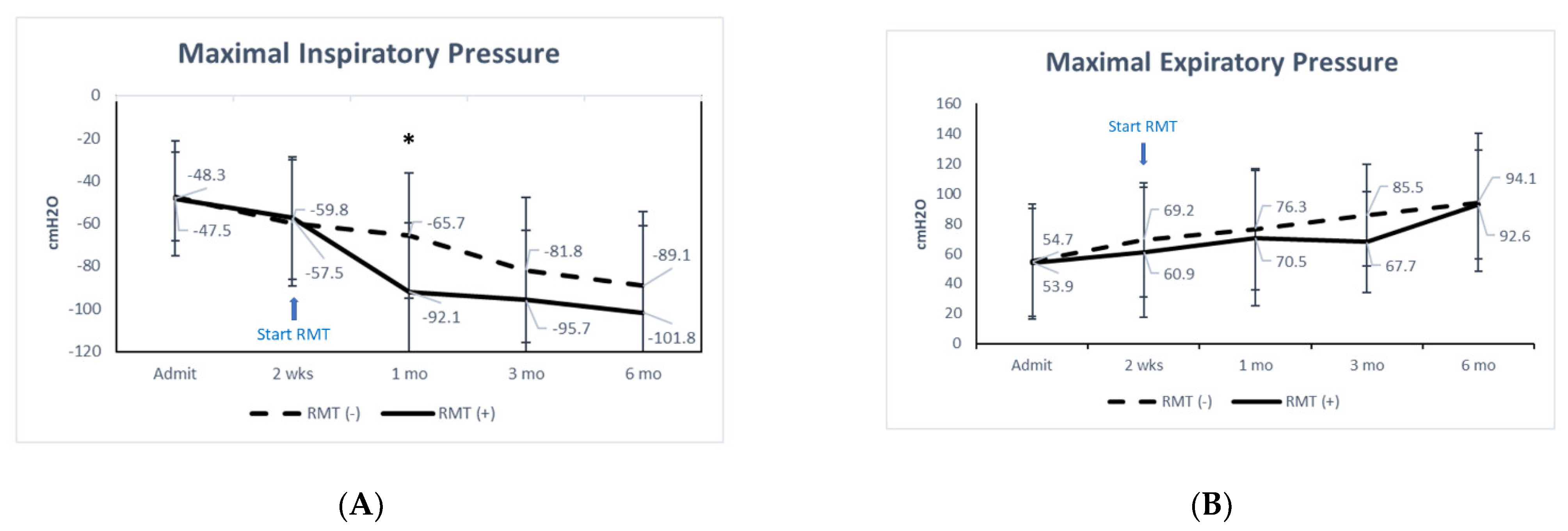
3.1. Baroreflex and Pulmonary Function
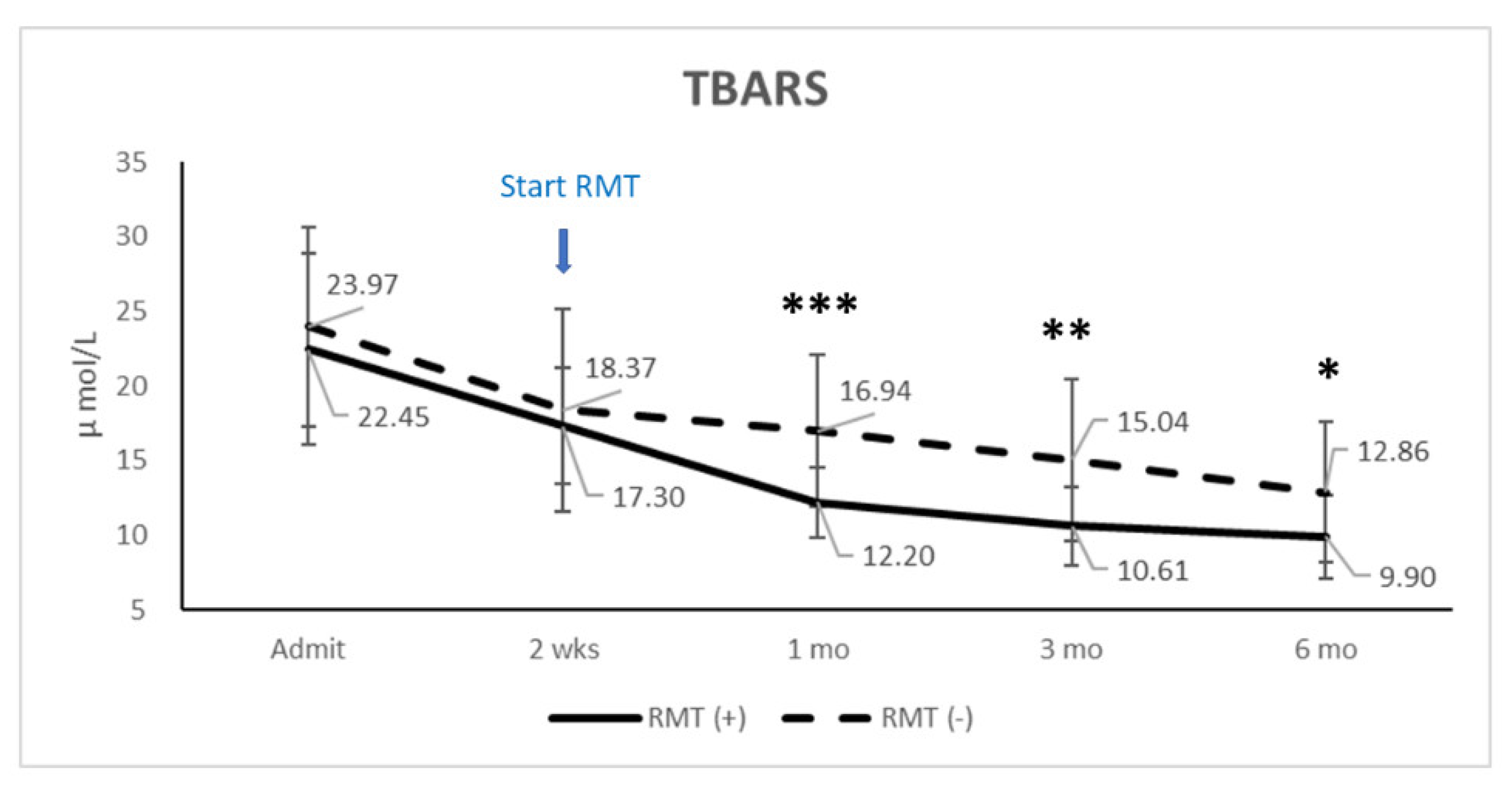
3.2. TBARS
4. Discussion
4.1. Autonomic Function
4.2. Respiratory Function
4.3. Injury Severity and Other Co-Mobility
4.4. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Garshick, E.; Kelley, A.; Cohen, S.A.; Garrison, A.; Tun, C.G.; Gagnon, D.; Brown, R. A prospective assessment of mortality in chronic spinal cord injury. Spinal Cord 2005, 43, 408–416. [Google Scholar] [CrossRef]
- Middleton, J.W.; Dayton, A.; Walsh, J.; Rutkowski, S.B.; Leong, G.; Duong, S. Life expectancy after spinal cord injury: A 50-year study. Spinal Cord 2012, 50, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Strauss, D.J.; Devivo, M.J.; Paculdo, D.R.; Shavelle, R.M. Trends in life expectancy after spinal cord injury. Arch. Phys. Med. Rehabil. 2006, 87, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.; DiMarco, A.F.; Hoit, J.D.; Garshick, E. Respiratory dysfunction and management in spinal cord injury. Respir. Care 2006, 51, 853–868; discussion 869–870. [Google Scholar]
- Aarabi, B.; Harrop, J.S.; Tator, C.H.; Alexander, M.; Dettori, J.R.; Grossman, R.G.; Fehlings, M.G.; Mirvis, S.E.; Shanmuganathan, K.; Zacherl, K.M.; et al. Predictors of pulmonary complications in blunt traumatic spinal cord injury. J. Neurosurg. Spine 2012, 17, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Draghici, A.E.; Taylor, J.A. Baroreflex autonomic control in human spinal cord injury: Physiology, measurement, and potential alterations. Auton Neurosci. 2018, 209, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.B.; Brown, R.; Tun, C.G.; Gagnon, D.; Garshick, E. Determinants of forced expiratory volume in 1 second (fev1), forced vital capacity (fvc), and fev1/fvc in chronic spinal cord injury. Arch. Phys. Med. Rehabil. 2006, 87, 1327–1333. [Google Scholar] [CrossRef][Green Version]
- Fishburn, M.J.; Marino, R.J.; Ditunno, J.F., Jr. Atelectasis and pneumonia in acute spinal cord injury. Arch. Phys. Med. Rehabil. 1990, 71, 197–200. [Google Scholar]
- de Groot, P.C.; Bleeker, M.W.; van Kuppevelt, D.H.; van der Woude, L.H.; Hopman, M.T. Rapid and extensive arterial adaptations after spinal cord injury. Arch. Phys. Med. Rehabil. 2006, 87, 688–696. [Google Scholar] [CrossRef]
- Raurich, J.M.; Rialp, G.; Llompart-Pou, J.A.; Ayestaran, I.; Perez-Barcena, J.; Ibanez, J. Respiratory co(2) response in acute cervical spinal cord injury (co(2) response in spinal cord injury). Spinal Cord 2014, 52, 39–43. [Google Scholar] [CrossRef]
- Berney, S.; Bragge, P.; Granger, C.; Opdam, H.; Denehy, L. The acute respiratory management of cervical spinal cord injury in the first 6 weeks after injury: A systematic review. Spinal Cord 2011, 49, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, M.B.; Nantwi, K.; Goshgarian, H.G. Effect of spinal cord injury on the respiratory system: Basic research and current clinical treatment options. J. Spinal Cord Med. 2007, 30, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Baydur, A.; Adkins, R.H.; Milic-Emili, J. Lung mechanics in individuals with spinal cord injury: Effects of injury level and posture. J. Appl. Physiol. (1985) 2001, 90, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Linn, W.S.; Adkins, R.H.; Gong, H., Jr.; Waters, R.L. Pulmonary function in chronic spinal cord injury: A cross-sectional survey of 222 southern california adult outpatients. Arch. Phys. Med. Rehabil. 2000, 81, 757–763. [Google Scholar] [CrossRef]
- Nogues, M.A.; Benarroch, E. Abnormalities of respiratory control and the respiratory motor unit. Neurologist 2008, 14, 273–288. [Google Scholar] [CrossRef] [PubMed]
- Legg Ditterline, B.E.; Aslan, S.C.; Randall, D.C.; Harkema, S.J.; Castillo, C.; Ovechkin, A.V. Effects of respiratory training on heart rate variability and baroreflex sensitivity in individuals with chronic spinal cord injury. Arch. Phys. Med. Rehabil. 2018, 99, 423–432. [Google Scholar] [CrossRef]
- Berlowitz, D.J.; Tamplin, J. Respiratory muscle training for cervical spinal cord injury. Cochrane Database Syst. Rev. 2013, CD008507. [Google Scholar] [CrossRef]
- Karadalli, M.N.; Bosnak-Guclu, M.; Camcioglu, B.; Kokturk, N.; Turktas, H. Effects of inspiratory muscle training in subjects with sarcoidosis: A randomized controlled clinical trial. Respir. Care 2016, 61, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Berlowitz, D.J.; Wadsworth, B.; Ross, J. Respiratory problems and management in people with spinal cord injury. Breathe (Sheff) 2016, 12, 328–340. [Google Scholar] [CrossRef]
- Warburton, D.E.; Eng, J.J.; Krassioukov, A.; Sproule, S.; the, S.R.T. Cardiovascular health and exercise rehabilitation in spinal cord injury. Top. Spinal Cord Inj. Rehabil. 2007, 13, 98–122. [Google Scholar] [CrossRef] [PubMed]
- Visavadiya, N.P.; Patel, S.P.; VanRooyen, J.L.; Sullivan, P.G.; Rabchevsky, A.G. Cellular and subcellular oxidative stress parameters following severe spinal cord injury. Redox Biol. 2016, 8, 59–67. [Google Scholar] [CrossRef]
- Azbill, R.D.; Mu, X.; Bruce-Keller, A.J.; Mattson, M.P.; Springer, J.E. Impaired mitochondrial function, oxidative stress and altered antioxidant enzyme activities following traumatic spinal cord injury. Brain Res. 1997, 765, 283–290. [Google Scholar] [CrossRef]
- Vaziri, N.D.; Lee, Y.S.; Lin, C.Y.; Lin, V.W.; Sindhu, R.K. Nad(p)h oxidase, superoxide dismutase, catalase, glutathione peroxidase and nitric oxide synthase expression in subacute spinal cord injury. Brain Res. 2004, 995, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Bissett, B.M.; Leditschke, I.A.; Neeman, T.; Boots, R.; Paratz, J. Inspiratory muscle training to enhance recovery from mechanical ventilation: A randomised trial. Thorax 2016, 71, 812–819. [Google Scholar] [CrossRef]
- Marini, J.J.; Smith, T.C.; Lamb, V.J. External work output and force generation during synchronized intermittent mechanical ventilation. Effect of machine assistance on breathing effort. Am. Rev. Respir. Dis. 1988, 138, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.J. Measurement of respiratory muscle strength. Respir. Med. 1995, 89, 529–535. [Google Scholar] [CrossRef]
- Ratnovsky, A.; Elad, D.; Halpern, P. Mechanics of respiratory muscles. Respir. Physiol. Neurobiol. 2008, 163, 82–89. [Google Scholar]
- MacIntyre, N.R.; Cook, D.J.; Ely, E.W., Jr.; Epstein, S.K.; Fink, J.B.; Heffner, J.E.; Hess, D.; Hubmayer, R.D.; Scheinhorn, D.J.; American College of Chest, P.; et al. Evidence-based guidelines for weaning and discontinuing ventilatory support: A collective task force facilitated by the american college of chest physicians; the american association for respiratory care; and the american college of critical care medicine. Chest 2001, 120, 375S–395S. [Google Scholar]
- Yang, K.L. Reproducibility of weaning parameters. A need for standardization. Chest 1992, 102, 1829–1832. [Google Scholar] [CrossRef]
- Parreiras de Menezes, K.K.; Nascimento, L.R.; Ada, L.; Avelino, P.R.; Polese, J.C.; Mota Alvarenga, M.T.; Barbosa, M.H.; Teixeira-Salmela, L.F. High-intensity respiratory muscle training improves strength and dyspnea poststroke: A double-blind randomized trial. Arch. Phys. Med. Rehabil. 2019, 100, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Marco, E.; Ramirez-Sarmiento, A.L.; Coloma, A.; Sartor, M.; Comin-Colet, J.; Vila, J.; Enjuanes, C.; Bruguera, J.; Escalada, F.; Gea, J.; et al. High-intensity vs. Sham inspiratory muscle training in patients with chronic heart failure: A prospective randomized trial. Eur. J. Heart Fail. 2013, 15, 892–901. [Google Scholar] [CrossRef] [PubMed]
- Menezes, K.K.P.; Nascimento, L.R.; Polese, J.C.; Ada, L.; Teixeira-Salmela, L.F. Effect of high-intensity home-based respiratory muscle training on strength of respiratory muscles following a stroke: A protocol for a randomized controlled trial. Braz J. Phys. Ther. 2017, 21, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Muza, S.R.; Silverman, M.T.; Gilmore, G.C.; Hellerstein, H.K.; Kelsen, S.G. Comparison of scales used to quantitate the sense of effort to breathe in patients with chronic obstructive pulmonary disease. Am. Rev. Respir. Dis. 1990, 141, 909–913. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.J.; Close, L.; Gillon, S.C.; Molassiotis, A.; Lee, P.H.; Farquhar, M.C.; Breathlessness Research Interest, G. Use of the modified borg scale and numerical rating scale to measure chronic breathlessness: A pooled data analysis. Eur. Respir. J. 2016, 47, 1861–1864. [Google Scholar] [CrossRef] [PubMed]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Low, P.A. Testing the autonomic nervous system. Semin. Neurol. 2003, 23, 407–421. [Google Scholar] [PubMed]
- Parati, G.; Di Rienzo, M.; Mancia, G. How to measure baroreflex sensitivity: From the cardiovascular laboratory to daily life. J. Hypertens. 2000, 18, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Malberg, H.; Wessel, N.; Hasart, A.; Osterziel, K.J.; Voss, A. Advanced analysis of spontaneous baroreflex sensitivity, blood pressure and heart rate variability in patients with dilated cardiomyopathy. Clin. Sci. (London) 2002, 102, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Task force of the european society of cardiology and the north american society of pacing and electrophysiology. Circulation 1996, 93, 1043–1065. [CrossRef]
- Zimmermann, M.; Meyer, N. Annexin v/7-aad staining in keratinocytes. Methods Mol. Biol. 2011, 740, 57–63. [Google Scholar] [PubMed]
- McCord, J.M.; Fridovich, I. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J. Biol. Chem. 1969, 244, 6049–6055. [Google Scholar] [CrossRef]
- Benzel, E.C.; Lancon, J.; Kesterson, L.; Hadden, T. Cervical laminectomy and dentate ligament section for cervical spondylotic myelopathy. J. Spinal Disord. 1991, 4, 286–295. [Google Scholar] [CrossRef]
- Baker, S.P.; O’Neill, B.; Haddon, W., Jr.; Long, W.B. The injury severity score: A method for describing patients with multiple injuries and evaluating emergency care. J. Trauma 1974, 14, 187–196. [Google Scholar] [CrossRef]
- Beck, A.T.; Brown, G.K.; Steer, R.A. Psychometric characteristics of the scale for suicide ideation with psychiatric outpatients. Behav. Res. Ther. 1997, 35, 1039–1046. [Google Scholar] [CrossRef]
- Ware, J.E., Jr.; Sherbourne, C.D. The mos 36-item short-form health survey (sf-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Aslan, S.C.; Randall, D.C.; Krassioukov, A.V.; Phillips, A.; Ovechkin, A.V. Respiratory training improves blood pressure regulation in individuals with chronic spinal cord injury. Arch. Phys. Med. Rehabil. 2016, 97, 964–973. [Google Scholar] [CrossRef]
- Studinger, P.; Goldstein, R.; Taylor, J.A. Mechanical and neural contributions to hysteresis in the cardiac vagal limb of the arterial baroreflex. J. Physiol. 2007, 583, 1041–1048. [Google Scholar] [CrossRef]
- Phillips, A.A.; Krassioukov, A.V.; Ainslie, P.N.; Cote, A.T.; Warburton, D.E. Increased central arterial stiffness explains baroreflex dysfunction in spinal cord injury. J. Neurotrauma 2014, 31, 1122–1128. [Google Scholar] [CrossRef]
- Jan, Y.K.; Anderson, M.; Soltani, J.; Burns, S.; Foreman, R.D. Comparison of changes in heart rate variability and sacral skin perfusion in response to postural changes in people with spinal cord injury. J. Rehabil. Res. Dev. 2013, 50, 203–214. [Google Scholar] [CrossRef]
- Burgess, D.E.; Hundley, J.C.; Li, S.G.; Randall, D.C.; Brown, D.R. First-order differential-delay equation for the baroreflex predicts the 0.4-hz blood pressure rhythm in rats. Am. J. Physiol. 1997, 273, R1878–R1884. [Google Scholar] [CrossRef]
- Inoue, K.; Ogata, H.; Hayano, J.; Miyake, S.; Kamada, T.; Kuno, M.; Kumashiro, M. Assessment of autonomic function in traumatic quadriplegic and paraplegic patients by spectral analysis of heart rate variability. J. Auton. Nerv. Syst. 1995, 54, 225–234. [Google Scholar] [CrossRef]
- Ondrusova, K.; Svacinova, J.; Javorka, M.; Novak, J.; Novakova, M.; Novakova, Z. Impaired baroreflex function during orthostatic challenge in patients after spinal cord injury. J. Neurotrauma 2017, 34, 3381–3387. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.C.; Chen, K.Y.; Lin, Y.T.; Chen, W.F.; Liaw, M.Y.; Lin, Y.J.; Shih, F.Y.; Hsu, S.Y.; Tsai, N.W.; Lin, M.C.; et al. Factors associated with prolonged mechanical ventilation and re-ventilation in acute cervical spinal cord injury patients. Spine (Phila Pa 1976) 2020, 45, E515–E524. [Google Scholar] [CrossRef]
- Ushiku, C.; Suda, K.; Matsumoto, S.; Komatsu, M.; Takahata, M.; Iwasaki, N.; Minami, A. Time course of respiratory dysfunction and motor paralysis for 12 weeks in cervical spinal cord injury without bone injury. Spine Surg. Relat. Res. 2019, 3, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Schilero, G.J.; Spungen, A.M.; Bauman, W.A.; Radulovic, M.; Lesser, M. Pulmonary function and spinal cord injury. Respir. Physiol. Neurobiol. 2009, 166, 129–141. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, A.F. Restoration of respiratory muscle function following spinal cord injury. Review of electrical and magnetic stimulation techniques. Respir. Physiol. Neurobiol. 2005, 147, 273–287. [Google Scholar] [CrossRef]
- Khong, P.; Lazzaro, A.; Mobbs, R. Phrenic nerve stimulation: The australian experience. J. Clin. Neurosci. 2010, 17, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Onders, R.P.; Dimarco, A.F.; Ignagni, A.R.; Aiyar, H.; Mortimer, J.T. Mapping the phrenic nerve motor point: The key to a successful laparoscopic diaphragm pacing system in the first human series. Surgery 2004, 136, 819–826. [Google Scholar] [CrossRef]
- Raab, A.M.; Krebs, J.; Pfister, M.; Perret, C.; Hopman, M.; Mueller, G. Respiratory muscle training in individuals with spinal cord injury: Effect of training intensity and -volume on improvements in respiratory muscle strength. Spinal Cord 2019, 57, 482–489. [Google Scholar] [CrossRef]
- Shin, J.C.; Han, E.Y.; Cho, K.H.; Im, S.H. Improvement in pulmonary function with short-term rehabilitation treatment in spinal cord injury patients. Sci. Rep. 2019, 9, 17091. [Google Scholar] [CrossRef]
- Jaja, B.N.R.; Jiang, F.; Badhiwala, J.H.; Schar, R.; Kurpad, S.; Grossman, R.G.; Harrop, J.S.; Guest, J.D.; Toups, E.G.; Shaffrey, C.I.; et al. Association of pneumonia, wound infection, and sepsis with clinical outcomes after acute traumatic spinal cord injury. J. Neurotrauma 2019, 36, 3044–3050. [Google Scholar] [CrossRef] [PubMed]
- Khorasanizadeh, M.; Yousefifard, M.; Eskian, M.; Lu, Y.; Chalangari, M.; Harrop, J.S.; Jazayeri, S.B.; Seyedpour, S.; Khodaei, B.; Hosseini, M.; et al. Neurological recovery following traumatic spinal cord injury: A systematic review and meta-analysis. J. Neurosurg. Spine 2019, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Stolzmann, K.L.; Gagnon, D.R.; Brown, R.; Tun, C.G.; Garshick, E. Longitudinal change in fev1 and fvc in chronic spinal cord injury. Am. J. Respir. Crit. Care Med. 2008, 177, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Almenoff, P.L.; Spungen, A.M.; Lesser, M.; Bauman, W.A. Pulmonary function survey in spinal cord injury: Influences of smoking and level and completeness of injury. Lung 1995, 173, 297–306. [Google Scholar] [CrossRef]
- Torres, S.; Salgado-Ceballos, H.; Torres, J.L.; Orozco-Suarez, S.; Diaz-Ruiz, A.; Martinez, A.; Rivera-Cruz, M.; Rios, C.; Lara, A.; Collado, C.; et al. Early metabolic reactivation versus antioxidant therapy after a traumatic spinal cord injury in adult rats. Neuropathology 2010, 30, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Kahraman, S.; Duz, B.; Kayali, H.; Korkmaz, A.; Oter, S.; Aydin, A.; Sayal, A. Effects of methylprednisolone and hyperbaric oxygen on oxidative status after experimental spinal cord injury: A comparative study in rats. Neurochem. Res. 2007, 32, 1547–1551. [Google Scholar] [CrossRef] [PubMed]
- Muballe, K.D.; Sewani-Rusike, C.R.; Longo-Mbenza, B.; Iputo, J. Predictors of recovery in moderate to severe traumatic brain injury. J. Neurosurg. 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.C.; Lin, Y.J.; Shih, F.Y.; Chang, H.W.; Su, Y.J.; Cheng, B.C.; Su, C.M.; Tsai, N.W.; Chang, Y.T.; Kwan, A.L.; et al. The role of serial oxidative stress levels in acute traumatic brain injury and as predictors of outcome. World Neurosurg. 2016, 87, 463–470. [Google Scholar] [CrossRef]
- Yang, T.; Kong, B.; Gu, J.W.; Kuang, Y.Q.; Cheng, L.; Yang, W.T.; Xia, X.; Shu, H.F. Anti-apoptotic and anti-oxidative roles of quercetin after traumatic brain injury. Cell. Mol. Neurobiol. 2014, 34, 797–804. [Google Scholar] [CrossRef]
- Wagner, K.R.; Hua, Y.; de Courten-Myers, G.M.; Broderick, J.P.; Nishimura, R.N.; Lu, S.Y.; Dwyer, B.E. Tin-mesoporphyrin, a potent heme oxygenase inhibitor, for treatment of intracerebral hemorrhage: In vivo and in vitro studies. Cell. Mol. Biol. (Noisy-le-grand) 2000, 46, 597–608. [Google Scholar]
- Chen, C.Y.; Wu, R.W.; Tsai, N.W.; Lee, M.S.; Lin, W.C.; Hsu, M.C.; Huang, C.C.; Lai, Y.R.; Kung, C.T.; Wang, H.C.; et al. Increased circulating endothelial progenitor cells and improved short-term outcomes in acute non-cardioembolic stroke after hyperbaric oxygen therapy. J. Transl. Med. 2018, 16, 255. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.C.; Chou, K.H.; Lee, P.L.; Huang, Y.C.; Tsai, N.W.; Chen, H.L.; Cheng, K.Y.; Wang, H.C.; Lin, T.K.; Li, S.H.; et al. Brain mediators of systemic oxidative stress on perceptual impairments in parkinson’s disease. J. Transl. Med. 2015, 13, 386. [Google Scholar] [CrossRef] [PubMed]
| Parameters | RMT (N = 20) | No RMT (N = 24) | p-Value | Odds Ratio | 95% CI (Lower, Upper) |
|---|---|---|---|---|---|
| Age (y) | 46.1 ± 14.0 | 44.8 ± 15.5 | 0.788 | −7.857, 10.291 | |
| Male | 15 | 21 | 0.436 | 1.500 | 0.774, 2.906 |
| Body mass index | 25.8 ± 7.6 | 25.3 ± 4.8 | 0.762 | −3.240, 4.391 | |
| Level of SCI | 1.000 | 1.133 | 0.548, 2.343 | ||
| 15 | 19 | |||
| 5 | 5 | |||
| Injury Severity Score at admission | |||||
| 20 (16, 21) | 17 (16, 20) | 0.196 | ||
| 4 (4, 4) | 4 (4, 4) | 0.944 | ||
| 0 (0, 0) | 0 (0, 0) | 0.563 | ||
| Neurosurgical intervention | |||||
| 18 | 20 | 0.673 | 0.704 | 0.216, 2.291 |
| 12 | 11 | 0.382 | 0.730 | 0.373, 1.428 |
| 15 | 17 | 1.000 | 0.889 | 0.414, 1.909 |
| JOA score at admission, Median (IQR) | 5 (4, 6) | 6 (4, 12) | 0.539 | ||
| JOA recovery rate at 6 months (%) | 53.2 ± 33.3 | 62.8 ± 29.9 | 0.333 | −29.266, 10.154 | |
| ICU stay (days) | 4.8 ± 3.3 | 5.6 ± 5.0 | 0.646 | −3.960, 2.494 | |
| Hospitalization in acute department (days) | 12.1 ± 5.3 | 20.5 ± 12.5 | 0.012 | −15.130, −1.519 | |
| SF-36 before discharged | |||||
| 17.6 ± 12.0 | 20.3 ± 19.4 | 0.233 | −5.461, 13.857 | |
| 39.6 ± 16.4 | 47.8 ± 12.2 | 0.113 | −1.455, 11.033 | |
| SF-36 at 6 months | |||||
| 13.5 ± 8.5 | 32.5 ± 25.8 | 0.046 | −37.679, −0.447 | |
| 32.4 ± 5.3 | 50.2 ± 16.2 | 0.006 | −29.450, −6.121 | |
| BDI before discharged | 27.6 ± 10.5 | 22.3 ± 9.2 | 0.112 | −1.315, 12.065 | |
| BDI at 6 months | 25.7 ± 12.2 | 19.6 ± 13.0 | 0.259 | −4.761, 16.918 | |
| Respiratory function at time before RMT | |||||
| −57.5 ± 28.8 | −59.8 ± 38.1 | 0.355 | −16.573, 21.263 | |
| 60.9 ± 38.1 | 69.2 ± 43.1 | 0.946 | −34.624, 18.103 | |
| 15.3 ± 3.7 | 14.3 ± 4.6 | 0.885 | −3.383, 6.379 | |
| 10.3 ± 3.1 | 10.6 ± 3.0 | 0.702 | −5.404, 4.297 | |
| 0.75 ± 0.22 | 0.80 ± 0.29 | 0.379 | −0.424, 0.195 | |
| 31.7 ± 13.6 | 22.7 ± 14.4 | 0.752 | −14.537, 25.990 | |
| TBARS at admission | 22.45 ± 6.40 | 23.97 ± 6.67 | 0.459 | −5.625, 2.586 | |
| TBARS at 6 months | 9.90 ± 2.78 | 12.86 ± 4.72 | 0.024 | −5.511, −0.404 |
| RMT Group (n = 20) | Non-RMT Group (n = 24) | p¥ between RMT and without RMT Groups at the Time of 6 Months Follow-Up | |||
|---|---|---|---|---|---|
| Baseline | Follow-Up | Baseline | Follow-Up | ||
| Cardiovascular autonomic function | |||||
| 9.76 ± 2.07 | 13.09 ± 2.88 * | 9.65 ± 3.85 | 10.30 ± 3.03 | 0.017 |
| 1.54 ± 0.33 | 1.47 ± 0.30 | 1.54 ± 0.32 | 1.50 ± 0.29 | 0.794 |
| 2.20 ± 1.50 | 2.77 ± 1.68* | 2.16 ± 1.39 | 2.10 ± 1.48 | 0.274 |
| 11.39 ± 5.00 | 12.40 ± 6.87 | 10.73 ± 6.59 | 11.75 ± 7.20 | 0.810 |
| 1.44 ± 0.57 | 1.15 ± 0.42* | 1.50 ± 0.71 | 1.46 ± 0.62 | 0.132 |
| Pulmonary function parameters | |||||
| −57.5 ± 28.8 | −101.8 ± 41.0 * | −59.8 ± 29.2 | −89.1 ± 34.8 * | 0.358 |
| 60.9 ± 38.1 | 92.6 ± 45.9 * | 69.2 ± 43.1 | 94.1 ± 36.3 * | 0.925 |
| 15.3 ± 3.7 | 15.8 ± 6.8 | 14.3 ± 4.6 | 13.2 ± 3.3 | 0.269 |
| 10.3 ± 3.1 | 14.8 ± 5.5 * | 10.6 ± 3.0 | 10.2 ± 4.9 | 0.073 |
| 0.75 ± 0.22 | 1.14 ± 0.35 * | 0.80 ± 0.29 | 0.88 ± 0.31 | 0.005 |
| 21.7 ± 13.6 | 12.7 ± 5.2 * | 22.7 ± 14.4 | 32.7 ± 22.6 | 0.031 |
| Borg scale | |||||
| 1.33 ± 2.30 | 0.50 ± 0.71 | 1.08 ± 0.49 | 1.10 ± 0.55 | 0.109 |
| 2.67 ± 2.08 | 1.67 ± 1.53 | 3.50 ± 1.05 | 2.20 ± 1.30 | 0.616 |
| 1.33 ± 0.58 | 1.25 ± 1.26 | 2.41 ± 1.36 | 1.06 ± 1.07 | 0.779 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.-C.; Lin, Y.-T.; Huang, C.-C.; Lin, M.-C.; Liaw, M.-Y.; Lu, C.-H. Effects of Respiratory Muscle Training on Baroreflex Sensitivity, Respiratory Function, and Serum Oxidative Stress in Acute Cervical Spinal Cord Injury. J. Pers. Med. 2021, 11, 377. https://doi.org/10.3390/jpm11050377
Wang H-C, Lin Y-T, Huang C-C, Lin M-C, Liaw M-Y, Lu C-H. Effects of Respiratory Muscle Training on Baroreflex Sensitivity, Respiratory Function, and Serum Oxidative Stress in Acute Cervical Spinal Cord Injury. Journal of Personalized Medicine. 2021; 11(5):377. https://doi.org/10.3390/jpm11050377
Chicago/Turabian StyleWang, Hung-Chen, Yu-Tsai Lin, Chih-Cheng Huang, Meng-Chih Lin, Mei-Yun Liaw, and Cheng-Hsien Lu. 2021. "Effects of Respiratory Muscle Training on Baroreflex Sensitivity, Respiratory Function, and Serum Oxidative Stress in Acute Cervical Spinal Cord Injury" Journal of Personalized Medicine 11, no. 5: 377. https://doi.org/10.3390/jpm11050377
APA StyleWang, H.-C., Lin, Y.-T., Huang, C.-C., Lin, M.-C., Liaw, M.-Y., & Lu, C.-H. (2021). Effects of Respiratory Muscle Training on Baroreflex Sensitivity, Respiratory Function, and Serum Oxidative Stress in Acute Cervical Spinal Cord Injury. Journal of Personalized Medicine, 11(5), 377. https://doi.org/10.3390/jpm11050377







