Risk of Typical Diabetes-Associated Complications in Different Clusters of Diabetic Patients: Analysis of Nine Risk Factors
Abstract
1. Background
2. Methods
2.1. Data
2.2. Cohort
2.3. Risk Factors and Complications
2.4. Identification of Clusters
2.5. Cluster-Based Relative Risk Analysis
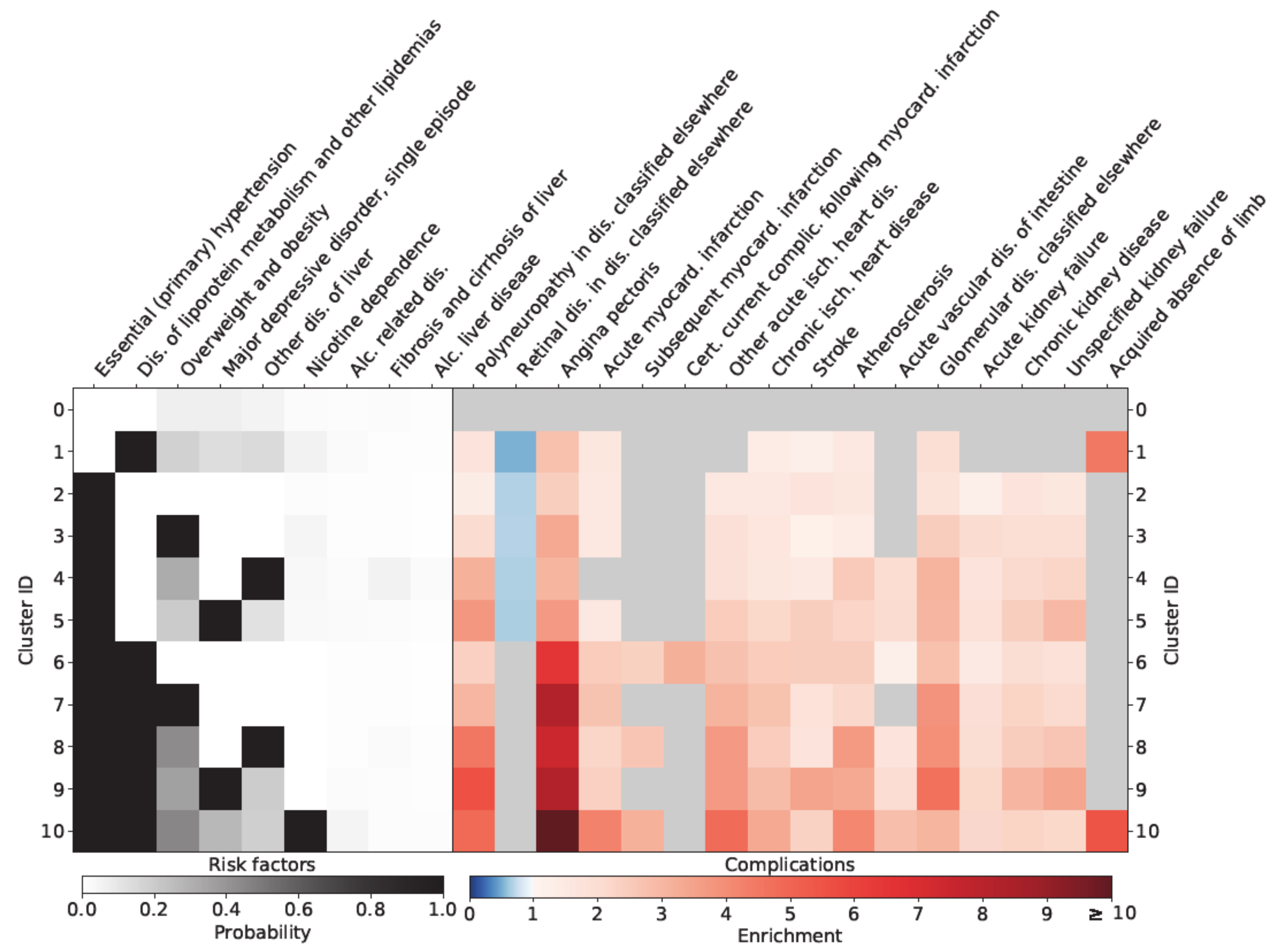
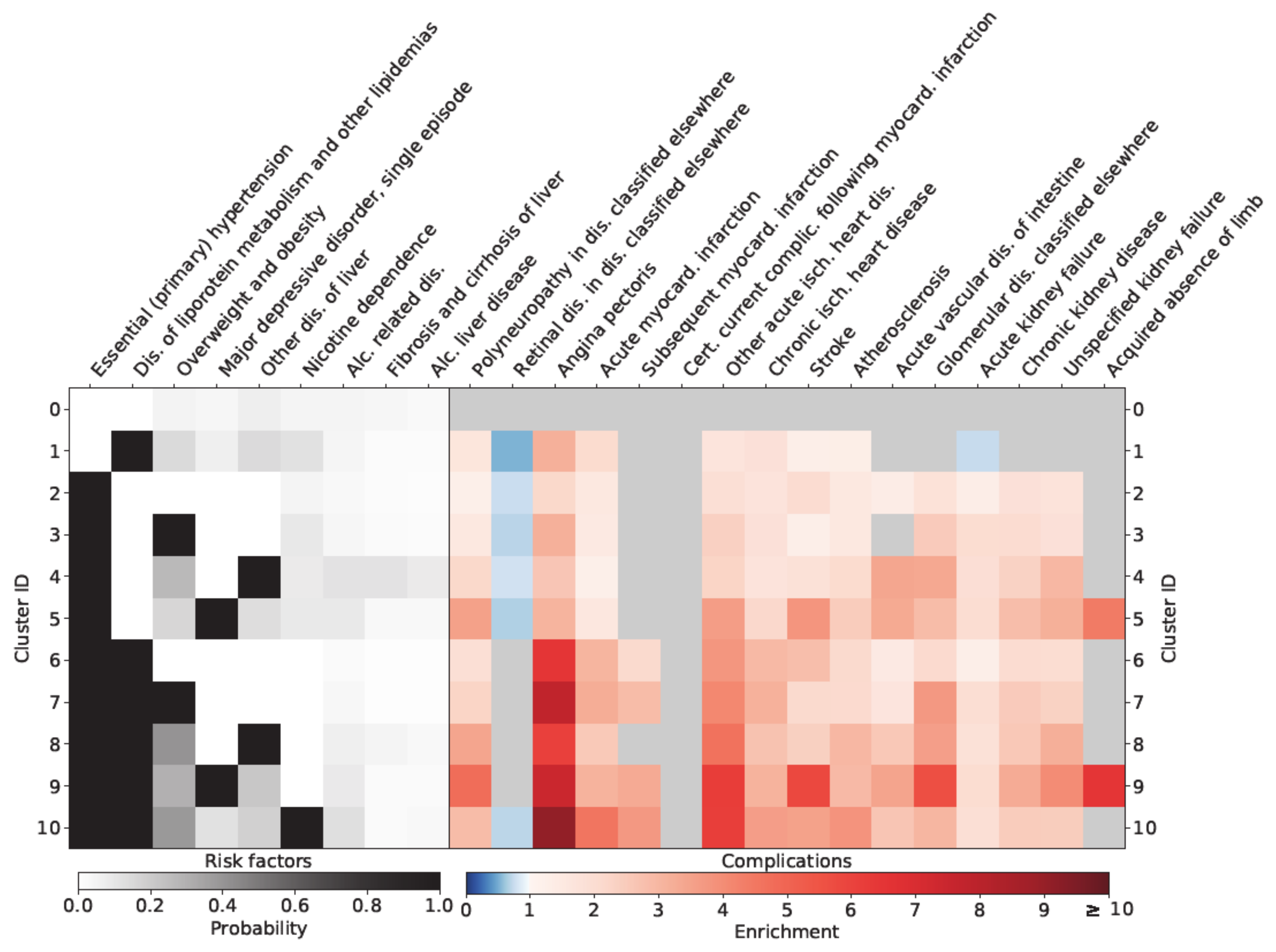
3. Results
Sex-Specific Relationships
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef]
- Alonso-Morán, E.; Orueta, J.F.; Esteban, J.I.F.; Axpe, J.M.A.; González, M.L.M.; Polanco, N.T.; Loiola, P.E.; Gaztambide, S.; Nuño-Solinís, R. Multimorbidity in people with type 2 diabetes in the Basque Country (Spain): Prevalence, comorbidity clusters and comparison with other chronic patients. Eur. J. Intern. Med. 2015, 26, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Uddin, S.; Srinivasan, U. Comorbidity network for chronic disease: A novel approach to understand type 2 diabetes progression. Int. J. Med. Inform. 2018, 115, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; ADVANCE Collaborative Group. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): A randomised controlled trial. Lancet 2007, 370, 829–840. [Google Scholar] [CrossRef]
- Colhoun, H.M.; Betteridge, D.J.; Durrington, P.N.; A Hitman, G.; Neil, H.A.W.; Livingstone, S.J.; Thomason, M.J.; Mackness, M.I.; Charlton-Menys, V.; Fuller, J.H. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): Multicentre randomised placebo-controlled trial. Lancet 2004, 364, 685–696. [Google Scholar] [CrossRef]
- Stamler, J.; Vaccaro, O.; Neaton, J.D.; Wentworth, D. The multiple risk factor intervention trial research group diabetes, other risk factors, and 12-yr cardiovascular mortality for men screened in the multiple risk factor intervention Trial. Diabetes Care 1993, 16, 434–444. [Google Scholar] [CrossRef] [PubMed]
- Kearney, P.M.; Blackwell, L.; Collins, R.; Keech, A.; Simes, J.; Peto, R.; Armitage, J.; Baigent, C. Efficacy of cholesterol-lowering therapy in 18,686 people with diabetes in 14 randomised trials of statins: A meta-analysis. Lancet 2008, 371, 117–125. [Google Scholar] [PubMed]
- Novak, M.; Mucsi, I.; Rhee, C.M.; Streja, E.; Lu, J.L.; Kalantar-Zadeh, K.; Molnar, M.Z.; Kovesdy, C.P. Increased risk of incident chronic kidney disease, cardiovascular disease, and mortality in patients with diabetes with comorbid depression. Diabetes Care 2016, 39, 1940–1947. [Google Scholar] [CrossRef]
- Wild, S.H.; Walker, J.J.; Morling, J.R.; McAllister, D.A.; Colhoun, H.M.; Farran, B.; McGurnaghan, S.; McCrimmon, R.; Read, S.H.; Sattar, N.; et al. Cardiovascular disease, cancer, and mortality among people with type 2 diabetes and alcoholic or nonalcoholic fatty liver disease hospital admission. Diabetes Care 2018, 41, 341–347. [Google Scholar] [CrossRef]
- Wong, T.Y.; Cheung, C.M.; Larsen, M.; Sharma, S.; Simó, R. Diabetic retinopathy. Nat. Rev. Dis. Primers. 2016, 2, 16012. [Google Scholar] [CrossRef]
- Ahlqvist, E.; Storm, P.; Käräjämäki, A.; Martinell, M.; Dorkhan, M.; Carlsson, A.; Vikman, P.; Prasad, R.B.; Almgren, P.; Wessman, Y.; et al. Novel subgroups of adult-onset diabetes and their association with outcomes: A data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol. 2018, 6, 361–369. [Google Scholar] [CrossRef]
- Dennis, J.M.; Shields, B.M.; E Henley, W.; Jones, A.G.; Hattersley, A.T. Disease progression and treatment response in data-driven subgroups of type 2 diabetes compared with models based on simple clinical features: An analysis using clinical trial data. Lancet Diabetes Endocrinol. 2019, 7, 442–451. [Google Scholar] [CrossRef]
- Kahkoska, A.R.; Geybels, M.S.; Klein, K.R.; Kreiner, F.F.; Marx, N.; Nauck, M.A.; Pratley, R.E.; Wolthers, B.O.; Buse, J.B. Validation of distinct type 2 diabetes clusters and their association with diabetes complications in the DEVOTE, LEADER and SUSTAIN -6 cardiovascular outcomes trials. Diabetes Obes. Metab. 2020, 22, 1537–1547. [Google Scholar] [CrossRef]
- Tanabe, H.; Saito, H.; Kudo, A.; Machii, N.; Hirai, H.; Maimaituxun, G.; Tanaka, K.; Masuzaki, H.; Watanabe, T.; Asahi, K.; et al. Factors associated with risk of diabetic complications in novel cluster-based diabetes subgroups: A Japanese retrospective cohort study. J. Clin. Med. 2020, 9, 2083. [Google Scholar] [CrossRef]
- Zaharia, O.P.; Strassburger, K.; Strom, A.; Bönhof, G.J.; Karusheva, Y.; Antoniou, S.; Bódis, K.; Markgraf, D.F.; Burkart, V.; Müssig, K.; et al. Risk of diabetes-associated diseases in subgroups of patients with recent-onset diabetes: A 5-year follow-up study. Lancet Diabetes Endocrinol. 2019, 7, 684–694. [Google Scholar] [CrossRef]
- Ryu, K.S.; Kang, H.Y.J.; Lee, S.; Park, H.W.; You, N.Y.; Kim, J.H.; Hwangbo, Y.; Choi, K.S.; Cha, H.S. Screening model for estimating undiagnosed diabetes among people with a family history of diabetes mellitus: A KNHANES-based study. Int. J. Environ. Res. Public Heal. 2020, 17, 8903. [Google Scholar] [CrossRef]
- Nedyalkova, M.; Madurga, S.; Simeonov, V. Combinatorial k-means clustering as a machine learning tool applied to diabetes mellitus type 2. Int. J. Environ. Res. Public Heal. 2021, 18, 1919. [Google Scholar] [CrossRef] [PubMed]
- MacQueen, J. Some methods for classification and analysis of multivariate observations. In Proceedings of the 5th Berkeley Symposium on Mathematical Statistics and Probability, Berkeley, CA, USA, 1 January 1967; University of California Press: Berkeley, CA, USA, 1967; pp. 281–297. [Google Scholar]
- Ward, J.J. Hierarchical grouping to optimize an objective function. J. Am. Stat. Assoc. 1963, 58, 236–244. [Google Scholar] [CrossRef]
- Haug, N.; Deischinger, C.; Gyimesi, M.; Kautzky-Willer, A.; Thurner, S.; Klimek, P. High-risk multimorbidity patterns on the road to cardiovascular mortality. BMC Med. 2020, 18, 44. [Google Scholar] [CrossRef] [PubMed]
- Haug, N.; Sorger, J.; Gisinger, T.; Gyimesi, M.; Kautzky-Willer, A.; Thurner, S.; Klimek, P. Decompression of multimorbidity along the disease trajectories of diabetes mellitus patients. Front. Physiol. 2021, 11, 612604. [Google Scholar] [CrossRef]
- Chavent, M.; Lechevallier, Y.; Briant, O. DIVCLUS-T: A monothetic divisive hierarchical clustering method. Comput. Stat. Data Anal. 2007, 52, 687–701. [Google Scholar] [CrossRef]
- Group UPDSU. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998, 352, 837–853. [Google Scholar] [CrossRef]
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998, 352, 854–865. [Google Scholar] [CrossRef]
- Baigent, C.; Keech, A.C.; Kearney, P.M.; Blackwell, L.; Buck, G.; Pollicino, C.; Kirby, A.; Sourjina, T.; Peto, R.; Collins, R.; et al. Efficacy and safety of cholesterol-lowering treatment: Prospective meta-analysis of data from 90 056 participants in 14 randomised trials of statins. Lancet 2005, 366, 1267–1278. [Google Scholar]
- Scott, R.S.; Obrien, R.T.; Fulcher, G.; Pardy, C.; Demden, M.C.; Tse, D.; Taskinen, M.-R.; Ehnholm, C.; Keech, A.C. Effects of fenofibrate treatment on cardiovascular disease risk in 9,795 individuals with type 2 diabetes and various components of the metabolic syndrome: The fenofibrate intervention and event lowering in diabetes (FIELD) study. Diabetes Care 2008, 32, 493–498. [Google Scholar] [CrossRef]
- Raman, R.; Gupta, A.; Krishna, S.; Kulothungan, V.; Sharma, T. Prevalence and risk factors for diabetic microvascular complications in newly diagnosed type II diabetes mellitus. Sankara Nethralaya Diabetic Retinopathy Epidemiology And Molecular Genetic Study (SN-DREAMS, report 27). J. Diabetes Complicat. 2012, 26, 123–128. [Google Scholar] [CrossRef]
- Nowakowska, M.; Zghebi, S.S.; Ashcroft, D.M.; Buchan, I.; Chew-Graham, C.; Holt, T.; Mallen, C.; Van Marwijk, H.; Peek, N.; Perera-Salazar, R.; et al. The comorbidity burden of type 2 diabetes mellitus: Patterns, clusters and predictions from a large English primary care cohort. BMC Med. 2019, 17, 145. [Google Scholar] [CrossRef]
- Piette, J.D.; Kerr, E.A. The impact of comorbid chronic conditions on diabetes care. Diabetes Care 2006, 29, 725–731. [Google Scholar] [CrossRef]
- Jiang, G.; Luk, A.O.; Tam, C.H.T.; Lau, E.S.; Ozaki, R.; Chow, E.Y.K.; Kong, A.P.S.; Lim, C.K.P.; Lee, K.F.; Siu, S.C.; et al. Obesity, clinical, and genetic predictors for glycemic progression in Chinese patients with type 2 diabetes: A cohort study using the Hong Kong Diabetes Register and Hong Kong Diabetes Biobank. PLoS Med. 2020, 17, e1003209. [Google Scholar] [CrossRef]
- Zhu, P.; Pan, X.-F.; Sheng, L.; Chen, H.; Pan, A. Cigarette smoking, diabetes, and diabetes complications: Call for urgent action. Curr. Diabetes Rep. 2017, 17, 78. [Google Scholar] [CrossRef]
- Zhang, P.; Lu, J.; Jing, Y.; Tang, S.; Zhu, D.; Bi, Y. Global epidemiology of diabetic foot ulceration: A systematic review and meta-analysis. Ann. Med. 2017, 49, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Chen, Y.; Yang, W.; Gao, X.; Han, X.; Ji, L. The association of smoking and risk of diabetic retinopathy in patients with type 1 and type 2 diabetes: A meta-analysis. Endocrine 2018, 62, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Petrak, F.; Baumeister, H.; Skinner, T.C.; Brown, A.; Holt, R.I.G. Depression and diabetes: Treatment and health-care delivery. Lancet Diabetes Endocrinol. 2015, 3, 472–485. [Google Scholar] [CrossRef]
- Targher, G.; Lonardo, A.; Byrne, C.D. Nonalcoholic fatty liver disease and chronic vascular complications of diabetes mellitus. Nat. Rev. Endocrinol. 2018, 14, 99–114. [Google Scholar] [CrossRef]
- Anjana, R.M.; Baskar, V.; Nair, A.T.N.; Jebarani, S.; Siddiqui, M.K.; Pradeepa, R.; Unnikrishnan, R.; Palmer, C.; Pearson, E.; Mohan, V. Novel subgroups of type 2 diabetes and their association with microvascular outcomes in an Asian Indian population: A data-driven cluster analysis: The INSPIRED study. BMJ Open Diabetes Res. Care 2020, 8, e001506. [Google Scholar] [CrossRef]
- Kanellakis, S.; Mavrogianni, C.; Karatzi, K.; Lindstrom, J.; Cardon, G.; Iotova, V.; Wikström, K.; Shadid, S.; Moreno, L.A.; Tsochev, K.; et al. Development and validation of two self-reported tools for insulin resistance and hypertension risk assessment in a european cohort: The Feel4Diabetes-study. Nutrients 2020, 12, 960. [Google Scholar] [CrossRef]
- Dorner, T.E.; Brath, H.; Kautzky-Willer, A. Sex-specific trends in smoking prevalence over seven years in different Austrian populations: Results of a time-series cross-sectional analysis. BMJ Open 2020, 10, e035235. [Google Scholar] [CrossRef]
- Kautzky-Willer, A.; Harreiter, J.; Pacini, G. Sex and gender differences in risk, pathophysiology and complications of type 2 diabetes mellitus. Endocr. Rev. 2016, 37, 278–316. [Google Scholar] [CrossRef]

| Cohort Size | 195,575 |
| Ratio of Males | 50,3% |
| Minimum Age of Patients in 2014 | 60 y |
| Median Age Males in 2014 (Lower-Upper Quartile) | 74 (67–82) y |
| Median Age Females in 2014 (Lower-Upper Quartile) | 82 (73–89) y |
| Cluster ID | Number of Patients | Female Ratio | Mean Age Females/y | Mean Age Males/y |
|---|---|---|---|---|
| 0 | 37,899 | 0.47 | 81 (72–91) | 75 (66–83) |
| 1 | 7124 | 0.42 | 76 (67–84) | 71 (64–77) |
| 2 | 63,518 | 0.52 | 83 (75–91) | 77 (69–84) |
| 3 | 10,600 | 0.54 | 76 (68–83) | 72 (65–77) |
| 4 | 6844 | 0.45 | 79 (71–87) | 74 (67–81) |
| 5 | 9057 | 0.70 | 82 (75–90) | 78 (69–86) |
| 6 | 29,621 | 0.45 | 80 (74–88) | 75 (68–81) |
| 7 | 10,399 | 0.48 | 76 (69–82) | 72 (66–77) |
| 8 | 6740 | 0.43 | 77 (70–84) | 73 (67–78) |
| 9 | 7778 | 0.68 | 80 (73–88) | 75 (68–82) |
| 10 | 5995 | 0.27 | 72 (65–76) | 70 (64–74) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leutner, M.; Haug, N.; Bellach, L.; Dervic, E.; Kautzky, A.; Klimek, P.; Kautzky-Willer, A. Risk of Typical Diabetes-Associated Complications in Different Clusters of Diabetic Patients: Analysis of Nine Risk Factors. J. Pers. Med. 2021, 11, 328. https://doi.org/10.3390/jpm11050328
Leutner M, Haug N, Bellach L, Dervic E, Kautzky A, Klimek P, Kautzky-Willer A. Risk of Typical Diabetes-Associated Complications in Different Clusters of Diabetic Patients: Analysis of Nine Risk Factors. Journal of Personalized Medicine. 2021; 11(5):328. https://doi.org/10.3390/jpm11050328
Chicago/Turabian StyleLeutner, Michael, Nils Haug, Luise Bellach, Elma Dervic, Alexander Kautzky, Peter Klimek, and Alexandra Kautzky-Willer. 2021. "Risk of Typical Diabetes-Associated Complications in Different Clusters of Diabetic Patients: Analysis of Nine Risk Factors" Journal of Personalized Medicine 11, no. 5: 328. https://doi.org/10.3390/jpm11050328
APA StyleLeutner, M., Haug, N., Bellach, L., Dervic, E., Kautzky, A., Klimek, P., & Kautzky-Willer, A. (2021). Risk of Typical Diabetes-Associated Complications in Different Clusters of Diabetic Patients: Analysis of Nine Risk Factors. Journal of Personalized Medicine, 11(5), 328. https://doi.org/10.3390/jpm11050328









