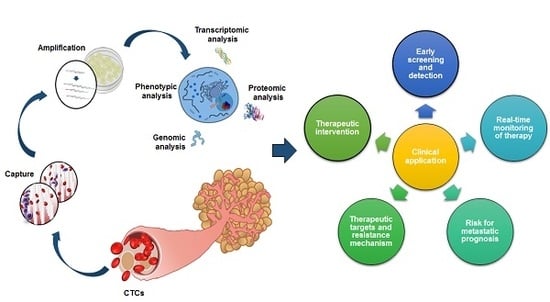
Perspective on Cancer Therapeutics Utilizing Analysis of Circulating Tumor Cells
Abstract
1. Cancer Screening and Diagnosis
2. Cancer Therapeutics and Circulating Tumor Cells (CTCs)
3. Concept for Circulating Tumor Cells (CTCs) Analysis
3.1. DNA-Based Genome Analysis
3.2. RNA-Based Gene Expression Analysis
3.3. Protein-Based Analysis
3.4. Glycosylation-Based CTC Analysis
3.5. Functional Analysis with CTC Culture
4. Clinical Application of Circulating Tumor Cells (CTCs) Analysis
5. Limitation of Circulating Tumor Cells (CTCs) Analysis for Clinical Applications
6. Conclusions
Conflicts of Interest
References
- Roganovic, D.; Djilas, D.; Vujnovic, S.; Pavic, D.; Stojanov, D. Breast mri, digital mammography and breast tomosynthesis: Comparison of three methods for early detection of breast cancer. Bosn. J. Basic Med. Sci. 2015, 15, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Wellings, E.; Vassiliades, L.; Abdalla, R. Breast cancer screening for high-risk patients of different ages and risk—Which modality is most effective? Cureus 2016, 8, e945. [Google Scholar] [CrossRef] [PubMed]
- U.S. Preventive Services Task Force; Bibbins-Domingo, K.; Grossman, D.C.; Curry, S.J.; Davidson, K.W.; Epling, J.W., Jr.; Garcia, F.A.R.; Gillman, M.W.; Harper, D.M.; Kemper, A.R.; et al. Screening for colorectal cancer: Us preventive services task force recommendation statement. JAMA 2016, 315, 2564–2575. [Google Scholar]
- Sithambaram, S.; Hilmi, I.; Goh, K.L. The diagnostic accuracy of the m2 pyruvate kinase quick stool test—A rapid office based assay test for the detection of colorectal cancer. PLoS ONE 2015, 10, e0131616. [Google Scholar] [CrossRef] [PubMed]
- Song, L.L.; Li, Y.M. Current noninvasive tests for colorectal cancer screening: An overview of colorectal cancer screening tests. World J. Gastrointest. Oncol. 2016, 8, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Bell, N.; Connor Gorber, S.; Shane, A.; Joffres, M.; Singh, H.; Dickinson, J.; Shaw, E.; Dunfield, L.; Tonelli, M.; Canadian Task Force on Preventive Health Care. Recommendations on screening for prostate cancer with the prostate-specific antigen test. CMAJ 2014, 186, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.N.; Diefenbach, M.A.; Valdimarsdottir, H.; Chen, T.; Hall, S.J.; Thompson, H.S. Pros and cons of prostate cancer screening: Associations with screening knowledge and attitudes among urban african american men. J. Natl. Med. Assoc. 2010, 102, 174–182. [Google Scholar] [CrossRef]
- Mokulis, J.; Thompson, I.I. Screening for prostate cancer: Pros, cons, and reality. Cancer Control 1995, 2, 15–21. [Google Scholar] [PubMed]
- Bao, P.Q.; Johnson, J.C.; Lindsey, E.H.; Schwartz, D.A.; Arildsen, R.C.; Grzeszczak, E.; Parikh, A.A.; Merchant, N.B. Endoscopic ultrasound and computed tomography predictors of pancreatic cancer resectability. J. Gastrointest. Surg. 2008, 12, 10–16; discussion 16. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.C. Radiation risk from medical imaging. Mayo Clin. Proc. 2010, 85, 1142–1146; quiz 1146. [Google Scholar] [CrossRef] [PubMed]
- Morris, E.; Feig, S.A.; Drexler, M.; Lehman, C. Implications of overdiagnosis: Impact on screening mammography practices. Popul. Health Manag. 2015, 18 (Suppl. S1), S3–S11. [Google Scholar] [CrossRef] [PubMed]
- Defrank, J.T.; Brewer, N. A model of the influence of false-positive mammography screening results on subsequent screening. Health Psychol. Rev. 2010, 4, 112–127. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Buchbinder, E.I.; Desai, A. Ctla-4 and pd-1 pathways: Similarities, differences, and implications of their inhibition. Am. J. Clin. Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Menezes, M.E.; Bhatia, S.; Wang, X.Y.; Emdad, L.; Sarkar, D.; Fisher, P.B. Gene therapies for cancer: Strategies, challenges and successes. J. Cell. Physiol. 2015, 230, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Rein, D.T.; Breidenbach, M.; Curiel, D.T. Current developments in adenovirus-based cancer gene therapy. Future Oncol. 2006, 2, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Sanna, V.; Pala, N.; Sechi, M. Targeted therapy using nanotechnology: Focus on cancer. Int. J. Nanomed. 2014, 9, 467–483. [Google Scholar]
- Oouchida, K.; Ieiri, S.; Kenmotsu, H.; Tomikawa, M.; Hashizume, M. [Robotic surgery for cancer treatment]. Gan Kagaku Ryoho Cancer Chemother. 2012, 39, 1–7. [Google Scholar]
- Yu, N.; Zhou, J.; Cui, F.; Tang, X. Circulating tumor cells in lung cancer: Detection methods and clinical applications. Lung 2015, 193, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Sheth, R.A.; Hesketh, R.; Deipolyi, A.R.; Oklu, R. Circulating tumor cells: Personalized medicine in interventional oncology? J. Vasc. Interv. Radiol. 2013, 24, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Skelley, A.M.; Merdek, K.D.; Sprott, K.M.; Jiang, C.; Pierceall, W.E.; Lin, J.; Stocum, M.; Carney, W.P.; Smirnov, D.A. Microfluidics and circulating tumor cells. J. Mol. Diagn. 2013, 15, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Harouaka, R.A.; Nisic, M.; Zheng, S.Y. Circulating tumor cell enrichment based on physical properties. J. Lab. Autom. 2013, 18, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Stott, S.; Toner, M.; Maheswaran, S.; Haber, D.A. Circulating tumor cells: Approaches to isolation and characterization. J. Cell Biol. 2011, 192, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Lapin, M.; Tjensvoll, K.; Oltedal, S.; Buhl, T.; Gilje, B.; Smaaland, R.; Nordgard, O. Mindec-an enhanced negative depletion strategy for circulating tumour cell enrichment. Sci. Rep. 2016, 6, 28929. [Google Scholar] [CrossRef] [PubMed]
- Satelli, A.; Brownlee, Z.; Mitra, A.; Meng, Q.H.; Li, S. Circulating tumor cell enumeration with a combination of epithelial cell adhesion molecule- and cell-surface vimentin-based methods for monitoring breast cancer therapeutic response. Clin. Chem. 2015, 61, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Sabeh, F.; Ota, I.; Holmbeck, K.; Birkedal-Hansen, H.; Soloway, P.; Balbin, M.; Lopez-Otin, C.; Shapiro, S.; Inada, M.; Krane, S.; et al. Tumor cell traffic through the extracellular matrix is controlled by the membrane-anchored collagenase mt1-mmp. J. Cell Biol. 2004, 167, 769–781. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Dicker, D.T.; Matthew, E.; El-Deiry, W.S.; Alpaugh, R.K. Circulating tumor cells: Silent predictors of metastasis. F1000Res 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Yokota, T. Are kras/braf mutations potent prognostic and/or predictive biomarkers in colorectal cancers? Anticancer Agents Med. Chem. 2012, 12, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Sato, K. Microdevice in cellular pathology: Microfluidic platforms for fluorescence in situ hybridization and analysis of circulating tumor cells. Anal. Sci. 2015, 31, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Gasch, C.; Pantel, K.; Riethdorf, S. Whole genome amplification in genomic analysis of single circulating tumor cells. Methods Mol. Biol. 2015, 1347, 221–232. [Google Scholar] [PubMed]
- Pantel, K.; Speicher, M.R. The biology of circulating tumor cells. Oncogene 2016, 35, 1216–1224. [Google Scholar] [CrossRef] [PubMed]
- Mostert, B.; Sieuwerts, A.M.; Kraan, J.; Bolt-de Vries, J.; van der Spoel, P.; van Galen, A.; Peeters, D.J.; Dirix, L.Y.; Seynaeve, C.M.; Jager, A.; et al. Gene expression profiles in circulating tumor cells to predict prognosis in metastatic breast cancer patients. Ann. Oncol. 2015, 26, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Barriere, G.; Fici, P.; Gallerani, G.; Fabbri, F.; Zoli, W.; Rigaud, M. Circulating tumor cells and epithelial, mesenchymal and stemness markers: Characterization of cell subpopulations. Ann. Transl. Med. 2014, 2, 109. [Google Scholar] [PubMed]
- Cho, W.J.; Oliveira, D.S.; Najy, A.J.; Mainetti, L.E.; Aoun, H.D.; Cher, M.L.; Heath, E.; Kim, H.R.; Bonfil, R.D. Gene expression analysis of bone metastasis and circulating tumor cells from metastatic castrate-resistant prostate cancer patients. J. Transl. Med. 2016, 14, 72. [Google Scholar] [CrossRef] [PubMed]
- Tomlins, S.A.; Mehra, R.; Rhodes, D.R.; Shah, R.B.; Rubin, M.A.; Bruening, E.; Makarov, V.; Chinnaiyan, A.M. Whole transcriptome amplification for gene expression profiling and development of molecular archives. Neoplasia 2006, 8, 153–162. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Payne, R.E.; Wang, F.; Su, N.; Krell, J.; Zebrowski, A.; Yague, E.; Ma, X.J.; Luo, Y.; Coombes, R.C. Viable circulating tumour cell detection using multiplex rna in situ hybridisation predicts progression-free survival in metastatic breast cancer patients. Br. J. Cancer 2012, 106, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Wang, L.; Tian, X. Application of circulating tumor cells scope technique on circulating tumor cell research. Mol. Cell. Ther. 2014, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Cheng, B.R.; He, Z.B.; Wang, S.Y.; Wang, Z.M.; Sun, M.; Song, H.B.; Fang, Y.; Chen, F.F.; Xiong, B. Capture and identification of heterogeneous circulating tumor cells using transparent nanomaterials and quantum dots-based multiplexed imaging. J. Cancer 2016, 7, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Hong, B.; Zu, Y. Detecting circulating tumor cells: Current challenges and new trends. Theranostics 2013, 3, 377–394. [Google Scholar] [CrossRef] [PubMed]
- Horm, T.M.; Schroeder, J.A. Muc1 and metastatic cancer: Expression, function and therapeutic targeting. Cell Adh. Migr. 2013, 7, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.; Han, C.; Wang, D.; Zhao, X.; Jin, G.; Shen, H. Detection of circulating tumor cells: Clinical relevance of a novel metastatic tumor marker. Exp. Ther. Med. 2011, 2, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Handerson, T.; Camp, R.; Harigopal, M.; Rimm, D.; Pawelek, J. Beta1,6-branched oligosaccharides are increased in lymph node metastases and predict poor outcome in breast carcinoma. Clin. Cancer Res. 2005, 11, 2969–2973. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liu, X.; Hsieh, B.; Bruce, R.; Somlo, G.; Huang, J.; Sambucetti, L. Exploring glycan markers for immunotyping and precision-targeting of breast circulating tumor cells. Arch. Med. Res. 2015, 46, 642–650. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Banford, S.; Timson, D.J. Udp-n-acetyl-d-galactosamine:Polypeptide n-acetylgalactosaminyltransferase-6 (pp-galnac-t6): Role in cancer and prospects as a drug target. Curr. Cancer Drug Targets 2017, 17, 53–61. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ao, Z.; Liu, X. Fiber-optic array scanning technology (fast) for detection and molecular characterization of circulating tumor cells. Methods Mol. Biol. 2017, 1634, 235–246. [Google Scholar] [PubMed]
- Monzavi-Karbassi, B.; Pashov, A.; Kieber-Emmons, T. Tumor-associated glycans and immune surveillance. Vaccines 2013, 1, 174–203. [Google Scholar] [CrossRef] [PubMed]
- Praharaj, P.P.; Bhutia, S.K.; Nagrath, S.; Bitting, R.L.; Deep, G. Circulating tumor cell-derived organoids: Current challenges and promises in medical research and precision medicine. Biochim. Biophys. Acta 2018, 1869, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Maheswaran, S.; Haber, D.A. Ex vivo culture of ctcs: An emerging resource to guide cancer therapy. Cancer Res. 2015, 75, 2411–2415. [Google Scholar] [CrossRef] [PubMed]
- De Mattos-Arruda, L.; Cortes, J.; Santarpia, L.; Vivancos, A.; Tabernero, J.; Reis-Filho, J.S.; Seoane, J. Circulating tumour cells and cell-free DNA as tools for managing breast cancer. Nat. Rev. Clin. Oncol. 2013, 10, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Alix-Panabieres, C.; Pantel, K. Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov. 2016, 6, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Gorin, M.A.; Verdone, J.E.; van der Toom, E.; Bivalacqua, T.J.; Allaf, M.E.; Pienta, K.J. Circulating tumour cells as biomarkers of prostate, bladder, and kidney cancer. Nat. Rev. Urol. 2017, 14, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, F.; Yoneda, K.; Kondo, N.; Hashimoto, M.; Takuwa, T.; Matsumoto, S.; Okumura, Y.; Rahman, S.; Tsubota, N.; Tsujimura, T.; et al. Circulating tumor cell as a diagnostic marker in primary lung cancer. Clin. Cancer Res. 2009, 15, 6980–6986. [Google Scholar] [CrossRef] [PubMed]
- Ilie, M.; Hofman, V.; Long, E.; Bordone, O.; Selva, E.; Washetine, K.; Marquette, C.H.; Hofman, P. Current challenges for detection of circulating tumor cells and cell-free circulating nucleic acids, and their characterization in non-small cell lung carcinoma patients. What is the best blood substrate for personalized medicine? Ann. Transl. Med. 2014, 2, 107. [Google Scholar] [PubMed]
- Shahneh, F.Z. Current challenges in metastasis: Disseminated and circulating tumor cells detection. Hum. Antibodies 2013, 22, 77–85. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, K.-Y.; Kim, E.K.; Park, M.H.; Kim, H.M. Perspective on Cancer Therapeutics Utilizing Analysis of Circulating Tumor Cells. Diagnostics 2018, 8, 23. https://doi.org/10.3390/diagnostics8020023
Jeong K-Y, Kim EK, Park MH, Kim HM. Perspective on Cancer Therapeutics Utilizing Analysis of Circulating Tumor Cells. Diagnostics. 2018; 8(2):23. https://doi.org/10.3390/diagnostics8020023
Chicago/Turabian StyleJeong, Keun-Yeong, Eun Kyung Kim, Min Hee Park, and Hwan Mook Kim. 2018. "Perspective on Cancer Therapeutics Utilizing Analysis of Circulating Tumor Cells" Diagnostics 8, no. 2: 23. https://doi.org/10.3390/diagnostics8020023
APA StyleJeong, K.-Y., Kim, E. K., Park, M. H., & Kim, H. M. (2018). Perspective on Cancer Therapeutics Utilizing Analysis of Circulating Tumor Cells. Diagnostics, 8(2), 23. https://doi.org/10.3390/diagnostics8020023