The Influence of Sex, Ear, and Age on Auditory Brainstem Responses Recorded with the NeuroAudio System
Abstract
1. Introduction
2. Material and Methods
2.1. Ethics Statement
2.2. Participants
2.3. Procedures
3. Results
3.1. Statistical Analysis
3.2. Effect of Sex
3.3. Effect of Age
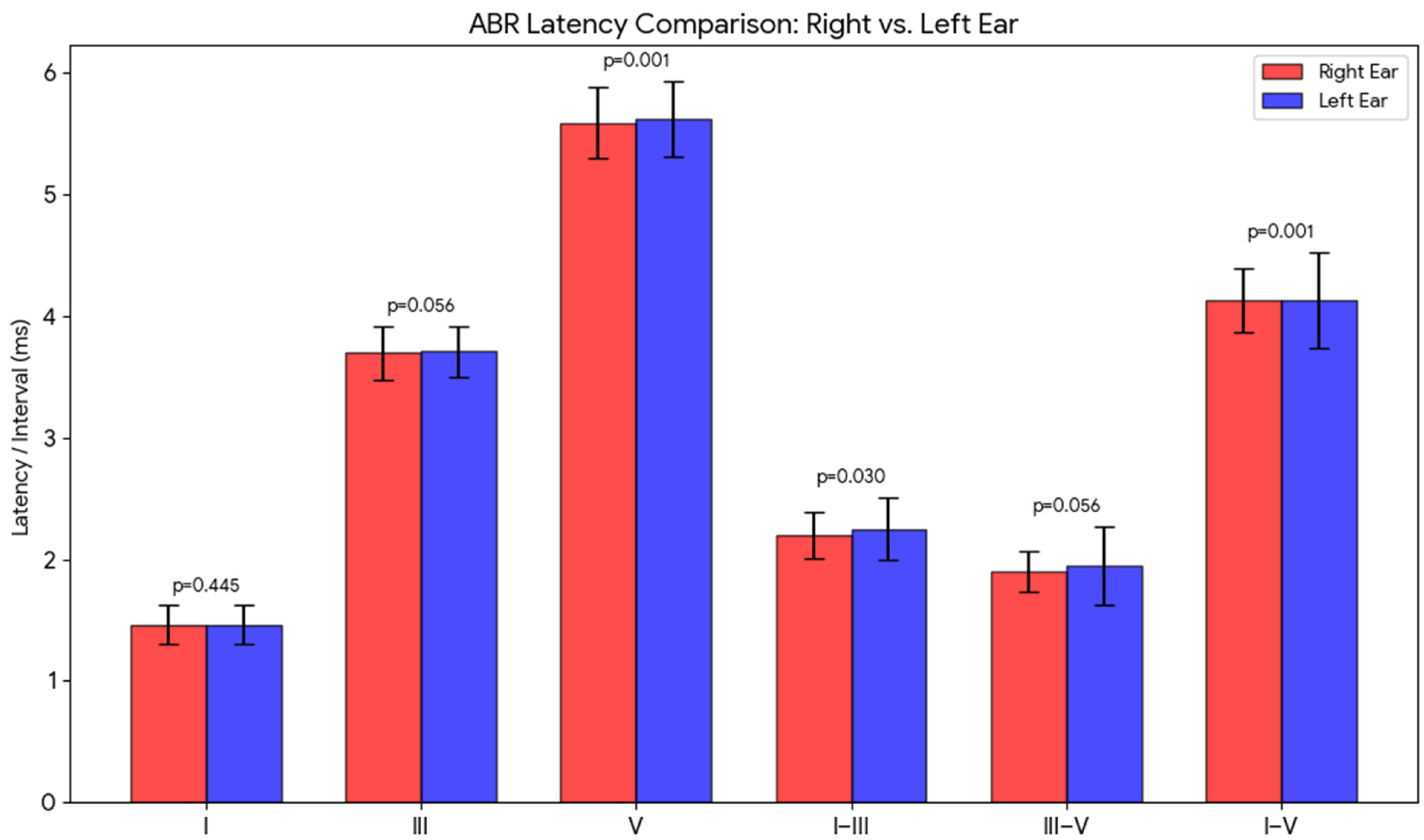
3.4. Effect of Stimulus Ear
3.5. Expected Normative Values
4. Discussion
4.1. Effect of Sex
4.2. Effect of Age
4.3. Effect of Stimulus Ear
4.4. The Influence of Sex, Ear, and Age on the ABR
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABR | Auditory Brainstem Response |
| daPa | Decapascal |
| dB | decibels |
| dBHL | Decibel Hearing Level |
| dBnHL | Decibel Normalized Hearing Level |
| EPM | Paulista School of Medicine |
| ER–3A | Etymotic Research Insert Earphones Model 3A |
| Fz | Frontal Zero Electrode Position |
| FFR | Frequency Following Response |
| GSI | Grason-Stadler Inc. |
| Hz | Hertz |
| IAD | Interaural Asymetry Difference |
| ICF | Informed Consent Form |
| IEC-645 | International Electrotechnical Commission Standard 645 |
| ISO-389 | International Organization for Standardization 389 |
| kHz | Kilohertz |
| kΩ | Kilo-ohm |
| LE | Left Ear |
| M | mean |
| MN | Minnesota |
| Mmhos | Millimhos |
| Ms | millisecond |
| RE | Right Ear |
| SD | Standard Deviation |
| SRT | Speech Recognition Threshold |
| SPSS | Statistical Package for the Social Sciences |
| TM | Trademark |
| UNIFESP | Federal University of São Paulo |
| USA | United States of America |
| WHO | World Health Organization |
| µV | Microvolt |
References
- Singh, R.; Vates, E. Brainstem auditory evoked response test. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK597358 (accessed on 30 January 2026).
- Hall, J.W., III. eHandbook of Auditory Evoked Responses; Kindle Direct Publishing: New York, NY, USA, 2015; Available online: https://www.amazon.com/eHandbook-Auditory-Evoked-Responses-Principles-ebook/dp/B0145G2FFM (accessed on 5 January 2026).
- Sanfins, M.D.; Colella-Santos, M.F.; Ferrazoli, N.; Rezende, A.; Donadon, C.; Gos, E.; Skarżyński, P.H. Latency and interpeak interval values of auditory brainstem response in 73 individuals with normal hearing. Med. Sci. Monit. 2022, 28, e937847. [Google Scholar] [CrossRef] [PubMed]
- Chhajed, M.; Madaan, P.; Saini, L.; Kaur, S.; Udani, V. Normative values for auditory brainstem responses and wave V thresholds in Indian neonates and infants. Indian J. Otolaryngol. Head Neck Surg. 2022, 74, 3658–3661. [Google Scholar] [CrossRef] [PubMed]
- Gorga, M.P.; Reiland, J.K.; Beauchaine, K.A.; Worthington, D.W.; Jesteadt, W. Auditory brainstem response from graduates of an intensive care nursery: Normal patterns of response. J. Speech Hear. Res. 1987, 30, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Gorga, M.P.; Kaminiski, J.R.; Beauchaine, K.L.; Jesteadt, W.; Neely, S.T. Auditory brainstem response from children three months to three years of age: Normal patterns of response II. J. Speech Hear. Res. 1989, 32, 281–288. [Google Scholar] [CrossRef] [PubMed]
- McFadden, D.; Champlin, C.A.; Pho, M.H.; Pasanen, E.G.; Maloney, M.M.; Leshikar, E.M. Auditory evoked potentials: Differences by sex, race, and menstrual cycle and correlations with common psychoacoustical tasks. PLoS ONE 2021, 16, e0251363. [Google Scholar] [CrossRef] [PubMed]
- Krizman, J.; Rotondo, E.K.; Nicol, T.; Kraus, N.; Bieszczad, K.M. Sex differences in auditory processing vary across estrous cycle. Sci. Rep. 2021, 11, 22898. [Google Scholar] [CrossRef] [PubMed]
- Tasman, A.; Hahn, T.; Maiste, A. Menstrual cycle synchronized changes in the brain stem auditory evoked potentials and visual evoked potentials. Biol. Psychiatry 1999, 45, 1516–1519. [Google Scholar] [CrossRef] [PubMed]
- Jerger, J.; Hall, J. Effects of age and sex on auditory brainstem response. Arch. Otolaryngol. 1980, 106, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Başoğlu, İ.; Belgin, E.; Ölçek, G.; Başoğlu, Y. Age and sex differences in speech FFR suggest early central auditory aging. Sci. Rep. 2025, 15, 40319. [Google Scholar] [CrossRef] [PubMed]
- Villavisanis, D.F.; Berson, E.R.; Lauer, A.M.; Cosetti, M.K.; Schrode, K.M. Sex-based differences in hearing loss: Perspectives from non-clinical research to clinical outcomes. Otol. Neurotol. 2020, 41, 290–298. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. World Report on Hearing; World Health Organization: Geneva, Switzerland, 2021; p. 252. Available online: https://www.who.int/publications/i/item/9789240020481 (accessed on 27 October 2025).
- Jerger, J. Clinical experience with impedance audiometry. Arch. Otolaryngol. 1970, 92, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Jasper, H.H. The ten-twenty electrode system of the International Federation. Electroencephalogr. Clin. Neurophysiol. 1958, 10, 371–375. [Google Scholar] [CrossRef]
- Krizman, J.; Skoe, E.; Kraus, N. Sex differences in auditory subcortical function. Clin. Neurophysiol. 2012, 123, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Lotfi, Y.; Abdollahi, F.Z. Age and gender effects on auditory brain stem response (ABR). Iran. Rehabil. J. 2012, 10, 25–29. [Google Scholar]
- Hoormann, J.; Falkenstein, M.; Hohnsbein, J.; Blanke, L. The human frequency-following response (FFR): Normal variability and relation to the click-evoked brainstem response. Hear. Res. 1992, 59, 179–188. [Google Scholar] [CrossRef]
- Kırbac, A.; Turkyılmaz, M.D.; Yağcıoglu, S. Gender effects on binaural speech auditory brainstem response. J. Int. Adv. Otol. 2022, 18, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Aoyagi, M.; Kim, Y.; Yokoyama, J.; Kiren, T.; Suzuki, Y.; Koike, Y. Head size as a basis of gender difference in the latency of the brainstem auditory-evoked response. Int. J. Audiol. 1990, 29, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Trune, D.R.; Mitchell, C.; Phillips, D.S. The relative importance of head size, gender and age on the auditory brainstem response. Hear. Res. 1988, 32, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Tremere, L.A.; Pinaud, R. Brain-generated estradiol drives long-term optimization of auditory coding to enhance the discrimination of communication signals. J. Neurosci. 2011, 31, 3271–3289. [Google Scholar] [CrossRef] [PubMed]
- Esteves, M.C.B.N.; Dell, A.H.B.; Arruda, G.V.; Dell, A.R.; Nardi, J.C. Brainstem evoked response audiometry in normal hearing subjects. Braz. J. Otorhinolaryngol. 2009, 75, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Allison, T.; Wood, C.C.; Goff, W.R. Brain stem auditory, pattern-reversal visual, and short-latency somatosensory evoked potentials: Latencies in relation to age, sex, and brain and body size. Electroencephalogr. Clin. Neurophysiol. 1983, 55, 619–636. [Google Scholar] [CrossRef] [PubMed]
- Boer, J.; Hardy, A.; Krumbholz, K. Could tailored chirp stimuli benefit measurement of the supra threshold auditory brainstem wave I response? J. Assoc. Res. Otolaryngol. 2022, 23, 787–802. [Google Scholar] [CrossRef] [PubMed]
- Torre, P., 3rd; Giraldo, M. Age-related changes in auditory brainstem response wave latencies and amplitudes in a clinical population. Am. J. Audiol. 2018, 27, 145–156. [Google Scholar] [CrossRef]
- Wu, Y.H.; Ho, K.Y.; Lin, Y.Y. Structural and functional neuroimaging correlates of age-related auditory decline: Focus on cochlear and brainstem integrity. Neuroimage Clin. 2020, 28, 102415. [Google Scholar] [CrossRef]
- Spitzer, E.; White-Schwoch, T.; Carr, K.W.; Skoe, E.; Kraus, N. Continued maturation of the click-evoked auditory brainstem response in preschoolers. J. Am. Acad. Audiol. 2015, 26, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Rotteveel, J.J.; Colon, E.J.; Stegeman, D.F.; Visser, Y.M. The maturation of the central auditory conduction in preterm infants until three months post term. I. Composite group averages of brainstem (ABR) and middle latency (MLR) auditory evoked responses. Hear. Res. 1987, 28, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.J.; Khurana, D.S.; Kothare, S.V. Normative data for brainstem auditory-evoked potentials and middle latency auditory-evoked potentials in children. Clin. Neurophysiol. 2009, 120, 522–532. [Google Scholar] [CrossRef]
- Sharma, M.; Bist, S.S.; Kumar, S. Age-related maturation of wave V latency of auditory brainstem response in children. J. Audiol. Otol. 2016, 20, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Henry, K.R. Sex- and age-related elevation of cochlear nerve envelope response and auditory brainstem response thresholds in C57BL/6 mice. Hear. Res. 2002, 165, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.A.; McGaffigan, P.M. Right-left asymmetries in the human brain stem: Auditory evoked potentials. Electroencephalogr. Clin. Neurophysiol. 1983, 55, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Belin, P.; Zilbovicius, M.; Crozier, S.; Thivard, L.; Fontaine, A.; Masure, M.C.; Samson, Y. Lateralization of speech and auditory temporal processing. J. Cogn. Neurosci. 1998, 10, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Sininger, Y.S.; Cone-Wesson, B. Asymmetric cochlear processing mimics the effects of tinnitus and hearing loss on sound localization. Hear. Res. 2006, 212, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Hugdahl, K.; Brønnick, K.; Kyllingsbæk, S.; Law, I.; Gade, A.; Paulson, O.B. Brain activation during dichotic presentations of consonant-vowel and musical instrument stimuli: A 15O-PET study. Neuropsychologia 1999, 37, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Tadros, S.F.; Frisina, S.T.; Mapes, F.; Kim, S.; Frisina, D.R.; Frisina, R.D. Loss of peripheral right-ear advantage in age-related hearing loss. Audiol. Neurootol. 2005, 10, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Morita, K.; Guo, Y.; Toi, T. Age-related asymmetry in left-right ears of sound lateralization with respect to four different rise times. Front. Neurosci. 2023, 17, 1249119. [Google Scholar] [CrossRef] [PubMed]
- Morita, K.; Shiroki, M.; Iwaki, S.; Toi, T. Mismatch negativity linked with sound lateralization in elderly individuals. Proc. Meet. Acoust. 2023, 50, 050003. [Google Scholar] [CrossRef]
- Musiek, F.E.; Baran, J.A. Auditory brainstem and middle latency evoked response sensitivity near threshold. Semin. Hear. 2022, 43, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Chiappa, K.H. Evoked Potentials in Clinical Medicine, 3rd ed.; Lippincott-Raven: Philadelphia, PA, USA, 1997; Available online: https://www.amazon.com.br/Evoked-Potentials-Clinical-Medicine-Chiappa/dp/0397516592 (accessed on 19 March 2026).
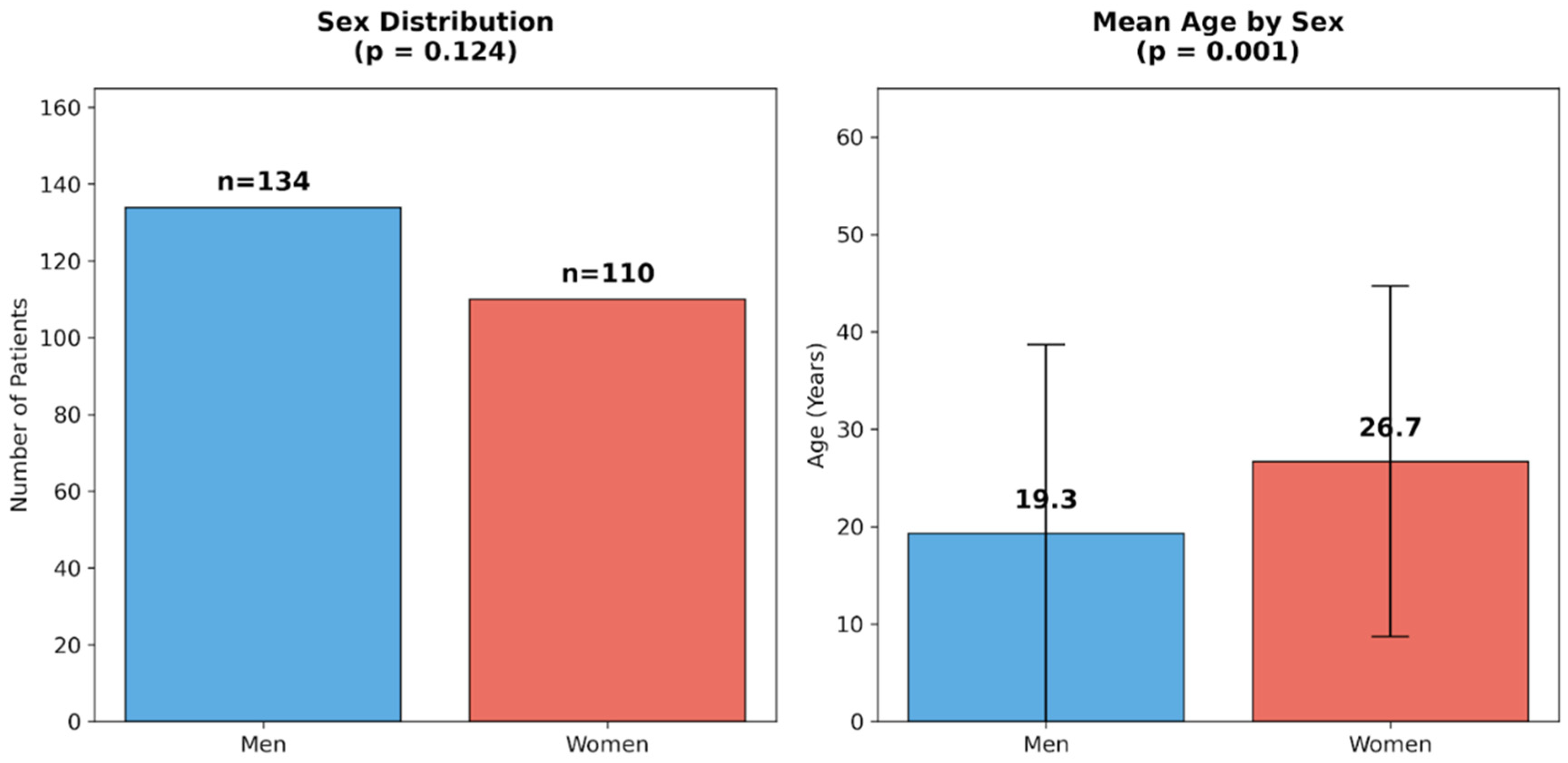

| Men (n = 134) | Women (n = 110) | U | p | |||
|---|---|---|---|---|---|---|
| M | SD | M | SD | |||
| RE I | 1.47 | 0.15 | 1.46 | 0.16 | 6999.0 | 0.499 |
| RE III | 3.73 | 0.18 | 3.65 | 0.25 | 5297.5 | <0.001 |
| RE V | 5.65 | 0.25 | 5.55 | 0.32 | 4992.5 | <0.001 |
| RE I–III | 2.26 | 0.16 | 2.19 | 0.23 | 5109.0 | <0.001 |
| RE III–V | 1.93 | 0.16 | 1.86 | 0.16 | 5609.5 | 0.001 |
| RE I–V | 4.19 | 0.22 | 4.05 | 0.28 | 4561.0 | <0.001 |
| LE I | 1.46 | 0.15 | 1.46 | 0.17 | 7027.5 | 0.930 |
| LE III | 3.76 | 0.19 | 3.64 | 0.21 | 4691.0 | <0.001 |
| LE V | 5.71 | 0.29 | 5.52 | 0.31 | 4315.0 | <0.001 |
| LE I–III | 2.32 | 0.31 | 2.18 | 0.18 | 4250.0 | <0.001 |
| LE III–V | 2.07 | 0.38 | 1.88 | 0.20 | 5388.0 | 0.002 |
| LEI I–V | 4.20 | 0.45 | 4.05 | 0.29 | 4128.5 | <0.001 |
| Age Group | RE_V | LE_V |
|---|---|---|
| 3–17 years | −0.30 ** | −0.34 *** |
| 18–49 years | 0.29 ** | 0.28 ** |
| 50+ years | 0.13 | 0.23 |
| Age | |
|---|---|
| RE_I | 0.21 ** |
| RE_III | −0.09 |
| RE_V | −0.09 |
| RE_I_III | −0.31 *** |
| RE_III_V | −0.05 |
| RE_I_V | −0.23 *** |
| LE_I | 0.33 *** |
| LE_III | −0.03 |
| LE_V | −0.14 * |
| LE_I_III | −0.30 *** |
| LE_III_V | −0.19 ** |
| LE_I_V | −0.32 *** |
| Right Ear (n = 244) | Left Ear (n = 244) | W | p | |||
|---|---|---|---|---|---|---|
| M | SD | M | SD | |||
| I | 1.46 | 0.16 | 1.46 | 0.16 | 0.77 | 0.445 |
| III | 3.70 | 0.22 | 3.71 | 0.21 | 1.91 | 0.056 |
| V | 5.59 | 0.29 | 5.62 | 0.31 | 3.30 | 0.001 |
| I–III | 2.20 | 0.19 | 2.25 | 0.26 | 2.17 | 0.030 |
| III–V | 1.90 | 0.17 | 1.95 | 0.32 | 1.91 | 0.056 |
| I–V | 4.13 | 0.26 | 4.13 | 0.39 | 3.24 | 0.001 |
| Right Ear (n = 244) | Left Ear (n = 244) | W | p | |||
|---|---|---|---|---|---|---|
| M | SD | M | SD | |||
| I | 0.36 | 0.18 | 0.37 | 0.19 | 0.82 | 0.412 |
| V | 0.50 | 0.20 | 0.51 | 0.24 | 0.72 | 0.472 |
| V/I | 1.66 | 0.89 | 1.64 | 1.10 | 0.62 | 0.538 |
| IAD V | 0.08 | 0.07 | 0.08 | 0.07 | 0.00 | >0.999 |
| Ear | Gender | Age Group | I | III | V | I–III | III–V | I–V |
|---|---|---|---|---|---|---|---|---|
| Right | M | 3–17 | 1.24–1.62 | 3.52–3.95 | 5.17–5.99 | 2.08–2.49 | 1.66–2.16 | 3.76–4.54 |
| 18–49 | 1.28–1.79 | 3.49–3.98 | 5.38–5.90 | 2.01–2.41 | 1.69–2.13 | 4.03–4.28 | ||
| 50+ | 1.36–1.88 | 3.45–4.09 | 5.38–6.04 | 1.93–2.50 | 1.78–2.06 | 3.82–4.40 | ||
| F | 3–17 | 1.28–1.59 | 3.38–4.03 | 5.06–5.97 | 1.98–2.46 | 1.65–2.10 | 3.61–4.43 | |
| 18–49 | 1.24–1.62 | 3.32–3.82 | 5.01–5.80 | 1.94–2.34 | 1.62–2.05 | 3.69–4.26 | ||
| 50+ | 1.24–1.65 | 3.50–3.96 | 5.32–5.94 | 1.83–2.52 | 1.75–2.10 | 3.93–4.20 | ||
| Left | M | 3–17 | 1.27–1.59 | 3.51–4.02 | 5.22–6.06 | 2.06–2.58 | 1.69–2.18 | 3.81–4.62 |
| 18–49 | 1.34–1.68 | 3.53–4.03 | 5.38–5.90 | 2.04–2.47 | 1.72–2.07 | 3.88–4.47 | ||
| 50+ | 1.28–1.85 | 3.51–4.22 | 5.45–6.19 | 2.09–2.42 | 1.69–2.15 | 4.00–4.36 | ||
| F | 3–17 | 1.24–1.66 | 3.41–3.88 | 5.20–5.92 | 1.94–2.41 | 1.71–2.11 | 3.75–4.47 | |
| 18–49 | 1.20–1.76 | 3.25–3.89 | 5.03–5.77 | 1.97–2.35 | 1.63–2.11 | 3.61–4.25 | ||
| 50+ | 1.34–1.84 | 3.50–4.02 | 5.38–5.90 | 1.84–2.50 | 1.64–2.12 | 3.94–4.30 |
| Ear | Gender | Age Group | I | III | V | I–III | III–V | I–V |
|---|---|---|---|---|---|---|---|---|
| Right | M | 3–17 | 1.17–1.71 | 3.37–4.08 | 5.10–6.20 | 1.97–2.61 | 1.61–2.25 | 3.74–4.69 |
| 18–49 | 1.24–1.80 | 3.40–4.05 | 5.20–6.03 | 1.93–2.45 | 1.56–2.33 | 3.74–4.49 | ||
| 50+ | 1.25–1.90 | 3.44–4.16 | 5.31–6.16 | 1.91–2.54 | 1.69–2.17 | 3.79–4.53 | ||
| F | 3–17 | 1.15–1.74 | 3.05–4.39 | 4.79–6.42 | 1.66–2.89 | 1.50–2.27 | 3.40–4.91 | |
| 18–49 | 1.11–1.80 | 3.25–3.94 | 4.97–5.89 | 1.86–2.42 | 1.55–2.11 | 3.61–4.33 | ||
| 50+ | 1.17–1.87 | 3.31–4.17 | 5.23–6.08 | 1.72–2.71 | 1.67–2.16 | 3.75–4.51 | ||
| Left | M | 3–17 | 1.15–1.70 | 3.35–4.13 | 5.07–6.37 | 1.64–3.06 | 1.16–2.96 | 3.14–5.31 |
| 18–49 | 1.22–1.80 | 3.38–4.14 | 5.23–6.05 | 1.89–2.61 | 1.63–2.11 | 3.73–4.52 | ||
| 50+ | 1.27–1.91 | 3.44–4.22 | 5.35–6.24 | 1.97–2.53 | 1.59–2.33 | 3.88–4.54 | ||
| F | 3–17 | 1.14–1.74 | 3.21–4.10 | 4.89–6.32 | 1.88–2.56 | 1.46–2.44 | 3.44–4.88 | |
| 18–49 | 1.13–1.80 | 3.20–4.01 | 4.95–5.92 | 1.79–2.49 | 1.53–2.12 | 3.53–4.41 | ||
| 50+ | 1.09–2.06 | 3.41–4.18 | 5.20–6.22 | 1.81–2.63 | 1.56–2.27 | 3.86–4.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sanfins, M.D.; Santillo, M.E.A.; Martins, M.F.P.; Silva, D.L.d.S.; Gos, E.; Skarzynski, P.H.; Hall, J.W., III. The Influence of Sex, Ear, and Age on Auditory Brainstem Responses Recorded with the NeuroAudio System. Diagnostics 2026, 16, 971. https://doi.org/10.3390/diagnostics16070971
Sanfins MD, Santillo MEA, Martins MFP, Silva DLdS, Gos E, Skarzynski PH, Hall JW III. The Influence of Sex, Ear, and Age on Auditory Brainstem Responses Recorded with the NeuroAudio System. Diagnostics. 2026; 16(7):971. https://doi.org/10.3390/diagnostics16070971
Chicago/Turabian StyleSanfins, Milaine Dominici, Maria Eduarda Aidar Santillo, Mariana Ferreira Pires Martins, Diego Lourenço dos Santos Silva, Elzbieta Gos, Piotr Henryk Skarzynski, and James W. Hall, III. 2026. "The Influence of Sex, Ear, and Age on Auditory Brainstem Responses Recorded with the NeuroAudio System" Diagnostics 16, no. 7: 971. https://doi.org/10.3390/diagnostics16070971
APA StyleSanfins, M. D., Santillo, M. E. A., Martins, M. F. P., Silva, D. L. d. S., Gos, E., Skarzynski, P. H., & Hall, J. W., III. (2026). The Influence of Sex, Ear, and Age on Auditory Brainstem Responses Recorded with the NeuroAudio System. Diagnostics, 16(7), 971. https://doi.org/10.3390/diagnostics16070971









