Dynamic Ultrasound of the Infrapatellar Fat Pad for Detecting Anterior Cruciate Ligament Deficiency: A Biomechanical Cadaveric Proof-of-Concept Study
Abstract
1. Introduction
2. Materials and Methods
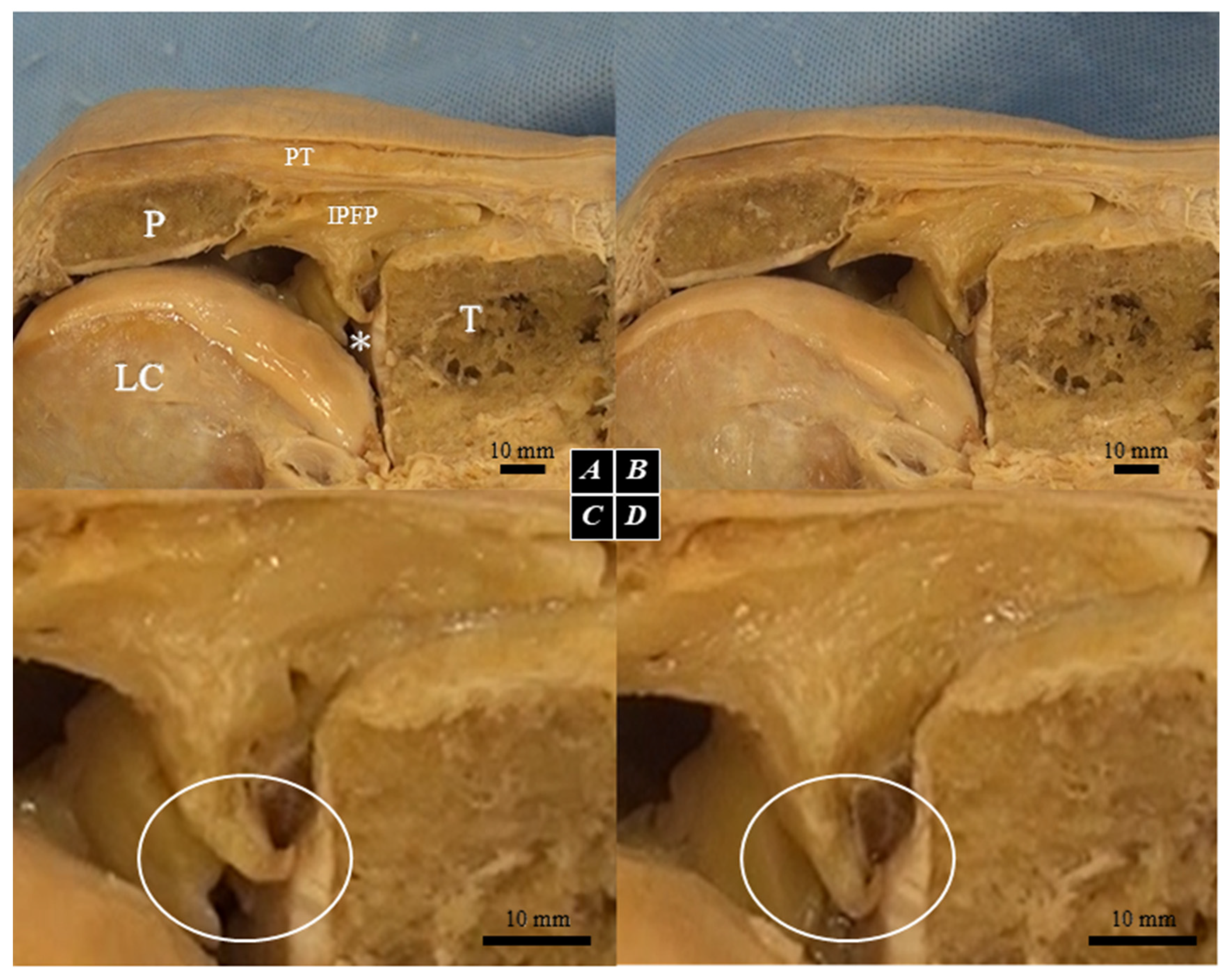
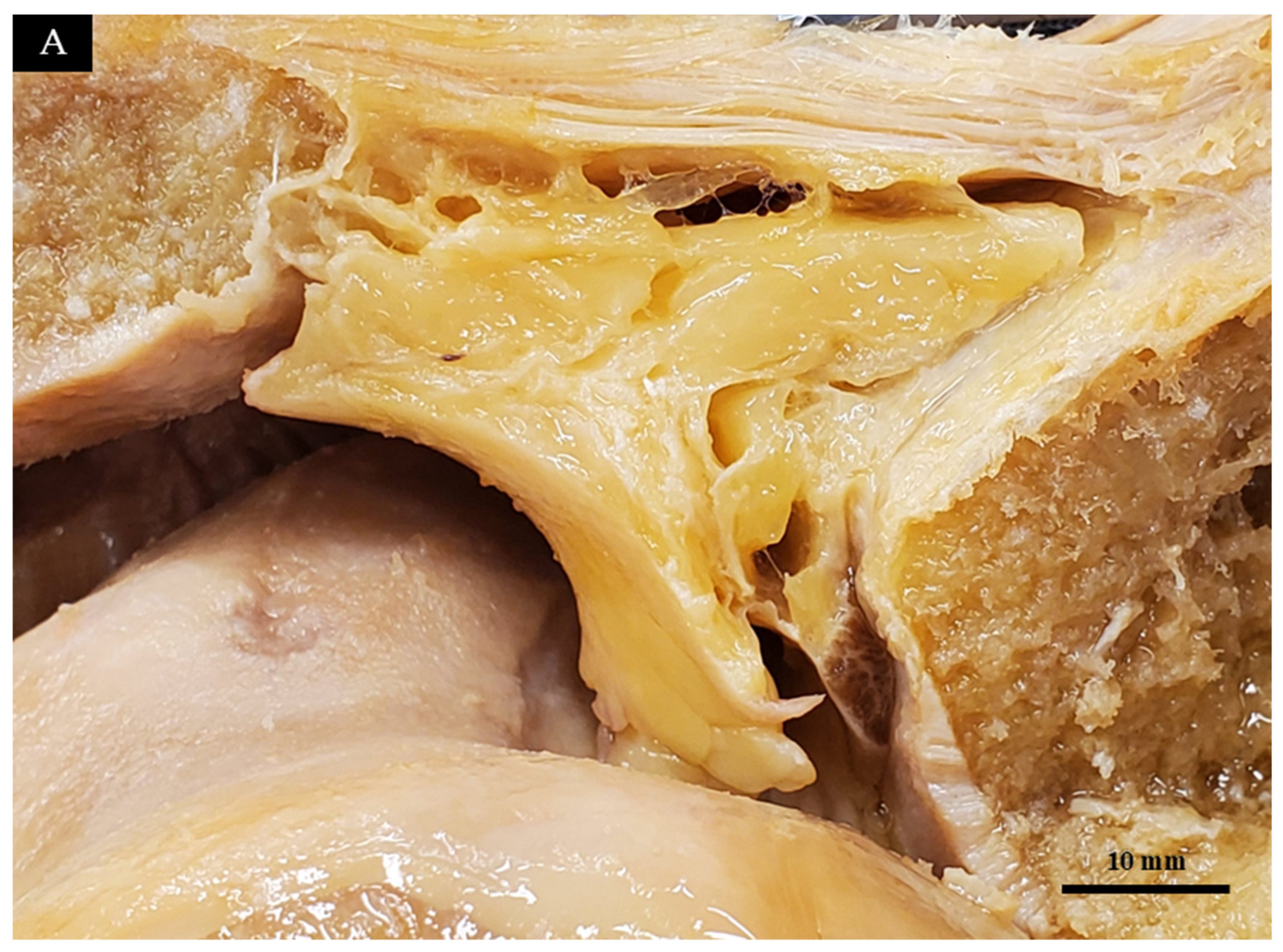
2.1. Identification of the Target Region (Macroscopic and Histological Assessment)
2.2. Thiel-Embalmed Specimens and Demographic Justification
2.3. Ultrasound-Guided Marking and Potential Biases
2.4. Passive Flexion Protocol and Surgical Interventions
2.5. Quantitative Kinematic Assessment
2.6. Statistical Analysis
2.7. Ethics Statement
3. Results
3.1. Histological Findings
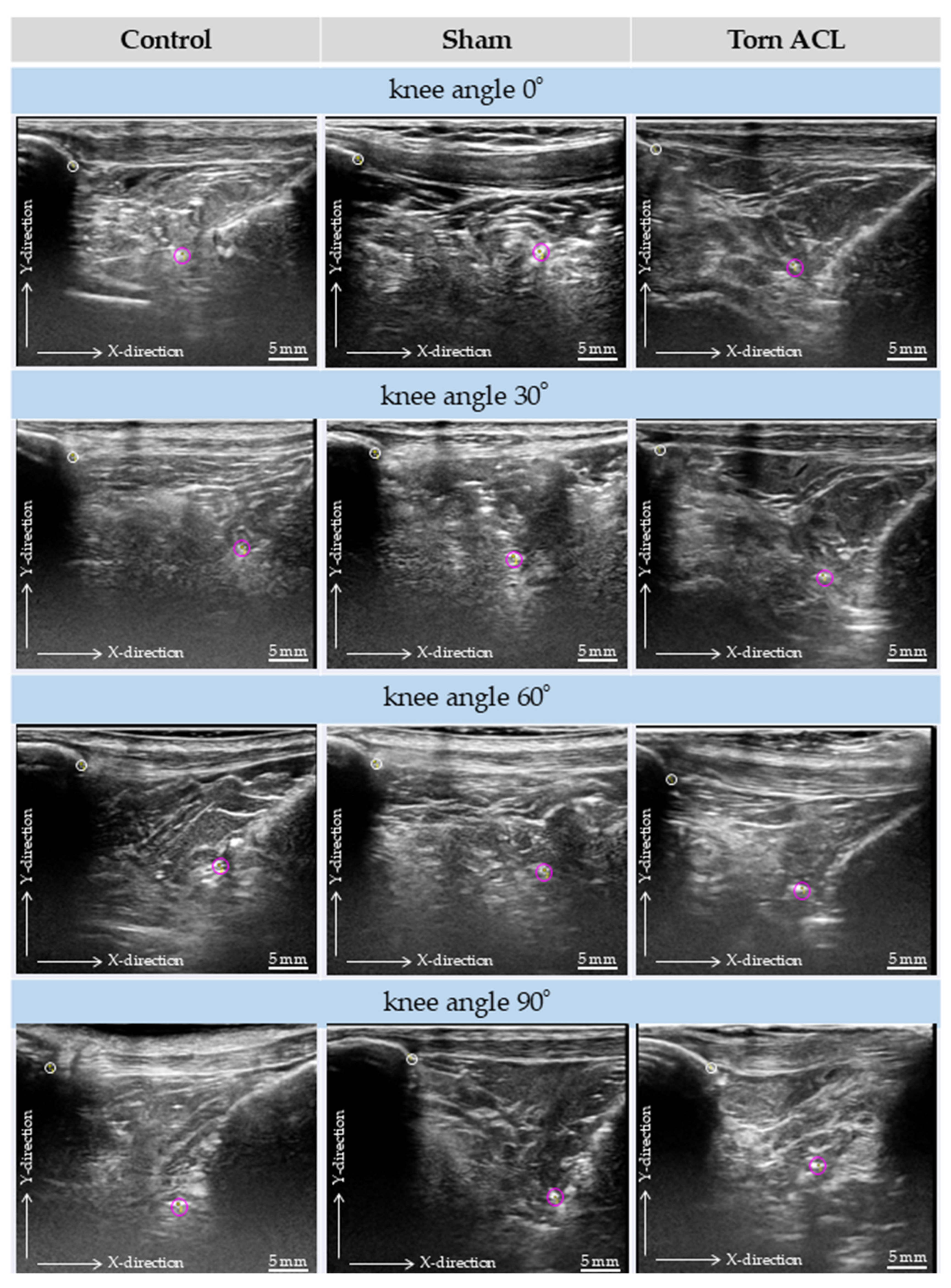
3.2. Qualitative Ultrasound Observations
3.3. Quantitative Kinematic Assessment and Reliability
3.4. Primary Endpoints and Statistical Analysis
3.5. Macroscopic Verification of the Marker Position
4. Discussion
4.1. Anatomical and Histological Basis for AFC-SEG Compliance
4.2. Biomechanical Mechanisms
4.3. Exploratory Clinical Workflow for ACL Screening
4.4. Limitations
4.5. Future Directions (Translational Plan)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACL | Anterior Cruciate Ligament |
| AFC-SEG | Adipo-Follicular Segment |
| ANOVA | Analysis of Variance |
| AUC | Area Under the Receiver Operating Characteristic Curve |
| CIs | Confidence Intervals |
| DSV-SEG | Deep Synovial Segment |
| H&E | Hematoxylin and Eosin |
| ICC | Intraclass Correlation Coefficient |
| IPFP | Infrapatellar Fat Pad |
| IPP | Infrapatellar Plica |
| LMM | Linear Mixed-Effects Model |
| mm | Millimeters |
| MRI | Magnetic Resonance Imaging |
| ROI | Region of Interest |
| SEG | Segment |
| SPD-SEG | Superficial Distal Segment |
| SPP-SEG | Superficial Proximal Segment |
| Δ | Interval Displacement |
References
- Colombet, P.; Dejour, D.; Panisset, J.C.; Siebold, R. Current concept of partial anterior cruciate ligament ruptures. Orthop. Traumatol. Surg. Res. 2010, 96, S109–S118. [Google Scholar] [CrossRef] [PubMed]
- Dejour, D.; Ntagiopoulos, P.G.; Saggin, P.R.; Panisset, J.C. The diagnostic value of clinical tests, magnetic resonance imaging, and instrumented laxity in the differentiation of complete versus partial anterior cruciate ligament tears. Arthroscopy 2013, 29, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Kondo, E.; Merican, A.M.; Yasuda, K.; Amis, A.A. Biomechanical analysis of knee laxity with isolated anteromedial or posterolateral bundle-deficient anterior cruciate ligament. Arthroscopy 2014, 30, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Siebold, R.; Fu, F.H. Assessment and augmentation of symptomatic anteromedial or posterolateral bundle tears of the anterior cruciate ligament. Arthroscopy 2008, 24, 1289–1298. [Google Scholar] [CrossRef]
- Li, Z.; Ren, S.; Zhou, R.; Jiang, X.; You, T.; Li, C.; Zhang, W. Deep Learning-Based Magnetic Resonance Imaging Image Features for Diagnosis of Anterior Cruciate Ligament Injury. J. Health Eng. 2021, 2021, 4076175. [Google Scholar] [CrossRef]
- Lee, S.H.; Yun, S.J. Feasibility of point-of-care knee ultrasonography for diagnosing anterior cruciate and posterior cruciate ligament tears in the ED. Am. J. Emerg. Med. 2020, 38, 237–242. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, A.; Kumar, P. Functional Ultrasonography in Diagnosing Anterior Cruciate Ligament Injury as Compared to Magnetic Resonance Imaging. Indian J. Orthop. 2018, 52, 638–644. [Google Scholar] [CrossRef]
- Konar, S.; Das, R.; Das, S. An Assessment of the Role of Diagnostic Ultrasound in Anterior Cruciate Ligament Tear and Its Correlation with MRI Findings. Int. J. Acad. Med. Pharm. 2025, 7, 1458–1464. [Google Scholar]
- Palm, H.G.; Bergenthal, G.; Ehry, P.; Schwarz, W.; Schmidt, R.; Friemert, B. Functional ultrasonography in the diagnosis of acute anterior cruciate ligament injuries: A field study. Knee 2009, 16, 441–446. [Google Scholar] [CrossRef]
- Wasilczyk, C. The Value of Ultrasound Diagnostic Imaging of Anterior Crucial Ligament Tears Verified Using Experimental and Arthroscopic Investigations. Diagnostics 2024, 14, 305. [Google Scholar] [CrossRef]
- Sakakibara, Y.; Mathur, V.; Osei-Hwedieh, D.O.; Bhimani, R.; Taseh, A.; Ashkani-Esfahani, S.; Tanaka, M.J. Diagnostic Accuracy and Reliability of Dynamic Handheld Ultrasound Testing in Detecting Anterior Cruciate Ligament Tears: A Cadaveric Study. Arch. Bone Jt. Surg. 2025, 13, 823–830. [Google Scholar]
- Gallagher, J.; Tierney, P.; Murray, P.; O’Brien, M. The infrapatellar fat pad: Anatomy and clinical correlations. Knee Surg. Sports Traumatol. Arthrosc. 2005, 13, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Bombaci, H.; Marasli, M.K.; Akinci, O.; Ozogul, M. MRI-based evaluation of tibial tunnel proximity to the anterior horn of the lateral meniscus in remnant-preserving anterior cruciate ligament reconstruction. Acta Orthop. Traumatol. Turc. 2025, 59, 280–285. [Google Scholar] [CrossRef]
- Yeo, I.-S.; Hong, J.-E.; Yang, H.-M. Histomorphometric analysis of anterior cruciate ligament bundles and anatomical insights into injury mechanisms. Sci. Rep. 2025, 15, 6762. [Google Scholar] [CrossRef]
- Pehlivanoglu, G.; Yildiz, K.I.; Albayrak, K.; Cakir, T.; Aykut, U.S.; Ozkul, B. Evaluating Tibial Tunnel Landmarks in Anterior Cruciate Ligament Reconstruction: Remnant Versus Lateral Meniscus Anterior Horn. J. Clin. Med. 2025, 14, 8096. [Google Scholar] [CrossRef]
- Shao, J.; Zhang, J.; Ren, S.; Liu, P.; Ma, Y.; Ao, Y. Better coverage of the ACL tibial footprint and less injury to the anterior root of the lateral meniscus using a rounded-rectangular tibial tunnel in ACL reconstruction: A cadaveric study. Orthop. J. Sports Med. 2022, 10, 23259671221083581. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- Hardaker, W.T., Jr.; Whipple, T.L.; Bassett, F.H., 3rd. Diagnosis and treatment of the plica syndrome of the knee. J. Bone Jt. Surg. Am. 1980, 62, 221–225. [Google Scholar] [CrossRef]
- Jacobson, J.A.; Lenchik, L.; Ruhoy, M.K.; Schweitzer, M.E.; Resnick, D. MR imaging of the infrapatellar fat pad of Hoffa. Radiographics 1997, 17, 675–691. [Google Scholar] [CrossRef]
- Smith, P.N.; Refshauge, K.M.; Scarvell, J.M. Development of the concepts of knee kinematics. Arch. Phys. Med. Rehabil. 2003, 84, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, Y. Regional Differences in Infrapatellar Fat Pad Stiffness with Changes in Knee Flexion Angle: A Quantitative Analysis Using Shear Wave Elastography. Cureus 2026, 18, e101476. [Google Scholar] [CrossRef]
- Ishii, Y.; Sugimoto, M.; Nekomoto, A.; Nakamae, A.; Zhu, K.; Hashizume, T.; Matsumura, K.; Nakashima, Y.; Takahashi, M.; Adachi, N. Restricted morphological changes in infrapatellar fat pad during walking is revealed as a dynamics feature in symptomatic knee osteoarthritis. J. Med. Ultrason. 2026, 53, 97–104. [Google Scholar] [CrossRef] [PubMed]

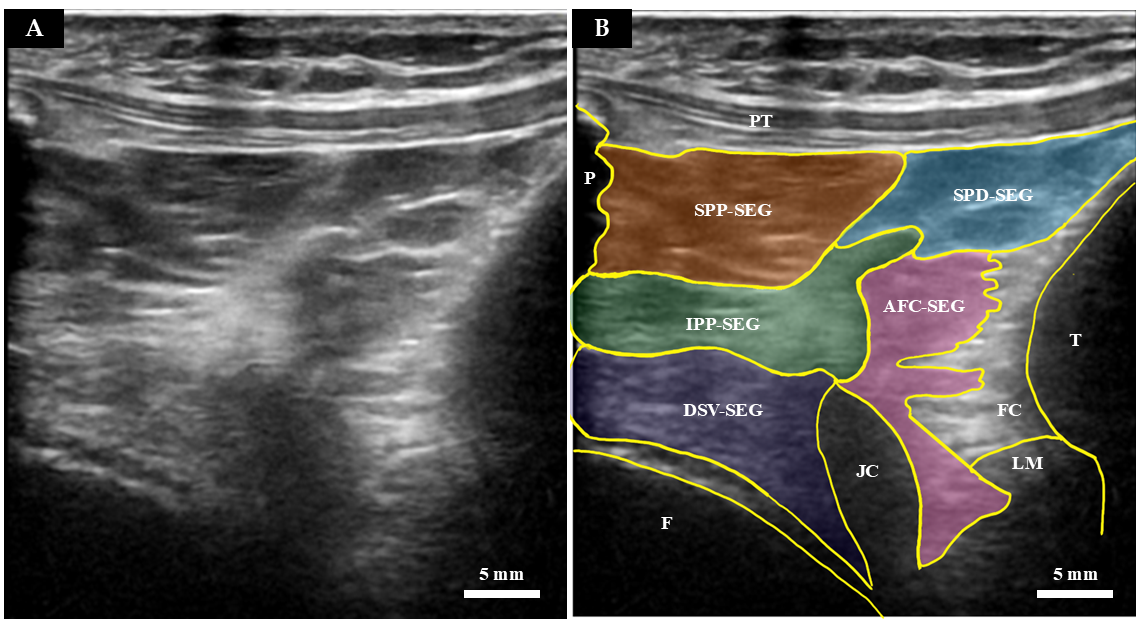


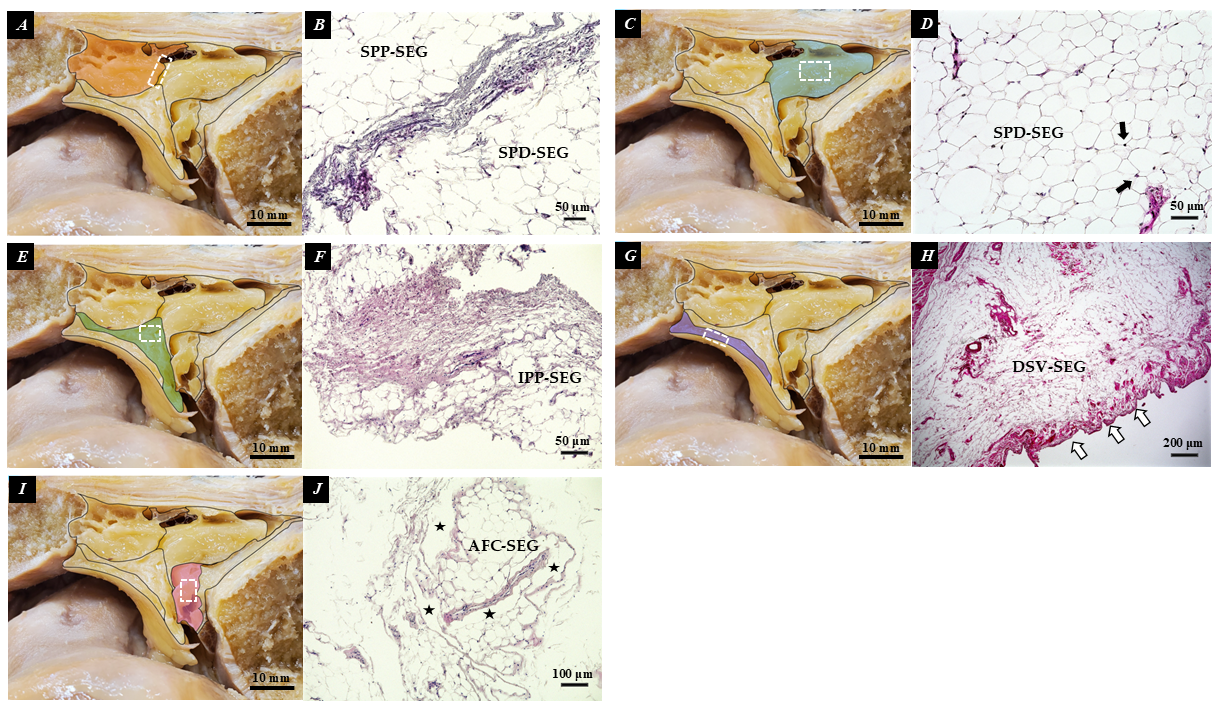








| Segment Abbreviation | Full Name | Macroscopic Anatomical Location | Histological and Structural Features |
|---|---|---|---|
| SPP-SEG | Superficial Proximal Segment | Connects the patella directly to the superficial layer of the patellar tendon. | Densely packed adipocytes. Separated from the SPD-SEG by connective tissue septa. |
| SPD-SEG | Superficial Distal Segment | Bounded by the patellar tendon, deep infrapatellar bursa, and tibia. | Adipocytes present. Linked to the tibia via the complex connective tissue of the joint capsule. |
| IPP-SEG | Infrapatellar Plica Segment | Regarded as the extended tissue of the infrapatellar plica (ligamentum mucosum). | High ratio of dense connective tissue mixed with adipocytes. Structurally less deformable. |
| DSV-SEG | Deep Synovial Segment | Deepest portion near the joint cavity. Separated from the patella by a horizontal cleft. | Characterized by a synovial cell lining on its articular surface. |
| AFC-SEG [Target Region] | Adipo-Follicular Segment | Bounded by SPD-SEG, IPP-SEG, and tibia. Connected to the anterior horns of the menisci. | Contains unique follicle-like spaces. Highly compliant and susceptible to joint pressure changes. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tokuda, Y.; Nakamura, T.; Shiraishi, Y.; Hori, K.; Okuda, H.; Ozaki, N. Dynamic Ultrasound of the Infrapatellar Fat Pad for Detecting Anterior Cruciate Ligament Deficiency: A Biomechanical Cadaveric Proof-of-Concept Study. Diagnostics 2026, 16, 1097. https://doi.org/10.3390/diagnostics16071097
Tokuda Y, Nakamura T, Shiraishi Y, Hori K, Okuda H, Ozaki N. Dynamic Ultrasound of the Infrapatellar Fat Pad for Detecting Anterior Cruciate Ligament Deficiency: A Biomechanical Cadaveric Proof-of-Concept Study. Diagnostics. 2026; 16(7):1097. https://doi.org/10.3390/diagnostics16071097
Chicago/Turabian StyleTokuda, Yoshiyuki, Tsuneo Nakamura, Yoshitake Shiraishi, Kiyomi Hori, Hiroaki Okuda, and Noriyuki Ozaki. 2026. "Dynamic Ultrasound of the Infrapatellar Fat Pad for Detecting Anterior Cruciate Ligament Deficiency: A Biomechanical Cadaveric Proof-of-Concept Study" Diagnostics 16, no. 7: 1097. https://doi.org/10.3390/diagnostics16071097
APA StyleTokuda, Y., Nakamura, T., Shiraishi, Y., Hori, K., Okuda, H., & Ozaki, N. (2026). Dynamic Ultrasound of the Infrapatellar Fat Pad for Detecting Anterior Cruciate Ligament Deficiency: A Biomechanical Cadaveric Proof-of-Concept Study. Diagnostics, 16(7), 1097. https://doi.org/10.3390/diagnostics16071097






