Patient and Healthcare Provider Barriers in the LDCT Lung Cancer Screening Continuum
Abstract
1. Introduction
1.1. Background
1.2. Differentiation Between Uptake and Longitudinal Adherence
1.3. Objectives
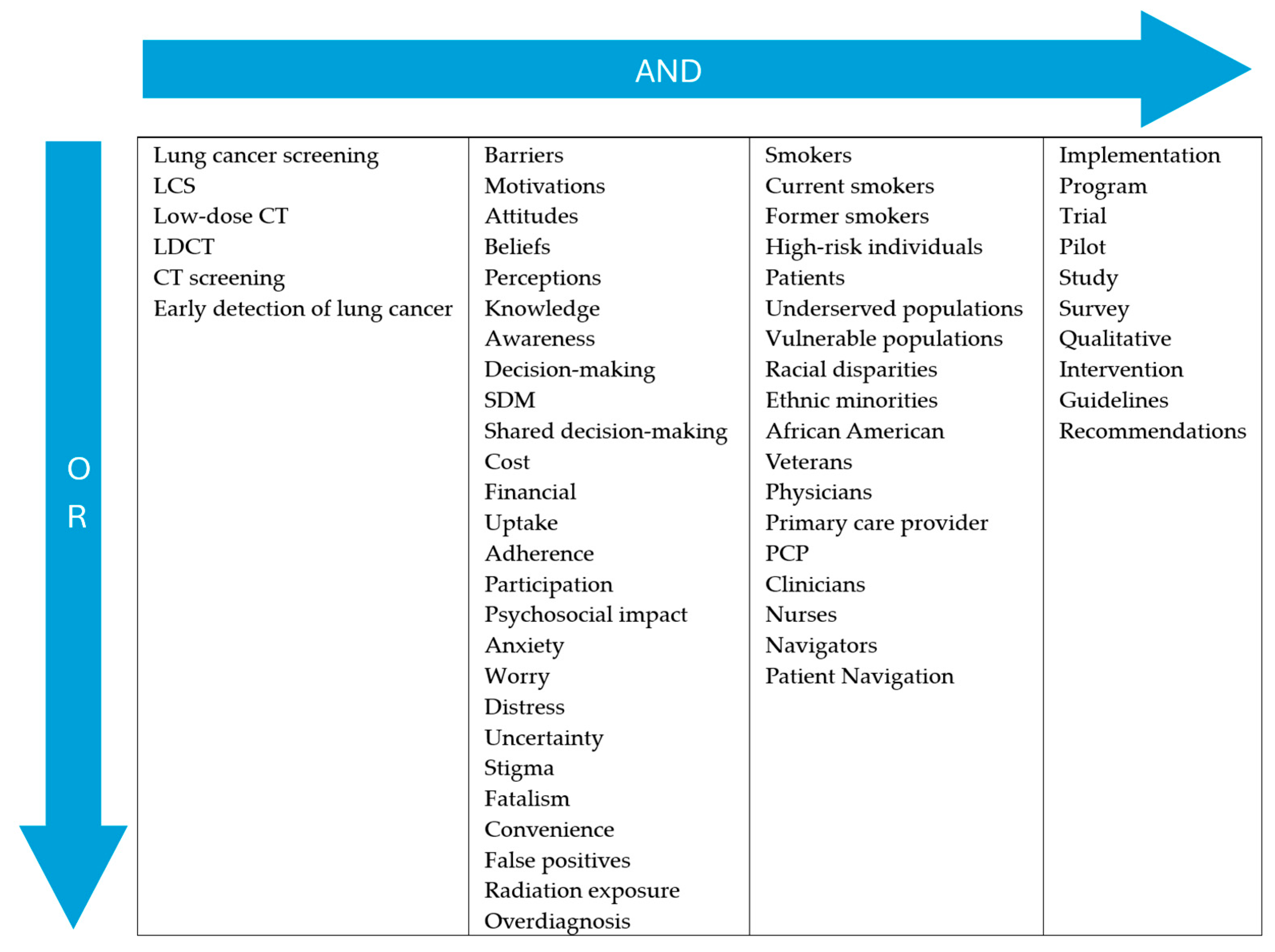
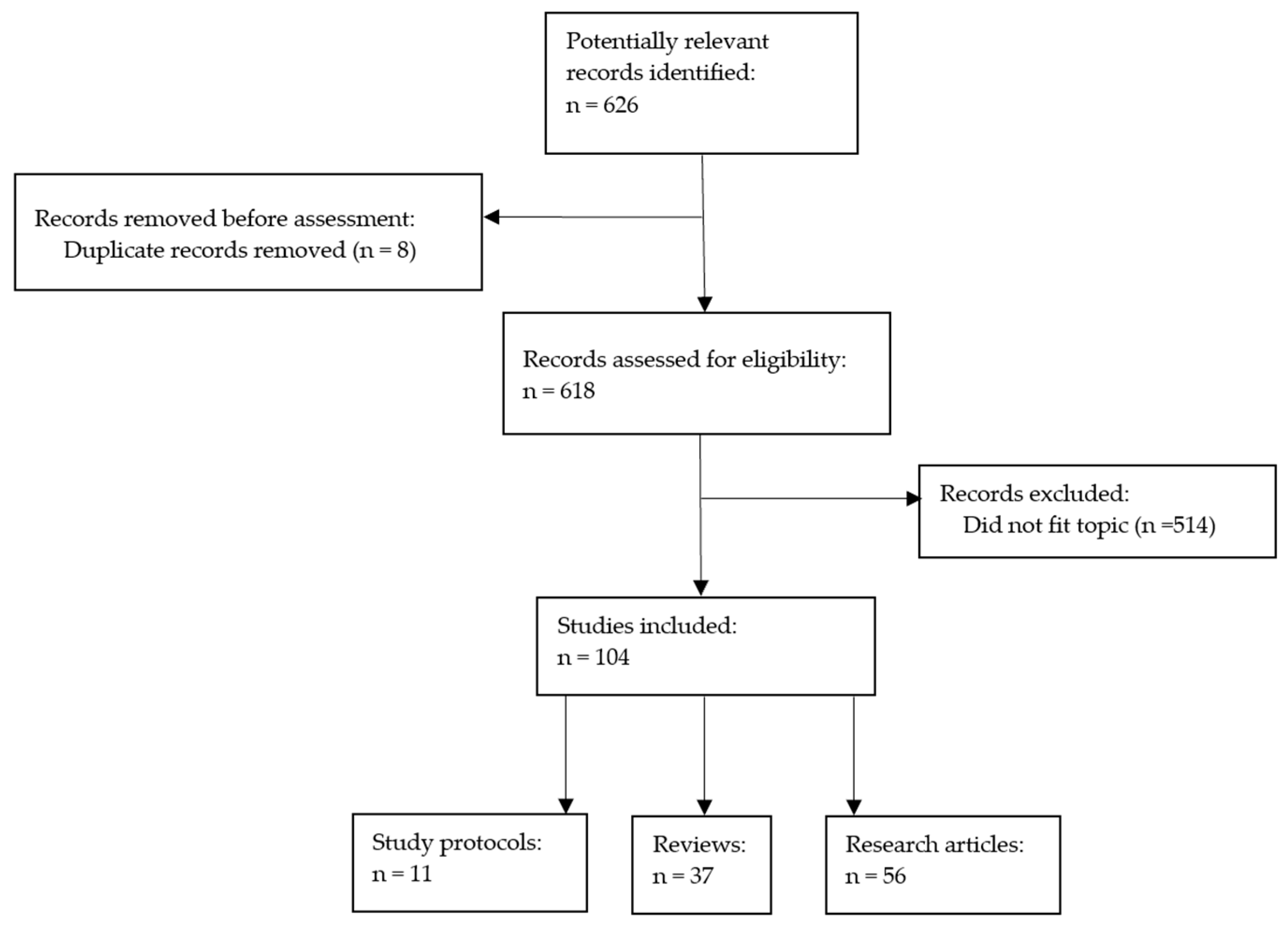
2. Methodology: A Systematized Search Strategy
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
2.4. Selection Process
3. Patient-Level Factors: Psychosocial and Socioeconomic Barriers
3.1. Knowledge and Awareness
3.1.1. Lack of Awareness About Eligibility and the Concept of Screening
3.1.2. Misconceptions That “No Symptoms” Means “No Screening Needed”
3.2. Psychosocial Beliefs and Stigma
3.2.1. Fear and Fatalism
| Study Population | Barrier or Reason for Delaying or Avoiding Screening | Explanation | Prevalence (%) | |
|---|---|---|---|---|
| Smokers aged 55–80 [46] | Current smoker | Perceiving current smoking status as a reason to delay | 62.2% | |
| Lack of knowledge | About the test | 56.4% | ||
| Worry about result | Anxiety regarding potential findings | 56.1% | ||
| Lack of symptoms | Belief that lack of symptoms means screening is unnecessary | 45.0% | ||
| High cost | Concern regarding financial burden | 38.1% | ||
| Stigma/blame | Worry about being blamed for having smoked | 36.9% | ||
| Fear of harm from LDCT | Fear of radiation effects on health | 9.0% | ||
| Current smokers declining screening [63] | Cost/no insurance | Cited as reason for not getting screened | 33.3% | |
| Fear of finding cancer | Afraid to find out whether they had lung cancer | 33.3% | ||
| Older smokers (aged ≥ 55) [66] | Fear of finding cancer | “Is afraid CT scan will find cancer” | 51.3% | |
| Radiation fear | “Afraid radiation could cause lung cancer” | 39.1% | ||
| Fear of scanning process | “Scared of CT scans” (general fear of the procedure) | 32.8% | ||
| Fatalism | Belief that “The treatment is more of a suffering than the disease itself” | 47.4% | ||
| Current/former Smokers (Pakistan) [43] | Health anxiety | “Screening would only make you feel more anxious about your health” | 68.6% | |
| Financial cost | “Lung cancer screening is too expensive for you to afford” | 61.4% | ||
| Fear of positive result | “You’re afraid of a positive result” | 52.6% | ||
| Fear of hospitals/scanners | “Fear of hospitals and CT scanners prevent you from screening” | 39.1% | ||
| LCS program participants (racial disparities) [65] | Cost | Percentage rating cost as a “Very Important” factor in decision-making | Black | 58.4% |
| White | 37.8% | |||
| Convenience | Percentage rating convenience as a “Very Important” factor in decision-making | Black | 60.0% | |
| White | 26.8% | |||
| Risk of disease | Percentage rating risk of disease as a “Very Important” factor in decision-making | Black | 61.4% | |
| White | 45.1% | |||
3.2.2. Stigma
3.3. The Smoking “Teachable Moment”
3.3.1. How Screening Impacts Motivation to Quit Smoking
3.3.2. Mixed Evidence on Negative Scans: “License to Smoke” vs. Motivation to Quit
3.3.3. Impact of Abnormal Findings and the Necessity of Integration
3.4. Financial and Insurance Barriers
3.4.1. Cost Concerns Persist Despite Coverage Expansion
3.4.2. The “Diagnostic Gap” and Downstream Financial Toxicity
3.4.3. Coverage Gaps and Variation
3.4.4. Indirect Costs, Administrative Barriers, and Mitigation Strategies
3.5. Screening Disparities in Black/African American Populations and the Role of Educational Attainment
3.5.1. Adherence Among African American Populations and the “African American Smoking Paradox”
3.5.2. Disparities Among Those with Lower Educational Attainment and SES
3.5.3. Race and Screening Program Structure
4. Provider-Level Factors: The Gatekeepers
4.1. Knowledge and Guideline Familiarity
4.1.1. Gaps in Primary Care Provider (PCP) Knowledge Regarding Eligibility Criteria Leading to Under-Referral
4.1.2. Fundamental Misunderstandings of the Screening Concept
4.1.3. Confusion Regarding Management of Incidental Findings
4.1.4. Complexity of Incidental and Non-Target Findings
4.2. Time Constraints in SDM
4.2.1. Universal Time Pressures in Primary Care Practice
4.2.2. U.S.-Specific Regulatory Constraints: The CMS SDM Requirement
4.2.3. Structural and Organizational Modifiers of Time Constraints
4.3. Quality of Communication
4.3.1. Clinician Overemphasis on Benefits vs. Harms
4.3.2. Reliance on “Check-Box” SDM Rather than Meaningful Dialogue
4.4. Therapeutic Alliance
4.4.1. The Role of Trust in the Provider as a Primary Motivator for Screening Completion
4.4.2. Deference to Provider Judgment for Mitigating Distress and Ensuring Adherence
4.4.3. Addressing Mistrust and Disparities
5. Facilitators and Interventions: Strategies to Improve Adherence
5.1. Patient Navigation
5.1.1. Reducing Barriers and Bridging Gaps in SDOH
5.1.2. Impact on Uptake, Adherence, and Equity
5.1.3. Efficacy in Vulnerable Populations
5.1.4. Distinction from System-Level Coordinators
5.2. DA Effectiveness
5.2.1. Video and Web-Based DAs
5.2.2. Increasing Knowledge, Decisional Quality, and Preparedness
5.2.3. Mixed Evidence on Screening Uptake
5.2.4. Tailoring Tools to Smoking Status and Specific Barriers
5.3. Targeted Outreach
5.3.1. Culturally Sensitive Materials and “Citizen Scientists”
5.3.2. CHWs
5.3.3. Mobile Screening Units
5.3.4. Rebranding as a “Lung Health Check” (LHC)
5.3.5. Social Media and Digital Outreach
5.3.6. Accessibility and Health Literacy
5.4. Communication Strategies and Therapeutic Alliance
5.4.1. Therapeutic Alliance and Provider Trust
5.4.2. Person-Centered Communication
5.4.3. Addressing Medical Mistrust
5.5. Leveraging the Screening Process as a “Teachable Moment”
6. Limitations
7. Priorities for Future Research
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CHW | Community health worker |
| CMS | Centers for Medicare & Medicaid Services |
| CXR | Chest X-ray |
| DA | Decision aid |
| DANTE | Detection And screening of early lung cancer |
| DLCST | Danish Lung Cancer Screening Trial |
| EHR | Electronic health record |
| FBTA | Facebook targeted advertisements |
| GP | General practitioner |
| HR | Hazard ratio |
| ITALUNG | Italian Lung Cancer Screening |
| LCS | Lung cancer screening |
| LDCT | Low-dose computed tomography |
| LHC | Lung Health Check |
| Lung-RADS | Lung Imaging Reporting and Data System |
| LSUT | Lung Screen Uptake Trial |
| LUSI | German Lung Cancer Screening Intervention |
| MILD | Multicentric Italian Lung Detection |
| NLST | National Lung Screening Trial |
| NRT | Nicotine replacement therapy |
| OR | Odds ratio |
| PCP | Primary care provider |
| RR | Relative risk |
| SDM | Shared decision-making |
| SDOH | Social determinants of health |
| SES | Socioeconomic status |
| UKLS | UK Lung Cancer Screening Pilot |
| USPSTF | U.S. Preventive Services Task Force |
| VHA | Veterans Health Administration |
References
- Ferlay, J.; Shin, H.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of Worldwide Burden of Cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef]
- Ferlay, J.; Steliarova-Foucher, E.; Lortet-Tieulent, J.; Rosso, S.; Coebergh, J.W.W.; Comber, H.; Forman, D.; Bray, F. Cancer Incidence and Mortality Patterns in Europe: Estimates for 40 Countries in 2012. Eur. J. Cancer 2013, 49, 1374–1403. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Ervik, M.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.; Forman, D.; Bray, D. GLOBOCAN 2012: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012 v1.0; International Agency for Research on Cancer (IARC): Lyon, France, 2013; ISBN 978-92-832-2447-1. [Google Scholar]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Garshell, J.; Miller, D.; Altekruse, S.F.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; et al. SEER Cancer Statistics Review, 1975–2012; National Cancer Institute: Bethesda, MD, USA, 2014.
- United States Census Bureau. Intercensal Estimates of the Resident Population, 2000–2010; United States Census Bureau: Washington, DC, USA, 2010.
- Eurostat. Population on 1 January by Age and Sex; Eurostat: Luxembourg, 2015. [Google Scholar]
- The National Lung Screening Trial Research Team Reduced Lung-Cancer Mortality with Low-Dose Computed Tomographic Screening. N. Engl. J. Med. 2011, 365, 395–409. [CrossRef] [PubMed]
- Bach, P.B.; Mirkin, J.N.; Oliver, T.K.; Azzoli, C.G.; Berry, D.A.; Brawley, O.W.; Byers, T.; Colditz, G.A.; Gould, M.K.; Jett, J.R.; et al. Benefits and Harms of CT Screening for Lung Cancer: A Systematic Review. JAMA 2012, 307, 2418. [Google Scholar] [CrossRef] [PubMed]
- Tammemägi, M.C.; Katki, H.A.; Hocking, W.G.; Church, T.R.; Caporaso, N.; Kvale, P.A.; Chaturvedi, A.K.; Silvestri, G.A.; Riley, T.L.; Commins, J.; et al. Selection Criteria for Lung-Cancer Screening. N. Engl. J. Med. 2013, 368, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Gage, J.C.; Schiffman, M.; Katki, H.A.; Castle, P.E.; Fetterman, B.; Wentzensen, N.; Poitras, N.E.; Lorey, T.; Cheung, L.C.; Kinney, W.K. Reassurance Against Future Risk of Precancer and Cancer Conferred by a Negative Human Papillomavirus Test. JNCI J. Natl. Cancer Inst. 2014, 106, dju153. [Google Scholar] [CrossRef]
- Oudkerk, M.; Devaraj, A.; Vliegenthart, R.; Henzler, T.; Prosch, H.; Heussel, C.P.; Bastarrika, G.; Sverzellati, N.; Mascalchi, M.; Delorme, S.; et al. European Position Statement on Lung Cancer Screening. Lancet Oncol. 2017, 18, e754–e766. [Google Scholar] [CrossRef] [PubMed]
- De Koning, H.J.; Van Der Aalst, C.M.; De Jong, P.A.; Scholten, E.T.; Nackaerts, K.; Heuvelmans, M.A.; Lammers, J.-W.J.; Weenink, C.; Yousaf-Khan, U.; Horeweg, N.; et al. Reduced Lung-Cancer Mortality with Volume CT Screening in a Randomized Trial. N. Engl. J. Med. 2020, 382, 503–513. [Google Scholar] [CrossRef]
- McKee, B.J.; McKee, A.B.; Regis, S.M. Low-Dose Computed Tomography Screening for Lung Cancer in a Clinical Setting: Essential Elements of a Screening Program. J. Thorac. Imaging 2015, 30, 115–129. [Google Scholar] [CrossRef]
- Bonney, A.; Malouf, R.; Marchal, C.; Manners, D.; Fong, K.M.; Marshall, H.M.; Irving, L.B.; Manser, R. Impact of Low-Dose Computed Tomography (LDCT) Screening on Lung Cancer-Related Mortality. Cochrane Database Syst. Rev. 2022, 2022, CD013829. [Google Scholar] [CrossRef]
- Lancaster, H.L.; Heuvelmans, M.A.; Oudkerk, M. Low-Dose Computed Tomography Lung Cancer Screening: Clinical Evidence and Implementation Research. J. Intern. Med. 2022, 292, 68–80. [Google Scholar] [CrossRef]
- Van Hal, G.; Diab Garcia, P. Lung Cancer Screening: Targeting the Hard to Reach—A Review. Transl. Lung Cancer Res. 2021, 10, 2309–2322. [Google Scholar] [CrossRef]
- Infante, M.; Cavuto, S.; Lutman, F.R.; Brambilla, G.; Chiesa, G.; Ceresoli, G.; Passera, E.; Angeli, E.; Chiarenza, M.; Aranzulla, G.; et al. A Randomized Study of Lung Cancer Screening with Spiral Computed Tomography. Am. J. Respir. Crit. Care Med. 2009, 180, 445–453. [Google Scholar] [CrossRef]
- Pedersen, J.H.; Ashraf, H.; Dirksen, A.; Bach, K.; Hansen, H.; Toennesen, P.; Thorsen, H.; Brodersen, J.; Skov, B.G.; Døssing, M.; et al. The Danish Randomized Lung Cancer CT Screening Trial—Overall Design and Results of the Prevalence Round. J. Thorac. Oncol. 2009, 4, 608–614. [Google Scholar] [CrossRef]
- Becker, N.; Motsch, E.; Trotter, A.; Heussel, C.P.; Dienemann, H.; Schnabel, P.A.; Kauczor, H.; Maldonado, S.G.; Miller, A.B.; Kaaks, R.; et al. Lung Cancer Mortality Reduction by LDCT Screening—Results from the Randomized German LUSI Trial. Int. J. Cancer 2020, 146, 1503–1513. [Google Scholar] [CrossRef]
- Field, J.K.; Vulkan, D.; Davies, M.P.A.; Baldwin, D.R.; Brain, K.E.; Devaraj, A.; Eisen, T.; Gosney, J.; Green, B.A.; Holemans, J.A.; et al. Lung Cancer Mortality Reduction by LDCT Screening: UKLS Randomised Trial Results and International Meta-Analysis. Lancet Reg. Health–Eur. 2021, 10, 100179. [Google Scholar] [CrossRef]
- Hunger, T.; Wanka-Pail, E.; Brix, G.; Griebel, J. Lung Cancer Screening with Low-Dose CT in Smokers: A Systematic Review and Meta-Analysis. Diagnostics 2021, 11, 1040. [Google Scholar] [CrossRef] [PubMed]
- Sands, J.; Tammemägi, M.C.; Couraud, S.; Baldwin, D.R.; Borondy-Kitts, A.; Yankelevitz, D.; Lewis, J.; Grannis, F.; Kauczor, H.-U.; Von Stackelberg, O.; et al. Lung Screening Benefits and Challenges: A Review of The Data and Outline for Implementation. J. Thorac. Oncol. 2021, 16, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Veronesi, G.; Baldwin, D.; Henschke, C.; Ghislandi, S.; Iavicoli, S.; Oudkerk, M.; De Koning, H.; Shemesh, J.; Field, J.; Zulueta, J.; et al. Recommendations for Implementing Lung Cancer Screening with Low-Dose Computed Tomography in Europe. Cancers 2020, 12, 1672. [Google Scholar] [CrossRef]
- O’Dowd, E.L.; Baldwin, D.R. Lung Cancer Screening—Low Dose CT for Lung Cancer Screening: Recent Trial Results and next Steps. Br. J. Radiol. 2018, 91, 20170460. [Google Scholar] [CrossRef] [PubMed]
- Field, J.K.; Duffy, S.W.; Baldwin, D.R.; Whynes, D.K.; Devaraj, A.; Brain, K.E.; Eisen, T.; Gosney, J.; Green, B.A.; Holemans, J.A.; et al. UK Lung Cancer RCT Pilot Screening Trial: Baseline Findings from the Screening Arm Provide Evidence for the Potential Implementation of Lung Cancer Screening. Thorax 2016, 71, 161–170. [Google Scholar] [CrossRef]
- Castro, S.; Sosa, E.; Lozano, V.; Akhtar, A.; Love, K.; Duffels, J.; Raz, D.J.; Kim, J.Y.; Sun, V.; Erhunmwunsee, L. The Impact of Income and Education on Lung Cancer Screening Utilization, Eligibility, and Outcomes: A Narrative Review of Socioeconomic Disparities in Lung Cancer Screening. J. Thorac. Dis. 2021, 13, 3745–3757. [Google Scholar] [CrossRef] [PubMed]
- Karanth, S.D.; Divaker, J.; Blair, M.; Gray, J.E.; Hochhegger, B.; Kobetz, E.; Machuca, T.; Rackauskas, M.; Yang, D.; Rodriguez, E.; et al. Rethinking How Mobile Units Can Catalyze Progress on Lung Cancer Screening: A Scoping Review of What We Have Learned. J. Thorac. Dis. 2024, 16, 7143–7154. [Google Scholar] [CrossRef]
- Shusted, C.S.; Barta, J.A.; Lake, M.; Brawer, R.; Ruane, B.; Giamboy, T.E.; Sundaram, B.; Evans, N.R.; Myers, R.E.; Kane, G.C. The Case for Patient Navigation in Lung Cancer Screening in Vulnerable Populations: A Systematic Review. Popul. Health Manag. 2019, 22, 347–361. [Google Scholar] [CrossRef] [PubMed]
- Adnan, S.M.; Chin, K.; Ma, G.X.; Erkmen, C.P. A Narrative Review of the Social Determinants of Lung Cancer Screening: Knowledge Gaps and Controversies. Curr. Chall. Thorac. Surg. 2023, 5, 41. [Google Scholar] [CrossRef]
- Haddad, D.N.; Sandler, K.L.; Henderson, L.M.; Rivera, M.P.; Aldrich, M.C. Disparities in Lung Cancer Screening: A Review. Ann. Am. Thorac. Soc. 2020, 17, 399–405. [Google Scholar] [CrossRef]
- Shehata, D.G.; Pan, J.M.; Pan, Z.; Vigneswaran, J.; Contreras, N.; Rodriguez, E.; Sakowitz, S.; Magarinos, J.; Pereira, S.; Wilder, F.G.; et al. Equity and Opportunities in Lung Cancer Care—Addressing Disparities, Challenges, and Pathways Forward. Cancers 2025, 17, 1347. [Google Scholar] [CrossRef]
- Lam, S.; Bai, C.; Baldwin, D.R.; Chen, Y.; Connolly, C.; De Koning, H.; Heuvelmans, M.A.; Hu, P.; Kazerooni, E.A.; Lancaster, H.L.; et al. Current and Future Perspectives on Computed Tomography Screening for Lung Cancer: A Roadmap from 2023 to 2027 from the International Association for the Study of Lung Cancer. J. Thorac. Oncol. 2024, 19, 36–51. [Google Scholar] [CrossRef] [PubMed]
- Nunez, E.R.; Steiling, K.A.; Litle, V.R. Opportunities and Challenges in Lung Cancer Screening Implementation: A Narrative Review. Curr. Chall. Thorac. Surg. 2023, 5, 1. [Google Scholar] [CrossRef]
- Rankin, N.M.; McWilliams, A.; Marshall, H.M. Lung Cancer Screening Implementation: Complexities and Priorities. Respirology 2020, 25, 5–23. [Google Scholar] [CrossRef]
- Erkmen, C.P.; Dako, F.; Moore, R.; Dass, C.; Weiner, M.G.; Kaiser, L.R.; Ma, G.X. Adherence to Annual Lung Cancer Screening with Low-Dose CT Scan in a Diverse Population. Cancer Causes Control 2021, 32, 291–298. [Google Scholar] [CrossRef]
- Hirsch, E.A.; New, M.L.; Brown, S.P.; Barón, A.E.; Malkoski, S.P. Patient Reminders and Longitudinal Adherence to Lung Cancer Screening in an Academic Setting. Ann. Am. Thorac. Soc. 2019, 16, 1329–1332. [Google Scholar] [CrossRef]
- Sow, M.K.-S.; Howlett, J.; Bell, R.; Odell, D.; Ekeke, C.N. A Narrative Review of Lung Cancer Screening: From Adoption to Maturity. AME Surg. J. 2024, 4, 22. [Google Scholar] [CrossRef]
- Kim, R.Y.; Rendle, K.A.; Mitra, N.; Saia, C.A.; Neslund-Dudas, C.; Greenlee, R.T.; Burnett-Hartman, A.N.; Honda, S.A.; Simoff, M.J.; Schapira, M.M.; et al. Racial Disparities in Adherence to Annual Lung Cancer Screening and Recommended Follow-Up Care: A Multicenter Cohort Study. Ann. Am. Thorac. Soc. 2022, 19, 1561–1569. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute (NCI). National Lung Screening Trial A Randomized Trial Comparing Low-Dose Helical CT With Chest Xray for Lung Cancer; clinicaltrials.gov, 2014. Available online: https://clinicaltrials.gov/study/NCT00047385?tab=history&a=25#version-content-panel (accessed on 26 November 2025).
- Carter-Bawa, L.; Banerjee, S.C.; Comer, R.S.; Kale, M.S.; King, J.C.; Leopold, K.T.; Monahan, P.O.; Ostroff, J.S.; Slaven, J.E.; Valenzona, F.; et al. Leveraging Social Media to Increase Lung Cancer Screening Awareness, Knowledge and Uptake among High-Risk Populations (The INSPIRE-Lung Study): Study Protocol of Design and Methods of a Community-Based Randomized Controlled Trial. BMC Public Health 2023, 23, 975. [Google Scholar] [CrossRef]
- Carter-Bawa, L.; Slaven, J.E., Jr.; Monahan, P.O.; Brandzel, S.; Gao, H.; Wernli, K.J.; Lafata, J.E.; Rawl, S.M. Unpacking the Relationship Between Shared Decision-Making and Decisional Quality, Decision to Screen, and Screening Completion in Lung Cancer Screening. Patient Educ. Couns. 2024, 122, 108143. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.X.; Baggett, T.P.; Pandharipande, P.V.; Park, E.R.; Percac-Lima, S.; Shepard, J.-A.O.; Fintelmann, F.J.; Flores, E.J. Barriers to Lung Cancer Screening Engagement from the Patient and Provider Perspective. Radiology 2019, 290, 278–287. [Google Scholar] [CrossRef]
- Abbasi, A.; Siddiqi, R.; Owais, A.; Laeeq, T.; Ali, S.N.; Mushahid, Z.; Ahsan, S.M.; Jatoi, A.S.; Abbasi, A.; Butt, I.; et al. Prevalence and Barriers to Lung Cancer Screening in Karachi, Pakistan: A Cross-Sectional Survey of Smokers and Physicians. Cureus 2017, 9, e1248. [Google Scholar] [CrossRef]
- Sharma, A.; Kasza, K.; Hyland, A.; Cummings, K.M.; Bansal-Travers, M.; Fong, G.T.; O’Connor, R.J. Awareness and Interest in Lung Cancer Screening among Current and Former Smokers: Findings from the ITC United States Survey. Cancer Causes Control 2019, 30, 733–745. [Google Scholar] [CrossRef]
- Gressard, L.; DeGroff, A.S.; Richards, T.B.; Melillo, S.; Kish-Doto, J.; Heminger, C.L.; Rohan, E.A.; Allen, K.G. A Qualitative Analysis of Smokers’ Perceptions About Lung Cancer Screening. BMC Public Health 2017, 17, 589. [Google Scholar] [CrossRef]
- Raz, D.J.; Wu, G.; Nelson, R.A.; Sun, V.; Wu, S.; Alem, A.; Haupt, E.C.; Ismail, M.H.; Gould, M.K. Perceptions and Utilization of Lung Cancer Screening Among Smokers Enrolled in a Tobacco Cessation Program. Clin. Lung Cancer 2019, 20, e115–e122. [Google Scholar] [CrossRef] [PubMed]
- Tan, N.Q.P.; Lowenstein, L.M.; Douglas, E.E.; Silva, J.; Bershad, J.M.; An, J.; Shete, S.S.; Steinberg, M.B.; Ferrante, J.M.; Clark, E.C.; et al. The TELEhealth Shared Decision-Making COaching and Navigation in Primary carE (TELESCOPE) Intervention: A Study Protocol for Delivering Shared Decision-Making for Lung Cancer Screening by Patient Navigators. BMC Prim. Care 2024, 25, 373. [Google Scholar] [CrossRef]
- Crothers, K.; Kross, E.K.; Reisch, L.M.; Shahrir, S.; Slatore, C.; Zeliadt, S.B.; Triplette, M.; Meza, R.; Elmore, J.G. Patients’ Attitudes Regarding Lung Cancer Screening and Decision Aids. A Survey and Focus Group Study. Ann. Am. Thorac. Soc. 2016, 13, 1992–2001. [Google Scholar] [CrossRef]
- Navuluri, N.; Lanford, T.; Shapiro, A.; Krishnan, G.; Johnson, A.B.; Riley, I.L.; Zullig, L.L.; Cox, C.E.; Shofer, S. Barriers and Facilitators Impacting Lung Cancer Screening Uptake Among Black Veterans: A Qualitative Study. J. Natl. Compr. Cancer Netw. 2024, 22, 231–236. [Google Scholar] [CrossRef]
- Wiener, R.S.; Clark, J.A.; Koppelman, E.; Bolton, R.; Fix, G.M.; Slatore, C.G.; Kathuria, H. Patient vs Clinician Perspectives on Communication About Results of Lung Cancer Screening. Chest 2020, 158, 1240–1249. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, L.L.; Vadagam, P.; Vanderpoel, J.; Cohen, C.; Lewing, B.; Tkacz, J. Disparities in Lung Cancer: A Targeted Literature Review Examining Lung Cancer Screening, Diagnosis, Treatment, and Survival Outcomes in the United States. J. Racial Ethn. Health Disparities 2024, 11, 1489–1500. [Google Scholar] [CrossRef]
- Watson, K.S.; Siegel, L.D.; Henderson, V.A.; Murray, M.; Chukwudozie, I.B.; Odell, D.; Stinson, J.; Ituah, O.; Ben Levi, J.; Fitzgibbon, M.L.; et al. The SHARED Project: A Novel Approach to Engaging African American Men to Address Lung Cancer Disparities. Am. J. Mens Health 2020, 14, 1557988320958934. [Google Scholar] [CrossRef]
- Toubat, O.; David, E.A. A Narrative Review of Lung Cancer Screening in Underserved Populations. Curr. Chall. Thorac. Surg. 2023, 5, 6. [Google Scholar] [CrossRef]
- Mazières, J.; Pujol, J.-L.; Kalampalikis, N.; Bouvry, D.; Quoix, E.; Filleron, T.; Targowla, N.; Jodelet, D.; Milia, J.; Milleron, B. Perception of Lung Cancer among the General Population and Comparison with Other Cancers. J. Thorac. Oncol. 2015, 10, 420–425. [Google Scholar] [CrossRef]
- Rodríguez-Rabassa, M.S.; Simmons, V.N.; Vega, A.; Moreno, D.; Irizarry-Ramos, J.; Quinn, G.P. Perceptions of and Barriers to Lung Cancer Screening Among Physicians in Puerto Rico: A Qualitative Study. J. Health Care Poor Underserved 2020, 31, 973–991. [Google Scholar] [CrossRef] [PubMed]
- Lam, A.C.L.; Aggarwal, R.; Cheung, S.; Stewart, E.L.; Darling, G.; Lam, S.; Xu, W.; Liu, G.; Kavanagh, J. Predictors of Participant Nonadherence in Lung Cancer Screening Programs: A Systematic Review and Meta-Analysis. Lung Cancer 2020, 146, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Katsampouris, E.; Creamer, A.W.; Prendecki, R.; Clark, E.; Dickson, J.L.; Lee, R.; Janes, S.M.; Duffy, S.W.; Quaife, S.L. Patients’ Experiences of, and Psychological Responses to, Surveillance for Pulmonary Nodules Detected Through Lung Cancer Screening. BMJ Open Respir. Res. 2025, 12, e002498. [Google Scholar] [CrossRef]
- Quaife, S.L.; Ruparel, M.; Beeken, R.J.; McEwen, A.; Isitt, J.; Nolan, G.; Sennett, K.; Baldwin, D.R.; Duffy, S.W.; Janes, S.M.; et al. The Lung Screen Uptake Trial (LSUT): Protocol for a Randomised Controlled Demonstration Lung Cancer Screening Pilot Testing a Targeted Invitation Strategy for High Risk and ‘Hard-to-Reach’ Patients. BMC Cancer 2016, 16, 281. [Google Scholar] [CrossRef]
- Carter-Harris, L.; Brandzel, S.; Wernli, K.J.; Roth, J.A.; Buist, D.S.M. A Qualitative Study Exploring Why Individuals Opt out of Lung Cancer Screening. Fam. Pract. 2017, 34, 239–244. [Google Scholar] [CrossRef]
- Ali, N.; Lifford, K.J.; Carter, B.; McRonald, F.; Yadegarfar, G.; Baldwin, D.R.; Weller, D.; Hansell, D.M.; Duffy, S.W.; Field, J.K.; et al. Barriers to Uptake Among High-Risk Individuals Declining Participation in Lung Cancer Screening: A Mixed Methods Analysis of the UK Lung Cancer Screening (UKLS) Trial. BMJ Open 2015, 5, e008254. [Google Scholar] [CrossRef]
- Melzer, A.C.; Golden, S.E.; Ono, S.S.; Datta, S.; Crothers, K.; Slatore, C.G. What Exactly Is Shared Decision-Making? A Qualitative Study of Shared Decision-Making in Lung Cancer Screening. J. Gen. Intern. Med. 2020, 35, 546–553. [Google Scholar] [CrossRef]
- Luce, C.; Palazzo, L.; Anderson, M.L.; Carter-Bawa, L.; Gao, H.; Green, B.B.; Ralston, J.D.; Rogers, K.; Su, Y.-R.; Tuzzio, L.; et al. A Pragmatic Randomized Clinical Trial of Multilevel Interventions to Improve Adherence to Lung Cancer Screening (The Larch Study): Study Protocol. Contemp. Clin. Trials 2024, 140, 107495. [Google Scholar] [CrossRef]
- Delmerico, J.; Hyland, A.; Celestino, P.; Reid, M.; Cummings, K.M. Patient Willingness and Barriers to Receiving a CT Scan for Lung Cancer Screening. Lung Cancer 2014, 84, 307–309. [Google Scholar] [CrossRef]
- Cavers, D.; Nelson, M.; Rostron, J.; Robb, K.A.; Brown, L.R.; Campbell, C.; Akram, A.R.; Dickie, G.; Mackean, M.; Van Beek, E.J.R.; et al. Understanding Patient Barriers and Facilitators to Uptake of Lung Screening Using Low Dose Computed Tomography: A Mixed Methods Scoping Review of the Current Literature. Respir. Res. 2022, 23, 374. [Google Scholar] [CrossRef] [PubMed]
- Barta, J.A.; Shusted, C.S.; Ruane, B.; Pimpinelli, M.; McIntire, R.K.; Zeigler-Johnson, C.; Myers, R.E.; Evans, N.R.; Kane, G.C.; Juon, H.-S. Racial Differences in Lung Cancer Screening Beliefs and Screening Adherence. Clin. Lung Cancer 2021, 22, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Cataldo, J.K. High-Risk Older Smokers’ Perceptions, Attitudes, and Beliefs about Lung Cancer Screening. Cancer Med. 2016, 5, 753–759. [Google Scholar] [CrossRef]
- Crosbie, P.A.; Gabe, R.; Simmonds, I.; Kennedy, M.; Rogerson, S.; Ahmed, N.; Baldwin, D.R.; Booton, R.; Cochrane, A.; Darby, M.; et al. Yorkshire Lung Screening Trial (YLST): Protocol for a Randomised Controlled Trial to Evaluate Invitation to Community-Based Low-Dose CT Screening for Lung Cancer versus Usual Care in a Targeted Population at Risk. BMJ Open 2020, 10, e037075. [Google Scholar] [CrossRef]
- Slatore, C.G.; Wiener, R.S. Pulmonary Nodules. Chest 2018, 153, 1004–1015. [Google Scholar] [CrossRef]
- Wu, G.X.; Raz, D.J.; Brown, L.; Sun, V. Psychological Burden Associated with Lung Cancer Screening: A Systematic Review. Clin. Lung Cancer 2016, 17, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.C.; Hamann, H.A.; Browning, T.; Santini, N.O.; Abbara, S.; Balis, D.S.; Chiu, H.; Moran, B.A.; McGuire, M.; Gerber, D.E. Stakeholder Engagement to Initiate Lung Cancer Screening in an Urban Safety-Net Health System. Healthcare 2020, 8, 100370. [Google Scholar] [CrossRef]
- Gerber, D.E.; Hamann, H.A.; Santini, N.O.; Abbara, S.; Chiu, H.; McGuire, M.; Quirk, L.; Zhu, H.; Lee, S.J.C. Patient Navigation for Lung Cancer Screening in an Urban Safety-Net System: Protocol for a Pragmatic Randomized Clinical Trial. Contemp. Clin. Trials 2017, 60, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Brown-Johnson, C.G.; Berrean, B.; Cataldo, J.K. Development and Usability Evaluation of the mHealth Tool for Lung Cancer (mHealth TLC): A Virtual World Health Game for Lung Cancer Patients. Patient Educ. Couns. 2015, 98, 506–511. [Google Scholar] [CrossRef]
- Greene, P.A.; Sayre, G.; Heffner, J.L.; Klein, D.E.; Krebs, P.; Au, D.H.; Zeliadt, S.B. Challenges to Educating Smokers About Lung Cancer Screening: A Qualitative Study of Decision Making Experiences in Primary Care. J. Cancer Educ. 2019, 34, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Ostroff, J.S.; Shelley, D.R.; Chichester, L.-A.; King, J.C.; Li, Y.; Schofield, E.; Ciupek, A.; Criswell, A.; Acharya, R.; Banerjee, S.C.; et al. Study Protocol of a Multiphase Optimization Strategy Trial (MOST) for Delivery of Smoking Cessation Treatment in Lung Cancer Screening Settings. Trials 2022, 23, 664. [Google Scholar] [CrossRef]
- Maguire, R.; Lewis, L.; Kotronoulas, G.; McPhelim, J.; Milroy, R.; Cataldo, J. Lung Cancer Stigma: A Concept with Consequences for Patients. Cancer Rep. 2019, 2, e1201. [Google Scholar] [CrossRef]
- Green, D.B.; Pua, B.B.; Crawford, C.B.; Abbey, G.N.; Drexler, I.R.; Legasto, A.C.; Gruden, J.F. Screening for Lung Cancer: Communicating with Patients. Am. J. Roentgenol. 2018, 210, 497–502. [Google Scholar] [CrossRef]
- Williams, R.M.; Cordon, M.; Eyestone, E.; Smith, L.; Luta, G.; McKee, B.J.; Regis, S.M.; Abrams, D.B.; Niaura, R.S.; Stanton, C.A.; et al. Improved Motivation and Readiness to Quit Shortly After Lung Cancer Screening: Evidence for a Teachable Moment. Cancer 2022, 128, 1976–1986. [Google Scholar] [CrossRef]
- Moizs, M.; Bajzik, G.; Lelovics, Z.; Rakvács, M.; Strausz, J.; Repa, I. First Result of Differentiated Communication—To Smokers and Non-Smokers—In Order to Increase the Voluntary Participation Rate in Lung Screening. BMC Public Health 2013, 13, 914. [Google Scholar] [CrossRef]
- Quaife, S.L.; Waller, J.; Dickson, J.L.; Brain, K.E.; Kurtidu, C.; McCabe, J.; Hackshaw, A.; Duffy, S.W.; Janes, S.M. Psychological Targets for Lung Cancer Screening Uptake: A Prospective Longitudinal Cohort Study. J. Thorac. Oncol. 2021, 16, 2016–2028. [Google Scholar] [CrossRef]
- Martini, K.; Chassagnon, G.; Frauenfelder, T.; Revel, M.-P. Ongoing Challenges in Implementation of Lung Cancer Screening. Transl. Lung Cancer Res. 2021, 10, 2347–2355. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Williams, R.M.; Eyestone, E.; Cordon, M.; Smith, L.; Davis, K.; Luta, G.; Anderson, E.D.; McKee, B.; Batlle, J.; et al. Predictors of Attrition in a Smoking Cessation Trial Conducted in the Lung Cancer Screening Setting. Contemp. Clin. Trials 2021, 106, 106429. [Google Scholar] [CrossRef]
- Robichaux, C.; Anderson, M.; Freese, R.; Stately, A.; Begnaud, A. Lung Cancer Screening Outreach Program in an Urban Native American Clinic. J. Prim. Care Community Health 2023, 14, 21501319231212312. [Google Scholar] [CrossRef]
- Park, E.R.; Gareen, I.F.; Jain, A.; Ostroff, J.S.; Duan, F.; Sicks, J.D.; Rakowski, W.; Diefenbach, M.; Rigotti, N.A. Examining Whether Lung Screening Changes Risk Perceptions: National Lung Screening Trial Participants at 1-year Follow-up. Cancer 2013, 119, 1306–1313. [Google Scholar] [CrossRef]
- Cheng, Y.I.; Davies, M.P.A.; Liu, D.; Li, W.; Field, J.K. Implementation Planning for Lung Cancer Screening in China. Precis. Clin. Med. 2019, 2, 13–44. [Google Scholar] [CrossRef] [PubMed]
- Lowenstein, L.M.; Shih, Y.-C.T.; Minnix, J.; Lopez-Olivo, M.A.; Maki, K.G.; Kypriotakis, G.; Leal, V.B.; Shete, S.S.; Fox, J.; Nishi, S.P.; et al. A Protocol for a Cluster Randomized Trial of Care Delivery Models to Improve the Quality of Smoking Cessation and Shared Decision Making for Lung Cancer Screening. Contemp. Clin. Trials 2023, 128, 107141. [Google Scholar] [CrossRef] [PubMed]
- Antigua-Made, A.; Nguyen, S.; Rashidi, A.; Chen, W.-P.; Ziogas, A.; Sadigh, G. Lung Cancer Screening Completion Among Patients Using Decision Aids: A Systematic Review and Meta-Analysis. Cancer Causes Control 2025, 36, 871–885. [Google Scholar] [CrossRef]
- Haas, J.S.; Atlas, S.J.; Wright, A.; Orav, E.J.; Aman, D.G.; Breslau, E.S.; Burdick, T.E.; Carpenter, E.; Chang, F.; Dang, T.; et al. Multilevel Follow-up of Cancer Screening (mFOCUS): Protocol for a Multilevel Intervention to Improve the Follow-up of Abnormal Cancer Screening Test Results. Contemp. Clin. Trials 2021, 109, 106533. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.C.; Lee, J.; Zhu, H.; Chen, P.M.; Wahid, U.; Hamann, H.A.; Bhalla, S.; Cardenas, R.C.; Natchimuthu, V.S.; Johnson, D.H.; et al. Assessing Barriers and Facilitators to Lung Cancer Screening: Initial Findings from a Patient Navigation Intervention. Popul. Health Manag. 2023, 26, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Adler, D.H.; Wood, N.; Fiscella, K.; Rivera, M.P.; Hernandez-Romero, B.; Chamberlin, S.; Abar, B. Increasing Uptake of Lung Cancer Screening Among Emergency Department Patients: A Pilot Study. J. Emerg. Med. 2024, 67, e164–e176. [Google Scholar] [CrossRef]
- Tanner, N.T.; Egede, L.E.; Shamblin, C.; Gebregziabher, M.; Silvestri, G.A. Attitudes and Beliefs Toward Lung Cancer Screening Among US Veterans. Chest 2013, 144, 1783–1787. [Google Scholar] [CrossRef]
- Zarei Jalalabadi, N.; Rahimi, B.; Foroumandi, M.; Lackey, A.; Peiman, S. Willingness to Participate in a Lung Cancer Screening Program: Patients’ Attitudes Towards United States Preventive Services Taskforce (USPSTF) Recommendations. Eur. J. Intern. Med. 2022, 98, 128–129. [Google Scholar] [CrossRef]
- Núñez, E.R.; Ito Fukunaga, M.; Stevens, G.A.; Yang, J.K.; Reid, S.E.; Spiegel, J.L.; Ingemi, M.R.; Wiener, R.S. Review of Interventions That Improve Uptake of Lung Cancer Screening. Chest 2024, 166, 632–648. [Google Scholar] [CrossRef]
- Monestime, S.; Rigney, M.; Phipps, N.; Carlson, C.; Alongi, T.; Redding, M.; King, J.C. Health Inequities Across the Lung Cancer Care Continuum in Ten Marginalized Populations: A Narrative Review. J. Thorac. Dis. 2023, 15, 6345–6361. [Google Scholar] [CrossRef]
- Lopez-Olivo, M.A.; Maki, K.G.; Choi, N.J.; Hoffman, R.M.; Shih, Y.-C.T.; Lowenstein, L.M.; Hicklen, R.S.; Volk, R.J. Patient Adherence to Screening for Lung Cancer in the US: A Systematic Review and Meta-Analysis. JAMA Netw. Open 2020, 3, e2025102. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Fu, M.; Ding, R.; Inoue, K.; Jeon, C.Y.; Hsu, W.; Aberle, D.R.; Prosper, A.E. Patient Adherence to Lung CT Screening Reporting & Data System–Recommended Screening Intervals in the United States: A Systematic Review and Meta-Analysis. J. Thorac. Oncol. 2022, 17, 38–55. [Google Scholar] [CrossRef]
- Neslund-Dudas, C.; Tang, A.; Alleman, E.; Zarins, K.R.; Li, P.; Simoff, M.J.; Lafata, J.E.; Rendle, K.A.; Hartman, A.N.B.; Honda, S.A.; et al. Uptake of Lung Cancer Screening CT After a Provider Order for Screening in the PROSPR-Lung Consortium. J. Gen. Intern. Med. 2024, 39, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Hochheimer, C.J.; Sabo, R.T.; Tong, S.T.; Westfall, M.; Wolver, S.E.; Carney, S.; Day, T.; Krist, A.H. Practice, Clinician, and Patient Factors Associated with the Adoption of Lung Cancer Screening. J. Med. Screen. 2021, 28, 158–162. [Google Scholar] [CrossRef]
- Hall, D.L.; Lennes, I.T.; Carr, A.; Eusebio, J.R.; Yeh, G.Y.; Park, E.R. Lung Cancer Screening Uncertainty Among Patients Undergoing LDCT. Am. J. Health Behav. 2018, 42, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Kanodra, N.M.; Pope, C.; Halbert, C.H.; Silvestri, G.A.; Rice, L.J.; Tanner, N.T. Primary Care Provider and Patient Perspectives on Lung Cancer Screening. A Qualitative Study. Ann. Am. Thorac. Soc. 2016, 13, 1977–1982. [Google Scholar] [CrossRef] [PubMed]
- Rajupet, S.; Doshi, D.; Wisnivesky, J.P.; Lin, J.J. Attitudes About Lung Cancer Screening: Primary Care Providers Versus Specialists. Clin. Lung Cancer 2017, 18, e417–e423. [Google Scholar] [CrossRef]
- Couraud, S.; Girard, N.; Erpeldinger, S.; Gueyffier, F.; Devouassoux, G.; Llorca, G.; Souquet, P.J. Physicians’ Knowledge and Practice of Lung Cancer Screening: A Cross-Sectional Survey Comparing General Practitioners, Thoracic Oncologists, and Pulmonologists in France. Clin. Lung Cancer 2013, 14, 574–580. [Google Scholar] [CrossRef]
- Khanna, A.; Fix, G.M.; McCullough, M.B.; Herbst, A.N.; Li, M.; Miano, D.; Barker, A.M.; Wiener, R.S. Implementing Shared Decision-Making for Lung Cancer Screening across a Veterans Health Administration Hospital Network: A Hybrid Effectiveness–Implementation Study Protocol. Ann. Am. Thorac. Soc. 2022, 19, 476–483. [Google Scholar] [CrossRef]
- Guldbrandt, L.M.; Rasmussen, T.R.; Rasmussen, F.; Vedsted, P. Implementing Direct Access to Low-Dose Computed Tomography in General Practice—Method, Adaption and Outcome. PLoS ONE 2014, 9, e112162. [Google Scholar] [CrossRef]
- Wiener, R.S.; Koppelman, E.; Bolton, R.; Lasser, K.E.; Borrelli, B.; Au, D.H.; Slatore, C.G.; Clark, J.A.; Kathuria, H. Patient and Clinician Perspectives on Shared Decision-Making in Early Adopting Lung Cancer Screening Programs: A Qualitative Study. J. Gen. Intern. Med. 2018, 33, 1035–1042. [Google Scholar] [CrossRef]
- Volk, R.J.; Myers, R.E.; Arenberg, D.; Caverly, T.J.; Hoffman, R.M.; Katki, H.A.; Mazzone, P.J.; Moulton, B.W.; Reuland, D.S.; Tanner, N.T.; et al. The American Cancer Society National Lung Cancer Roundtable Strategic Plan: Current Challenges and Future Directions for Shared Decision Making for Lung Cancer Screening. Cancer 2024, 130, 3996–4011. [Google Scholar] [CrossRef]
- Parker, K.; Colhoun, S.; Bartholomew, K.; Sandiford, P.; Lewis, C.; Milne, D.; McKeage, M.; McKree Jansen, R.; Fong, K.M.; Marshall, H.; et al. Invitation Methods for Indigenous New Zealand Māori in Lung Cancer Screening: Protocol for a Pragmatic Cluster Randomized Controlled Trial. PLoS ONE 2023, 18, e0281420. [Google Scholar] [CrossRef]
- Roth, J.A.; Carter-Harris, L.; Brandzel, S.; Buist, D.S.M.; Wernli, K.J. A Qualitative Study Exploring Patient Motivations for Screening for Lung Cancer. PLoS ONE 2018, 13, e0196758. [Google Scholar] [CrossRef] [PubMed]
- Quaife, S.L.; Janes, S.M.; Brain, K.E. The Person behind the Nodule: A Narrative Review of the Psychological Impact of Lung Cancer Screening. Transl. Lung Cancer Res. 2021, 10, 2427–2440. [Google Scholar] [CrossRef]
- Sayani, A.; Ali, M.A.; Dey, P.; Corrado, A.M.; Ziegler, C.; Nicholson, E.; Lofters, A. Interventions Designed to Increase the Uptake of Lung Cancer Screening: An Equity-Oriented Scoping Review. JTO Clin. Res. Rep. 2023, 4, 100469. [Google Scholar] [CrossRef]
- Percac-Lima, S.; Ashburner, J.M.; Rigotti, N.A.; Park, E.R.; Chang, Y.; Kuchukhidze, S.; Atlas, S.J. Patient Navigation for Lung Cancer Screening Among Current Smokers in Community Health Centers a Randomized Controlled Trial. Cancer Med. 2018, 7, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Baggett, T.P.; Sporn, N.; Barbosa Teixeira, J.; Rodriguez, E.C.; Anandakugan, N.; Little, B.R.; Chang, Y.; Park, E.R.; Rigotti, N.A.; Fine, D.R. Homelessness, Patient Navigation, and Lung Cancer Screening in a Health Center Setting: A Subgroup Analysis of a Randomized Clinical Trial. JAMA Netw. Open 2025, 8, e2519780. [Google Scholar] [CrossRef]
- Schapira, M.M.; Hubbard, R.A.; Whittle, J.; Vachani, A.; Kaminstein, D.; Chhatre, S.; Rodriguez, K.L.; Bastian, L.A.; Kravetz, J.D.; Asan, O.; et al. Lung Cancer Screening Decision Aid Designed for a Primary Care Setting: A Randomized Clinical Trial. JAMA Netw. Open 2023, 6, e2330452. [Google Scholar] [CrossRef] [PubMed]
- Carter-Harris, L.; Comer, R.S.; Slaven, J.E., II; Monahan, P.O.; Vode, E.; Hanna, N.H.; Ceppa, D.P.; Rawl, S.M. Computer-Tailored Decision Support Tool for Lung Cancer Screening: Community-Based Pilot Randomized Controlled Trial. J. Med. Internet Res. 2020, 22, e17050. [Google Scholar] [CrossRef]
- Sferra, S.R.; Cheng, J.S.; Boynton, Z.; DiSesa, V.; Kaiser, L.R.; Ma, G.X.; Erkmen, C.P. Aiding Shared Decision Making in Lung Cancer Screening: Two Decision Tools. J. Public Health 2021, 43, 673–680. [Google Scholar] [CrossRef]
- Volk, R.J.; Lowenstein, L.M.; Leal, V.B.; Escoto, K.H.; Cantor, S.B.; Munden, R.F.; Rabius, V.A.; Bailey, L.; Cinciripini, P.M.; Lin, H.; et al. Effect of a Patient Decision Aid on Lung Cancer Screening Decision-Making by Persons Who Smoke: A Randomized Clinical Trial. JAMA Netw. Open 2020, 3, e1920362. [Google Scholar] [CrossRef]
- Han, P.K.J.; Lary, C.; Black, A.; Gutheil, C.; Mandeville, H.; Yahwak, J.; Fukunaga, M. Effects of Personalized Risk Information on Patients Referred for Lung Cancer Screening with Low-Dose CT. Med. Decis. Mak. 2019, 39, 950–961. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.A.; Bonnet, K.; Schlundt, D.G.; Byerly, S.; Lindsell, C.J.; Henschke, C.I.; Yankelevitz, D.F.; York, S.J.; Hendler, F.; Dittus, R.S.; et al. Rural Barriers and Facilitators of Lung Cancer Screening Program Implementation in the Veterans Health Administration: A Qualitative Study. Front. Health Serv. 2023, 3, 1209720. [Google Scholar] [CrossRef]
- Wait, S.; Alvarez-Rosete, A.; Osama, T.; Bancroft, D.; Cornelissen, R.; Marušić, A.; Garrido, P.; Adamek, M.; Van Meerbeeck, J.; Snoeckx, A.; et al. Implementing Lung Cancer Screening in Europe: Taking a Systems Approach. JTO Clin. Res. Rep. 2022, 3, 100329. [Google Scholar] [CrossRef]
- Quaife, S.L.; Ruparel, M.; Dickson, J.L.; Beeken, R.J.; McEwen, A.; Baldwin, D.R.; Bhowmik, A.; Navani, N.; Sennett, K.; Duffy, S.W.; et al. Lung Screen Uptake Trial (LSUT): Randomized Controlled Clinical Trial Testing Targeted Invitation Materials. Am. J. Respir. Crit. Care Med. 2020, 201, 965–975. [Google Scholar] [CrossRef]
- Jessup, D.L.; Glover, M., IV; Daye, D.; Banzi, L.; Jones, P.; Choy, G.; Shepard, J.-A.O.; Flores, E.J. Implementation of Digital Awareness Strategies to Engage Patients and Providers in a Lung Cancer Screening Program: Retrospective Study. J. Med. Internet Res. 2018, 20, e52. [Google Scholar] [CrossRef]
- Dickson, J.L.; Hall, H.; Horst, C.; Tisi, S.; Verghese, P.; Mullin, A.-M.; Teague, J.; Farrelly, L.; Bowyer, V.; Gyertson, K.; et al. Uptake of Invitations to a Lung Health Check Offering Low-Dose CT Lung Cancer Screening among an Ethnically and Socioeconomically Diverse Population at Risk of Lung Cancer in the UK (SUMMIT): A Prospective, Longitudinal Cohort Study. Lancet Public Health 2023, 8, e130–e140. [Google Scholar] [CrossRef] [PubMed]
- Kalinke, L.; Thakrar, R.; Janes, S.M. The Promises and Challenges of Early Non-Small Cell Lung Cancer Detection: Patient Perceptions, Low-Dose CT Screening, Bronchoscopy and Biomarkers. Mol. Oncol. 2021, 15, 2544–2564. [Google Scholar] [CrossRef] [PubMed]
| Trial Name | Number of Participants | Screening Interval/Duration | Follow-Up Period | Lung Cancer Mortality Reduction (Primary Outcome) | Refs |
|---|---|---|---|---|---|
| NLST (National Lung Screening Trial) | 53,454 | Annual (3 rounds) | Median 6.5 years | 20% reduction (vs. chest X-ray) | [13,14,22] |
| NELSON Trial | 15,822 | Increasing intervals (0, 1, 2, and 2.5 years; 4 rounds) | 10 years | 24–26% reduction in men (vs. no screening); larger reduction observed in women (39–61%) | [14,22,23] |
| MILD (Multicentric Italian Lung Detection) | 4099 | Annual or Biennial (median 6 annual or 3 biennial screens) | 10 years | 39% reduction at 10 years (vs. no screening); no difference found between annual vs. biennial intervals | [22,23,24] |
| LUSI (German Lung Cancer Screening Intervention) | 4052 | Annual (5 rounds) | Median 8.8 years | No statistically significant reduction overall; significant reduction observed in women (HR 0.31) | [14,22,23] |
| DANTE (detection and screening of early lung cancer) | 2472 | Annual (5 rounds) | Median 8.4 years | No statistically significant reduction | [14,22] |
| DLCST (Danish Lung Cancer Screening Trial) | 4104 | Annual (5 rounds) | Median 9.5 years | No statistically significant reduction | [14,22] |
| ITALUNG (Italian Lung Cancer Screening) | 3206 | Annual (4 rounds) | Median 8.5 years | No statistically significant reduction (30% reduction observed but not statistically significant) | [14,22] |
| UKLS (UK Lung Cancer Screening Pilot) | 4055 | Single screen (1 round) | Median 10 years | Non-significant reduction in primary analysis due to pilot design/sample size (RR 0.86) | [14,22,25] |
| Barrier Domain | Specific Themes | Examples |
|---|---|---|
| Individual/psychological | Knowledge avoidance and fear of disease | Anxiety regarding a potential diagnosis; Preference for ignorance over knowing bad news; Fear of treatment outcomes. |
| False positive worry | Anxiety regarding the stress of potential misdiagnosis or inconclusive results. | |
| Fear of screening procedure | Claustrophobia or anxiety regarding the physical scan. | |
| Denial of risk | Belief that lack of symptoms equates to health. | |
| System/practical | Cost & insurance misunderstanding | Real or perceived financial burden; Confusion regarding coverage; Inability to pay copays. |
| Logistical barriers | Lack of time; Conflicts with work; Inconvenience of location. | |
| Confusion around results | Frustration with inconclusive findings or the “runaround” of diagnostic testing. | |
| Cultural and beliefs | Fatalistic beliefs and perceived low value | Belief that screening makes no difference to the outcome; “What I don’t know won’t hurt me” attitude; Skepticism about benefits. |
| Distrust | Suspicion of medical system motives (profit over care); Feeling marginalized or unheard by doctors. |
| Domain | Metric/Population | Pre-Screening (T0)/Control Group | Post-Screening (T1)/Intervention Group | Notes | Ref. |
|---|---|---|---|---|---|
| Attitudes and motivation | Readiness to quit | 32.9% (Ready in next 30 days) | N/A | 25.7% of participants reported increased readiness to quit following screening (p < 0.001). | [77] |
| Motivation to quit score (mean, scale 1–10) | 6.5 (SD 2.3) | 6.7 (SD 2.3) | Statistically significant increase in motivation (p < 0.05). | [77] | |
| Consumption | Cigarettes per day (mean) | 18.2 (SD 9.0) | 16.7 (SD 9.1) | Statistically significant reduction in daily cigarette consumption (p < 0.001). | [77] |
| Decreased smoking (study of 1060 adults) | N/A | 45% | 45% of smokers decreased smoking after the first screening; this was more typical in younger participants (<65 years). | [78] | |
| Cessation rates (trial data) | Mayo Clinic screening program (longitudinal study) | 5% to 7% (General population historical rate) | 14% (Year 1) 22% (Year 2) 24% (Year 3) | Screening program participants exceeded general population quit rates; 98% of former smokers (>1 yr) remained tobacco-free. | [13] |
| NELSON Trial (2-year follow-up) | N/A | 16.6% (Screening arm) (control arm was 19.1%) | While rates were high, no significant difference was found between screening and control arms in this specific analysis. | [13,24] | |
| UKLS Trial (2-year follow-up) | 21% (Control arm) | 24% (Screening arm) | Net trial quit rate of 22% was significantly higher than the UK general population rate of 4%. | [24] | |
| UKLS Trial (2 weeks post-randomization) | N/A | RR 2.16 (vs. control) | Significantly higher quit rate in the LDCT screening group compared with control (95% CI 1.47 to 3.18). | [14] | |
| Impact of test result | Abnormal/positive screen | N/A | 41.9% (with 3 positive screens); 24.2% (with 1 positive screen) | Positive correlation between the number of positive results and smoking abstinence; suspicious findings were associated with higher cessation. | [13] |
| Negative screen (false reassurance) | N/A | 19.8% quit rate (with no positive exams) | Evidence argues against a “permission to smoke” phenomenon; negative screens did not lead to lower abstinence compared to general population rates. | [13,24] |
| Predictor | Comparison | Association with Adherence/Nonadherence | Refs. |
|---|---|---|---|
| Smoking Status | Current vs. Former Smokers | Nonadherence: Current smokers were significantly more likely to be nonadherent compared to former smokers (RR 1.23; 95% CI 1.09–1.40). | [56,65,95] |
| Sex | Female vs. male | Mixed/Nonadherence: Meta-analysis found no significant difference (RR 0.99; 95% CI 0.85–1.15). However, individual studies have shown mixed results, with some finding males more adherent. | [56,96] |
| Race | White vs. non-white/Black | Adherence: White patients were twice as likely to adhere to screening compared to patients of other races (OR 2.0; 95% CI 1.6–2.6). Black patients demonstrated lower adherence to annual screening (aRR 0.73 in decentralized programs, without dedicated tracking staff like screening coordinators or navigators). | [38,94] |
| Education | College vs. no college/high school | Adherence: Completion of 4 or more years of college was associated with increased adherence (OR 1.5; 95% CI 1.1–2.1). Education > High School diploma associated with higher adherence (OR 1.87). | [65,94] |
| Screening result | Lung-RADS 3/4 (suspicious) vs. Lung-RADS 1/2 (negative) | Adherence: Patients with suspicious or abnormal findings were significantly more adherent to follow-up than those with negative screens (OR 3.8 for Lung-RADS 3; OR 14.0 for Lung-RADS 4). | [95] |
| Insurance type | Medicare/private vs. Medicaid/uninsured | Adherence: Patients with Medicare were more likely to adhere compared to Medicaid (OR 2.23) or dual-eligible patients. Uninsured patients or self-pay cohorts had higher nonadherence rates. | [56,65] |
| Program structure | Centralized vs. decentralized | Adherence: Centralized programs (with navigation/tracking) showed higher adherence (76.1% vs. 34.8%) and mitigated racial disparities compared to decentralized models. | [28,38] |
| Comorbidities | COPD diagnosis vs. No COPD | Adherence: Patients with a diagnosis of COPD were more likely to complete screening uptake/adherence (OR 1.13; 95% CI 1.06–1.20). | [96] |
| Attitude/Practice Metric | Primary Care Providers/General Practitioners (PCP/GP) | Specialists (Pulm/Onc) | p-Value | Refs |
|---|---|---|---|---|
| Able to identify appropriate patients | 63.8% | 93.5% | <0.01 | [100] |
| Feel comfortable counseling patients | 51.4% | 82.8% | 0.01 | [100] |
| Have sufficient time to counsel | 14.3% | 50.0% | <0.01 | [100] |
| Confused about applying guidelines | 63.8% | 35.5% | 0.01 | [100] |
| Believe yearly screening is feasible | 27.5% | 86.7% | <0.01 | [100] |
| Believe screening is NOT cost-effective | 8.6% | 29.0% | 0.01 | [100] |
| Aware that LDCT is an effective test | 18.0% | 81.0% (Onc) | <0.0001 | [101] |
| Currently propose screening in practice | 20.0% | 53.0% (Pulm) | <0.001 | [101] |
| Use inappropriate screening test (e.g., CXR) | 93.0% | 44.0% (Pulm) | <0.0001 | [101] |
| Recommend correct annual screening interval | 7.0% | 76.0% | <0.0001 | [101] |
| Propose screening for “all smokers” (Incorrect criteria) | 55.0% | 25.0% | 0.04 | [101] |
| Believe tobacco control MUST be associated with screening | 52.0% | 80–86% | <0.0001 | [101] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Anghel, R.; Folea, A.-R.; Moga, V.-L.; Pavel, C.; Troncotă, D.; Dumitru, C.-O.; Șerban, A.-I.; Bîlteanu, L. Patient and Healthcare Provider Barriers in the LDCT Lung Cancer Screening Continuum. Diagnostics 2026, 16, 1092. https://doi.org/10.3390/diagnostics16071092
Anghel R, Folea A-R, Moga V-L, Pavel C, Troncotă D, Dumitru C-O, Șerban A-I, Bîlteanu L. Patient and Healthcare Provider Barriers in the LDCT Lung Cancer Screening Continuum. Diagnostics. 2026; 16(7):1092. https://doi.org/10.3390/diagnostics16071092
Chicago/Turabian StyleAnghel, Rodica, Antonia-Ruxandra Folea, Vlad-Luca Moga, Cristian Pavel, Diana Troncotă, Corneliu-Octavian Dumitru, Andreea-Iren Șerban, and Liviu Bîlteanu. 2026. "Patient and Healthcare Provider Barriers in the LDCT Lung Cancer Screening Continuum" Diagnostics 16, no. 7: 1092. https://doi.org/10.3390/diagnostics16071092
APA StyleAnghel, R., Folea, A.-R., Moga, V.-L., Pavel, C., Troncotă, D., Dumitru, C.-O., Șerban, A.-I., & Bîlteanu, L. (2026). Patient and Healthcare Provider Barriers in the LDCT Lung Cancer Screening Continuum. Diagnostics, 16(7), 1092. https://doi.org/10.3390/diagnostics16071092






