Unravelling Sarcopenia in Chronic Kidney Disease: From Pathogenesis to Diagnosis and Therapeutics
Abstract
1. Introduction
2. Search Methodology
3. Pathogenetic Factors and Molecular Implications
3.1. Chronic Low-Grade Inflammation
3.2. Oxidative Stress
3.3. Hormonal and Metabolic Changes
3.4. Epigenetic Changes
3.5. Nutritional Parameters
3.6. Physical Inactivity
3.7. Gut Dysbiosis
4. Diagnosis
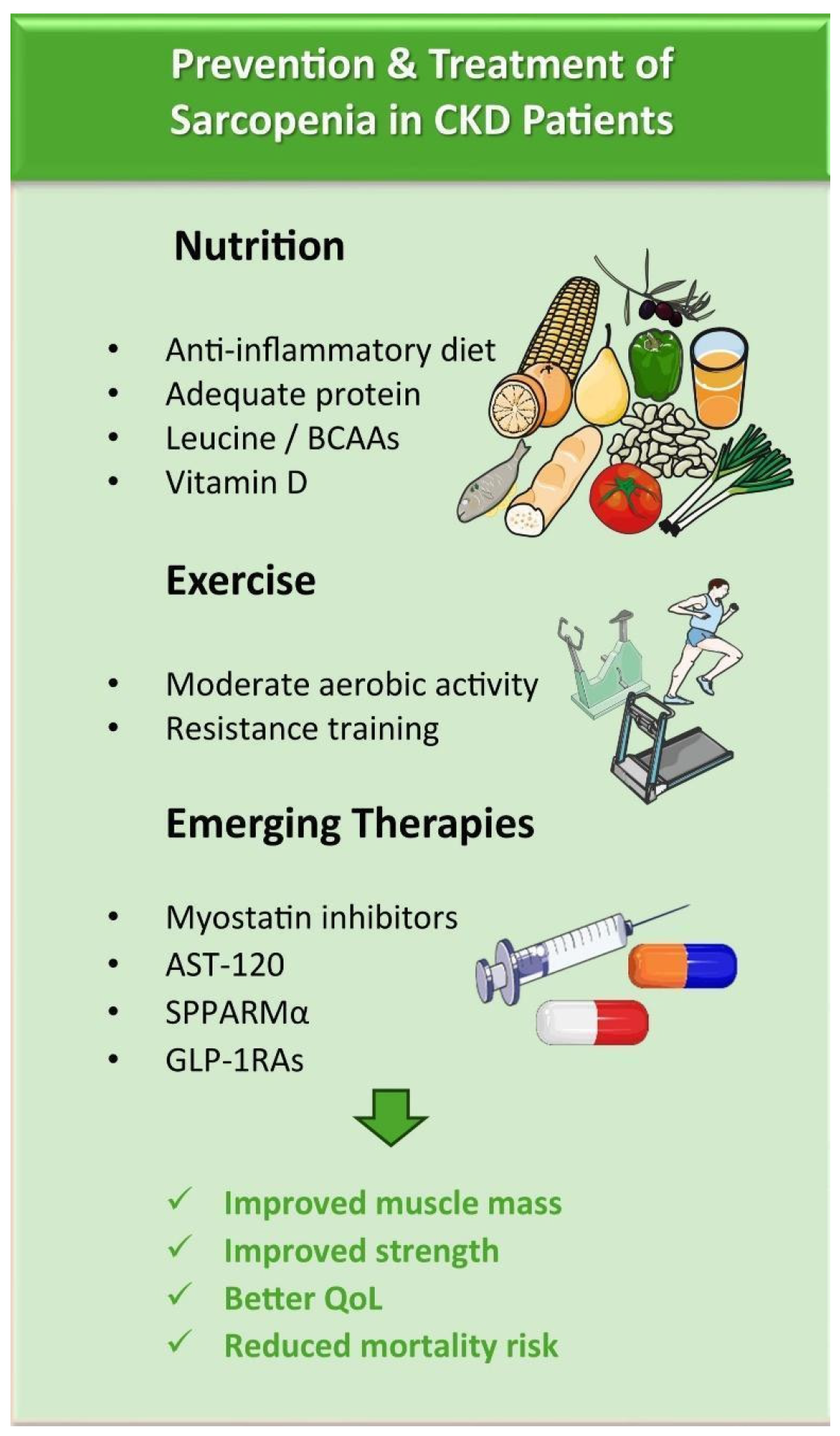
5. The Importance of Nutritional Interventions and Physical Exercise
6. Potential Therapeutics
7. Future Perspectives
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024, 105, S1–S197. [Google Scholar] [CrossRef]
- GBD 2023 Chronic Kidney Disease Collaborators. Global, regional, and national burden of chronic kidney disease in adults, 1990-2023, and its attributable risk factors: A systematic analysis for the Global Burden of Disease Study 2023. Lancet 2025, 406, 2461–2482. [Google Scholar] [CrossRef]
- Ferro, C.J.; Wanner, C.; Luyckx, V.; Stevens, K.; Cerqueira, S.; Darwish, R.; Fernandez, B.F.; Fiel, D.; Filev, R.; Grieger, M.; et al. ABCDE to identify and prevent chronic kidney disease: A call to action. Nephrol. Dial. Transplant. 2025, 40, 1786–1798. [Google Scholar] [CrossRef]
- Duarte, M.P.; Almeida, L.S.; Neri, S.G.R.; Oliveira, J.S.; Wilkinson, T.J.; Ribeiro, H.S.; Lima, R.M. Prevalence of sarcopenia in patients with chronic kidney disease: A global systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2024, 15, 501–512. [Google Scholar] [CrossRef]
- Tsai, C.C.; Wang, P.C.; Hsiung, T.; Fan, Y.H.; Wu, J.T.; Kan, W.C.; Shiao, C.C. Sarcopenia in Chronic Kidney Disease: A Narrative Review from Pathophysiology to Therapeutic Approaches. Biomedicines 2025, 13, 352. [Google Scholar] [CrossRef]
- Caldiroli, L.; Molinari, P.; D’Alessandro, C.; Cupisti, A.; Alfieri, C.; Castellano, G.; Vettoretti, S. Osteosarcopenia in Chronic Kidney Disease: An Overlooked Syndrome? J. Cachexia Sarcopenia Muscle 2025, 16, e13787. [Google Scholar] [CrossRef]
- Hsieh, M.F.; Wang, S.I.; Hsieh, H.F.; Huang, M.Z.; Wang, H.H. Low muscle strength and mortality: Key risk factors in the National Health and Nutrition Examination Survey retrospective cohort study. Medicine 2025, 104, e45284. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Tu, H.; Zhang, Y.; Yang, S.; Yu, P.; Liu, J. Risks of All-Cause Mortality in Adults With Chronic Kidney Disease With Sarcopenia or Obesity: A Population-Based Study. J. Cachexia Sarcopenia Muscle 2025, 16, e13828. [Google Scholar] [CrossRef]
- Geladari, E.; Alexopoulos, T.; Kontogianni, M.D.; Vasilieva, L.; Mani, I.; Alexopoulou, A. Mechanisms of sarcopenia in liver cirrhosis and the role of myokines. Ann. Gastroenterol. 2023, 36, 392–404. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Liang, Y.; Sun, P.; Fang, X.; Sun, Q. Molecular and Cellular Mechanisms Linking Chronic Kidney Disease and Sarcopenia in Aging: An Integrated Perspective. Clin. Interv. Aging 2025, 20, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Price, S.R.; Wang, X.H. Protein-energy wasting in chronic kidney disease: Mechanisms responsible for loss of muscle mass and function. Kidney Res. Clin. Pract. 2025, 44, 726–740. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.Y.; Chen, J.H.; Chiang, C.K.; Hung, K.Y. Pathophysiology and potential treatment of uremic sarcopenia. Kidney Res. Clin. Pract. 2025, 45, 160–173. [Google Scholar] [CrossRef]
- Chao, C.T.; Kovesdy, C.P.; Merchant, R.A. Sarcopenia, sarcopenic obesity, and frailty in individuals with chronic kidney disease: A comprehensive review. Kidney Res. Clin. Pract. 2025, 45, 174. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G.; Cao, J.; Wang, X.H.; He, W.; Wang, B. The gut microbiome, chronic kidney disease, and sarcopenia. Cell Commun. Signal. 2024, 22, 558. [Google Scholar] [CrossRef]
- Li, S.; Cao, H.; Gao, Z.; Liang, Y.; Ma, Y.; Liu, S.; Wang, L.; Wei, W. Association between chronic kidney disease and sarcopenia and emerging treatment strategies. Front. Nutr. 2025, 12, 1699218. [Google Scholar] [CrossRef]
- Kounatidis, D.; Vallianou, N.; Evangelopoulos, A.; Vlahodimitris, I.; Grivakou, E.; Kotsi, E.; Dimitriou, K.; Skourtis, A.; Mourouzis, I. SGLT-2 Inhibitors and the Inflammasome: What’s Next in the 21st Century? Nutrients 2023, 15, 2294. [Google Scholar] [CrossRef]
- Shang, T.; Xu, H.; Yao, X.; Zhao, Z.; Niu, X.; Shen, Y.; Chen, B.; Sun, H. Dual role of macrophages in skeletal muscle atrophy: Mechanisms and therapeutic strategies. Biochem. Pharmacol. 2026, 244, 117589. [Google Scholar] [CrossRef]
- Sarapultsev, A.; Gusev, E.; Komelkova, M.; Utepova, I.; Luo, S.; Hu, D. JAK-STAT signaling in inflammation and stress-related diseases: Implications for therapeutic interventions. Mol. Biomed. 2023, 4, 40. [Google Scholar] [CrossRef]
- Xu, H.; Brown, J.L.; Bhaskaran, S.; Van Remmen, H. Reactive oxygen species in the pathogenesis of sarcopenia. Free Radic. Biol. Med. 2025, 227, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Lian, D.; Chen, M.-M.; Wu, H.; Deng, S.; Hu, X. The Role of Oxidative Stress in Skeletal Muscle Myogenesis and Muscle Disease. Antioxidants 2022, 11, 755. [Google Scholar] [CrossRef]
- Duranti, G. Oxidative Stress and Skeletal Muscle Function. Int. J. Mol. Sci. 2023, 24, 10227. [Google Scholar] [CrossRef] [PubMed]
- Bellanti, F.; Coda, A.R.D.; Trecca, M.I.; Lo Buglio, A.; Serviddio, G.; Vendemiale, G. Redox Imbalance in Inflammation: The Interplay of Oxidative and Reductive Stress. Antioxidants 2025, 14, 656. [Google Scholar] [CrossRef]
- Souza, A.L.G.d.; Alves, A.L.R.; Martinez, C.G.; Sousa, J.C.d.; Kurtenbach, E. Biomarkers of Skeletal Muscle Atrophy Based on Atrogenes Evaluation: A Systematic Review and Meta-Analysis Study. Int. J. Mol. Sci. 2025, 26, 3516. [Google Scholar] [CrossRef]
- Gungor, O.; Ulu, S.; Hasbal, N.B.; Anker, S.D.; Kalantar-Zadeh, K. Effects of hormonal changes on sarcopenia in chronic kidney disease: Where are we now and what can we do? J. Cachexia Sarcopenia Muscle 2021, 12, 1380–1392. [Google Scholar] [CrossRef]
- Cesanelli, L.; Minderis, P.; Fokin, A.; Ratkevicius, A.; Satkunskiene, D.; Degens, H. Myostatin in Obesity: A Molecular Link Between Metabolic Dysfunction and Musculotendinous Remodeling. Int. J. Mol. Sci. 2026, 27, 967. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, M.; Negrón, R.; Neira-Maldonado, C.; Ponce-Fuentes, F.; Cuyul-Vásquez, I. Fasting Glycemia, Glycosylated Hemoglobin and Malnutrition Inflammation Are Associated With Sarcopenia in Older People With Chronic Kidney Disease Undergoing Hemodialysis Treatment: A Cross-Sectional Study. Cureus 2024, 16, e74432. [Google Scholar] [CrossRef]
- Gutiérrez-Calabrés, E.; Campillo, S.; Alcalde-Estévez, E.; Cuevas-Delgado, P.; Barbas, C.; García-Villoria, S.; Silvestre-Vargas, A.; Griera, M.; de Frutos, S.; Ruiz-Torres, M.P.; et al. Role of Calpains in Uremia-Related Functional and Structural Muscle Changes: Protective Effect of Calpastatin Overexpression. Cells 2025, 14, 1846. [Google Scholar] [CrossRef]
- Lee, S.M.; Han, M.Y.; Kim, S.H.; Cha, R.H.; Kang, S.H.; Kim, J.C.; An, W.S. Indoxyl Sulfate Might Play a Role in Sarcopenia, While Myostatin Is an Indicator of Muscle Mass in Patients with Chronic Kidney Disease: Analysis from the RECOVERY Study. Toxins 2022, 14, 660. [Google Scholar] [CrossRef]
- Hou, Y.C.; Liu, Y.M.; Liao, M.T.; Zheng, C.M.; Lu, C.L.; Liu, W.C.; Hung, K.C.; Lin, S.M.; Lu, K.C. Indoxyl sulfate mediates low handgrip strength and is predictive of high hospitalization rates in patients with end-stage renal disease. Front. Med. 2023, 10, 1023383. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Yoo, K.D.; Chao, C.T. Risk factors for incident or persistent SARC-F-defined sarcopenia in patients with end-stage kidney disease: A cohort study. J. Formos. Med. Assoc. 2025, in press. [Google Scholar] [CrossRef]
- Brenta, G. Kidney diseases and the thyroid: Interactions and consequences. Nat. Rev. Endocrinol. 2025, 22, 228–241. [Google Scholar] [CrossRef]
- Hernandez-Martinez, J.; Vásquez-Carrasco, E.; Cid-Calfucura, I.; Sandoval, C.; Herrera-Valenzuela, T.; Núñez-Espinosa, C.; Branco, B.H.M.; Valdés-Badilla, P. Effects of Concurrent Training on Biomarkers, Morphological Variables, and Physical Performance in People with Sarcopenic Obesity: A Meta-Analysis with Meta-Regression. Medicina 2025, 61, 1697. [Google Scholar] [CrossRef]
- Tuokkola, J.; Anderson, C.E.; Collins, S.; Pugh, P.; Vega, M.R.W.; Harmer, M.; Harshman, L.A.; Nelms, C.L.; Toole, B.; Desloovere, A.; et al. Assessment and management of magnesium and trace element status in children with CKD stages 2-5, on dialysis and post-transplantation: Clinical practice points from the Pediatric Renal Nutrition Taskforce. Pediatr. Nephrol. 2025, 40, 3301–3323. [Google Scholar] [CrossRef]
- Wang, J.; Enechojo, O.S.; Shi, Y.; Cao, D.; Guo, T.; Zhong, J.; Lin, Y.; Bian, Y.; Wang, X.; Liu, A. Epigenetics of sarcopenia: Insights into mechanisms and interventions for healthy muscle aging. Biomed. Pharmacother. 2026, 195, 118960. [Google Scholar] [CrossRef]
- Uzun Ayar, C.; Güiza, F.; Derese, I.; Van den Berghe, G.; Vanhorebeek, I. Association Between Abnormal DNA Methylation and Altered Transcriptome in Muscle Five Years After Critical Illness. J. Cachexia Sarcopenia Muscle 2026, 17, 70170. [Google Scholar] [CrossRef]
- Wei, S.; Shu, Z.; Li, X.; Zhong, S.; Xiao, L.; Shen, R.; Lu, X.; He, L.; Zhang, Y.; Quan, Y.; et al. Air pollution exacerbates cardiovascular-kidney-metabolic syndrome and sarcopenia comorbidity via shared genetic-epigenetic mechanisms: A multi-omics and Mendelian Randomization study. Metabolism 2026, 175, 156452. [Google Scholar] [CrossRef] [PubMed]
- Ammous, F.; Peterson, M.D.; Mitchell, C.; Faul, J.D. Physical Activity Is Associated With Decreased Epigenetic Aging: Findings From the Health and Retirement Study. J. Cachexia Sarcopenia Muscle 2025, 16, e13873. [Google Scholar] [CrossRef]
- Castelli, S.; Aiello, G.; Aiello, V.; Massimino, E.; Pieri, M.; Amoah, I.; Lombardo, M.; Tripodi, G.; Baldelli, S. Nutritional Strategies and Aging: Current Evidence and Future Directions. Molecules 2026, 31, 756. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, S.; Morioka, T.; Morioka, F.; Mori, K.; Emoto, M. Zinc Deficiency in Chronic Kidney Disease and Hemodialysis: Insights from Basic Research to Clinical Implications. Nutrients 2025, 17, 2191. [Google Scholar] [CrossRef]
- Sunsandee, N.; Thimachai, P.; Satirapoj, B.; Supasyndh, O. Anti-sarcopenic effect of leucine-enriched branched-chain amino acid supplementation among elderly chronic kidney disease patients: A double-blinded randomized controlled trial. Int. Urol. Nephrol. 2025, 57, 3811–3819. [Google Scholar] [CrossRef] [PubMed]
- Hryciuk, M.; Heleniak, Z.; Małgorzewicz, S.; Kowalski, K.; Antosiewicz, J.; Koelmer, A.; Żmijewski, M.; Dębska-Ślizień, A. Assessment of Vitamin D Metabolism Disorders in Hemodialysis Patients. Nutrients 2025, 17, 774. [Google Scholar] [CrossRef]
- Harmer, M.J.; Wootton, S.A.; Gilbert, R.D.; Anderson, C.E. Nutritional Characterisation of Childhood Chronic Kidney Disease: Trace Element Malnutrition in Paediatric Renal Disease (TeMPeReD) Study. Nutrients 2025, 17, 535. [Google Scholar] [CrossRef]
- Carballo-Casla, A.; Avesani, C.M.; Beridze, G.; Ortolá, R.; García-Esquinas, E.; Lopez-Garcia, E.; Dai, L.; Dunk, M.M.; Stenvinkel, P.; Lindholm, B.; et al. Protein Intake and Mortality in Older Adults With Chronic Kidney Disease. JAMA Netw. Open 2024, 7, e2426577. [Google Scholar] [CrossRef]
- Todoroki, H.; Takayanagi, T.; Morikawa, R.; Asada, Y.; Hidaka, S.; Yoshino, Y.; Hiratsuka, I.; Shibata, M.; Wada, A.; Asai, S.; et al. Effects of High-Protein Nutritional Guidance on Sarcopenia-Related Parameters in Individuals Aged ≥ 75 Years with Type 2 Diabetes: An Exploratory Single-Arm Pre-Post Intervention Study. Nutrients 2025, 17, 3459. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zeng, Y.; Lv, W.; Fu, P.; Yuan, H. Prevalence and severity of sarcopenia in patients on maintenance hemodialysis: A cross-sectional study. BMC Nephrol. 2024, 25, 385. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Yin, X.; Huang, L.; Zhang, H. The “adult inactivity triad” in patients with chronic kidney disease: A review. Front. Med. 2023, 10, 1160450. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, A.; Cordeiro, A.C.; Prado, C.M.; Lindholm, B.; Stenvinkel, P.; Avesani, C.M. Myosteatosis is associated with adiposity, metabolic derangements and mortality in patients with chronic kidney disease. Eur. J. Clin. Nutr. 2025, 79, 475–483. [Google Scholar] [CrossRef]
- Wang, X.H.; Mitch, W.E.; Price, S.R. Pathophysiological mechanisms leading to muscle loss in chronic kidney disease. Nat. Rev. Nephrol. 2022, 18, 138–152. [Google Scholar] [CrossRef]
- Vallianou, N.G.; Kounatidis, D.; Psallida, S.; Vythoulkas-Biotis, N.; Adamou, A.; Zachariadou, T.; Kargioti, S.; Karampela, I.; Dalamaga, M. NAFLD/MASLD and the Gut-Liver Axis: From Pathogenesis to Treatment Options. Metabolites 2024, 14, 366. [Google Scholar] [CrossRef]
- Kounatidis, D.C.; Vallianou, N.G. Decoding the Spectrum of Anorexia Nervosa: Clinical Impact, Molecular Insights, and Therapeutic Perspectives. Biomolecules 2025, 15, 1559. [Google Scholar] [CrossRef]
- Lustgarten, M.S. The kidney-gut-muscle axis in end-stage renal disease is similarly represented in older adults. Nutrients 2020, 12, 106. [Google Scholar] [CrossRef]
- Sokolova, A.V.; Dragunov, D.O.; Arutyunov, G.P. Plasma Short-Chain Fatty Acids and Cytokine Profiles in Chronic Kidney Disease: A Potential Pathophysiological Link. Int. J. Mol. Sci. 2026, 27, 550. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Han, S.; Park, H.; Hong, Y.A.; Hwang, Y.; Chang, Y.K. Abdominal computed tomography-assessed muscle quality and its prognostic value in patients with advanced chronic kidney disease initiating hemodialysis. PLoS ONE 2025, 20, e0334929. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.T.; Wei, T.S.; Ching, C.T. Validation of Ultrasound Measurement of Vastus Lateralis for Appendicular Skeletal Muscle Mass in Chronic Kidney Disease Patients with Hemodialysis. Diagnostics 2024, 14, 2600. [Google Scholar] [CrossRef]
- Wilkinson, T.J.; Gore, E.F.; Vadaszy, N.; Nixon, D.G.D.; Watson, E.L.; Smith, A.C. Utility of Ultrasound as a Valid and Accurate Diagnostic Tool for Sarcopenia: Sex-Specific Cutoff Values in Chronic Kidney Disease. J. Ultrasound Med. 2021, 40, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Malmstrom, T.K.; Morley, J.E. SARC-F: A simple questionnaire to rapidly diagnose sarcopenia. J. Am. Med. Dir. Assoc. 2013, 14, 531–532. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Gao, C.; Wang, X.; Ma, X.; Xie, J.; Yu, H.; Yang, Z.; Chen, Z.; Chen, X. Validity of the SARC-F questionnaire in assessing sarcopenia in patients with chronic kidney disease: A cross-sectional study. Front. Med. 2023, 10, 1188971. [Google Scholar] [CrossRef]
- Yao, T.; Dai, J.; Sang, Z.; Huang, F.; Wang, Y.; Han, S. Respiratory sarcopenia as a mortality predictor for chronic kidney disease: A dual-cohort longitudinal study. Ren. Fail. 2025, 47, 2587375. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, H.; Bai, Y.; Huang, L.; Zhong, Y.; Li, Y. Gait Speed and All-Cause Mortality in Whole-Spectrum Chronic Kidney Disease: A Systematic Review and Meta-Analysis Included 6217 Participants. J. Cachexia Sarcopenia Muscle 2025, 16, e13739. [Google Scholar] [CrossRef]
- Jiang, K.; Slee, A.; Davenport, A. Screening Tests for Sarcopenia in Patients with Chronic Kidney Disease. Nutr. Clin. Pract. 2021, 36, 1049–1052. [Google Scholar] [CrossRef] [PubMed]
- Potok, O.A.; Katz, R.; Bansal, N.; Langlo, K.A.; Hallan, S.I. Using the Difference Between Estimated Glomerular Filtration Rate by Cystatin C and Creatinine to Improve Mortality Risk Prediction in Elderly Patients With CKD in the HUNT Study. Kidney Med. 2025, 8, 101129. [Google Scholar] [CrossRef]
- Guerrero-Romero, F.; Simental-Mendıa, L.E.; Gonzalez-Ortiz, M.; Martınez-Abundis, E.; Ramos-Zavala, M.G.; Hernández-González, S.O.; Jacques-Camarena, O.; Rodríguez-Morán, M. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic-hyperinsulinemic clamp. J. Clin. Endocrinol. Metab. 2010, 95, 3347–3351. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, F.; Li, W. Association between triglyceride-glucose index and sarcopenia in patients with chronic kidney disease. Front. Endocrinol. 2025, 16, 1626241. [Google Scholar] [CrossRef]
- Zhao, Y.; Hu, Z.; Dong, H.; Zhu, L.; Xia, Q.; Lei, C.; Peng, X.; Zhu, K.; Li, J.; Liu, X. Development and validation of nomogram and machine learning models to predict sarcopenia in patients with chronic kidney disease. Sci. Rep. 2025, 15, 42370. [Google Scholar] [CrossRef]
- Zheng, W.H.; Hu, Y.G.; Yu, D.X.; Huang, H.B. Creatinine-to-cystatin C ratio as muscle assessment tool and predictive value for mortality and sarcopenia in patients with chronic kidney disease: A meta-analysis. Front. Nutr. 2025, 12, 1655488. [Google Scholar] [CrossRef]
- Zheng, W.H.; Zhu, Y.B.; Yao, Y.; Huang, H.B. Serum creatinine/cystatin C ratio as a muscle mass evaluating tool and prognostic indicator for hospitalized patients: A meta-analysis. Front. Med. 2022, 9, 1058464. [Google Scholar] [CrossRef]
- Chen, D.C.; Shlipak, M.G.; Scherzer, R.; Bauer, S.R.; Potok, O.A.; Rifkin, D.E.; Ix, J.H.; Muiru, A.N.; Hsu, C.Y.; Estrella, M.M. Association of Intraindividual Difference in Estimated Glomerular Filtration Rate by Creatinine vs Cystatin C and End-stage Kidney Disease and Mortality. JAMA Netw. Open 2022, 5, e2148940. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, D.; de Souza, V.A.; Souza, G.C.; Suassuna, L.F.; Bastos, M.G.; Reboredo, M.M.; da Silva Fernandes, N.M. Sarcopenic obesity in predialysis chronic kidney disease: Muscle strength is a predictor of mortality and disease progression in a six-year prospective cohort. PLoS ONE 2025, 20, e0318773, Erratum in PLoS ONE 2025, 20, e0332435. [Google Scholar] [CrossRef]
- Gohda, T.; Kamei, N.; Tanaka, M.; Furuhashi, M.; Sato, T.; Kubota, M.; Sanuki, M.; Koshida, T.; Hagiwara, S.; Suzuki, Y.; et al. Association of Difference Between eGFR From Cystatin C and Creatinine and Serum GDF-15 With Adverse Outcomes in Diabetes Mellitus. J. Cachexia Sarcopenia Muscle 2025, 16, e70011. [Google Scholar] [CrossRef]
- Afzal, O.; Afzal, M.; Khan, N.H.; Altharawi, A.; Alamri, M.A.; Alossaimi, M.A.; Altamimi, A.S.A.; Goud, P. GDF-15 as an integrative cardiometabolic biomarker. Clin. Chim. Acta 2026, 583, 120839. [Google Scholar] [CrossRef]
- Wang, D.; Day, E.A.; Townsend, L.K.; Djordjevic, D.; Jørgensen, S.B.; Steinberg, G.R. GDF15: Emerging biology and therapeutic applications for obesity and cardiometabolic disease. Nat. Rev. Endocrinol. 2021, 17, 592–607. [Google Scholar] [CrossRef]
- Hale, C.; Véniant, M.M. Growth differentiation factor 15 as a potential therapeutic for treating obesity. Mol. Metab. 2021, 46, 101117. [Google Scholar] [CrossRef]
- Yu, X.; Pu, H.; Voss, M. Overview of anti-inflammatory diets and their promising effects on non-communicable diseases. Br. J. Nutr. 2024, 132, 898–918. [Google Scholar] [CrossRef]
- Moldovan, D.; Rusu, C.C.; Potra, A.R.; Tirinescu, D.; Ticala, M.; Maslyennikov, Y.; Bărar, A.A.; Urs, A.; Kacso, I.M. Nutritional Intervention and Musculoskeletal Health in Chronic Kidney Disease. Nutrients 2025, 17, 896. [Google Scholar] [CrossRef]
- Kim, J.W.; Yang, S.J. Dietary Patterns, Kidney Function, and Sarcopenia in Chronic Kidney Disease. Nutrients 2025, 17, 404. [Google Scholar] [CrossRef]
- Mansouri, F.; Shateri, Z.; Jahromi, S.E.; Mahmudi-Zadeh, M.; Nouri, M.; Babajafari, S. Association between pro-vegetarian dietary patterns and the risk of protein-energy wasting and sarcopenia in patients with chronic kidney disease. J. Health Popul. Nutr. 2024, 43, 110. [Google Scholar] [CrossRef] [PubMed]
- Rahbar Saadat, Y.; Abbasi, A.; Hejazian, S.S.; Hekmatshoar, Y.; Ardalan, M.; Farnood, F.; Zununi Vahed, S. Combating chronic kidney disease-associated cachexia: A literature review of recent therapeutic approaches. BMC Nephrol. 2025, 26, 133. [Google Scholar] [CrossRef]
- Ikizler, T.A.; Burrowes, J.D.; Byham-Gray, L.D.; Campbell, K.L.; Carrero, J.-J.; Chan, W.; Fouque, D.; Friedman, A.N.; Ghaddar, S.; Goldstein-Fuchs, D.J.; et al. KDOQI Clinical Practice Guideline for Nutrition in CKD: 2020 Update. Am. J. Kidney Dis. 2020, 76, S1–S107. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Y.; Elsurer Afsar, R.; Sussman-Dabach, E.J.; White, J.A.; MacLaughlin, H.; Ikizler, T.A. Vitamin Supplement Use in Patients With CKD: Worth the Pill Burden? Am. J. Kidney Dis. 2024, 83, 370–385. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, Y.; Baciga, F.; Bulighin, F.; Amicone, M.; Mosconi, G.; Storari, A.; Brugnano, R.; Pozzato, M.; Motta, D.; D’Alessandro, C.; et al. Physical activity and exercise in chronic kidney disease: Consensus statements from the Physical Exercise Working Group of the Italian Society of Nephrology. J. Nephrol. 2024, 37, 1735–1765. [Google Scholar] [CrossRef]
- Iwamoto, Y.; Yamakoshi, S.; Sekimoto, A.; Hosomi, K.; Toyama, T.; Saito, Y.; Kunisawa, J.; Takahashi, N.; Mishima, E.; Sato, E. Lactoferrin attenuates renal fibrosis and uremic sarcopenia in a mouse model of adenine-induced chronic kidney disease. J. Nutr. Biochem. 2025, 146, 110039. [Google Scholar] [CrossRef]
- Akhter, A.; Md Sheikh, A.; Yoshino, J.; Kanda, T.; Nagai, A.; Matsuo, M.; Yano, S. Inhibiting Myostatin Expression by the Antisense Oligonucleotides Improves Muscle Wasting in a Chronic Kidney Disease Mouse Model. Int. J. Mol. Sci. 2025, 26, 3098. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, S.; Jang, H.; Jung, S.; Jung, M.H.; Yun, J.W.; Jeon, H.; Kim, H.J.; Chang, S.H.; Lee, E.J.; et al. Transcriptional Intermediary Factor 1γ-Induced Irisin in Skeletal Muscle Attenuates Renal Fibrosis in Diabetic Nephropathy. J. Cachexia Sarcopenia Muscle 2025, 16, e13810. [Google Scholar] [CrossRef]
- Cha, R.H. Pharmacologic therapeutics in sarcopenia with chronic kidney disease. Kidney Res. Clin. Pract. 2024, 43, 143–155. [Google Scholar] [CrossRef]
- Cha, R.H.; Kang, S.H.; Han, M.Y.; An, W.S.; Kim, S.H.; Kim, J.C. Effects of AST-120 on muscle health and quality of life in chronic kidney disease patients: Results of RECOVERY study. J. Cachexia Sarcopenia Muscle 2022, 13, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Mae, Y.; Takata, T.; Taniguchi, S.; Fujino, Y.; Kageyama, K.; Hanada, H.; Iyama, T.; Sugihara, T.; Isomoto, H. Selective peroxisome proliferator-activated receptor-α modulator improves hypertriglyceridemia and muscle quality in patients with chronic kidney disease: A retrospective observational study. Clin. Nutr. ESPEN 2025, 65, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Llinares-Arvelo, V.; Martínez-Alberto, C.E.; Gonzále-Luis, A.; Macía-Heras, M.; Siverio-Morales, O.; Navarro-González, J.F.; Donate-Correa, J. Targeting Sarcopenia in CKD: The Emerging Role of GLP-1 Receptor Agonists. Int. J. Mol. Sci. 2025, 26, 8096. [Google Scholar] [CrossRef]
- Sattar, N.; Neeland, I.J.; Dahlqvist Leinhard, O.; Fernández Landó, L.; Bray, R.; Linge, J.; Rodriguez, A. Tirzepatide and muscle composition changes in people with type 2 diabetes (SURPASS-3 MRI): A post-hoc analysis of a randomised, open-label, parallel-group, phase 3 trial. Lancet Diabetes Endocrinol. 2025, 13, 482–493. [Google Scholar] [CrossRef] [PubMed]
- Prokopidis, K.; Daly, R.M.; Suetta, C. Weighing the risk of GLP-1 treatment in older adults: Should we be concerned about sarcopenic obesity? J. Nutr. Health Aging 2025, 29, 100652. [Google Scholar] [CrossRef]
- Bacci, M.; Angelotto, F.R.; Rosa, T.D.S.; De Araújo, T.B.; De Luca Corrêa, H.; De Deus, L.A.; Neves, R.V.P.; Reis, A.L.; Santos, R.L.D.; Da Silva Barbosa, J.M.; et al. Impact of Sarcopenia, Dynapenia, and Obesity on Muscle Strength and Quality in Chronic Kidney Disease Patients: A Sex-Specific Study. Healthcare 2025, 13, 1621. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, Y.; Chen, C.; Zhang, L.; Zhang, G. Association between chronic kidney disease and sarcopenia: A national cohort study and two-step Mendelian randomization analysis. Arch. Gerontol. Geriatr. 2026, 140, 106051. [Google Scholar] [CrossRef]
- Xiong, Y.; Jiang, X.; Zhong, Q.; Zhang, Y.; Zhang, H.; Liu, Z.; Wang, X. Possible sarcopenia and risk of chronic kidney disease: A four-year follow-up study and Mendelian randomization analysis. Endocr. Res. 2024, 49, 165–178. [Google Scholar] [CrossRef]
- Markewitz, N.; Wu, C.; Vandsburger, M.; Paredes, W.; Duran, S.; Islam, S.; Noehren, B.; Fry, C.S.; Peng, Q.; Abramowitz, M.K.; et al. Multiparametric quantitative magnetic resonance imaging of skeletal muscle in CKD. Am. J. Physiol. Ren. Physiol. 2025, 329, F99–F111. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Wang, S.; Chen, P.; Li, F.; Li, P.; Chen, Q.; Li, X.; Li, F.; Guo, S.; Zhang, J.; et al. Predictive model for sarcopenia in chronic kidney disease: A nomogram and machine learning approach using CHARLS data. Front. Med. 2025, 12, 1546988. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, J.; Zhang, W.; Chen, X.; Wu, Q.; Li, Y.; Ou, Y.; Liu, Y.; Feng, H.; Zhang, J.; et al. Device-Measured Physical Activity, Sedentary Behaviour and Risk of Chronic Kidney Diseases Across Levels of Grip Strength. J. Cachexia Sarcopenia Muscle 2025, 16, e13726. [Google Scholar] [CrossRef]
- Huang, Y.M.; Chen, W.M.; Chen, M.; Shia, B.C.; Wu, S.Y. Sarcopenia Is an Independent Risk Factor for Severe Diabetic Nephropathy in Type 2 Diabetes: A Long-Term Follow-Up Propensity Score-Matched Diabetes Cohort Study. J. Clin. Med. 2022, 11, 2992. [Google Scholar] [CrossRef]
- Wang, M.; You, L.; He, X.; Peng, Y.; Wang, R.; Zhang, Z.; Shu, J.; Zhang, P.; Sun, X.; Jia, L.; et al. Multiomics Analysis Reveals Therapeutic Targets for Chronic Kidney Disease With Sarcopenia. J. Cachexia Sarcopenia Muscle 2025, 16, e13696. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Jin, H.; Yang, J.; Zuo, Z.; Ou, R.; Hu, F.; Pu, L.; Dong, Y.; Wu, M.; Dong, B.; et al. Integrated Proteomic and Metabolomic Profiling for Developing Novel Plasma-Based Diagnostic Models of Sarcopenia. J. Cachexia Sarcopenia Muscle 2026, 17, e70188. [Google Scholar] [CrossRef]

| ProbableSarcopenia | Decreased Handgrip Strength or Chair Stand Test | Handgrip Strength <27 kg for Males and <16 kg for Females, as Assessed by a Dynamometer, and Chair Stand Test >15 s over 5 Tests |
| Definite Sarcopenia | Decreased handgrip strength or chair stand test plus low muscle quantity/quality as assessed by DEXA, BIA, CT or MRI | ASMM <20 kg for males and <15 kg for females ASMI <7 kg/m2 for males and <5.5 kg/m2 for females |
| Severe Sarcopenia | Definite Sarcopenia plus poor physical performance | A 6 m gait speed ≤0.8 m/s SFPB ≤8 points TUG ≥20 s, i.e., the time to stand up from a chair, walk for 3 m where there is a marker, then turn around, walk back and sit on the same chair. |
| STRENGTH | Difficulty in Lifting 10 Pounds None: 0 Points | Some: 1 Point | A lot: 2 Points |
| ASSISTANCE WITH WALKING | Needs None: 0 points | Some: 1 point | A lot: 2 points |
| RISING FROM A CHAIR | Difficulty None: 0 points | Some: 1 point | A lot: 2 points |
| CLIMBING 10 STAIRS | Difficulty None: 0 points | Some: 1 point | A lot: 2 points |
| FALLS | None: 0 points | Between 1 and 3: 1 points | 4 or more: 2 points |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Vallianou, N.G.; Evangelopoulos, A.A.; Christodoulatos, G.S.; Tantsi, I.; Mantouvalos, N.; Chatzis, D.; Stratigou, T.; Geladari, E.V.; Constantinou, K.; Tousis, A.; et al. Unravelling Sarcopenia in Chronic Kidney Disease: From Pathogenesis to Diagnosis and Therapeutics. Diagnostics 2026, 16, 1063. https://doi.org/10.3390/diagnostics16071063
Vallianou NG, Evangelopoulos AA, Christodoulatos GS, Tantsi I, Mantouvalos N, Chatzis D, Stratigou T, Geladari EV, Constantinou K, Tousis A, et al. Unravelling Sarcopenia in Chronic Kidney Disease: From Pathogenesis to Diagnosis and Therapeutics. Diagnostics. 2026; 16(7):1063. https://doi.org/10.3390/diagnostics16071063
Chicago/Turabian StyleVallianou, Natalia G., Apostolos A. Evangelopoulos, Gerasimos Socrates Christodoulatos, Ioanna Tantsi, Nikos Mantouvalos, Dimitrios Chatzis, Theodora Stratigou, Eleni V. Geladari, Kyriaki Constantinou, Alexandros Tousis, and et al. 2026. "Unravelling Sarcopenia in Chronic Kidney Disease: From Pathogenesis to Diagnosis and Therapeutics" Diagnostics 16, no. 7: 1063. https://doi.org/10.3390/diagnostics16071063
APA StyleVallianou, N. G., Evangelopoulos, A. A., Christodoulatos, G. S., Tantsi, I., Mantouvalos, N., Chatzis, D., Stratigou, T., Geladari, E. V., Constantinou, K., Tousis, A., & Kounatidis, D. C. (2026). Unravelling Sarcopenia in Chronic Kidney Disease: From Pathogenesis to Diagnosis and Therapeutics. Diagnostics, 16(7), 1063. https://doi.org/10.3390/diagnostics16071063







