Acute Airway Crisis in Mucopolysaccharidosis VI: Management Challenges
Abstract
1. Introduction and Clinical Significance
2. Ethical Issues
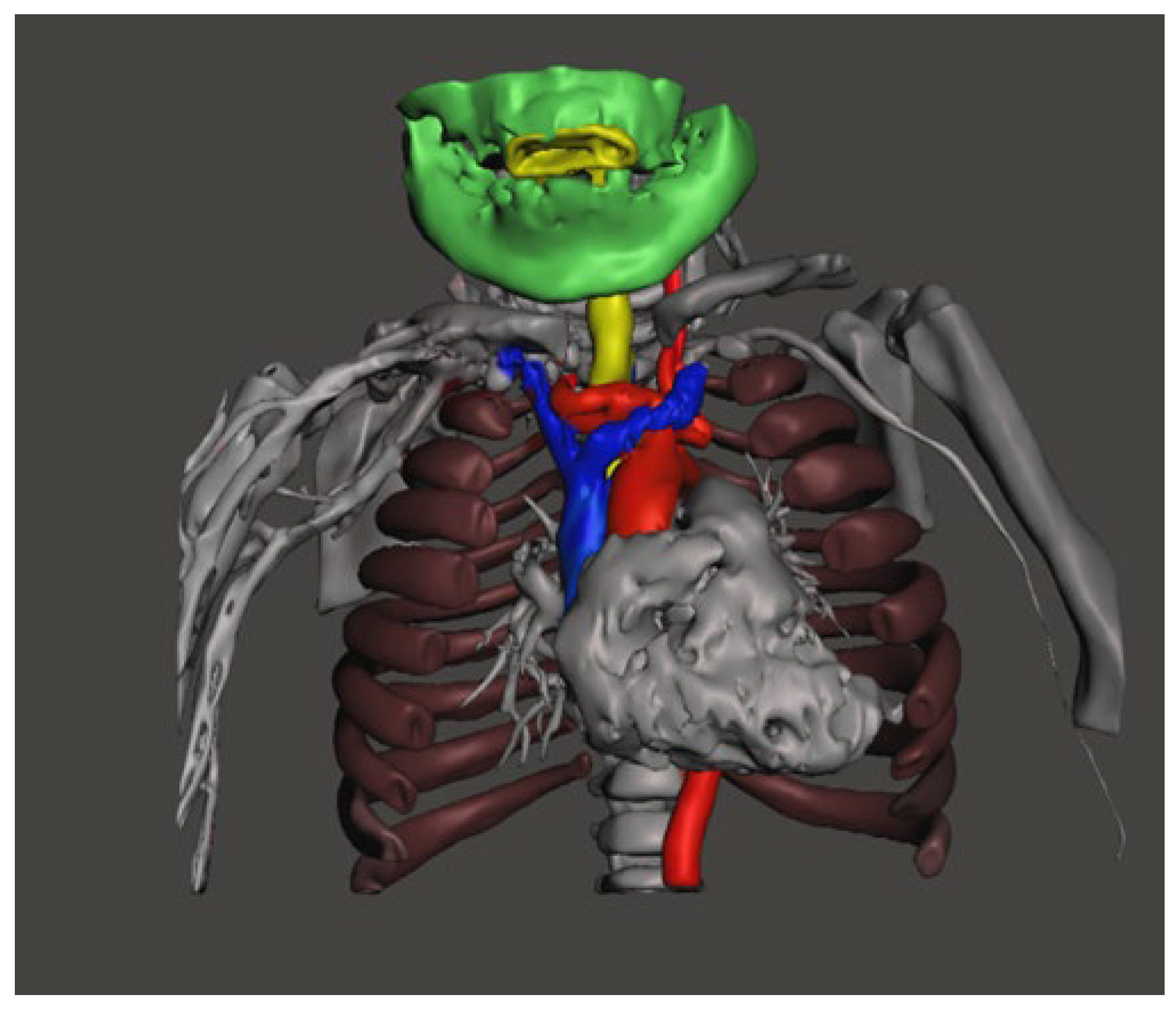
3. Case Presentation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MPS | Mucopolysaccharidosis |
| ARSB | Arylsulfatase B |
| HGMD | Human Gene Mutation Database |
| ENT | Ear, Nose and Throat |
| GAG | Glycosaminoglycans |
| ERT | Enzyme Replacement Therapy |
| BMI | Body Mass Index |
| FVC | Forced Vital Capacity |
| FEV1 | Forced Expiratory Volume in 1 s |
| CT | Computerised Tomography |
| 3D | Three Dimensions |
| LMA | Laryngeal Mask Airway |
| CPAP | Continuous Positive Airway Pressure |
References
- Mehta, A.B.; Winchester, B. Lysosomal Storage Disorders: A Practical Guide; Wiley-Blackwell Chicester: Chichester, UK, 2012. [Google Scholar]
- Muenzer, J. Overview of the mucopolysaccharidoses. Rheumatology 2011, 50, v4–v12. [Google Scholar] [CrossRef] [PubMed]
- Braunlin, E.A.; Harmatz, P.R.; Scarpa, M.; Furlanetto, B.; Kampmann, C.; Loehr, J.P.; Ponder, K.P.; Roberts, W.C.; Rosenfeld, H.M.; Giugliani, R. Cardiac disease in patients with mucopolysaccharidosis: Presentation, diagnosis and management. J. Inherit. Metab. Dis. 2011, 34, 1183–1197. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, E.; Muenzer, J. The mucopolysaccharidoses. In The Metabolic and Molecular Bases of Inherited Diseases, 8th ed.; Scriver, C.R., Beaudet, A.L., Sly, W.S., Valle, D., Childs, R., Kinzler, K.W., Eds.; McGraw-Hill: New York, NY, USA, 2001; pp. 3421–3452. [Google Scholar]
- Valayannopoulos, V.; Nicely, H.; Harmatz, P.; Turbeville, S. Mucopolysaccharidosis vi. Orphanet J. Rare Dis. 2010, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Natowicz, M.R.; Short, M.P.; Wang, Y.; Dickersin, G.R.; Gebhardt, M.C.; Rosenthal, D.I.; Sims, K.B.; Rosenberg, A.E. Clinical and biochemical manifestations of hyaluronidase deficiency. N. Engl. J. Med. 1996, 335, 1029–1033. [Google Scholar]
- Al Fahdi, I.; Singh, S.; Yadavalli, K.; Chatti, K.; Bhavani, G.S.; Girisha, K.M. Syndrome of the Month: ARSK—Related Mucopolysaccharidosis Type 10. Am. J. Med. Genet. Part A 2025, 197, e64210. [Google Scholar]
- Tulebayeva, A.; Mukhambetova, G.; Sharipova, M.; Tylki-Szymanska, A. The birth prevalence of mucopolysaccharidosis types I, II, III, IVA, VI, and VII in the Republic of Kazakhstan between 1984 and 2023. Diagnostics 2025, 15, 679. [Google Scholar] [CrossRef] [PubMed]
- D’Avanzo, F.; Zanetti, A.; De Filippis, C.; Tomanin, R. Mucopolysaccharidosis type VI, an updated overview of the disease. Int. J. Mol. Sci. 2021, 22, 13456. [Google Scholar] [CrossRef] [PubMed]
- Harmatz, P.; Shediac, R. Mucopolysaccharidosis VI: Pathophysiology, diagnosis and treatment. Front. Biosci. (Landmark Ed.) 2017, 22, 385–406. [Google Scholar] [CrossRef] [PubMed]
- Gadepalli, C.; Stepien, K.M.; Sharma, R.; Jovanovic, A.; Tol, G.; Bentley, A. Airway Abnormalities in Adult Mucopolysaccharidosis and Development of Salford Mucopolysaccharidosis Airway Score. J. Clin. Med. 2021, 10, 3275. [Google Scholar] [CrossRef] [PubMed]
- Karageorgos, L.; Brooks, D.A.; Pollard, A.; Melville, E.L.; Hein, L.K.; Clements, P.R.; Ketteridge, D.; Swiedler, S.J.; Beck, M.; Giugliani, R. Mutational analysis of 105 mucopolysaccharidosis type VI patients. Hum. Mutat. 2007, 28, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; El-Boghdadly, K.; Iliff, H.; Dua, G.; Higgs, A.; Huntington, M.; Mir, F.; Nouraei, S.R.; O’Sullivan, E.P.; Patel, A. Difficult Airway Society 2025 guidelines for management of unanticipated difficult tracheal intubation in adults. Br. J. Anaesth. 2025, 136, 283–307. [Google Scholar] [CrossRef] [PubMed]
- Akyol, M.U.; Alden, T.D.; Amartino, H.; Ashworth, J.; Belani, K.; Berger, K.I.; Borgo, A.; Braunlin, E.; Eto, Y.; Gold, J.I. Recommendations for the management of MPS IVA: Systematic evidence-and consensus-based guidance. Orphanet J. Rare Dis. 2019, 14, 137. [Google Scholar] [CrossRef] [PubMed]
- Nicolas-Jilwan, M.; AlSayed, M. Mucopolysaccharidoses: Overview of neuroimaging manifestations. Pediatr. Radiol. 2018, 48, 1503–1520. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Phillips, D.; Johnson, J.; Martin, K.; Randolph, L.M.; Rosenfeld, H.; Harmatz, P. Long-term outcomes of patients with mucopolysaccharidosis VI treated with galsulfase enzyme replacement therapy since infancy. Mol. Genet. Metab. 2021, 133, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Demis, A.A.; Oikonomidou, S.; Daglis, F.; Polymenakos, S.; Panagiotou, M. Double valve replacement in a patient with Maroteaux—Lamy syndrome as an ultimate team challenge. J. Cardiothorac. Surg. 2021, 16, 141. [Google Scholar] [CrossRef] [PubMed]
- Walker, P.; Rose, E.; Williams, J. Upper airways abnormalities and tracheal problems in Morquio’s disease. Thorax 2003, 58, 458–459. [Google Scholar] [CrossRef] [PubMed]
- Frawley, G.; Fuenzalida, D.; Donath, S.; Yaplito-Lee, J.; Peters, H. A retrospective audit of anesthetic techniques and complications in children with mucopolysaccharidoses. Pediatr. Anesth. 2012, 22, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, F.; Garavello, W. Tracheostomy in children with mucopolysaccharidosis: A systematic review. Int. J. Pediatr. Otorhinolaryngol. 2024, 187, 112167. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.-S.; Cho, D.-Y.; Ahn, K.M.; Jin, D.-K. Complications of tracheotomy in patients with mucopolysaccharidoses type II (Hunter syndrome). Int. J. Pediatr. Otorhinolaryngol. 2006, 70, 1765–1769. [Google Scholar] [CrossRef] [PubMed]
- Briscoe, S.; Martin Pintado, C.; Sutcliffe, K.; Melendez-Torres, G.; Garside, R.; Lawal, H.M.; Orr, N.; Shaw, L.; Thompson Coon, J. Evidence of inequities experienced by the rare disease community with respect to receipt of a diagnosis and access to services: A scoping review of UK and international evidence. Orphanet J. Rare Dis. 2025, 20, 303. [Google Scholar] [CrossRef] [PubMed]
- Valayannopoulos, V.; Wijburg, F.A. Therapy for the mucopolysaccharidoses. Rheumatology 2011, 50, v49–v59. [Google Scholar] [CrossRef] [PubMed]




| MPS Type (Eponym) | Incidence per 105 Live Births; Inheritance Pattern | Typical Age at Diagnosis | Typical Life Expectancy If Untreated | Enzyme Deficiency | GAG |
|---|---|---|---|---|---|
| MPS I Hurler (H) MPS I Hurler-Scheie (H-S) MPS I Scheie (S) | 0.11–1.67; AR | H: <1 year H-S: 3–8 years S: 10–20 years | H: death in childhood H-S: death in teens or early adulthood S: normal to slightly reduced lifespan | α-L-iduronidase | DS, HS |
| MPS II (Hunter) | 0.1–1.07; XR | 1–2 years when rapidly progressing | rapidly progressing: death <15 years, slowly progressing: death in adulthood | iduronate-2-sulfatase | DS, HS |
| MPS III (Sanfilippo) A-B-C-D | 0.39–1.89; AR | 4–6 years | death in puberty or early adulthood | heparan sulfamidase (A) N-acetyl-α-D-glucosaminidase (B) acetyl-CoA-α-glucosaminidase N-acetyltransferase (C) N-acetylglucosamine-6-sulfatase (D) | HS |
| MPS IV (Morquio) A-B | 0.15–0.47; AR | 1–3 years | death in childhood- middle age | N-acetylgalactosamine-6-sulfatase (A) β-galactosidase (B) | CS, KS (A) KS (B) |
| MPS VI (Maroteaux-Lamy) | 0–0.38; AR | rapidly progressing: 1–9 years, slowly progressing: >5 years | rapidly progressing: death in 2nd–3rd decade slowly progressing: death in 4–5th decade | N-acetylgalactosamine-4-sulfatase | DS |
| MPS VII (Sly) | 0–0.29; AR | neonatal to adulthood | death in infancy- 4th decade ** | β-D-glucuronidase | CS, DS, HS |
| MPS IX (Natowicz) * | unknown | adolescence | unknown | hyaluronidase | CS |
| MPS X *** | unknown AR | childhood-onset | unknown | Arylsulfatase K | DS |
| Airway Plan | Challenges | Consequence | Methods to Mitigate |
|---|---|---|---|
| Plan A Endo tracheal intubation | Reduced mouth opening, large tongue, high anterior larynx, and limited neck extension. A tortuous trachea with multiple narrowings can make passage of the endotracheal tube difficult. | Access to the larynx and passage of the endotracheal tube into the trachea will be difficult | Using a small endotracheal tube 1. Nasal intubation 2. Oral intubation with video laryngoscope or Hopkins rod telescope 3. Awake nasal or oral fiberoptic using an airway conduit |
| Plan B Laryngeal Mask airway (LMA) | Limited mouth opening, large tongue, high anterior larynx, bulky supraglottic | Inserting the LMA and securing a seal will be difficult | Using a reinforced LMA, which is more flexible to reach the anterior larynx |
| Plan C Bag and mask ventilation | Limited mouth opening, large tongue that can fall posteriorly occluding the airway, bulky oropharynx, bulky supraglottis | Inability to pass oxygen beyond the oropharynx due to obstruction | 1. Guedel’s airway to bypass the tongue base 2. Nasopharyngeal airway to bypass the epiglottis |
| Plan D Front of neck access | Short neck, limited extension, large head, small torso Large vessels in the thoracic inlet | Accessing the cervical trachea will be difficult, a large vessel catastrophic haemorrhage, and inserting the right-sized tracheostomy tube | Avoid tracheostomy; if attempted, perform high tracheostomy. Planning the right tracheostomy tube before the surgery |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tulebayeva, A.; Gadepalli, C.; Sharipova, M. Acute Airway Crisis in Mucopolysaccharidosis VI: Management Challenges. Diagnostics 2026, 16, 1009. https://doi.org/10.3390/diagnostics16071009
Tulebayeva A, Gadepalli C, Sharipova M. Acute Airway Crisis in Mucopolysaccharidosis VI: Management Challenges. Diagnostics. 2026; 16(7):1009. https://doi.org/10.3390/diagnostics16071009
Chicago/Turabian StyleTulebayeva, Assel, Chaitanya Gadepalli, and Maira Sharipova. 2026. "Acute Airway Crisis in Mucopolysaccharidosis VI: Management Challenges" Diagnostics 16, no. 7: 1009. https://doi.org/10.3390/diagnostics16071009
APA StyleTulebayeva, A., Gadepalli, C., & Sharipova, M. (2026). Acute Airway Crisis in Mucopolysaccharidosis VI: Management Challenges. Diagnostics, 16(7), 1009. https://doi.org/10.3390/diagnostics16071009






