The Diagnostic Challenge of Rapunzel Syndrome: Multimodal Imaging Findings in a Child
Abstract
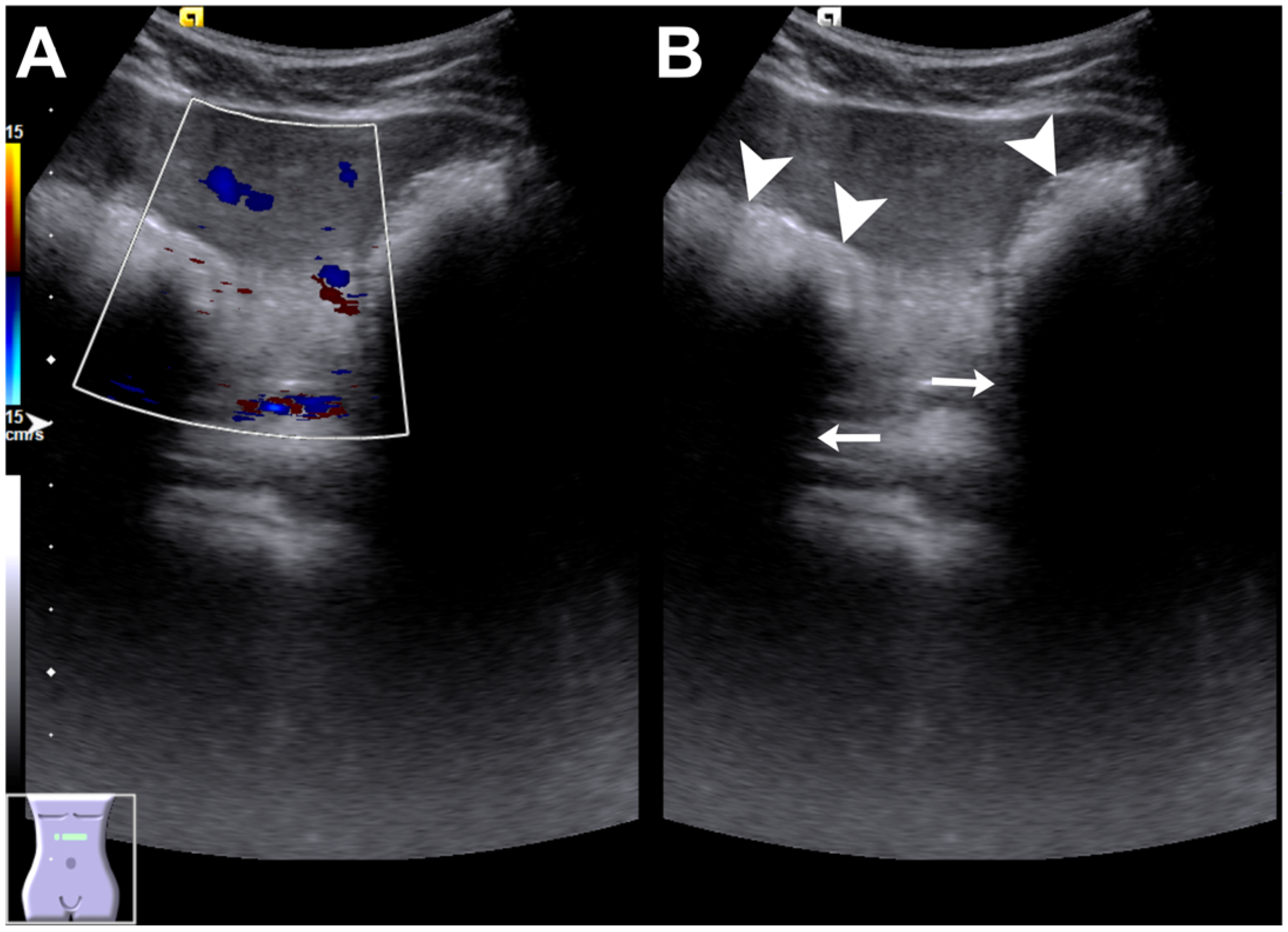
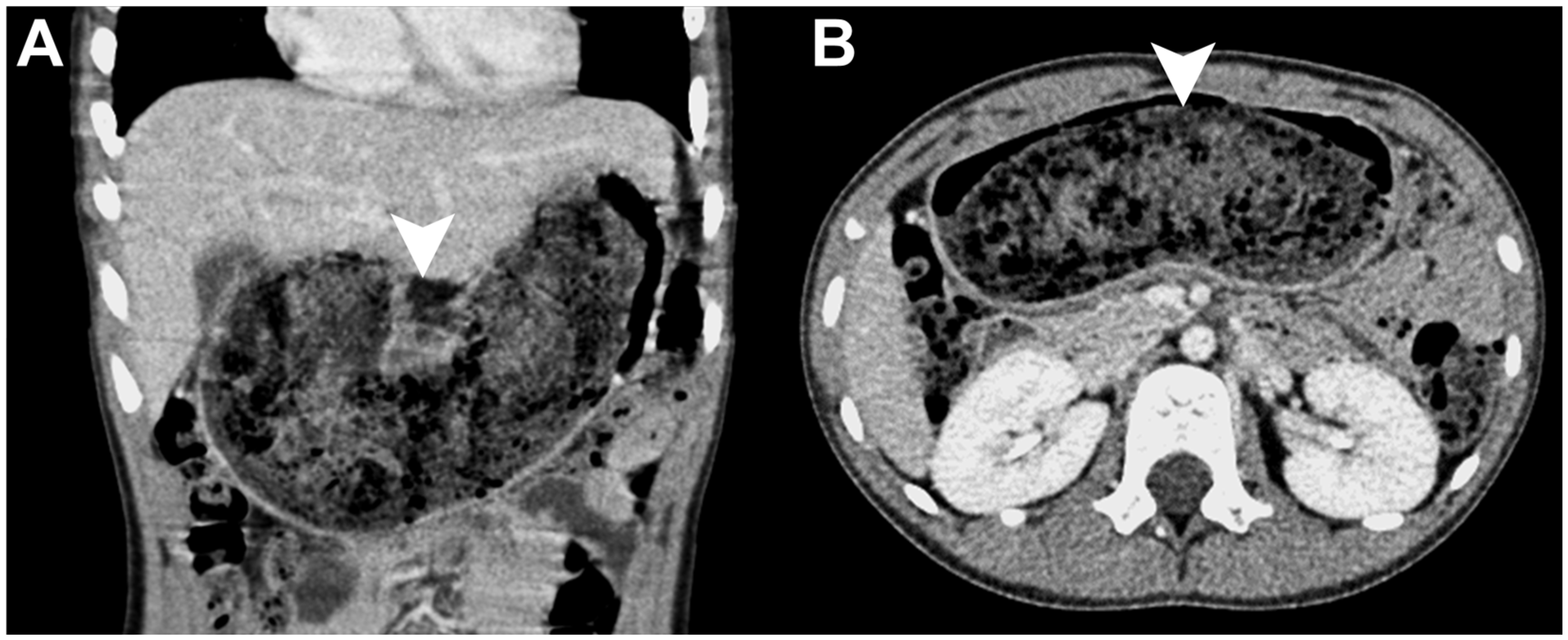


Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CT | Computed tomography |
| EGD | Esophagogastroduodenoscopy |
References
- Lisi, D.; D’Ovidio, V.; Lucidi, C. A Giant Trichobezoar Diagnosed by Ultrasound, Confirmed by Upper Endoscopy, and Managed by Minimally Invasive Laparoscopic Surgery. Clin. Gastroenterol. Hepatol. 2021, 19, e125. [Google Scholar] [CrossRef]
- Malik, N.; Malik, A.; Premkumar, A. A Rare but Significant Clinical Entity: Trichobezoar: 2388. Off. J. Am. Coll. Gastroenterol. ACG 2017, 112, S1301–S1302. [Google Scholar] [CrossRef]
- Ripollés, T.; García-Aguayo, J.; Martínez, M.J.; Gil, P. Gastrointestinal bezoars: Sonographic and CT characteristics. AJR Am. J. Roentgenol. 2001, 177, 65–69. [Google Scholar] [CrossRef]
- Sacco, M.A.; Gualtieri, S.; Princi, A.; Tarda, L.; Tarallo, A.P.; Calanna, L.; Lombardo, S.; Calafiore, J.; Gratteri, S.; Aquila, I. Rapunzel Syndrome: Clinical, Diagnostic and Forensic Aspects in Related Deaths—A Review of the Literature. J. Clin. Med. 2024, 13, 7464. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.J.; Park, J. Treatment of large gastric trichobezoar in children: Two case reports and literature review. Medicine 2023, 102, e33589. [Google Scholar] [CrossRef]
- Arshad, R.; Arshad, D.; Aamir, S.; Basharat, F.; Khan, H.; Hanif, M. 117 Double Trouble: Rapunzel Syndrome Presenting with Closed-Loop Intestinal Obstruction Between Two Synchronous Trichobezoars. Br. J. Surg. 2023, 110, znad241.439. [Google Scholar] [CrossRef]
- Ahmad, S.J.S.; Drvaric, I.; Ahmed, A.R.; Jakob, D.; Kyriazidis, I.P.; Pouwels, S.; Hajibandeh, S.; Tang, A.; Cripps, P.; Yang, W.; et al. From obstruction to ischaemia: A systematic review and meta-analysis on the diagnostic accuracy of CT scans in identifying small and large bowel obstruction, underlying causes and predicting critical complications in adults. BMJ Open 2025, 15, e103887. [Google Scholar] [CrossRef]
- Gorter, R.R.; Kneepkens, C.M.; Mattens, E.C.; Aronson, D.C.; Heij, H.A. Management of trichobezoar: Case report and literature review. Pediatr. Surg. Int. 2010, 26, 457–463. [Google Scholar] [CrossRef]
- Matsuoka, R.; Masuda, S.; Fujita, S.; Akiyama, N. Trichobezoar effectively treated with direct endoscopic injection of Coca-Cola: A case report. DEN Open 2024, 4, e283. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.-l.; Zhao, W.-c.; Wang, Y.-s. Endoscopic retrieval of gastric trichophytobezoar: Case report of a 12-year-old girl with trichophagia. Medicine 2017, 96, e5969. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Khan, I.-a.; Ullah, K.; Khan, S.; Wang, X.; Zhu, L.-p.; Rehman, M.u.M.; Chen, X.; Wang, B.-m. Etiological aspects of intragastric bezoars and its associations to the gastric function implications: A case report and a literature review. Medicine 2018, 97, e11320. [Google Scholar] [CrossRef] [PubMed]
- Alsafwah, S.; Alzein, M. Small bowel obstruction due to trichobezoar: Role of upper endoscopy in diagnosis. Gastrointest. Endosc. 2000, 52, 784–786. [Google Scholar] [CrossRef] [PubMed]
- Ladas, S.D.; Kamberoglou, D.; Karamanolis, G.; Vlachogiannakos, J.; Zouboulis-Vafiadis, I. Systematic review: Coca-Cola can effectively dissolve gastric phytobezoars as a first-line treatment. Aliment. Pharmacol. Ther. 2013, 37, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Varanko, A.; Saha, S.; Chilkoti, A. Recent trends in protein and peptide-based biomaterials for advanced drug delivery. Adv. Drug Deliv. Rev. 2020, 156, 133–187. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.B.; Talat, N.; Saleem, M. Gastrointestinal trichobezoar: An experience with 17 cases. J. Pediatr. Surg. 2020, 55, 2504–2509. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.R.; Hull, N.C.; Lee, E.Y.; Phillips, G.S. Pediatric Abdominal Masses: Imaging Guidelines and Recommendations. Radiol. Clin. N. Am. 2022, 60, 113–129. [Google Scholar] [CrossRef] [PubMed]
- Ranganath, S.H.; Lee, E.Y.; Eisenberg, R.L. Focal Cystic Abdominal Masses in Pediatric Patients. Am. J. Roentgenol. 2012, 199, W1–W16. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.D.; Grither, A.; Cohen, S.G.; Morales-Perez, L.; Rabiner, J.; Riera, A. Hair Today, OR Tomorrow: A Multicenter Case Series of Gastric Bezoars in Children Diagnosed With Point-of-Care Ultrasound. Pediatr. Emerg. Care 2025, 41, 63–67. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, C.-H.; Weng, S.-C. The Diagnostic Challenge of Rapunzel Syndrome: Multimodal Imaging Findings in a Child. Diagnostics 2026, 16, 940. https://doi.org/10.3390/diagnostics16060940
Wang C-H, Weng S-C. The Diagnostic Challenge of Rapunzel Syndrome: Multimodal Imaging Findings in a Child. Diagnostics. 2026; 16(6):940. https://doi.org/10.3390/diagnostics16060940
Chicago/Turabian StyleWang, Chih-Hao, and Shu-Chao Weng. 2026. "The Diagnostic Challenge of Rapunzel Syndrome: Multimodal Imaging Findings in a Child" Diagnostics 16, no. 6: 940. https://doi.org/10.3390/diagnostics16060940
APA StyleWang, C.-H., & Weng, S.-C. (2026). The Diagnostic Challenge of Rapunzel Syndrome: Multimodal Imaging Findings in a Child. Diagnostics, 16(6), 940. https://doi.org/10.3390/diagnostics16060940





