Molecular Pathology, Artificial Intelligence, and New Technologies in Hematologic Diagnostics: Translational Opportunities and Practical Considerations
Abstract
1. Introduction
2. Methods
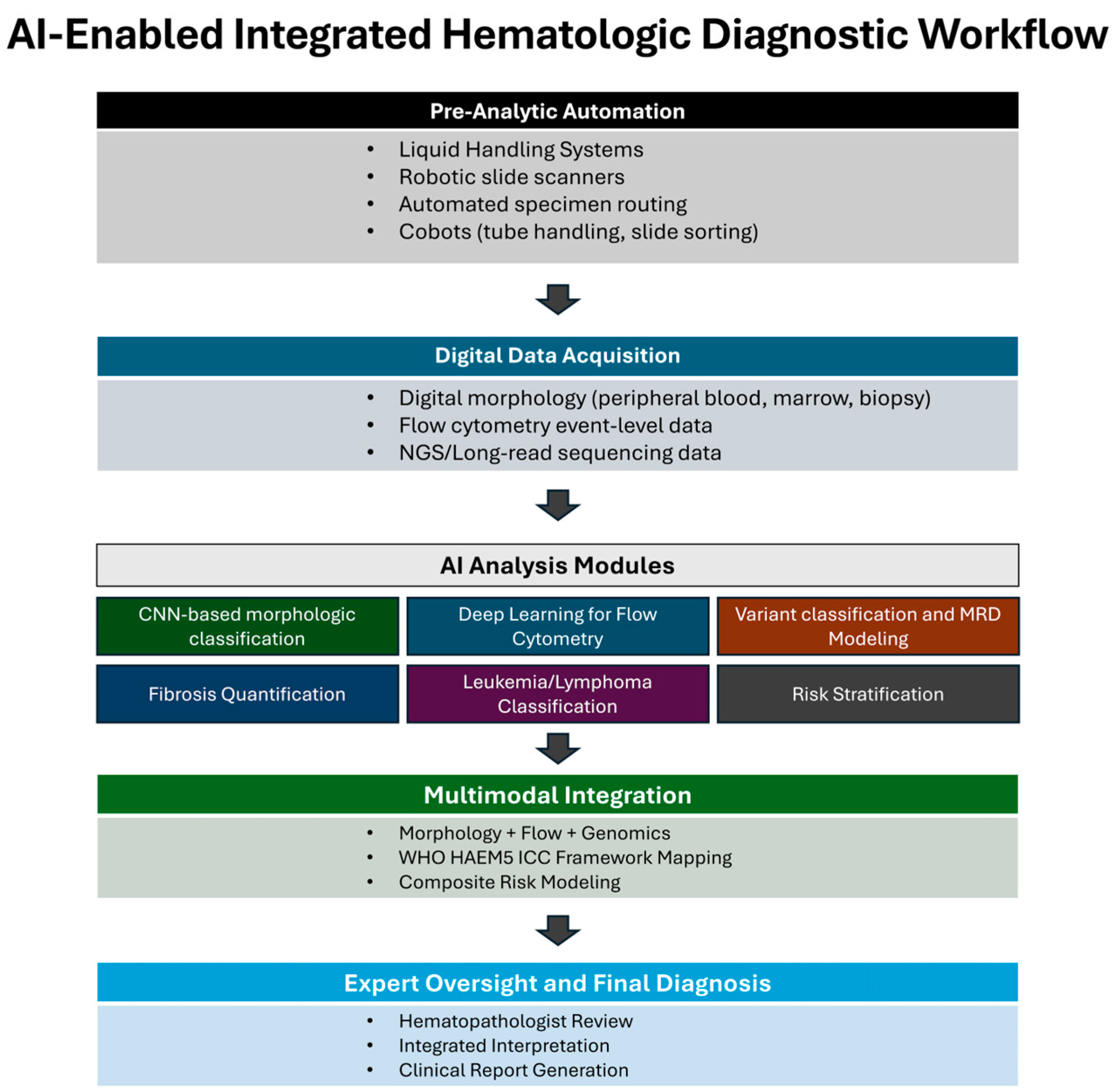
3. Results
3.1. Machine Learning Paradigms in Hematologic Diagnostics
3.2. Automation and Collaborative Robotics in Hematology and Molecular Pathology
| Application | Platform/Site | Domain | Key Points |
|---|---|---|---|
| Pre-analytic blood tube handling | UR5 cobots at Copenhagen University Hospital Gentofte | Core hematology | Two UR5 arms sort and load tubes into analyzers, processing ~3000 samples/day while maintaining a target of >90% results within one hour without additional staff. |
| High-throughput slide scanning cluster | Pramana Spectral HT cluster with robotic arm | Digital pathology | Four single-slide scanners around a central robotic handler; one cluster can scan >1000 slides/day with automated quality control and real-time error detection. |
| Large-scale precision medicine operations | Caris Life Sciences and Pramana partnership | Digital oncology | Caris integrates Pramana Spectral HT in a multi-scanner lab processing ~1.5 million slides/year; reported 100% scan success in a study of 867 slides with >98% agreement between automated and manual QC. |
| Academic pathology digitization | Vanderbilt University Medical Center and Pramana | Research and clinical digitization | Pramana system with four scanning compartments and a central robotic arm digitizes ~900 slides/day; pilot aims to scan 500,000 slides over three years for research and planning for primary digital diagnosis. |
| Hematopathology-specific AI collaboration | ARUP-Pramana partnership | Hematopathology and AI | Collaboration uses HT scanners and hematopathology expertise to develop AI algorithms for bone marrow biopsies and other hematopathology challenges, with an emphasis on edge AI deployment. |
3.3. Digital Morphology and AI in Peripheral Blood and Bone Marrow
3.4. AI in Flow Cytometry
3.5. AI in Molecular Pathology of Hematologic Neoplasms
3.6. Translational and Economic Considerations
| Domain | Representative Work | Use Case | Relevance |
|---|---|---|---|
| Peripheral blood and marrow smears | Matek 2019 [4]; Ahmed 2019 [17]; Goldgof 2023 [18] | Blast detection; leukemia subtype identification; marrow cell classification | Demonstrated that CNNs can reach human-level performance in blast recognition and assign leukemia subtypes from digital smears and marrow images; ensemble models can classify >20 marrow cell classes. |
| Marrow biopsies and fibrosis | Ryou 2023 [19]; Yu 2020 [20] | Quantitative fibrosis grading; MDS pattern analysis | ML-based fibrosis indices improve reproducibility and granularity; AI-supported histologic analysis aids in diagnosing MDS and linking morphology to genetic profiles. |
| Flow cytometry | Zhao 2020 [21]; Zhong 2022 [22]; Ng 2024 [5]; Spies 2025 [6] | Classification of B-cell neoplasms; acute leukemia diagnosis; AI implementation guidance | Deep learning on multiparameter flow data achieves hematologist-level classification; AI-assisted algorithms support acute leukemia diagnosis; expert frameworks guide AI deployment in flow cytometry. |
| MRD and longitudinal monitoring | Mocking 2025 [23]; Fuda 2023 [24] | MRD assessment in AML and other neoplasms | ML methods help standardize immunophenotypic MRD assessment and support risk-adapted treatment decisions. |
| Molecular risk models in myeloid neoplasms | Nazha 2021 [31]; Awada 2021 [32]; Al-Nusair 2025 [30] | Prognostic modeling in MDS and AML | Machine learning-based risk scores combine mutations, cytogenetics, and clinical data to refine prognosis and complement ICC- and WHO-based frameworks. |
| Morphology-genomics linkage | Kockwelp 2023 [35]; Yu 2020 [20] | Image-based prediction of mutations; morphology-mutation correlation | Image-based models predict therapy-relevant mutations in AML and link marrow histology to specific mutational profiles, suggesting that AI can bridge micro- and genomic scales. |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO Classification of Tumours Editorial Board. Haematolymphoid tumours. In WHO Classification of Tumours, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2024; Volume 11. [Google Scholar]
- Arber, D.A.; Orazi, A.; Hasserjian, R.P.; Borowitz, M.J.; Calvo, K.R.; Kvasnicka, H.-M.; Wang, S.A.; Bagg, A.; Barbui, T.; Branford, S.; et al. International consensus classification of myeloid neoplasms and acute leukemias: Integrating morphologic, clinical, and genomic data. Blood 2022, 140, 1200–1228. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.Y.; Lee, S.G.; Kim, T.H. Economic evaluation of total laboratory automation in the clinical laboratory of a tertiary care hospital. Ann. Lab. Med. 2022, 42, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Matek, C.; Schwarz, S.; Spiekermann, K.; Marr, C. Human-level recognition of blast cells in acute myeloid leukaemia with convolutional neural networks. Nat. Mach. Intell. 2019, 1, 538–544. [Google Scholar] [CrossRef]
- Ng, D.P.; Simonson, P.D.; Tarnok, A.; Lucas, F.; Kern, W.; Rolf, N.; Bogdanoski, G.; Green, C.; Brinkman, R.R.; Czechowska, K. Recommendations for using artificial intelligence in clinical flow cytometry. Cytom. Part B Clin. Cytom. 2024, 106, 228–238. [Google Scholar] [CrossRef]
- Spies, N.C.; Rangel, A.; English, P.; Morrison, M.; O’fallon, B.; Ng, D.P. Machine learning methods in clinical flow cytometry. Cancers 2025, 17, 483. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, O.; McVeigh, T.P. Increasing use of artificial intelligence in genomic medicine for cancer care: The promise and potential pitfalls. BJC Rep. 2025, 3, 20. [Google Scholar] [CrossRef]
- Universal Robots. Two UR5 Ensure Faster Delivery of Blood Sample Results at Gentofte Hospital. 2025. Available online: https://www.universal-robots.com/case-stories/gentofte-hospital/ (accessed on 9 November 2025).
- Pramana. High-Throughput Slide Scanning-As-A-Service Market Overview: Major Segments, Strategic Developments, and Leading Companies. Digital Pathology as a Service–High-Throughput Scanning. 2023. Available online: https://dataintelo.com/report/high-throughput-slide-scanning-as-a-service-market (accessed on 9 November 2025).
- Najdawi, M. Digital Pathology Aims to Unlock Secrets Hidden in Tissue Samples. 2025. Available online: https://news.vumc.org/2025/02/03/digital-pathology-aims-to-unlock-secrets-hidden-in-tissue-samples/ (accessed on 9 November 2025).
- Kratz, A.; Bengtsson, H.I.; Casey, J.E.; Keefe, J.M.; Beatrice, G.H.; Grzybek, D.Y.; Lewandrowski, K.B.; Van Cott, E.M. Performance evaluation of the CellaVision DM96 system: WBC differentials by automated digital image analysis supported by an artificial neural network. Am. J. Clin. Pathol. 2005, 124, 770–781. [Google Scholar] [CrossRef]
- Katz, B.; Feldman, M.D.; Tessema, M.; Benisty, D.; Toles, G.S.; Andre, A.; Shtreker, B.; Paz, F.M.; Edwards, J.; Jengehino, D.; et al. Evaluation of Scopio Labs X100 full field PBS: The first high-resolution full field viewing of peripheral blood specimens combined with artificial intelligence-based morphological analysis. Int. J. Lab. Hematol. 2021, 43, 1408–1416. [Google Scholar] [CrossRef]
- De Iuliis, V.; Chiatamone Ranieri, S. Performance evaluation of the Scopio Labs X100HT digital morphology analyzer and abnormal cell detection in peripheral blood smears. Int. J. Lab. Hematol. 2025, 48, 54–62. [Google Scholar] [CrossRef]
- Kratz, A.; Lee, S.; Zini, G.; Riedl, J.A.; Hur, M.; Machin, S.; The International Council for Standardization in Haematology. Digital morphology analyzers in hematology: ICSH review and recommendations. Int. J. Lab. Hematol. 2019, 41, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Kock, F.; Pontones, M.; Ghete, T.; Metzler, M.; Höfener, H. Selecting a digitization device for bone marrow aspirate smears. Lab. Investig. 2025, 105, 104114. [Google Scholar] [CrossRef]
- Available online: https://www.aruplab.com/news/03-11-2025/pramana-and-arup-partner-digitize-pathology-slides (accessed on 9 November 2025).
- Ahmed, N.; Yigit, A.; Isik, Z.; Alpkocak, A. Identification of leukemia subtypes from microscopic images using convolutional neural network. Diagnostics 2019, 9, 104. [Google Scholar] [CrossRef]
- Goldgof, G.M.; Sun, S.; Van Cleave, J.; Wang, L.; Lucas, F.; Brown, L.; Spector, J.D.; Boiocchi, L.; Baik, J.; Zhu, M.; et al. DeepHeme: A generalizable, bone marrow classifier with hematopathologist-level performance. bioRxiv 2023. [Google Scholar] [CrossRef]
- Ryou, H.; Sirinukunwattana, K.; Aberdeen, A.; Grindstaff, G.; Stolz, BJ.; Byrne, H.; Harrington, HA.; Sousos, N.; Godfrey, AL.; Harrison, CN.; et al. Continuous indexing of fibrosis (CIF): improving the assessment and classification of MPN patients. Leukemia 2023, 37, 348–358. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yu, D.; Zhang, H.; Song, Y.; Tao, Y.; Zhou, F.; Wang, Z.; Fu, R.; Sun, T.; Dong, H.; Gu, W.; et al. Artificial intelligence-based quantitative bone marrow pathology analysis for myeloproliferative neoplasms. Haematologica 2025, 110, 2691–2701. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhao, M.; Mallesh, N.; Höllein, A.; Schabath, R.; Haferlach, C.; Haferlach, T.; Elsner, F.; Lüling, H.; Krawitz, P.; Kern, W. Hematologist-level classification of mature B-cell neoplasm using deep learning on multiparameter flow cytometry data. Cytom. A 2020, 97, 1073–1080. [Google Scholar] [CrossRef]
- Zhong, P.; Hong, M.; He, H.; Zhang, J.; Chen, Y.; Wang, Z.; Chen, P.; Ouyang, J. Diagnosis of acute leukemia by multiparameter flow cytometry with the assistance of artificial intelligence. Diagnostics 2022, 12, 827. [Google Scholar] [CrossRef]
- Mocking, T.R.; van de Loosdrecht, A.A.; Cloos, J.; Bachas, C. Applications of machine learning for immunophenotypic measurable residual disease assessment in acute myeloid leukemia. HemaSphere 2025, 9, e70138. [Google Scholar] [CrossRef]
- Fuda, F.; Chen, M.; Chen, W.; Cox, A. Artificial intelligence in clinical multiparameter flow cytometry and mass cytometry—Key tools and progress. Semin. Diagn. Pathol. 2023, 40, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, L.S.; Holmes, J.; Taghiyev, A.F. The emergence of artificial intelligence-guided karyotyping: A review and reflection. Genes 2025, 16, 685. [Google Scholar] [CrossRef]
- Doig, K.D.; Perera, R.; Kankanige, Y.; Fellowes, A.; Li, J.; Lupat, R.; Thompson, E.R.; Blombery, P.; Fox, S.B. Using artificial intelligence (AI) to model clinical variant reporting for next generation sequencing (NGS) oncology assays. BioData Min. 2025, 18, 74. [Google Scholar] [CrossRef]
- Steinicke, T.L.; Benfatto, S.; Capilla-Guerra, M.R.; Monteleone, A.B.; Young, J.H.; Shankar, S.; Michaels, P.D.; Tsai, H.K.; Good, J.D.; Kreso, A.; et al. Rapid epigenomic classification of acute leukemia. Nat. Genet. 2025, 57, 2456–2467. [Google Scholar] [CrossRef] [PubMed]
- Short, N.J.; Aldoss, I.; DeAngelo, D.J.; Konopleva, M.; Leonard, J.; Logan, A.C.; Park, J.; Shah, B.; Stock, W.; Jabbour, E. Clinical use of measurable residual disease in adult ALL: Recommendations from a panel of US experts. Blood Adv. 2025, 9, 1442–1451. [Google Scholar] [CrossRef] [PubMed]
- Cloos, J.; Valk, P.J.; Thiede, C.; Döhner, K.; Roboz, G.J.; Wood, B.L.; Walter, R.B.; A Wang, S.; Wierzbowska, A.; Wei, A.H.; et al. 2025 update on MRD in acute myeloid leukemia: A consensus document from the ELN-DAVID MRD Working Party. Blood, 2025; in press. [Google Scholar] [CrossRef] [PubMed]
- Al-Nusair, J.; Lanino, L.; Durmaz, A.; Della Porta, M.G.; Zeidan, A.M.; Kewan, T. Artificial intelligence in myeloid malignancies: Clinical applications of machine learning in myelodysplastic syndromes and acute myeloid leukemia. Blood Rev. 2025, 74, 101340. [Google Scholar] [CrossRef] [PubMed]
- Nazha, A.; Komrokji, R.; Meggendorfer, M.; Jia, X.; Radakovich, N.; Shreve, J.; Hilton, C.B.; Nagata, Y.; Hamilton, B.K.; Mukherjee, S.; et al. Personalized prediction model to risk stratify patients with myelodysplastic syndromes. J. Clin. Oncol. 2021, 39, 3737–3746. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Awada, H.; Durmaz, A.; Gurnari, C.; Kishtagari, A.; Meggendorfer, M.; Kerr, C.M.; Kuzmanovic , T.; Durrani, J.; Shreve, J.; Nagata, Y. Machine learning integrates genomic signatures for subclassification beyond primary and secondary acute myeloid leukemia. Blood 2021, 138, 1885–1895. [Google Scholar] [CrossRef] [PubMed]
- Walker, B.A.; Mavrommatis, K.; Wardell, C.P.; Ashby, T.C.; Bauer, M.; Davies, F.; Rosenthal, A.; Wang, H.; Qu, P.; Hoering, A.; et al. A high-risk, double-hit, group of newly diagnosed myeloma identified by genomic analysis. Leukemia 2019, 33, 159–170. [Google Scholar] [CrossRef]
- van Eekelen, L.; Litjens, G.; Hebeda, K.M. Artificial intelligence in bone marrow histological diagnostics: Potential applications and challenges. Pathobiology 2024, 91, 8–17. [Google Scholar] [CrossRef]
- Kockwelp, J.; Thiele, S.; Bartsch, J.; Haalck, L.; Gromoll, J.; Schlatt, S.; Exeler, R.; Bleckmann, A.; Lenz, G.; Wolf, S.; et al. Deep learning predicts therapy-relevant genetics in acute myeloid leukemia from Pappenheim-stained bone marrow smears. Blood Adv. 2024, 8, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Ahlers, S.M.; Stratman, C.; Aridor, O.; Pantanowitz, L.; Fine, J.L.; Kuzmishin, J.A.; Montalto, M.C.; Parwani, A.V. Can digital pathology result in cost savings? A financial projection for digital pathology implementation at a large integrated health care organization. J. Pathol. Inform. 2014, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- Matias-Guiu, X.; Temprana-Salvador, J.; Lopez, P.G.; Kammerer-Jacquet, S.-F.; Rioux-Leclercq, N.; Clark, D.; Schürch, C.M.; Fend, F.; Mattern, S.; Snead, D.; et al. Implementing digital pathology: Qualitative and financial insights from eight leading European laboratories. Virchows Arch. 2025, 487, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Cheah, P.L.; Looi, L.M.; Horton, S. Cost analysis of operating an anatomic pathology laboratory in a middle-income country. Am. J. Clin. Pathol. 2018, 149, 1–7. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Alnoor, F.; Mukherjee, S.; Menon, M.P.; Ng, D.; Li, P.; Ohgami, R.S. Molecular Pathology, Artificial Intelligence, and New Technologies in Hematologic Diagnostics: Translational Opportunities and Practical Considerations. Diagnostics 2026, 16, 913. https://doi.org/10.3390/diagnostics16060913
Alnoor F, Mukherjee S, Menon MP, Ng D, Li P, Ohgami RS. Molecular Pathology, Artificial Intelligence, and New Technologies in Hematologic Diagnostics: Translational Opportunities and Practical Considerations. Diagnostics. 2026; 16(6):913. https://doi.org/10.3390/diagnostics16060913
Chicago/Turabian StyleAlnoor, Fnu, Shuvam Mukherjee, Madhu P. Menon, David Ng, Peng Li, and Robert S. Ohgami. 2026. "Molecular Pathology, Artificial Intelligence, and New Technologies in Hematologic Diagnostics: Translational Opportunities and Practical Considerations" Diagnostics 16, no. 6: 913. https://doi.org/10.3390/diagnostics16060913
APA StyleAlnoor, F., Mukherjee, S., Menon, M. P., Ng, D., Li, P., & Ohgami, R. S. (2026). Molecular Pathology, Artificial Intelligence, and New Technologies in Hematologic Diagnostics: Translational Opportunities and Practical Considerations. Diagnostics, 16(6), 913. https://doi.org/10.3390/diagnostics16060913






