Early Identification of Atherosclerosis in People Living with HIV by Coronary Computed Tomography Angiography
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design
2.2. Study Population
2.3. Definitions
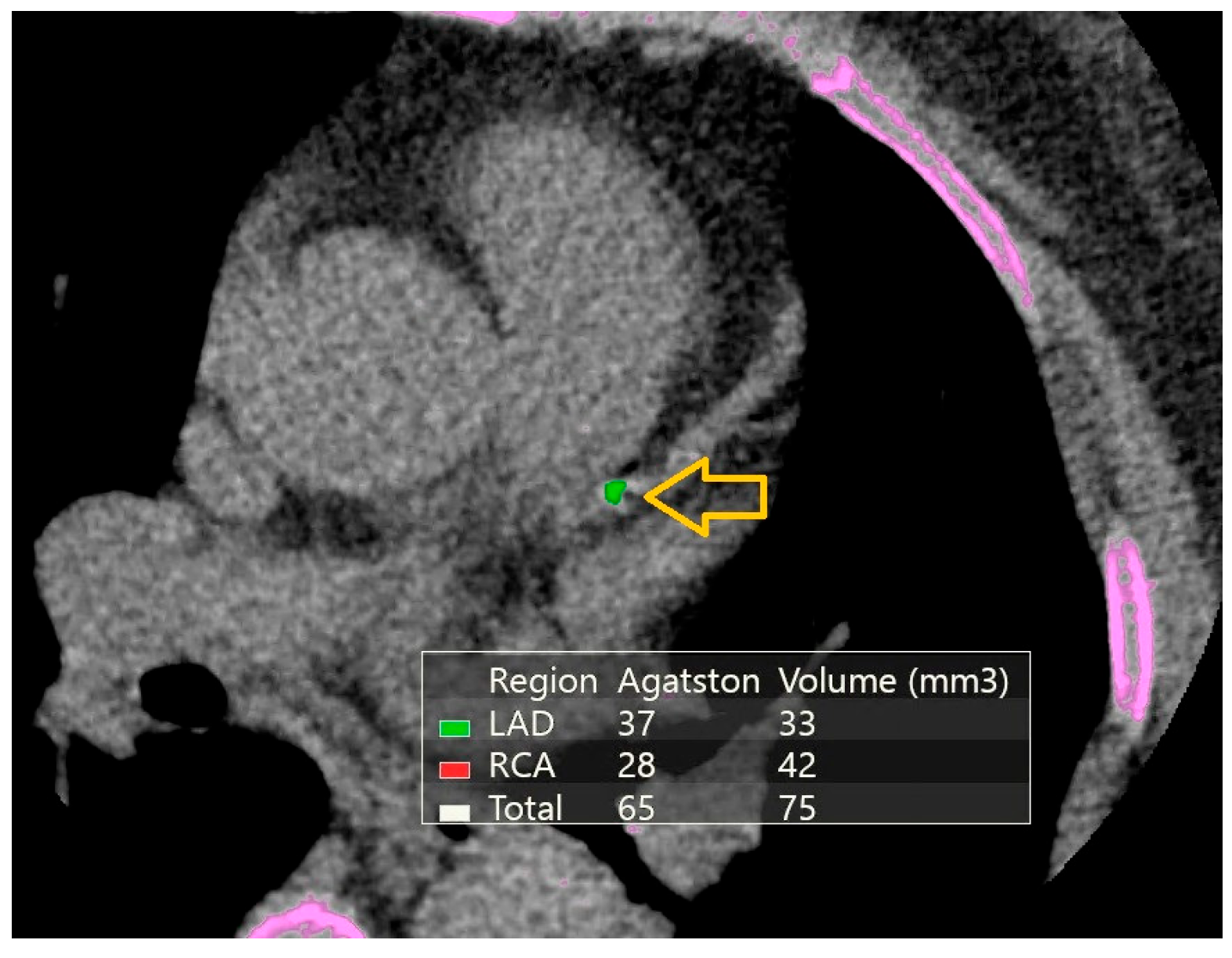
2.4. Cardiac Computed Tomography Angiography
2.5. Ethical Approval
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- UNAIDS. 2025. Available online: https://www.unaids.org/en/resources/fact-sheet (accessed on 10 November 2025).
- Grinspoon, S.K.; Fitch, K.V.; Overton, E.T.; Fichtenbaum, C.J.; Zanni, M.V.; Aberg, J.A.; Malvestutto, C.; Lu, M.T.; Currier, J.S.; Sponseller, C.A.; et al. Rationale and design of the Randomized Trial to Prevent Vascular Events in HIV (REPRIEVE). Am. Heart J. 2019, 212, 23–35. [Google Scholar] [CrossRef]
- Shah, A.S.; Stelzle, D.; Lee, K.K.; Beck, E.J.; Alam, S.; Clifford, S.; Longenecker, C.T.; Strachan, F.; Bagchi, S.; Whiteley, W.; et al. Global burden of atherosclerotic cardiovascular disease in people living with HIV: Systematic review and meta-analysis. Circulation 2018, 138, 1100–1112. [Google Scholar] [CrossRef]
- Feinstein, M.J.; Hsue, P.Y.; Benjamin, L.A.; Bloomfield, G.S.; Currier, J.S.; Freiberg, M.S.; Grinspoon, S.K.; Levin, J.; Longenecker, C.T.; Post, W.S.; et al. Characteristics, prevention, and management of cardiovascular disease in people living with HIV: A scientific statement from the American Heart Association. Circulation 2019, 140, e98–e124. [Google Scholar] [CrossRef]
- Fayos, M.; de las Revillas, F.A.; Quintanilla, V.G.; González-Rico, C.; Fariñas-Álvarez, C.; Parra, J.A.; Fariñas, M.C. Progression of subclinical cardiovascular disease in patients with HIV. Rev. Esp. Quimioter. 2024, 37, 341–350. [Google Scholar] [CrossRef]
- Hunt, P.W. HIV and inflammation: Mechanisms and consequences. Curr. HIV/AIDS Rep. 2012, 9, 139–147. [Google Scholar] [CrossRef]
- Foldyna, B.; Mayrhofer, T.; Zanni, M.V.; Lyass, A.; Barve, R.; Karady, J.; McCallum, S.; Burdo, T.H.; Fitch, K.V.; Paradis, K.; et al. Pericoronary adipose tissue density, inflammation, and subclinical coronary artery disease among people with HIV in the REPRIEVE cohort. Clin. Infect. Dis. 2023, 77, 1676–1686. [Google Scholar] [CrossRef]
- Paternò Raddusa, M.S.; Marino, A.; Celesia, B.M.; Spampinato, S.; Giarratana, C.; Venanzi Rullo, E.; Cacopardo, B.; Nunnari, G. Atherosclerosis and cardiovascular complications in people living with HIV: A focused review. Infect. Dis. Rep. 2024, 16, 846–863. [Google Scholar] [CrossRef] [PubMed]
- Hanna, D.B.; Moon, J.Y.; Haberlen, S.A.; French, A.L.; Palella, F.J., Jr.; Gange, S.J.; Witt, M.D.; Kassaye, S.; Lazar, J.M.; Tien, P.C.; et al. Carotid artery atherosclerosis is associated with mortality in HIV-positive women and men. AIDS 2018, 32, 2393–2403. [Google Scholar] [CrossRef]
- Hudson, J.A.; Majonga, E.D.; Ferrand, R.A.; Perel, P.; Alam, S.R.; Shah, A.S.V. Association of HIV infection with cardiovascular pathology based on advanced cardiovascular imaging: A systematic review. JAMA 2022, 328, 951–962. [Google Scholar] [CrossRef]
- Detrano, R.; Guerci, A.D.; Carr, J.J.; Bild, D.E.; Burke, G.; Folsom, A.R.; Liu, K.; Shea, S.; Szklo, M.; Bluemke, D.A.; et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N. Engl. J. Med. 2008, 358, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. J. Hypertens. 2018, 36, 1953–2041. [Google Scholar] [CrossRef] [PubMed]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- ElSayed, N.A.; Aleppo, G.; Aroda, V.R.; Bannuru, R.R.; Brown, F.M.; Bruemmer, D.; Collins, B.S.; Gaglia, J.L.; Hilliard, M.E.; Isaacs, D.; et al. Classification and diagnosis of diabetes: Standards of Care in Diabetes–2023. Diabetes Care 2023, 46, S19–S40. [Google Scholar] [CrossRef]
- Agatston, A.S.; Janowitz, W.R.; Hildner, F.J.; Zusmer, N.R.; Viamonte, M., Jr.; Detrano, R. Quantification of coronary artery calcium using ultrafast computed tomography. J. Am. Coll. Cardiol. 1990, 15, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Tarr, P.E.; Ledergerber, B.; Calmy, A.; Doco-Lecompte, T.; Marzel, A.; Weber, R.; Kaufmann, P.A.; Nkoulou, R.; Buechel, R.R.; Kovari, H.; et al. Subclinical coronary artery disease in Swiss HIV-positive and HIV-negative persons. Eur. Heart J. 2018, 39, 2147–2154. [Google Scholar] [CrossRef]
- Zhou, J.; Jin, S.; Ding, H.; Liu, H.; Huo, H.; Peng, X.; Hou, Y.; Geng, W.; Shang, H.; Liu, T. Quantification of subclinical plaque characteristics and perivascular fat using coronary computed tomography angiography among individuals with HIV. Quant. Imaging Med. Surg. 2024, 14, 4675–4687. [Google Scholar] [CrossRef]
- Knudsen, A.D.; Fuchs, A.; Benfield, T.; Køber, L.; Nordestgaard, B.G.; Afzal, S.; Kuhl, J.T.; Sigvardsen, P.E.; Suarez-Zdunek, M.A.; Gelpi, M.; et al. HIV is associated with subclinical coronary atherosclerosis: A prospective matched cohort study. Clin. Infect. Dis. 2025, 81, 84–91. [Google Scholar]
- Ferrer, P.; López, L.; Pérez, J.; Cabello, N.; Núñez, M.J.; Sagastagoitia, I.; Cotarelo, M.; de Isla, L.P.; Estrada, V. Subclinical atherosclerosis burden in carotid and femoral territories in HIV subjects. BMC Infect. Dis. 2024, 24, 932. [Google Scholar] [CrossRef]
- Lembas, A.; Załęski, A.; Peller, M.; Mikuła, T.; Wiercińska-Drapało, A. Human Immunodeficiency Virus as a Risk Factor for Cardiovascular Disease. Cardiovasc. Toxicol. 2024, 24, 1–14. [Google Scholar] [CrossRef]
- Perkins, M.V.; Joseph, S.B.; Dittmer, D.P.; Mackman, N. Cardiovascular Disease and Thrombosis in HIV Infection. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Illanes-Álvarez, F.; Márquez-Ruiz, D.; Cuesta-Sancho, S.; Campaña-Gómez, I.; Martín-Aspas, A.; Tinoco-Racero, I.; Márquez-Coello, M.; Girón-González, J.A. Persistent inflammatory activation in people living with HIV. Involvement in atherosclerosis. Front. Med. 2025, 12, 1621765. [Google Scholar] [CrossRef] [PubMed]
- Moran, A.E.; Forouzanfar, M.H.; Roth, G.A.; Mensah, G.A.; Ezzati, M.; Murray, C.J.; Naghavi, M. Temporal trends in ischemic heart disease mortality in 21 world regions, 1980 to 2010. Circulation 2014, 129, 1483–1492. [Google Scholar] [CrossRef] [PubMed]
- Triant, V.A.; Perez, J.; Regan, S.; Massaro, J.M.; Meigs, J.B.; Grinspoon, S.K.; Sr D’Agostino, R.B. Cardiovascular risk prediction functions underestimate risk in HIV infection. Circulation 2018, 137, 2203–2214. [Google Scholar] [CrossRef]
- D’Ascenzo, F.; Cerrato, E.; Calcagno, A.; Grossomarra, W.; Ballocca, F.; Omede, P.; Montefusco, A.; Veglia, S.; Barbero, U.; Gili, S.; et al. High prevalence at computed coronary tomography of non-calcified plaques in asymptomatic HIV patients treated with HAART: A meta-analysis. Atherosclerosis 2015, 240, 197–204. [Google Scholar] [CrossRef]
- Soares, C.; Samara, A.; Yuyun, M.F.; Echouffo-Tcheugui, J.B.; Masri, A.; Samara, A.; Morrison, A.R.; Lin, N.; Wu, W.C.; Erqou, S. Coronary artery calcification and plaque characteristics in people living with HIV: A systematic review and meta-analysis. J. Am. Heart Assoc. 2021, 10, e019291. [Google Scholar] [CrossRef]
- Post, W.S.; Budoff, M.; Kingsley, L.; Palella, F.J., Jr.; Witt, M.D.; Li, X.; George, R.T.; Brown, T.T.; Jacobson, L.P. Associations between HIV infection and subclinical coronary atherosclerosis. Ann. Intern. Med. 2014, 160, 458–467. [Google Scholar] [CrossRef]
- Pinto-Sietsma, S.J.; Velthuis, B.K.; Nurmohamed, N.S.; Vliegenthart, R.; Martens, F.M.A.C. Computed tomography and coronary artery calcium score for screening of coronary artery disease and cardiovascular risk management in asymptomatic individuals. Neth. Heart J. 2024, 32, 371–377. [Google Scholar] [PubMed]
- Hu, X.; Zhang, Y.; Zhang, T.; Li, W.; Han, J.; Zhang, X.; Meng, F. Echocardiographic assessment of left cardiac structure and function in antiretroviral therapy–naïve people living with HIV/AIDS. Immun. Inflamm. Dis. 2023, 11, e799. [Google Scholar] [CrossRef]
- Freiberg, M.S.; Chang, C.C.H.; Skanderson, M.; Patterson, O.V.; DuVall, S.L.; Brandt, C.A.; So-Armah, K.A.; Vasan, R.S.; Oursler, K.A.; Gottdiener, J.; et al. Association between HIV infection and the risk of heart failure with reduced ejection fraction and preserved ejection fraction. JAMA Cardiol. 2017, 2, 536–546. [Google Scholar] [CrossRef]
- Doria de Vasconcellos, H.; Post, W.S.; Ervin, A.M.; Haberlen, S.A.; Budoff, M.; Malvestutto, C.; Magnani, J.W.; Feinstein, M.J.; Brown, T.T.; Lima, J.A.; et al. Associations between HIV serostatus and cardiac structure and function evaluated by echocardiography in the Multicenter AIDS Cohort Study. J. Am. Heart Assoc. 2021, 10, e019709. [Google Scholar] [CrossRef]
- Liang, K.W.; Sheu, W.H.; Lee, W.J.; Wang, J.S.; Lee, W.L. Higher fasting glucose is associated with poorer survival in non-diabetic subjects with ischemia and non-obstructive coronary arteries. Sci. Rep. 2024, 14, 20681. [Google Scholar] [PubMed]
- Dykun, I.; Bayturan, O.; Carlo, J.; Nissen, S.E.; Kapadia, S.R.; Tuzcu, E.M.; Nicholls, S.J.; Puri, R. HbA1c, coronary atheroma progression and cardiovascular outcomes. Am. J. Prev. Cardiol. 2022, 9, 100317. [Google Scholar] [CrossRef] [PubMed]
- Fernández Soto, J.; Romero-Jiménez, M.J.; Alarcón García, J.C.; Bonet Estruch, E.; Sánchez Ramos, J.L.; Castaño López, M.Á. Predictors of subclinical atherosclerosis in HIV. BMC Infect. Dis. 2023, 23, 17. [Google Scholar] [CrossRef]



| Characteristics | PLWH (n = 74) | Controls (n = 112) | p Value |
|---|---|---|---|
| Age (years), median (IQR) | 35 (31.7–37) | 34 (32–35) | 0.104 a |
| Male, n (%) | 70 (94.6) | 97 (86.6) | 0.130 b |
| Smoking, n (%) | 37 (71.2) | 33 (55.9) | 0.144 b |
| Alcohol, n (%) | 5 (9.6) | 5 (8.5) | 1 b |
| Hypertension, n (%) | 7 (9.6) | 12 (13) | 0.656 b |
| Diabetes mellitus, n (%) | 4 (5.5) | 14 (15.1) | 0.086 b |
| Dyslipidemia, n (%) | 6 (8.2) | 8 (8.8) | 1 b |
| Metabolic parameters, median (IQR) | |||
| Total cholesterol (mg/dL) | 179.5 (143–210) | 182 (160–210) | 0.357 a |
| HDL cholesterol (mg/dL) | 39.9 (34.9–48.8) | 43.4 (36.1–52.3) | 0.030 a |
| LDL cholesterol (mg/dL) | 103 (84–131.7) | 103 (82–133) | 0.939 a |
| Triglycerides (mg/dL) | 145 (76.9–194) | 128 (76.4–220) | 0.926 a |
| Hemoglobin A1c (%) | 5.3 (5.0–5.6) | 5.5 (5.1–5.8) | 0.067 a |
| Fasting blood sugar (mg/dL) | 90 (83–99) | 91.9 (85–100) | 0.148 a |
| BMI (kg/m2) | 25.1 (21.7–28) | 25.8 (23.5–29.4) | 0.065 a |
| Inflammatory parameters, median (IQR) | |||
| CRP (mg/L) | 6 (3.1–10.5) | 2.9 (1–8) | 0.025 a |
| IL-6 (pg/mL) | 5.1 (4–5.5) | 3.1 (2–4.4) | 0.003 a |
| Parameter | PLWH (n = 74) | Controls (n = 112) | p Value |
|---|---|---|---|
| Presence of coronary plaque, n (%) | 15 (20.3) | 8 (7) | 0.015 a |
| Percentage of stenosis, median (IQR) | 40 (25–52) | 45 (40–60) | 0.330 b |
| Agatston calcium score, median (IQR) | 59.5 (33.5–206.2) | 87.5 (40–189.5) | 1 b |
| Calcium volume score, median (IQR) | 69 (39.7–206.5) | 86 (44–200.2) | 0.920 b |
| Coronary stenosis > 50%, n (%) | 4 (28.6) | 4 (50) | 0.386 a |
| LV Ejection fraction, median (IQR) | 59.5 (53.7–69.5) | 62 (54–67) | 0.910 b |
| End diastolic volume, median (IQR) | 130 (115–150) | 143 (130–168) | 0.036 b |
| End systolic volume, median (IQR) | 55 (36.5–70) | 56 (38–67) | 0.733 b |
| Stroke volume, median (IQR) | 81 (71–94.5) | 88 (74–102) | 0.317 b |
| Cardiac output, median (IQR) | 5.8 (5.1–6.7) | 6.6 (5.4–7.5) | 0.079 b |
| Myocardial mass, median (IQR) | 98 (86–114) | 104 (81–122) | 0.678 b |
| Myocardial volume, median (IQR) | 93.5 (81.7–108) | 98 (78–116) | 0.538 b |
| LV/RV regurgitation, median (IQR) | 25.5 (13.5–38) | 23 (12–33) | 0.439 b |
| Plaque Present (n = 23) | No Plaque (n = 163) | p Value | |
|---|---|---|---|
| Age (years), median (IQR) | 60 (46–74) | 34 (32–36) | 0.001 a |
| Male, n (%) | 22 (95.7) | 145 (89) | 0.476 b |
| Smoking, n (%) | 8 (66.7) | 62 (62.6) | 1 b |
| Hypertension, n (%) | 5 (21.7) | 14 (9.9) | 0.149 b |
| Diabetes mellitus, n (%) | 3 (13) | 15 (10.5) | 0.719 b |
| Dyslipidemia, n (%) | 4 (14.7) | 10 (7.1) | 0.112 b |
| Hemoglobin A1c (%), median (IQR) | 5.6 (5.3–5.9) | 5.4 (5–5.7) | 0.017 a |
| Fasting blood sugar (mg/dL), median (IQR) | 100 (95–127) | 90 (83.8–96.9) | <0.001 a |
| Total cholesterol (mg/dL), median (IQR) | 195 (174–216) | 177 (148–205) | 0.080 a |
| CD4/CD8 ratio, median (IQR) | 0.72 (0.3–1.3) | 1.08 (0.74–1.5) | 0.067 a |
| CRP (mg/L), median (IQR) | 5.2 (1.2–8.4) | 4.5 (1–8) | 0.816 a |
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
| OR (95% CI) | p | aOR (95% CI) | p | |
| HIV seropositivity | ||||
| No | - | - | - | - |
| Yes | 3.305 (1.32–8.25) | 0.010 | 5.5 (1.17–25.8) | 0.030 |
| Dyslipidemia | ||||
| No | - | - | - | - |
| Yes | 2.75 (0.78–9.6) | 0.113 | 1.08 (0.17–6.8) | 0.928 |
| Age | ||||
| <35 | - | - | - | - |
| ≥35 | 23.59 (8.33–66.76) | <0.001 | 21.7 (5.3–88) | <0.001 |
| HbA1c | 1.062 (0.77–1.46) | 0.714 | 1.28 (0.73–2.23) | 0.385 |
| Fasting blood sugar | 1.008 (0.99–1.01) | 0.126 | 1 (0.98–1.02) | 0.900 |
| HDL | 1.02 (0.99–1.05) | 0.057 | 1.02 (0.98–1.06) | 0.200 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Toygar Deniz, M.; Çakır, Ö.; Acar, B.; Çakmak, C.; Balcı, S.; Akhan, S. Early Identification of Atherosclerosis in People Living with HIV by Coronary Computed Tomography Angiography. Diagnostics 2026, 16, 893. https://doi.org/10.3390/diagnostics16060893
Toygar Deniz M, Çakır Ö, Acar B, Çakmak C, Balcı S, Akhan S. Early Identification of Atherosclerosis in People Living with HIV by Coronary Computed Tomography Angiography. Diagnostics. 2026; 16(6):893. https://doi.org/10.3390/diagnostics16060893
Chicago/Turabian StyleToygar Deniz, Müge, Özgür Çakır, Burak Acar, Cemile Çakmak, Sibel Balcı, and Sıla Akhan. 2026. "Early Identification of Atherosclerosis in People Living with HIV by Coronary Computed Tomography Angiography" Diagnostics 16, no. 6: 893. https://doi.org/10.3390/diagnostics16060893
APA StyleToygar Deniz, M., Çakır, Ö., Acar, B., Çakmak, C., Balcı, S., & Akhan, S. (2026). Early Identification of Atherosclerosis in People Living with HIV by Coronary Computed Tomography Angiography. Diagnostics, 16(6), 893. https://doi.org/10.3390/diagnostics16060893







