Seasonal Variations in the Risk of Outpatient Acute Kidney Injury in Patients with Chronic Kidney Disease
Abstract
1. Introduction
2. Method
2.1. Data Source
2.2. Study Population and Cohort Definition
2.3. Outcome Measures
2.4. Statistical Analysis
3. Result
3.1. Patient Characteristics
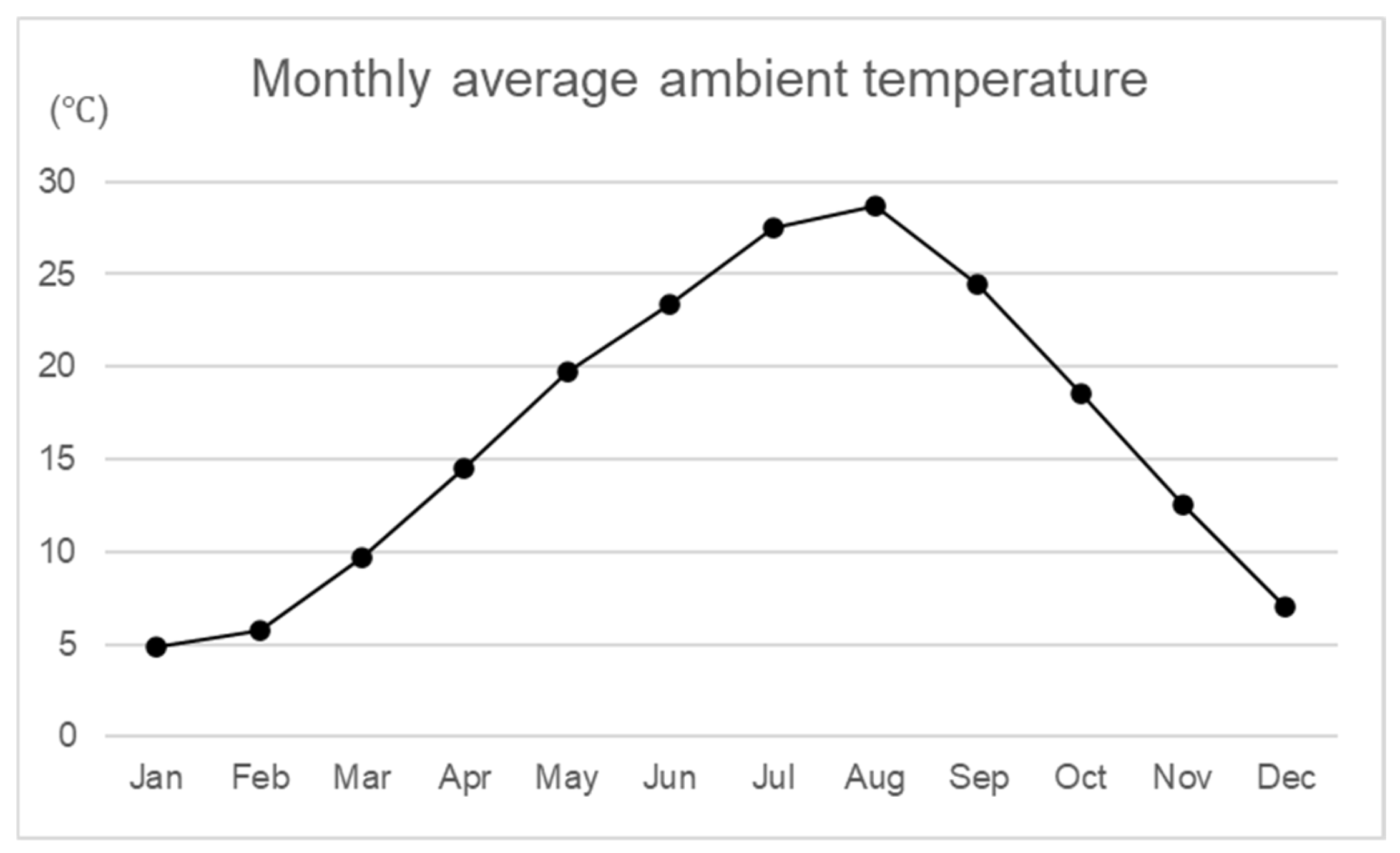
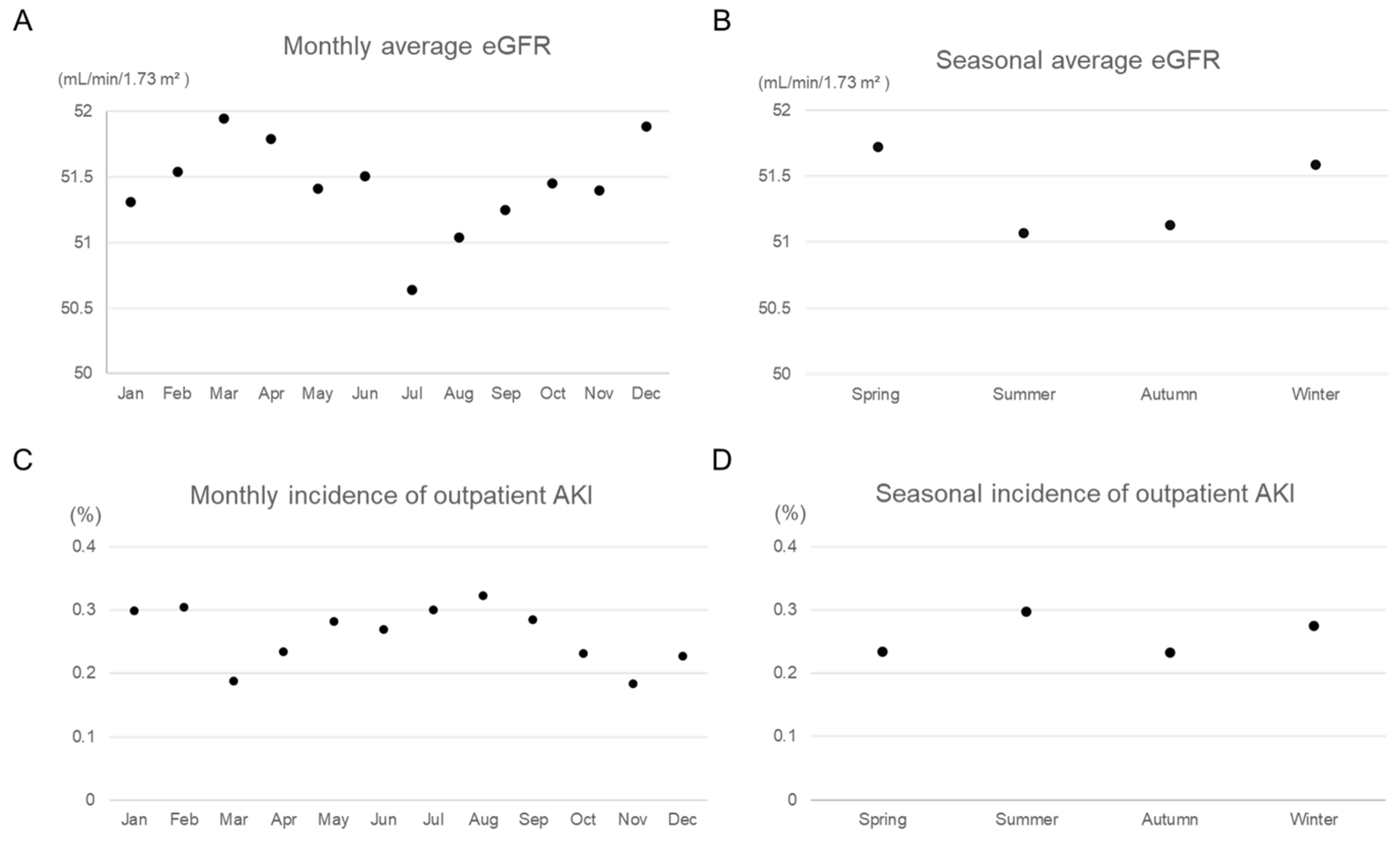
3.2. Seasonal and Monthly Variations in Outpatient AKI and Renal Parameters
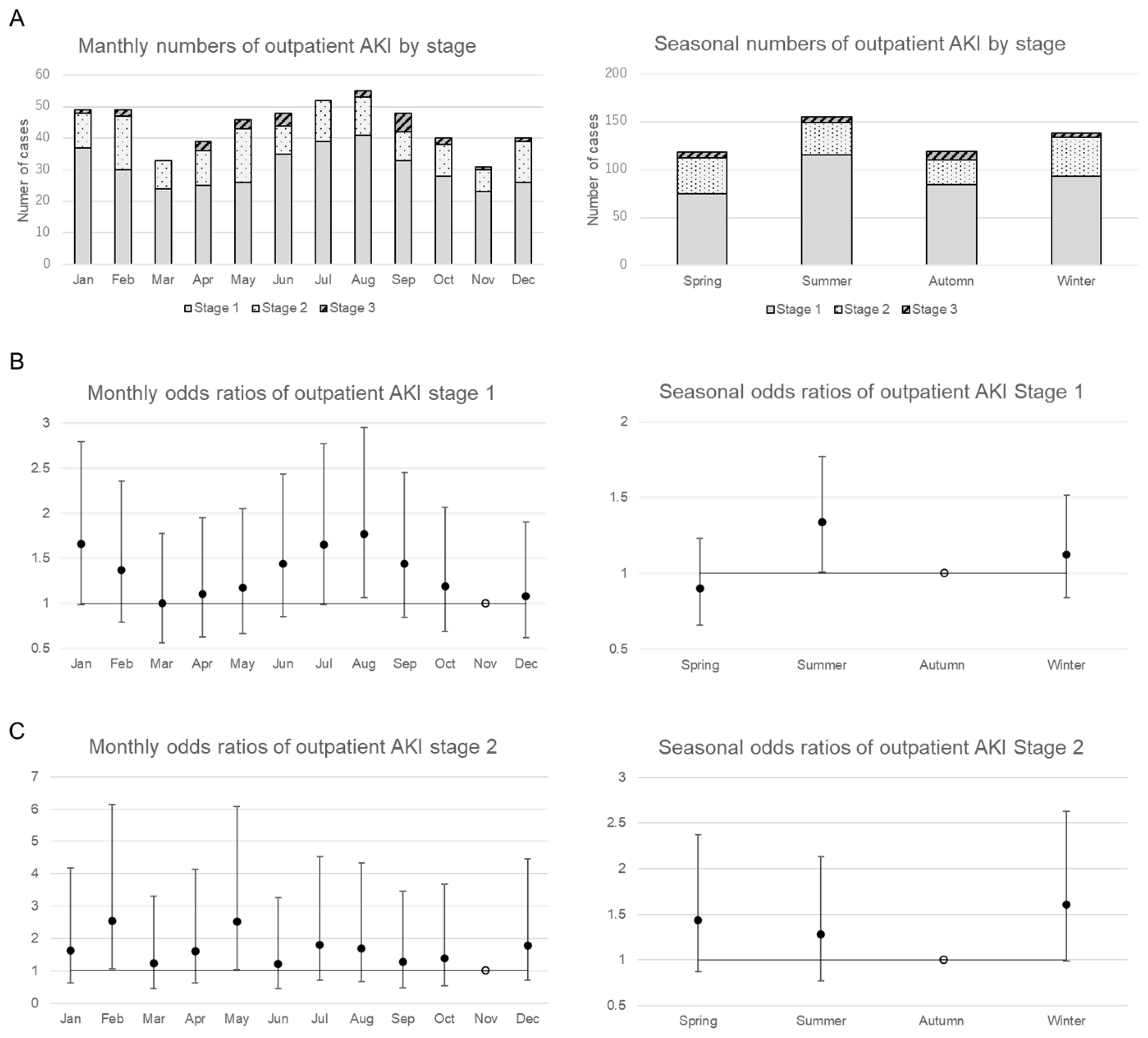
3.3. Subgroup Analyses of Outpatient AKI by AKI Stage, Age, and CKD Stage
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lameire, N.H.; Bagga, A.; Cruz, D.; de Maeseneer, J.; Endre, Z.; Kellum, J.A.; Liu, K.D.; Mehta, R.L.; Pannu, N.; Van Biesen, W.; et al. Acute kidney injury: An increasing global concern. Lancet 2013, 382, 170–179. [Google Scholar] [CrossRef]
- Hsu, R.K.; McCulloch, C.E.; Dudley, R.A.; Lo, L.J.; Hsu, C.Y. Temporal changes in incidence of dialysis-requiring AKI. J. Am. Soc. Nephrol. 2013, 24, 37–42. [Google Scholar] [CrossRef]
- Coca, S.G.; Yusuf, B.; Shlipak, M.G.; Garg, A.X.; Parikh, C.R. Long-term risk of mortality and other adverse outcomes after acute kidney injury: A systematic review and meta-analysis. Am. J. Kidney Dis. 2009, 53, 961–973. [Google Scholar] [CrossRef] [PubMed]
- Susantitaphong, P.; Cruz, D.N.; Cerda, J.; Abulfaraj, M.; Alqahtani, F.; Koulouridis, I.; Jaber, B.L. World incidence of AKI: A meta-analysis. Clin. J. Am. Soc. Nephrol. 2013, 8, 1482–1493. [Google Scholar] [CrossRef] [PubMed]
- Sawhney, S.; Mitchell, M.; Marks, A.; Fluck, N.; Black, C. Long-term prognosis after acute kidney injury (AKI): What is the role of baseline kidney function and recovery? A systematic review. BMJ Open 2015, 5, e006497. [Google Scholar] [CrossRef]
- Coca, S.G.; Singanamala, S.; Parikh, C.R. Chronic kidney disease after acute kidney injury: A systematic review and meta-analysis. Kidney Int. 2012, 81, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Collister, D.; Pannu, N.; Ye, F.; James, M.; Hemmelgarn, B.; Chui, B.; Manns, B.; Klarenbach, S.; Alberta Kidney Disease, N. Health Care Costs Associated with AKI. Clin. J. Am. Soc. Nephrol. 2017, 12, 1733–1743. [Google Scholar] [CrossRef]
- Leither, M.D.; Murphy, D.P.; Bicknese, L.; Reule, S.; Vock, D.M.; Ishani, A.; Foley, R.N.; Drawz, P.E. The impact of outpatient acute kidney injury on mortality and chronic kidney disease: A retrospective cohort study. Nephrol. Dial. Transplant. 2019, 34, 493–501. [Google Scholar] [CrossRef]
- Diamantidis, C.J.; Zepel, L.; Smith, V.A.; Brookhart, M.A.; Burks, E.; Bowling, C.B.; Maciejewski, M.L.; Wang, V. Epidemiology of Community-Acquired Acute Kidney Injury Among US Veterans. Am. J. Kidney Dis. 2023, 82, 300–310. [Google Scholar] [CrossRef]
- Holmes, J.; Rainer, T.; Geen, J.; Roberts, G.; May, K.; Wilson, N.; Williams, J.D.; Phillips, A.O.; on behalf of the Welsh AKI Steering Group. Acute Kidney Injury in the Era of the AKI E-Alert. Clin. J. Am. Soc. Nephrol. 2016, 11, 2123–2131. [Google Scholar] [CrossRef]
- Soto, K.; Campos, P.; Pinto, I.; Rodrigues, B.; Frade, F.; Papoila, A.L.; Devarajan, P. The risk of chronic kidney disease and mortality are increased after community-acquired acute kidney injury. Kidney Int. 2016, 90, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Stucker, F.; Ponte, B.; De la Fuente, V.; Alves, C.; Rutschmann, O.; Carballo, S.; Vuilleumier, N.; Martin, P.Y.; Perneger, T.; Saudan, P. Risk factors for community-acquired acute kidney injury in patients with and without chronic kidney injury and impact of its initial management on prognosis: A prospective observational study. BMC Nephrol. 2017, 18, 380. [Google Scholar] [CrossRef]
- Talabani, B.; Zouwail, S.; Pyart, R.D.; Meran, S.; Riley, S.G.; Phillips, A.O. Epidemiology and outcome of community-acquired acute kidney injury. Nephrology 2014, 19, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Yeh, H.C.; Ting, I.W.; Huang, H.C.; Chiang, H.Y.; Kuo, C.C. Acute Kidney Injury in the Outpatient Setting Associates with Risk of End-Stage Renal Disease and Death in Patients with CKD. Sci. Rep. 2019, 9, 17658. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.; Reule, S.; Vock, D.; Drawz, P. Acute Kidney Injury in the Outpatient Setting: Developing and Validating a Risk Prediction Model. Kidney Med. 2022, 4, 100376. [Google Scholar] [CrossRef]
- Ragnarsdotttir, T.H.; Kristjansdottir, M.; Gislason, G.; Sanchez-Brunete, V.; Tomasdottir, M.O.; Samuelsson, O.H.; Palsson, R.; Indridason, O.S. Prospective study of risk factors for community-acquired acute kidney injury. Eur. J. Intern. Med. 2025, 131, 83–88. [Google Scholar] [CrossRef]
- Selby, N.M. Acute kidney injury changes with the seasons. Nephrol. Dial. Transplant. 2018, 33, 1281–1283. [Google Scholar] [CrossRef]
- Iwagami, M.; Moriya, H.; Doi, K.; Yasunaga, H.; Isshiki, R.; Sato, I.; Mochida, Y.; Ishioka, K.; Ohtake, T.; Hidaka, S.; et al. Seasonality of acute kidney injury incidence and mortality among hospitalized patients. Nephrol. Dial. Transplant. 2018, 33, 1354–1362. [Google Scholar] [CrossRef]
- Phillips, D.; Young, O.; Holmes, J.; Allen, L.A.; Roberts, G.; Geen, J.; Williams, J.D.; Phillips, A.O.; On behalf of the Welsh AKI steering group. Seasonal pattern of incidence and outcome of Acute Kidney Injury: A national study of Welsh AKI electronic alerts. Int. J. Clin. Pract. 2017, 71, e13000. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024, 105, S117–S314. [CrossRef]
- KDIGO AKI Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Section 2: AKI Definition. Kidney Int. Suppl. 2012, 2, 19–36. [Google Scholar] [CrossRef]
- Japan Meteorological Agency. Monthly Climate Data for Okayama. Available online: https://www.data.jma.go.jp/stats/etrn/view/monthly_s3.php?block_no=47768&elm=monthly&prec_no=66 (accessed on 31 January 2026).
- Wong, E.; Peracha, J.; Pitcher, D.; Casula, A.; Steenkamp, R.; Medcalf, J.F.; Nitsch, D. Seasonal mortality trends for hospitalised patients with acute kidney injury across England. BMC Nephrol. 2023, 24, 144. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, G.; Gambaro, G.; Pertica, N.; Naticchia, A.; Bargagli, M.; Ferraro, P.M. Seasonality of acute kidney injury in a tertiary hospital academic center: An observational cohort study. Environ. Health 2021, 20, 8. [Google Scholar] [CrossRef]
- Li, J.; Zhou, Q.; Zhang, D.; Wang, J.; Yang, L. Seasonal variation in the detection rate and all-cause in-hospital mortality of AKI in China: A nationwide cohort study. Front. Public Health 2022, 10, 947185. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.E.; Lee, H.; Kim, J.; Lee, Y.K.; Kang, M.; Hijioka, Y.; Kim, H. Temperature as a risk factor of emergency department visits for acute kidney injury: A case-crossover study in Seoul, South Korea. Environ. Health 2019, 18, 55. [Google Scholar] [CrossRef]
- Bolt, H.; Suffel, A.; Matthewman, J.; Sandmann, F.; Tomlinson, L.; Eggo, R. Seasonality of acute kidney injury phenotypes in England: An unsupervised machine learning classification study of electronic health records. BMC Nephrol. 2023, 24, 234. [Google Scholar] [CrossRef]
- Baker, L.B. Physiology of sweat gland function: The roles of sweating and sweat composition in human health. Temperature 2019, 6, 211–259. [Google Scholar] [CrossRef]
- Chapman, C.L.; Johnson, B.D.; Parker, M.D.; Hostler, D.; Pryor, R.R.; Schlader, Z. Kidney physiology and pathophysiology during heat stress and the modification by exercise, dehydration, heat acclimation and aging. Temperature 2021, 8, 108–159. [Google Scholar] [CrossRef]
- Liu, J.; Varghese, B.M.; Hansen, A.; Borg, M.A.; Zhang, Y.; Driscoll, T.; Morgan, G.; Dear, K.; Gourley, M.; Capon, A.; et al. Hot weather as a risk factor for kidney disease outcomes: A systematic review and meta-analysis of epidemiological evidence. Sci. Total Environ. 2021, 801, 149806. [Google Scholar] [CrossRef]
- Lim, Y.H.; So, R.; Lee, C.; Hong, Y.C.; Park, M.; Kim, L.; Yoon, H.J. Ambient temperature and hospital admissions for acute kidney injury: A time-series analysis. Sci. Total Environ. 2018, 616–617, 1134–1138. [Google Scholar] [CrossRef]
- McTavish, R.K.; Richard, L.; McArthur, E.; Shariff, S.Z.; Acedillo, R.; Parikh, C.R.; Wald, R.; Wilk, P.; Garg, A.X. Association Between High Environmental Heat and Risk of Acute Kidney Injury Among Older Adults in a Northern Climate: A Matched Case-Control Study. Am. J. Kidney Dis. 2018, 71, 200–208. [Google Scholar] [CrossRef]
- Kario, K. Caution for winter morning surge in blood pressure: A possible link with cardiovascular risk in the elderly. Hypertension 2006, 47, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, S.; Wang, C.; Wang, B.; Guo, P. Effects of moderate strength cold air exposure on blood pressure and biochemical indicators among cardiovascular and cerebrovascular patients. Int. J. Environ. Res. Public Health 2014, 11, 2472–2487. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.N.; Shin, M.K.; Lim, Y.H.; Bae, S.; Kim, J.H.; Hwang, S.S.; Kim, M.J.; Oh, J.; Lim, H.; Choi, J.; et al. Associations of cold exposure with hospital admission and mortality due to acute kidney injury: A nationwide time-series study in Korea. Sci. Total Environ. 2023, 863, 160960. [Google Scholar] [CrossRef] [PubMed]
- De Giorgi, A.; Storari, A.; Rodriguez-Munoz, P.M.; Cappadona, R.; Lamberti, N.; Manfredini, F.; Lopez-Soto, P.J.; Manfredini, R.; Fabbian, F. Seasonal pattern in elderly hospitalized with acute kidney injury: A retrospective nationwide study in Italy. Int. Urol. Nephrol. 2022, 54, 3243–3253. [Google Scholar] [CrossRef]
- Mohammad, K.N.; Chan, E.Y.Y.; Lau, S.Y.; Lam, H.C.Y.; Goggins, W.B.; Chong, K.C. Relationship between acute kidney injury, seasonal influenza, and environmental factors: A 14-year retrospective analysis. Environ. Int. 2021, 153, 106521. [Google Scholar] [CrossRef]
- Shih, C.H.; Lee, Y.J.; Chao, P.W.; Kuo, S.C.; Ou, S.M.; Huang, H.M.; Chen, Y.T. Association between influenza vaccination and the reduced risk of acute kidney injury among older people: A nested case-control study. Eur. J. Intern. Med. 2018, 54, 65–69. [Google Scholar] [CrossRef]
- Yoo, Y.J.; Wilkins, K.J.; Alakwaa, F.; Liu, F.; Torre-Healy, L.A.; Krichevsky, S.; Hong, S.S.; Sakhuja, A.; Potu, C.K.; Saltz, J.H.; et al. Geographic and Temporal Trends in COVID-Associated Acute Kidney Injury in the National COVID Cohort Collaborative. Clin. J. Am. Soc. Nephrol. 2023, 18, 1006–1018. [Google Scholar] [CrossRef]
- Chan, L.; Chaudhary, K.; Saha, A.; Chauhan, K.; Vaid, A.; Zhao, S.; Paranjpe, I.; Somani, S.; Richter, F.; Miotto, R.; et al. AKI in Hospitalized Patients with COVID-19. J. Am. Soc. Nephrol. 2021, 32, 151–160. [Google Scholar] [CrossRef]
- Pan, Y.; Han, Y.; Zhou, C.; Zheng, J.; Zhao, L.; Ye, X.; He, Y.; Consortium, N.C. Assessing acute kidney injury risk after COVID vaccination and infection in a large cohort study. NPJ Vaccines 2024, 9, 213. [Google Scholar] [CrossRef]
- Tsai, P.Y.; Wang, Y.H.; Huang, J.Y.; Yang, S.F.; Wu, S.W. Risk of acute kidney injury and mortality in patients vaccinated against COVID-19. Int. J. Med. Sci. 2025, 22, 4483–4492. [Google Scholar] [CrossRef]
- Mehta, K.; Pajai, A.; Bhurke, S.; Shirkande, A.; Bhadade, R.; D’Souza, R. Acute Kidney Injury of Infectious Etiology in Monsoon Season: A Prospective Study Using Acute Kidney Injury Network Criteria. Indian. J. Nephrol. 2018, 28, 143–152. [Google Scholar] [CrossRef]
- Kupferman, J.; Ramirez-Rubio, O.; Amador, J.J.; Lopez-Pilarte, D.; Wilker, E.H.; Laws, R.L.; Sennett, C.; Robles, N.V.; Lau, J.L.; Salinas, A.J.; et al. Acute Kidney Injury in Sugarcane Workers at Risk for Mesoamerican Nephropathy. Am. J. Kidney Dis. 2018, 72, 475–482. [Google Scholar] [CrossRef]
- Johnson, R.J.; Wesseling, C.; Newman, L.S. Chronic Kidney Disease of Unknown Cause in Agricultural Communities. N. Engl. J. Med. 2019, 380, 1843–1852. [Google Scholar] [CrossRef]




| Total Number | n = 203,853 | n (%) |
|---|---|---|
| Sex | ||
| male | 117,490 | 57.6% |
| female | 86,363 | 42.3% |
| Age category (years) | ||
| <65 years | 94,056 | 46.1% |
| 65–74 years | 62,166 | 30.4% |
| ≥75 years | 47,631 | 23.3% |
| CKD stage | ||
| Stage 2 (60 ≤ eGFR < 90) | 76,215 | 17.7% |
| Stage 3 (30 ≤ eGFR < 60) | 91,544 | 44.9% |
| Stage 4 or 5 (eGFR < 30) | 36,094 | 37.3% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nakanoh, H.; Tsuji, K.; Fukushima, K.; Uchida, N.; Haraguchi, S.; Kitamura, S.; Wada, J. Seasonal Variations in the Risk of Outpatient Acute Kidney Injury in Patients with Chronic Kidney Disease. Diagnostics 2026, 16, 845. https://doi.org/10.3390/diagnostics16060845
Nakanoh H, Tsuji K, Fukushima K, Uchida N, Haraguchi S, Kitamura S, Wada J. Seasonal Variations in the Risk of Outpatient Acute Kidney Injury in Patients with Chronic Kidney Disease. Diagnostics. 2026; 16(6):845. https://doi.org/10.3390/diagnostics16060845
Chicago/Turabian StyleNakanoh, Hiroyuki, Kenji Tsuji, Kazuhiko Fukushima, Naruhiko Uchida, Soichiro Haraguchi, Shinji Kitamura, and Jun Wada. 2026. "Seasonal Variations in the Risk of Outpatient Acute Kidney Injury in Patients with Chronic Kidney Disease" Diagnostics 16, no. 6: 845. https://doi.org/10.3390/diagnostics16060845
APA StyleNakanoh, H., Tsuji, K., Fukushima, K., Uchida, N., Haraguchi, S., Kitamura, S., & Wada, J. (2026). Seasonal Variations in the Risk of Outpatient Acute Kidney Injury in Patients with Chronic Kidney Disease. Diagnostics, 16(6), 845. https://doi.org/10.3390/diagnostics16060845







