Layer-Specific Architecture and Nerve Innervation of the Popliteus Muscle: Neuroanatomical Basis for Precision-Guided Interventions for the Knee Joint
Abstract
1. Introduction
2. Materials and Methods
2.1. Dissection and Morphological Analysis of the Popliteus Muscle
2.2. Modified Sihler’s Staining
2.3. Ultrasound Examination
2.4. Histological Analysis: ATPase Staining
3. Results
3.1. Morphological Characteristics of the Popliteus Muscle
3.2. Innervation of the Popliteus Muscle
3.3. Intramuscular Nerve Distribution (Modified Sihler’s Staining)
3.4. Topographic Relationship with Nerve to Tibialis Posterior
3.5. Ultrasound Scanning Analysis
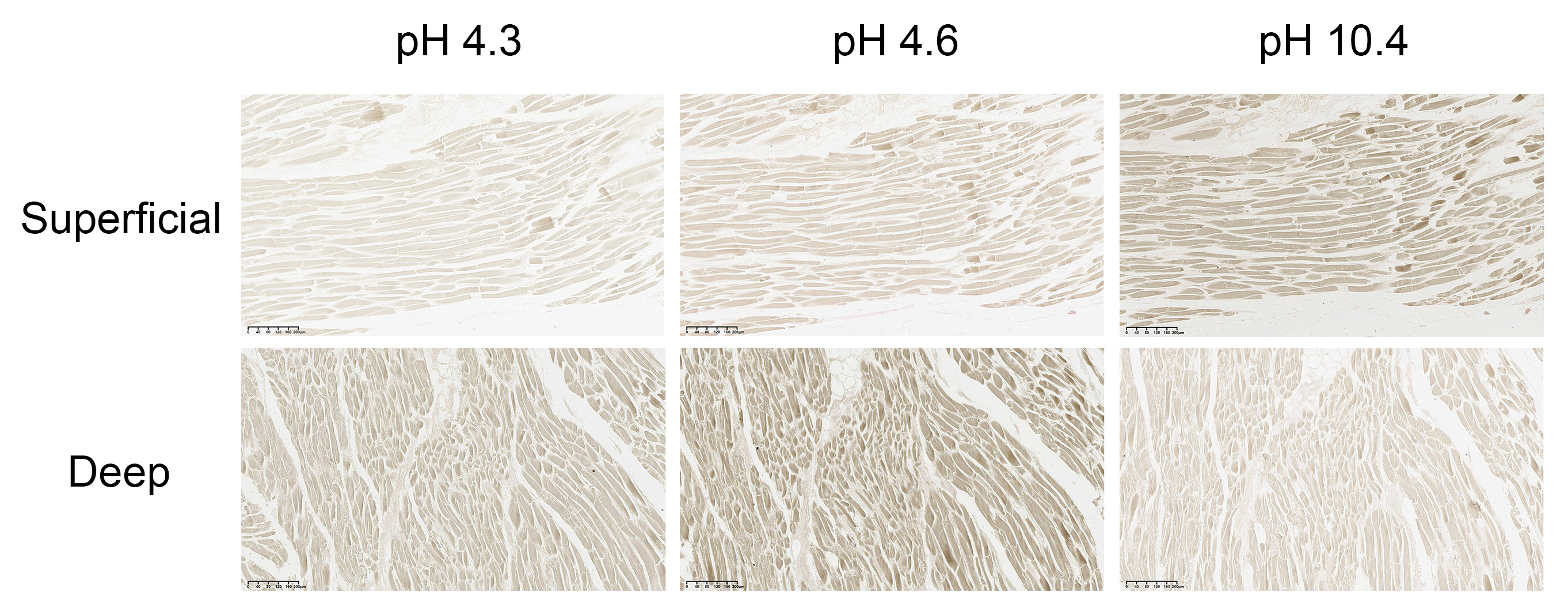
3.6. Histological Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BoNT | Botulinum toxin |
| PCL | Posterior cruciate ligament |
| PLC | Posterolateral complex |
| PM | Popliteus muscle |
| TP | Tibialis posterior |
References
- Grood, E.S.; Stowers, S.F.; Noyes, F.R. Limits of movement in the human knee. Effect of sectioning the posterior cruciate ligament and posterolateral structures. J. Bone Jt. Surg. 1988, 70, 88–97. [Google Scholar] [CrossRef]
- LaPrade, R.F.; Wozniczka, J.K.; Stellmaker, M.P.; Wijdicks, C.A. Analysis of the static function of the popliteus tendon and evaluation of an anatomic reconstruction: The “fifth ligament” of the knee. Am. J. Sports Med. 2010, 38, 543–549. [Google Scholar] [CrossRef]
- Shahane, S.A.; Ibbotson, C.; Strachan, R.; Bickerstaff, D.R. The popliteofibular ligament. An anatomical study of the posterolateral corner of the knee. J. Bone Jt. Surg. Br. 1999, 81, 636–642. [Google Scholar] [CrossRef] [PubMed]
- Zabrzyński, J.; Huri, G.; Yataganbaba, A.; Paczesny, Ł.; Szwedowski, D.; Zabrzyńska, A.; Łapaj, Ł.; Gagat, M.; Wiśniewski, M.; Pękala, P. Current concepts on the morphology of popliteus tendon and its clinical implications. Folia Morphol. 2021, 80, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Paraskevas, G.; Papaziogas, B.; Kitsoulis, P.; Spanidou, S. A study on the morphology of the popliteus muscle and arcuate popliteal ligament. Folia Morphol. 2006, 65, 381–384. [Google Scholar]
- Olewnik, Ł.; LaPrade, R.F.; Paulsen, F.; Gonera, B.; Kurtys, K.; Podgórski, M.; Aragonés, P.; Sanudo, J.R.; Polguj, M. A proposal for a new morphological classification of the popliteus muscle tendon with potential clinical and biomechanical significance. Sci. Rep. 2021, 11, 14434. [Google Scholar] [CrossRef]
- Ullrich, K.; Krudwig, W.K.; Witzel, U. Posterolateral aspect and stability of the knee joint. I. Anatomy and function of the popliteus muscle-tendon unit: An anatomical and biomechanical study. Knee Surg. Sports Traumatol. Arthrosc. 2002, 10, 86–90. [Google Scholar] [CrossRef]
- Pasque, C.; Noyes, F.R.; Gibbons, M.; Levy, M.; Grood, E. The role of the popliteofibular ligament and the tendon of popliteus in providing stability in the human knee. J. Bone Jt. Surg. Br. 2003, 85, 292–298. [Google Scholar] [CrossRef]
- Lephart, S.M.; Pincivero, D.M.; Rozzi, S.L. Proprioception of the ankle and knee. Sports Med. 1998, 25, 149–155. [Google Scholar] [CrossRef]
- Nyland, J.; Lachman, N.; Kocabey, Y.; Brosky, J.; Altun, R.; Caborn, D. Anatomy, function, and rehabilitation of the popliteus musculotendinous complex. J. Orthop. Sports Phys. Ther. 2005, 35, 165–179. [Google Scholar] [CrossRef]
- Kato, T.; Sasaki, A.; Nakazawa, K. Short-and long-latency afferent inhibition of the human leg motor cortex by h-reflex subthreshold electrical stimulation at the popliteal fossa. Exp. Brain Res. 2023, 241, 249–261. [Google Scholar] [CrossRef]
- Sass, P.; Hassan, G. Lower extremity abnormalities in children. Am. Fam. Physician 2003, 68, 461–468. [Google Scholar]
- Kozanek, M.; Fu, E.C.; Van de Velde, S.K.; Gill, T.J.; Li, G. Posterolateral structures of the knee in posterior cruciate ligament deficiency. Am. J. Sports Med. 2009, 37, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.F.; Condon, S.M.; de Lateur, B.J.; Smith, J.C. Gait abnormalities in tibial nerve paralysis: A biomechanical study. Arch. Phys. Med. Rehabil. 1985, 66, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Somanath, D.; Ramalingam, S. A comprehensive review of the anatomy of popliteus and its clinico-surgical relevance. J. Orthop. Traumatol. Rehabil. 2020, 12, 107–114. [Google Scholar]
- Podore, P.C. Popliteal entrapment syndrome: A report of tibial nerve entrapment. J. Vasc. Surg. 1985, 2, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Mastaglia, F.L. Tibial nerve entrapment in the popliteal fossa. Muscle Nerve 2000, 23, 1883–1886. [Google Scholar] [CrossRef]
- Cho, K.J.; Kang, S.; Ko, S.; Baek, J.; Kim, Y.; Park, N.K. Neurovascular compression caused by popliteus muscle enlargement without discrete trauma. Ann. Rehabil. Med. 2016, 40, 545–550. [Google Scholar] [CrossRef]
- Blake, S.M.; Treble, N.J. Popliteus tendon tenosynovitis. Br. J. Sports Med. 2005, 39, e42, discussion e42. [Google Scholar] [CrossRef]
- Gracies, J.M.; Lugassy, M.; Weisz, D.J.; Vecchio, M.; Flanagan, S.; Simpson, D.M. Botulinum toxin dilution and endplate targeting in spasticity: A double-blind controlled study. Arch. Phys. Med. Rehabil. 2009, 90, 9–16.e12. [Google Scholar] [CrossRef] [PubMed]
- Van Campenhout, A.; Molenaers, G. Localization of the motor endplate zone in human skeletal muscles of the lower limb: Anatomical guidelines for injection with botulinum toxin. Dev. Med. Child Neurol. 2011, 53, 108–119. [Google Scholar] [CrossRef]
- Arezzo, J.C. Possible mechanisms for the effects of botulinum toxin on pain. Clin. J. Pain 2002, 18, S125–S132. [Google Scholar] [CrossRef]
- Qerama, E.; Fuglsang-Frederiksen, A.; Jensen, T.S. The role of botulinum toxin in management of pain: An evidence-based review. Curr. Opin. Anaesthesiol. 2010, 23, 602–610. [Google Scholar] [CrossRef]
- Cohn, A.K.; Mains, D.B. Popliteal hiatus of the lateral meniscus. Anatomy and measurement at dissection of 10 specimens. Am. J. Sports Med. 1979, 7, 221–226. [Google Scholar] [CrossRef]
- Patel, D. Proximal approaches to arthroscopic surgery of the knee. Am. J. Sports Med. 1981, 9, 296–303. [Google Scholar] [CrossRef]
- Chahla, J.; James, E.W.; Cinque, M.E.; LaPrade, R.F. Midterm outcomes following anatomic-based popliteus tendon reconstructions. Knee Surg. Sports Traumatol. Arthrosc. 2018, 26, 812–818. [Google Scholar] [CrossRef]
- Jeon, A.; Kim, Y.G.; Sohn, Y.; Lee, J.H. Nerve and arterial supply pattern of the popliteus muscle and clinical implications. Biomed. Res. Int. 2022, 2022, 6980471. [Google Scholar] [CrossRef] [PubMed]
- Terry, G.C.; LaPrade, R.F. The posterolateral aspect of the knee. Anatomy and surgical approach. Am. J. Sports Med. 1996, 24, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Takeda, S.; Tajima, G.; Fujino, K.; Yan, J.; Kamei, Y.; Maruyama, M.; Kikuchi, S.; Doita, M. Morphology of the femoral insertion of the lateral collateral ligament and popliteus tendon. Knee Surg. Sports Traumatol. Arthrosc. 2015, 23, 3049–3054. [Google Scholar] [CrossRef]
- Iwanaga, J.; Singh, V.; Takeda, S.; Ogeng’o, J.; Kim, H.J.; Moryś, J.; Ravi, K.S.; Ribatti, D.; Trainor, P.A.; Sañudo, J.R.; et al. Standardized statement for the ethical use of human cadaveric tissues in anatomy research papers: Recommendations from anatomical journal editors-in-chief. Clin. Anat. 2022, 35, 526–528. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, H.J.; Yi, K.H.; Lee, K.W.; Gil, Y.C.; Kim, H.J. Ideal injection points for botulinum neurotoxin for pectoralis minor syndrome: A cadaveric study. Toxins 2023, 15, 603. [Google Scholar] [CrossRef]
- Mu, L.; Sanders, I. Sihler’s whole mount nerve staining technique: A review. Biotech. Histochem. 2010, 85, 19–42. [Google Scholar] [CrossRef]
- Guth, L.; Samaha, F.J. Procedure for the histochemical demonstration of actomyosin atpase. Exp. Neurol. 1970, 28, 365–367. [Google Scholar] [CrossRef] [PubMed]
- Lim, A.Y.; Pereira, B.P.; Kumar, V.P.; De Coninck, C.; Taki, C.; Baudet, J.; Merle, M. Intramuscular innervation of upper-limb skeletal muscles. Muscle Nerve 2004, 29, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Adidharma, W.; Khouri, A.N.; Lee, J.C.; Vanderboll, K.; Kung, T.A.; Cederna, P.S.; Kemp, S.W.P. Sensory nerve regeneration and reinnervation in muscle following peripheral nerve injury. Muscle Nerve 2022, 66, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Torell, F.; Dimitriou, M. Local muscle pressure stimulates the principal receptors for proprioception. Cell Rep. 2024, 43, 114699. [Google Scholar] [CrossRef]
- Korfage, J.A.; Koolstra, J.H.; Langenbach, G.E.; van Eijden, T.M. Fiber-type composition of the human jaw muscles—(part 1) origin and functional significance of fiber-type diversity. J. Dent. Res. 2005, 84, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Ledoux, W.R.; Hirsch, B.E.; Church, T.; Caunin, M. Pennation angles of the intrinsic muscles of the foot. J. Biomech. 2001, 34, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Sopher, R.S.; Amis, A.A.; Davies, D.C.; Jeffers, J.R. The influence of muscle pennation angle and cross-sectional area on contact forces in the ankle joint. J. Strain Anal. Eng. Des. 2017, 52, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Van Hooren, B.; Aagaard, P.; Monte, A.; Blazevich, A.J. The role of pennation angle and architectural gearing to rate of force development in dynamic and isometric muscle contractions. Scand. J. Med. Sci. Sports 2024, 34, e14639. [Google Scholar] [CrossRef]
- Schinhan, M.; Bijak, M.; Unger, E.; Nau, T. Electromyographic study of the popliteus muscle in the dynamic stabilization of the posterolateral corner structures of the knee. Am. J. Sports Med. 2011, 39, 173–179. [Google Scholar] [CrossRef]
- Rockenfeller, R.; Günther, M.; Clemente, C.J.; Dick, T.J.M. Rethinking the physiological cross-sectional area of skeletal muscle reveals the mechanical advantage of pennation. R. Soc. Open Sci. 2024, 11, 240037. [Google Scholar] [CrossRef]
- Dick, T.J.M.; Wakeling, J.M. Shifting gears: Dynamic muscle shape changes and force-velocity behavior in the medial gastrocnemius. J. Appl. Physiol. 2017, 123, 1433–1442. [Google Scholar] [CrossRef]
- Maltin, C.A. Muscle development and obesity: Is there a relationship? Organogenesis 2008, 4, 158–169. [Google Scholar] [CrossRef]
- Dong, H.; Tsai, S.Y. Mitochondrial properties in skeletal muscle fiber. Cells 2023, 12, 2183. [Google Scholar] [CrossRef]
- Watanabe, Y.; Moriya, H.; Takahashi, K.; Yamagata, M.; Sonoda, M.; Shimada, Y.; Tamaki, T. Functional anatomy of the posterolateral structures of the knee. Arthroscopy 1993, 9, 57–62. [Google Scholar] [CrossRef]
- De Maeseneer, M.; Shahabpour, M.; Vanderdood, K.; De Ridder, F.; Van Roy, F.; Osteaux, M. Posterolateral supporting structures of the knee: Findings on anatomic dissection, anatomic slices and mr images. Eur. Radiol. 2001, 11, 2170–2177. [Google Scholar] [CrossRef]
- Krudwig, W.K.; Witzel, U.; Ullrich, K. Posterolateral aspect and stability of the knee joint. II. Posterolateral instability and effect of isolated and combined posterolateral reconstruction on knee stability: A biomechanical study. Knee Surg. Sports Traumatol. Arthrosc. 2002, 10, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Brinkman, J.M.; Schwering, P.J.; Blankevoort, L.; Kooloos, J.G.; Luites, J.; Wymenga, A.B. The insertion geometry of the posterolateral corner of the knee. J. Bone Jt. Surg. Br. 2005, 87, 1364–1368. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, S.P.; More, S.R.; Riascos, R.F.; Lemos, D.F.; Swischuk, L.E. Comprehensive review of the anatomy, function, and imaging of the popliteus and associated pathologic conditions. Radiographics 2014, 34, 496–513. [Google Scholar] [CrossRef] [PubMed]
- Hopwood, H.J.; Bellinger, P.M.; Compton, H.R.; Bourne, M.N.; Minahan, C. The relevance of muscle fiber type to physical characteristics and performance in team-sport athletes. Int. J. Sports Physiol. Perform. 2023, 18, 223–230. [Google Scholar] [CrossRef]
- LaPrade, R.F.; Ly, T.V.; Wentorf, F.A.; Engebretsen, L. The posterolateral attachments of the knee: A qualitative and quantitative morphologic analysis of the fibular collateral ligament, popliteus tendon, popliteofibular ligament, and lateral gastrocnemius tendon. Am. J. Sports Med. 2003, 31, 854–860. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.G.; Kim, J.H.; Ha, J.K. The anatomy of the posterior and posterolateral corner of the knee. J. Korean Orthop. Assoc. 2005, 40, 70–75. [Google Scholar] [CrossRef]
- Song, Y.; Xiong, Y.; Yao, J.; Wang, H.; Tan, L.; Hu, X.; Zhang, S.; Wu, Y. Applied anatomy and three-dimensional visualization of the tendon-bone junctions of the knee joint posterolateral complex. Ann. Anat. 2020, 229, 151413. [Google Scholar] [CrossRef] [PubMed]
- Gibson, M.H.; Mills, J.G.; Johnson, G.E.; Downs, A.R. Popliteal entrapment syndrome. Ann. Surg. 1977, 185, 341–348. [Google Scholar] [CrossRef]
- Levien, L.J.; Veller, M.G. Popliteal artery entrapment syndrome: More common than previously recognized. J. Vasc. Surg. 1999, 30, 587–598. [Google Scholar] [CrossRef]
- Sinha, S.; Houghton, J.; Holt, P.J.; Thompson, M.M.; Loftus, I.M.; Hinchliffe, R.J. Popliteal entrapment syndrome. J. Vasc. Surg. 2012, 55, 252–262.e230. [Google Scholar] [CrossRef]
- Gandor, F.; Tisch, S.; Grabs, A.J.; Delaney, A.J.; Bester, L.; Darveniza, P. Botulinum toxin a in functional popliteal entrapment syndrome: A new approach to a difficult diagnosis. J. Neural. Transm. 2014, 121, 1297–1301. [Google Scholar] [CrossRef]
- Isner-Horobeti, M.E.; Muff, G.; Masat, J.; Daussin, J.L.; Dufour, S.P.; Lecocq, J. Botulinum toxin as a treatment for functional popliteal artery entrapment syndrome. Med. Sci. Sports Exerc. 2015, 47, 1124–1127. [Google Scholar] [CrossRef]
- Li, Y.H.; Leong, J.C. Intoeing gait in children. Hong Kong Med. J. 1999, 5, 360–366. [Google Scholar] [PubMed]
- Quinlan, J.F.; Webb, S.; McDonald, K.; Meikle, G.; McMahon, S.F. Isolated popliteus rupture at the musculo-tendinous junction. J. Knee Surg. 2011, 24, 137–140. [Google Scholar] [CrossRef]
- Davalos, E.A.; Barank, D.; Varma, R.K. Two cases of chronic knee pain caused by unusual injuries to the popliteus tendon. Joints 2016, 4, 62–64. [Google Scholar] [CrossRef][Green Version]
- Yi, K.H.; Lee, J.H.; Kim, G.Y.; Yoon, S.W.; Oh, W.; Kim, H.J. Novel anatomical proposal for botulinum neurotoxin injection targeting lateral canthal rhytids. Toxins 2022, 14, 462. [Google Scholar] [CrossRef]
- Kojovic, M.; Caronni, A.; Bologna, M.; Rothwell, J.C.; Bhatia, K.P.; Edwards, M.J. Botulinum toxin injections reduce associative plasticity in patients with primary dystonia. Mov. Disord. 2011, 26, 1282–1289. [Google Scholar] [CrossRef]
- Luvisetto, S. Botulinum toxin and neuronal regeneration after traumatic injury of central and peripheral nervous system. Toxins 2020, 12, 434. [Google Scholar] [CrossRef]
- Ohishi, T.; Takahashi, M.; Suzuki, D.; Matsuyama, Y. Arthroscopic approach to the posterior compartment of the knee using a posterior transseptal portal. World J. Orthop. 2015, 6, 505–512. [Google Scholar] [CrossRef]
- Frings, J.; Weiß, S.; Kolb, J.; Behrendt, P.; Frosch, K.H.; Krause, M. Arthroscopic anatomy of the posterolateral corner of the knee: Anatomic relations and arthroscopic approaches. Arch. Orthop. Trauma Surg. 2022, 142, 443–453. [Google Scholar] [CrossRef]
- Lapatki, B.G.; van Dijk, J.P.; van de Warrenburg, B.P.; Zwarts, M.J. Botulinum toxin has an increased effect when targeted toward the muscle’s endplate zone: A high-density surface emg guided study. Clin. Neurophysiol. 2011, 122, 1611–1616. [Google Scholar] [CrossRef]
- Kim, M.; Yeo, I.S.; Cho, T.H.; Hong, J.E.; Kim, S.H.; Yang, H.M. Peripheral cranio-spinal nerve communication for trapezius muscle control using axonal profiling through immunostaining. Sci. Rep. 2024, 14, 25266. [Google Scholar] [CrossRef]
- Iwanaga, J.; Singh, V.; Ohtsuka, A.; Hwang, Y.; Kim, H.J.; Moryś, J.; Ravi, K.S.; Ribatti, D.; Trainor, P.A.; Sañudo, J.R.; et al. Acknowledging the use of human cadaveric tissues in research papers: Recommendations from anatomical journal editors. Clin. Anat. 2021, 34, 2–4. [Google Scholar] [CrossRef]




| Sex | Side | Total (n = 32) | |||||
|---|---|---|---|---|---|---|---|
| Male (n = 14) | Female (n = 18) | p-Value | Left (n = 16) | Right (n = 16) | p-Value | ||
| Vertical length (a) | 109.63 ± 11.32 | 102.36 ± 13.66 | 0.16 | 105.78 ± 14.25 | 103.92 ± 12.61 | 0.57 | 104.78 ± 13.20 |
| Horizontal length (b) | 54.60 ± 4.64 | 49.76 ± 2.75 | 0.01 | 50.37 ± 3.90 | 52.26 ± 4.24 | 0.27 | 51.38 ± 4.13 |
| Length of insertion (c) | 68.66 ± 6.18 | 64.37 ± 13.17 | 0.22 | 64.43 ± 10.57 | 67.34 ± 11.79 | 0.42 | 65.98 ± 11.15 |
| Superior border (d) | 72.66 ± 9.29 | 63.31 ± 5.01 | 0.00 | 66.94 ± 9.59 | 66.70 ± 7.04 | 0.89 | 66.82 ± 8.19 |
| Inferior border (e) | 117.68 ± 10.50 | 107.20 ± 9.67 | 0.00 | 113.54 ± 13.32 | 109.01 ± 8.53 | 0.12 | 111.13 ± 11.09 |
| Angle (f) | 63.82 ± 5.77 | 64.96 ± 4.78 | 0.54 | 63.71 ± 3.80 | 65.26 ± 6.07 | 0.19 | 64.53 ± 5.11 |
| X-Axis (mm) | Y-Axis (mm) | Location (%) | |
|---|---|---|---|
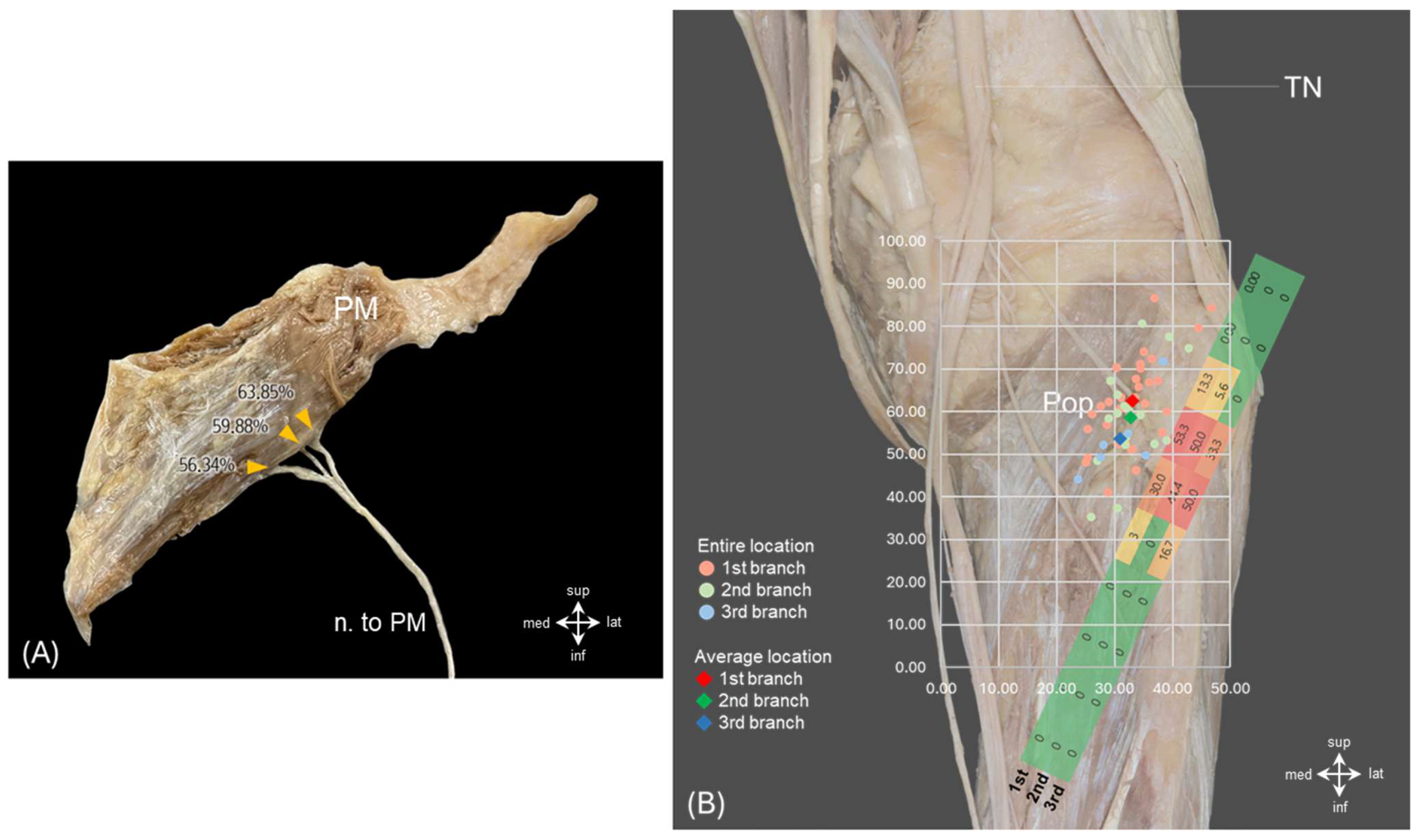
| n. to pop | |||
| 1st | 19.1 ± 4.8 | 28.5 ± 1.6 | 63.9 ± 6.7 |
| 2nd | 20.8 ± 4.4 | 35.7 ± 3.8 | 59.9 ± 5.8 |
| 3rd | 23.2 ± 4.5 | 37.1 ± 4.8 | 56.3 ± 7.8 |
| mean | 20.4 ± 4.4 | 32.7 ± 4.9 | 61.7 ± 6.9 |
| n. to tp | 25.8 ± 2.6 | 41.6 ± 5.2 | 51.3 ± 3.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kim, S.-J.; Lee, J.-H.; Yeo, I.-S. Layer-Specific Architecture and Nerve Innervation of the Popliteus Muscle: Neuroanatomical Basis for Precision-Guided Interventions for the Knee Joint. Diagnostics 2026, 16, 834. https://doi.org/10.3390/diagnostics16060834
Kim S-J, Lee J-H, Yeo I-S. Layer-Specific Architecture and Nerve Innervation of the Popliteus Muscle: Neuroanatomical Basis for Precision-Guided Interventions for the Knee Joint. Diagnostics. 2026; 16(6):834. https://doi.org/10.3390/diagnostics16060834
Chicago/Turabian StyleKim, Soo-Jung, Ji-Hyun Lee, and In-Seung Yeo. 2026. "Layer-Specific Architecture and Nerve Innervation of the Popliteus Muscle: Neuroanatomical Basis for Precision-Guided Interventions for the Knee Joint" Diagnostics 16, no. 6: 834. https://doi.org/10.3390/diagnostics16060834
APA StyleKim, S.-J., Lee, J.-H., & Yeo, I.-S. (2026). Layer-Specific Architecture and Nerve Innervation of the Popliteus Muscle: Neuroanatomical Basis for Precision-Guided Interventions for the Knee Joint. Diagnostics, 16(6), 834. https://doi.org/10.3390/diagnostics16060834







