Diagnostic Value of Serum VEGF-D in Lymphangioleiomyomatosis: Results of the First Prospective Study in the Russian Federation
Abstract
1. Introduction
1.1. Pathogenesis
1.2. Serum VEGF-D as a Biomarker
1.3. Studies on LAM and the Diagnostic Utility of VEGF-D
2. Materials and Methods
2.1. Study Design and Participants
2.2. Case Definition and Clinical Data Collection
2.3. Radiological Assessment
2.4. Biospecimen Collection and Preanalytical Handling
2.5. VEGF-D Measurement
2.6. Data Management and Statistical Analysis
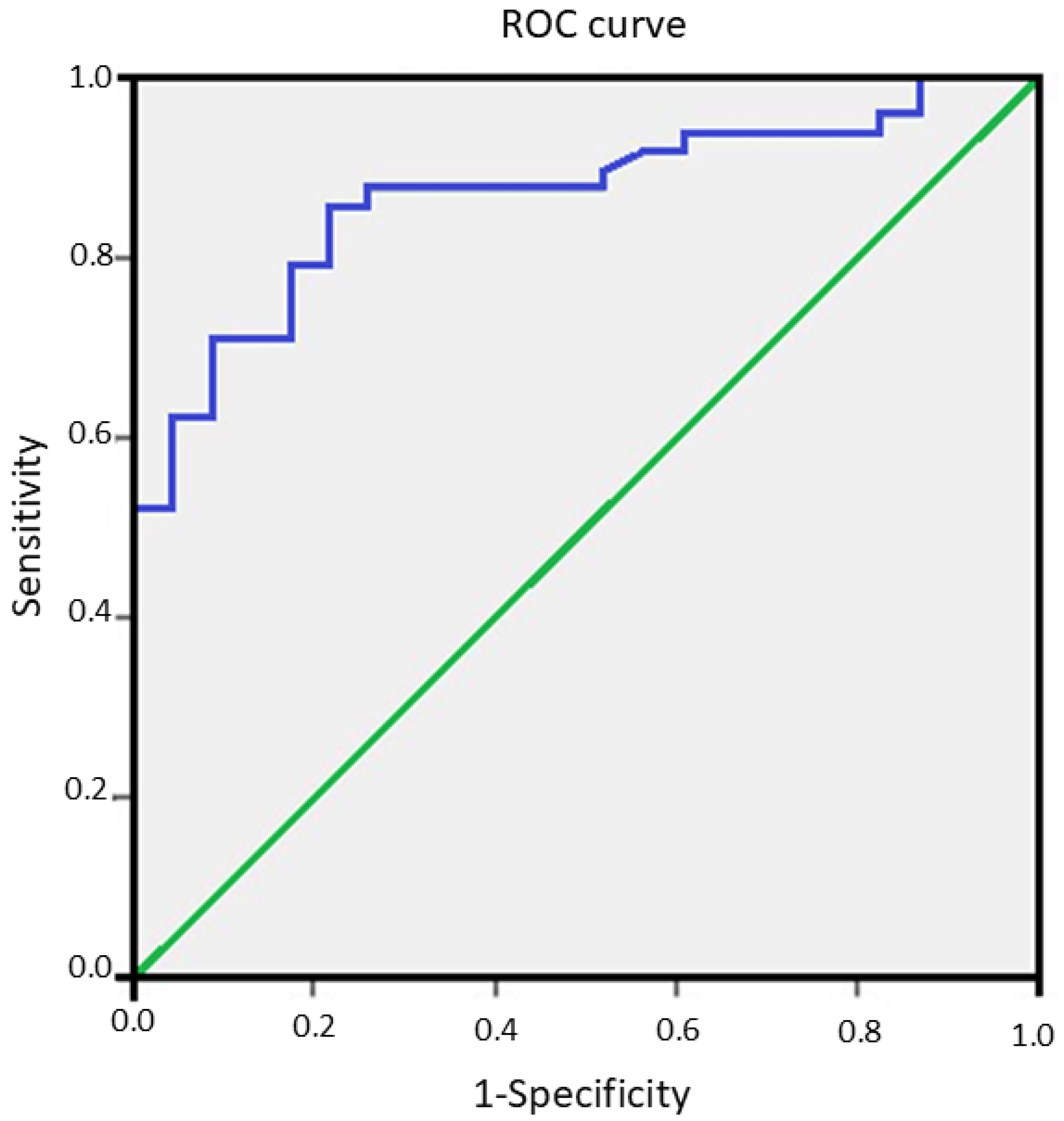
2.7. Diagnostic Performance and Threshold Selection
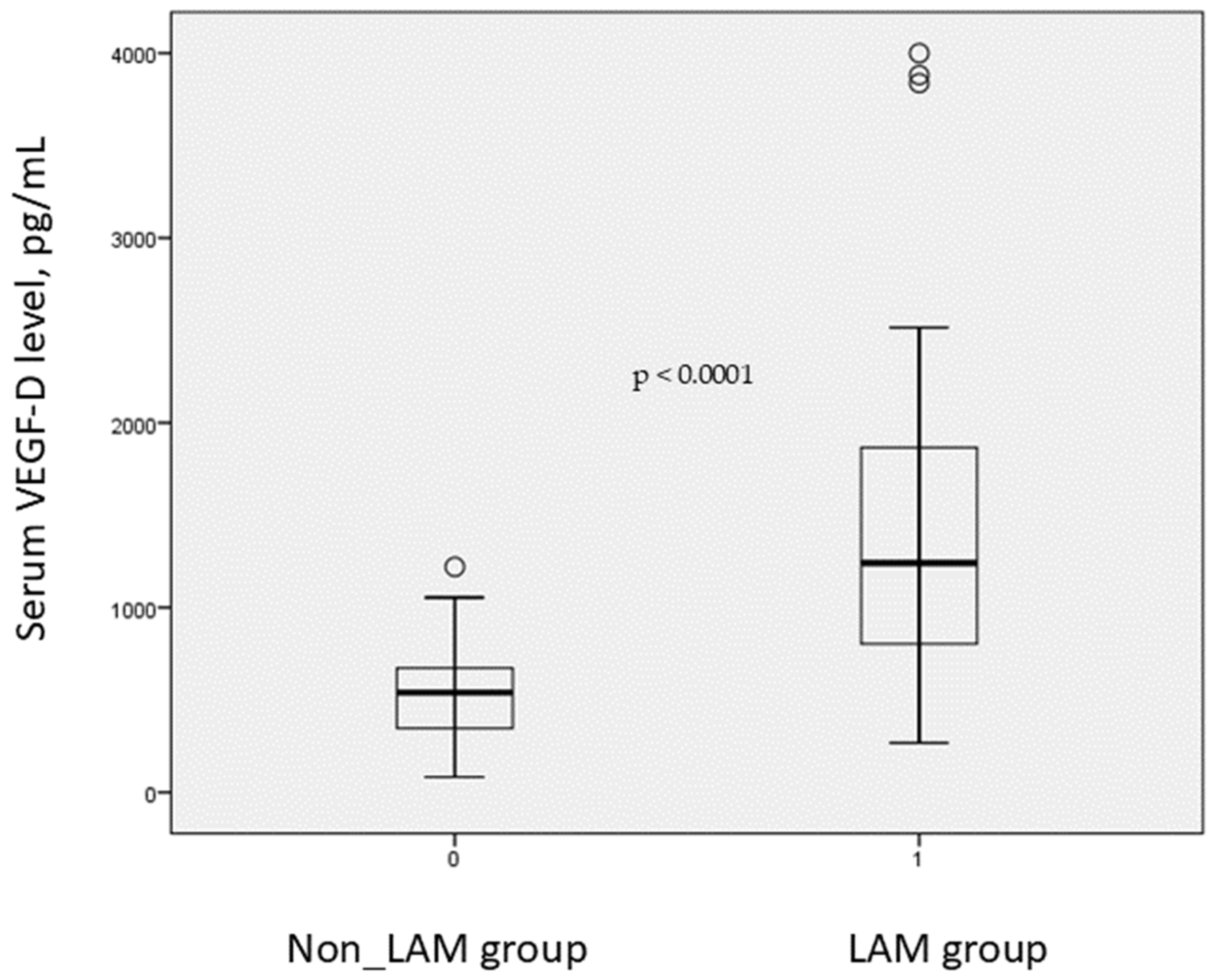
3. Results
4. Discussion
4.1. Diagnostic Utility
4.2. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McCormack, F.X.; Travis, W.D.; Colby, T.V.; Henske, E.P.; Moss, J. Lymphangioleiomyomatosis: Calling it what it is: A low-grade, destructive, metastasizing neoplasm. Am. J. Respir. Crit. Care Med. 2012, 186, 1210–1212. [Google Scholar] [CrossRef]
- Xu, K.F.; Lo, B.H. Lymphangioleiomyomatosis: Differential diagnosis and optimal management. Ther. Clin. Risk Manag. 2014, 10, 691–700. [Google Scholar] [CrossRef]
- Astrinidis, A.; Khare, L.; Carsillo, T.; Smolarek, T.; Au, K.-S.; Northrup, H.; Henske, E.P. Mutational analysis of the tuberous sclerosis gene TSC2 in patients with pulmonary lymphangioleiomyomatosis. J. Med. Genet. 2000, 37, 55–57. [Google Scholar] [CrossRef]
- Harknett, E.C.; Chang, W.Y.; Byrnes, S.; Johnson, J.; Lazor, R.; Cohen, M.; Gray, B.; Geiling, S.; Telford, H.; Tattersfield, A.; et al. Use of variability in national and regional data to estimate the prevalence of lymphangioleiomyomatosis. QJM 2011, 104, 971–979. [Google Scholar] [CrossRef]
- Lynn, E.; Forde, S.H.; Franciosi, A.N.; Bendstrup, E.; Veltkamp, M.; Wind, A.E.; Van Moorsel, C.H.M.; Lund, T.K.; Durheim, M.T.; Peeters, E.F.H.I.; et al. Updated prevalence of lymphangioleiomyomatosis in Europe. Am. J. Respir. Crit. Care Med. 2024, 209, 456–459. [Google Scholar] [CrossRef]
- Costello, L.C.; Hartman, T.E.; Ryu, J.H. High frequency of pulmonary lymphangioleiomyomatosis in women with tuberous sclerosis complex. Mayo Clin. Proc. 2000, 75, 591–594. [Google Scholar] [CrossRef]
- Obraztsova, K.; Basil, M.C.; Rue, R.; Sivakumar, A.; Lin, S.M.; Mukhitov, A.R.; Gritsiuta, A.I.; Evans, J.F.; Kopp, M.; Katzen, J.; et al. mTORC1 activation in lung mesenchyme drives sex- and age-dependent pulmonary structure and function decline. Nat. Commun. 2020, 11, 5640. [Google Scholar] [CrossRef]
- Wang, Q.; Luo, M.; Xiang, B.; Chen, S.; Ji, Y. The efficacy and safety of pharmacological treatments for lymphangioleiomyomatosis. Respir. Res. 2020, 21, 55. [Google Scholar] [CrossRef]
- Garami, A.; Zwartkruis, F.J.T.; Nobukuni, T.; Joaquin, M.; Roccio, M.; Stocker, H.; Kozma, S.C.; Hafen, E.; Bos, J.L.; Thomas, G. Insulin activation of Rheb, a mediator of mTOR/S6K/4E-BP signaling, is inhibited by TSC1 and 2. Mol. Cell 2003, 11, 1457–1466. [Google Scholar] [CrossRef]
- Dibble, C.C.; Elis, W.; Menon, S.; Qin, W.; Klekota, J.; Asara, J.M.; Finan, P.M.; Kwiatkowski, D.J.; Murphy, L.O.; Manning, B.D. TBC1D7 is a third subunit of the TSC1–TSC2 complex upstream of mTORC1. Mol. Cell 2012, 47, 535–546. [Google Scholar] [CrossRef]
- Bokhari, S.M.Z.; Hamar, P. Vascular endothelial growth factor-D (VEGF-D): An angiogenesis bypass in malignant tumors. Int. J. Mol. Sci. 2023, 24, 13317. [Google Scholar] [CrossRef]
- Yamada, Y.; Nezu, J.-I.; Shimane, M.; Hirata, Y. Molecular cloning of a novel vascular endothelial growth factor, VEGF-D. Genomics 1997, 42, 483–488. [Google Scholar] [CrossRef]
- Stacker, S.A.; Achen, M.G. Emerging roles for VEGF-D in human disease. Biomolecules 2018, 8, 1. [Google Scholar] [CrossRef]
- Seyama, K.; Kumasaka, T.; Souma, S.; Sato, T.; Kurihara, M.; Mitani, K.; Tominaga, S.; Fukuchi, Y. Vascular endothelial growth factor-D is increased in serum of patients with lymphangioleiomyomatosis. Lymphat. Res. Biol. 2006, 4, 143–152. [Google Scholar] [CrossRef]
- Young, L.R.; Inoue, Y.; McCormack, F.X. Diagnostic potential of serum VEGF-D for lymphangioleiomyomatosis. N. Engl. J. Med. 2008, 358, 199–200. [Google Scholar] [CrossRef]
- Young, L.R.; Vandyke, R.; Gulleman, P.M.; Inoue, Y.; Brown, K.K.; Schmidt, L.S.; Linehan, W.M.; Hajjar, F.; Kinder, B.W.; Trapnell, B.C.; et al. Serum vascular endothelial growth factor-D prospectively distinguishes lymphangioleiomyomatosis from other diseases. Chest 2010, 138, 674–681. [Google Scholar] [CrossRef]
- McCormack, F.X.; Inoue, Y.; Moss, J.; Singer, L.G.; Strange, C.; Nakata, K.; Barker, A.F.; Chapman, J.T.; Brantly, M.L.; Stocks, J.M.; et al. Efficacy and safety of sirolimus in lymphangioleiomyomatosis. N. Engl. J. Med. 2011, 364, 1595–1606. [Google Scholar] [CrossRef]
- Goldberg, H.J.; Harari, S.; Cottin, V.; Rosas, I.O.; Peters, E.; Biswal, S.; Cheng, Y.; Khindri, S.; Kovarik, J.M.; Ma, S.; et al. Everolimus for the treatment of lymphangioleiomyomatosis: A phase II study. Eur. Respir. J. 2015, 46, 783–794. [Google Scholar] [CrossRef]
- Pimenta, S.P.; Baldi, B.G.; Kairalla, R.A.; Carvalho, C.R. Doxiciclina em pacientes com linfangioleiomiomatose: biomarcadores e resposta funcional pulmonar. J. Bras. Pneumol. 2013, 39, 5–15. [Google Scholar] [CrossRef]
- Xu, K.F.; Zhang, P.; Tian, X.; Ma, A.; Li, X.; Zhou, J.; Zeng, N.; Gui, Y.-S.; Guo, Z.; Feng, R.; et al. The role of vascular endothelial growth factor-D in diagnosis of lymphangioleiomyomatosis (LAM). Respir. Med. 2013, 107, 263–268. [Google Scholar] [CrossRef]
- Gupta, N.; Finlay, G.A.; Kotloff, R.M.; Strange, C.; Wilson, K.C.; Young, L.R.; Taveira-DaSilva, A.M.; Johnson, S.R.; Cottin, V.; Sahn, S.A.; et al. Lymphangioleiomyomatosis diagnosis and management: High-resolution chest computed tomography, transbronchial lung biopsy, and pleural disease management. An official American Thoracic Society/Japanese Respiratory Society clinical practice guideline. Am. J. Respir. Crit. Care Med. 2017, 196, 1337–1348. [Google Scholar] [CrossRef] [PubMed]
- McCormack, F.X.; Gupta, N.; Finlay, G.R.; Young, L.R.; Taveira-DaSilva, A.M.; Glasgow, C.G.; Steagall, W.K.; Johnson, S.R.; Sahn, S.A.; Ryu, J.H.; et al. Official American Thoracic Society/Japanese Respiratory Society clinical practice guidelines: Lymphangioleiomyomatosis diagnosis and management. Am. J. Respir. Crit. Care Med. 2016, 194, 748–761. [Google Scholar] [CrossRef]
- Johnson, S.R.; Cordier, J.F.; Lazor, R.; Cottin, V.; Costabel, U.; Harari, S.; Reynaud-Gaubert, M.; Boehler, A.; Brauner, M.; Popper, H.; et al. European Respiratory Society guidelines for the diagnosis and management of lymphangioleiomyomatosis. Eur. Respir. J. 2010, 35, 14–26. [Google Scholar] [CrossRef]
- Li, M.; Zhu, W.-Y.; Wang, J.; Yang, X.-D.; Li, W.-M.; Wang, G. Diagnostic performance of VEGF-D for lymphangioleiomyomatosis: A meta-analysis. J. Bras. Pneumol. 2022, 48, e20210337. [Google Scholar] [CrossRef]
- Hirose, M.; Matsumuro, A.; Arai, T.; Sugimoto, C.; Akira, M.; Kitaichi, M.; Young, L.R.; McCormack, F.X.; Inoue, Y. Serum vascular endothelial growth factor-D as a diagnostic and therapeutic biomarker for lymphangioleiomyomatosis. PLoS ONE 2019, 14, e0212776. [Google Scholar] [CrossRef]
- O’Mahony, A.M.; Lynn, E.; Murphy, D.J.; Fabre, A.; McCarthy, C. Lymphangioleiomyomatosis: A clinical review. Breathe 2020, 16, 200007. [Google Scholar] [CrossRef] [PubMed]
- Amaral, A.F.; de Oliveira, M.R.; Dias, O.M.; Arimura, F.E.; Freitas, C.S.G.; Acencio, M.M.P.; de Alvarenga, V.A.; Kairalla, R.A.; Carvalho, C.R.R.; Baldi, B.G.; et al. Concentration of serum vascular endothelial growth factor (VEGF-D) and its correlation with functional and clinical parameters in patients with lymphangioleiomyomatosis from a Brazilian reference center. Lung 2019, 197, 139–146. [Google Scholar] [CrossRef]
- Taveira-DaSilva, A.M.; Jones, A.M.; Julien-Williams, P.; Stylianou, M.; Moss, J. Long-term effect of sirolimus on serum vascular endothelial growth factor D levels in patients with lymphangioleiomyomatosis. Chest 2018, 153, 124–132. [Google Scholar] [CrossRef]
- Hu, S.; Wu, X.; Xu, W.; Tian, X.; Yang, Y.; Wang, S.; Liu, S.; Xu, X.; Xu, K. Long-term efficacy and safety of sirolimus therapy in patients with lymphangioleiomyomatosis. Orphanet J. Rare Dis. 2019, 14, 206. [Google Scholar] [CrossRef] [PubMed]
- Revilla-López, E.; Ruiz de Miguel, V.; López-Meseguer, M.; Berastegui, C.; Boada-Pérez, M.; Mendoza-Valderrey, A.; Arjona-Peris, M.; Zapata-Ortega, M.; Monforte, V.; Bravo, C.; et al. Lymphangioleiomyomatosis: Searching for potential biomarkers. Front. Med. 2023, 10, 1079317. [Google Scholar] [CrossRef]
- Terraneo, S.; Lesma, E.; Ancona, S.; Imeri, G.; Palumbo, G.; Torre, O.; Giuliani, L.; Centanni, S.; Peron, A.; Tresoldi, S.; et al. Exploring the role of matrix metalloproteinases as biomarkers in sporadic lymphangioleiomyomatosis and tuberous sclerosis complex: A pilot study. Front. Med. 2021, 8, 605909. [Google Scholar] [CrossRef] [PubMed]
| LAM Group n = 48 | Non-LAM Group n = 23 | |
|---|---|---|
| Age, years mean ± SD | 48.2 ± 12.9 | 49.8 ±11.2 |
| BMI, kg/m2 mean ± SD | 24.8 ± 5.8 | 26.2 ± 4.9 |
| Smokers n (%) | 18 (38) | 5 (22) |
| Dyspnea n (%) | 25 (52) | 5 (22) |
| Chylothorax n (%) | 4 (8) | 0 |
| Pneumothorax n (%) | 15 (31) | 3 (13) |
| Hemoptysis n (%) | 3 (6) | 0 |
| Extrapulmonary manifestations (renal angiomyolipoma, lymphangioleiomyoma, lymphadenopathy) n (%) | 24 (50) | 3 (13) |
| VEGF-D * mean ± SD | 1425.0 ± 872.1 | 550.2 ± 276.5 |
| FEV1 *, % pred. mean ± SD | 77.2 ± 23.3 | 98.4 ± 13.9 |
| FVC *, % pred. mean ± SD | 90.3 ± 17.6 | 104.2 ± 13.7 |
| FEV1/FVC, % mean ± SD | 69.1 ± 16.1 | 77.7 ± 5.8 |
| DLco *, mL/min/mmHg mean ± SD | 66.3 ± 24.5 | 88.3 ± 14.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Makarova, M.; Baimakanova, G.; Belevskiy, A.; Chegodar, A.; Bilyalov, A.; Bodunova, N. Diagnostic Value of Serum VEGF-D in Lymphangioleiomyomatosis: Results of the First Prospective Study in the Russian Federation. Diagnostics 2026, 16, 533. https://doi.org/10.3390/diagnostics16040533
Makarova M, Baimakanova G, Belevskiy A, Chegodar A, Bilyalov A, Bodunova N. Diagnostic Value of Serum VEGF-D in Lymphangioleiomyomatosis: Results of the First Prospective Study in the Russian Federation. Diagnostics. 2026; 16(4):533. https://doi.org/10.3390/diagnostics16040533
Chicago/Turabian StyleMakarova, Marina, Gulsara Baimakanova, Andrey Belevskiy, Anzhelika Chegodar, Airat Bilyalov, and Natalia Bodunova. 2026. "Diagnostic Value of Serum VEGF-D in Lymphangioleiomyomatosis: Results of the First Prospective Study in the Russian Federation" Diagnostics 16, no. 4: 533. https://doi.org/10.3390/diagnostics16040533
APA StyleMakarova, M., Baimakanova, G., Belevskiy, A., Chegodar, A., Bilyalov, A., & Bodunova, N. (2026). Diagnostic Value of Serum VEGF-D in Lymphangioleiomyomatosis: Results of the First Prospective Study in the Russian Federation. Diagnostics, 16(4), 533. https://doi.org/10.3390/diagnostics16040533







