HF-Ultrasonography to Quantify Skin Atrophy in Patients with Inflammatory Rheumatic Diseases Treated with Courses of Glucocorticoids
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Demographic Data
2.3. Clinical Examination
2.4. High-Frequency Ultrasonography (HFUS)
2.5. Caliper Measurement of Skinfolds
2.6. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| GCs | glucocorticoids |
| HFUS | high-frequency ultrasonography |
| IRDs | inflammatory rheumatic diseases |
| ICC | intraclass correlation coefficients |
References
- Oray, M.; Abu Samra, K.; Ebrahimiadib, N.; Meese, H.; Foster, C.S. Long-term side effects of glucocorticoids. Expert. Opin. Drug Saf. 2016, 15, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Curtis, J.R.; Westfall, A.O.; Allison, J.; Bijlsma, J.W.; Freeman, A.; George, V.; Kovac, S.H.; Spettell, C.M.; Saag, K.G. Population-based assessment of adverse events associated with long-term glucocorticoid use. Arthritis Rheum. 2006, 55, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Amann, J.; Wessels, A.-M.; Breitenfeldt, F.; Huscher, D.; Bijlsma, J.W.J.; Jacobs, J.W.G.; Buttgereit, F. Quantifying cutaneous adverse effects of systemic glucocorticoids in patients with rheumatoid arthritis: A cross-sectional cohort study. Clin. Exp. Rheumatol. 2017, 35, 471–476. [Google Scholar] [PubMed]
- Polańska, A.; Dańczak-Pazdrowska, A.; Jałowska, M.; Żaba, R.; Adamski, Z. Current applications of high-frequency ultrasonography in dermatology. Postep. Dermatol. Alergol. 2017, 34, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Barnes, L.; Kaya, G.; Rollason, V. Topical corticosteroid-induced skin atrophy: A comprehensive review. Drug Saf. 2015, 38, 493–509. [Google Scholar] [CrossRef] [PubMed]
- Hesselstrand, R.; Carlestam, J.; Wildt, M.; Sandqvist, G.; Andréasson, K. High frequency ultrasound of skin involvement in systemic sclerosis—A follow-up study. Arthritis Res. Ther. 2015, 17, 329. [Google Scholar] [CrossRef] [PubMed]
- Naredo, E.; Pascau, J.; Damjanov, N.; Lepri, G.; Gordaliza, P.M.; Janta, I.; Ovalles-Bonilla, J.G.; López-Longo, F.J.; Matucci-Cerinic, M. Performance of ultra-high-frequency ultrasound in the evaluation of skin involvement in systemic sclerosis: A preliminary report. Rheumatology 2020, 59, 1671–1678. [Google Scholar] [CrossRef] [PubMed]
- Dykes, P.J.; Marks, R. Measurement of skin thickness: A comparison of two in vivo techniques with a conventional histometric method. J. Investig. Dermatol. 1977, 69, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Cicchetti, D.V. Multiple comparison methods: Establishing guidelines for their valid application in neuropsychological research. J. Clin. Exp. Neuropsychol. 1994, 16, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Battie, C.; Jitsukawa, S.; Bernerd, F.; Del Bino, S.; Marionnet, C.; Verschoore, M. New insights in photoaging, UVA induced damage and skin types. Exp. Dermatol. 2014, 23 (Suppl. S1), 7–12. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Landewé, R.B.M.; Bergstra, S.A.; Kerschbaumer, A.; Sepriano, A.; Aletaha, D.; Caporali, R.; Edwards, C.J.; Hyrich, K.L.; Pope, J.E.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update. Ann. Rheum. Dis. 2023, 82, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Huscher, D.; Thiele, K.; Gromnica-Ihle, E.; Hein, G.; Demary, W.; Dreher, R.; Zink, A.; Buttgereit, F. Dose-related patterns of glucocorticoid-induced side effects. Ann. Rheum. Dis. 2009, 68, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Stone, J.H.; McDowell, P.J.; Jayne, D.R.; Merkel, P.A.; Robson, J.; Patel, N.J.; Zhang, Y.; Yue, H.; Bekker, P.; Heaney, L.G. The glucocorticoid toxicity index: Measuring change in glucocorticoid toxicity over time. Semin. Arthritis Rheum. 2022, 55, 152010. [Google Scholar] [CrossRef] [PubMed]
- Olsen, L.O.; Takiwaki, H.; Serup, J. High-frequency ultrasound characterization of normal skin. Skin thickness and echographic density of 22 anatomical sites. Skin. Res. Technol. 1995, 1, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Brincat, M.; Kabalan, S.; Studd, J.; Moniz, C.; de Trafford, J.; Montgomery, J. A study of the decrease of skin collagen content, skin thickness, and bone mass in the postmenopausal woman. Obstet. Gynecol. 1987, 70, 840–845. [Google Scholar] [PubMed]
- Korting, H.C.; Unholzer, A.; Schäfer-Korting, M.; Tausch, I.; Gassmueller, J.; Nietsch, K.-H. Different skin thinning potential of equipotent medium-strength glucocorticoids. Skin. Pharmacol. Appl. Skin. Physiol. 2002, 15, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Cossmann, M.; Welzel, J. Evaluation of the atrophogenic potential of different glucocorticoids using optical coherence tomography, 20-MHz ultrasound and profilometry; a double-blind, placebo-controlled trial. Br. J. Dermatol. 2006, 155, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Lepri, G.; Hughes, M.; Allanore, Y.; Denton, C.P.; Furst, D.E.; Wang, Y.; Santiago, T.; Galetti, I.; Del Galdo, F.; Khanna, D.; et al. The role of skin ultrasound in systemic sclerosis: Looking below the surface to understand disease evolution. Lancet Rheumatol. 2023, 5, e422–e425. [Google Scholar] [CrossRef] [PubMed]
- Flower, V.A.; Barratt, S.L.; Hart, D.J.; Mackenzie, A.B.; Shipley, J.A.; Ward, S.G.; Pauling, J.D. High-frequency Ultrasound Assessment of Systemic Sclerosis Skin Involvement: Intraobserver Repeatability and Relationship with Clinician Assessment and Dermal Collagen Content. J. Rheumatol. 2021, 48, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Beiu, C.; Popa, L.G.; Bălăceanu-Gurău, B.; Iliescu, C.A.; Racoviță, A.; Popescu, M.N.; Mihai, M.M. Personalization of Minimally-Invasive Aesthetic Procedures with the Use of Ultrasound Compared to Alternative Imaging Modalities. Diagnostics 2023, 13, 3512. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- van der Goes, M.C.; Jacobs, J.W.G.; Boers, M.; Andrews, T.; Blom-Bakkers, M.A.M.; Buttgereit, F.; Caeyers, N.; Cutolo, M.; Silva, J.A.P.D.; Guillevin, L.; et al. Monitoring adverse events of low-dose glucocorticoid therapy: EULAR recommendations for clinical trials and daily practice. Ann. Rheum. Dis. 2010, 69, 1913–1919. [Google Scholar] [CrossRef]
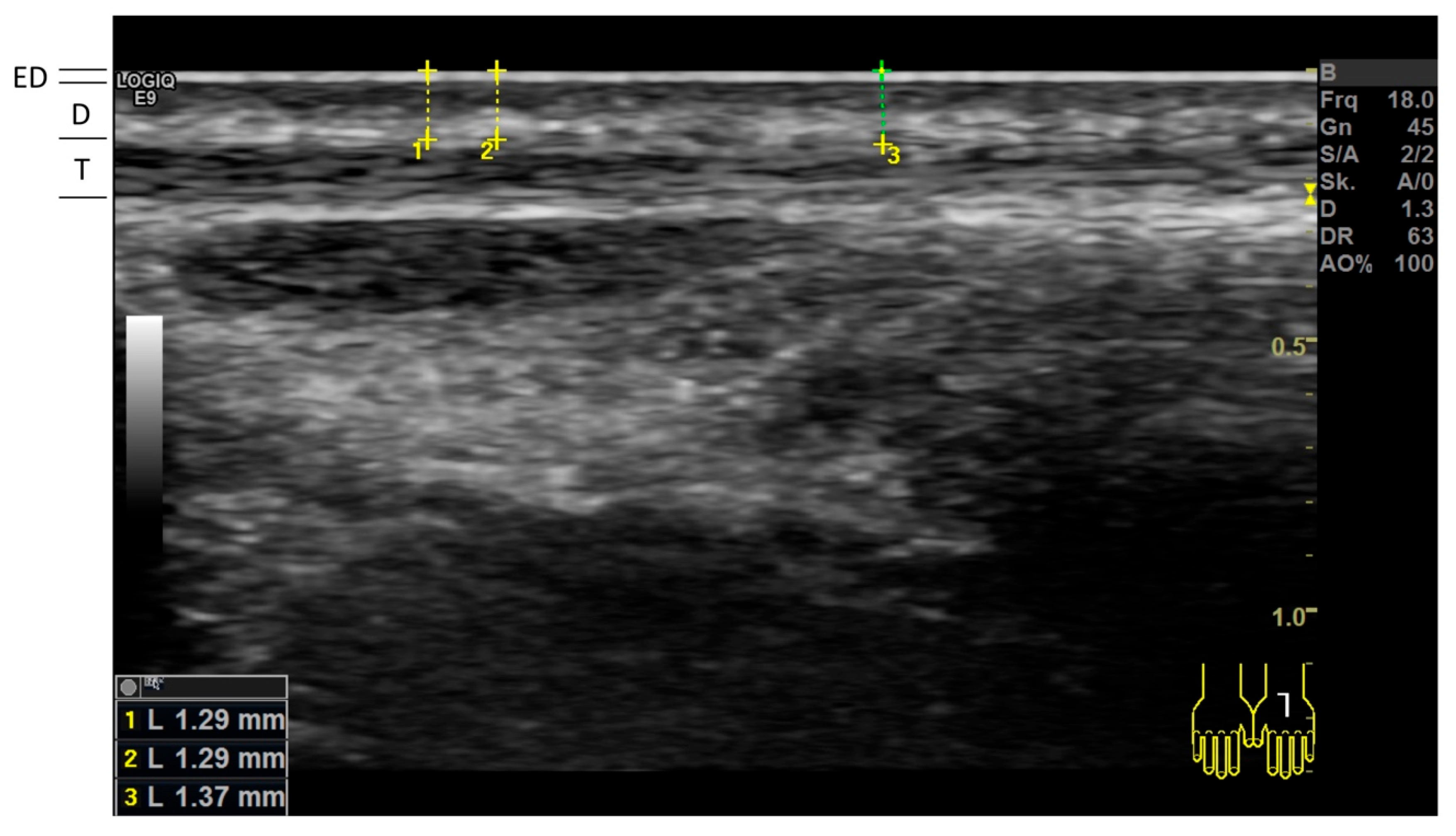
| Subjects Studied | Patients with IRDs and GC > 6 mo. | Subjects Without IRDs and No Prior GC | p-Value * |
|---|---|---|---|
| n = 53 | n = 53 | ||
| Demographics | |||
| Female sex, n (%) | 33 (62) | 32 (60) | 0.842 |
| Age in years, mean (±SD) | 66.4 (±10.0) | 65.9 (±11.3) | 0.813 |
| Age ≥ 70 years, n (%) | 21 (40) | 21 (40) | 1.000 |
| Body mass index in kg/m2, mean (±SD) | 26.7 (±4.6) | 27.3 (±5.3) | 0.503 |
| Duration since IRD diagnosis in years, median (range) | 9 (0–47) | - | - |
| Inflammatory rheumatic diseases (IRDs) | |||
| Rheumatoid arthritis | 26 (49) | - | - |
| Gouty arthritis | 2 (4) | - | - |
| Undifferentiated arthritis | 2 (4) | - | - |
| Psoriatic arthritis | 2 (4) | - | - |
| Spondyloarthritis | 2 (4) | - | - |
| Connective tissue diseases | 6 (11) | - | - |
| Polymyalgia rheumatica | 6 (11) | - | - |
| Giant cell arteritis | 5 (9) | - | - |
| Granulomatosis with polyangiitis | 1 (2) | - | - |
| Polyarteritis nodosa | 1 (2) | - | - |
| Patient-reported treatment with glucocorticoids (GC) | |||
| Any GC in previous week, n (%) | 52 (98) | - | - |
| Any GC in previous month, n (%) | 52 (98) | - | - |
| Any GC in previous year, n (%) | 52 (98) | - | - |
| Cumulative duration of GC treatment (years) | |||
| mean (±SD) | 10.4 (±10.0) | - | - |
| Average daily prednisolone-equivalent dose (mg) | |||
| mean (±SD) | 9.5 (±7.8) | - | - |
| Cumulative prednisolone dose (gram) | |||
| mean (±SD) | 30.5 (±38.5) | - | - |
| Selected patient-reported pre-existing conditions | |||
| Anticoagulation therapy, n (%) | 19 (36) | 7 (13) | 0.007 |
| Diabetes mellitus, n (%) | 9 (17) | 4 (8) | 0.139 |
| Arterial hypertension, n (%) | 40 (76) | 21 (34) | <0.001 |
| Hyperlipidemia, n (%) | 20 (38) | 9 (17) | 0.017 |
| Osteoporosis, n (%) | 31 (59) | 4 (8) | <0.001 |
| Cataract, n (%) | 16 (30) | 4 (8) | 0.003 |
| More than 3 upper respiratory infections per year, n (%) | 17 (32) | 1 (2) | <0.001 |
| Ever had an infection treated in the hospital, n (%) | 15 (28) | 2 (4) | 0.001 |
| Ever had an infection treated in intensive care, n (%) | 8 (15) | 0 (0) | 0.006 |
| Subjects Studied | Patients with IRD and GC > 6 mo. | Subjects Without IRD and No Prior GC | p-Value * |
|---|---|---|---|
| Clinical signs | n = 53 | n = 53 | |
| Skin atrophy | |||
| None | 0 (0) | 31 (58) | <0.001 |
| Mild | 0 (0) | 19 (36) | |
| Moderate | 34 (64) | 3 (6) | |
| Severe | 19 (36) | 0 (0) | |
| Purpura | |||
| None | 24 (45) | 49 (92) | <0.001 |
| Mild | 17 (32) | 4 (8) | |
| Severe | 12 (23) | 0 (0) | |
| Striae rubrae | |||
| None | 45 (85) | 44 (83) | 0.279 |
| Mild | 2 (4) | 6 (11) | |
| Severe | 6 (11) | 3 (6) | |
| Facies lunata | |||
| None | 42 (79) | 53 (100) | 0.001 |
| Mild | 5 (10) | 0 (0) | |
| Severe | 6 (11) | 0 (0) | |
| Buffalo hump | |||
| None | 41 (77) | 51 (96) | 0.010 |
| Mild | 8 (15) | 2 (4) | |
| Severe | 4 (8) | 0 (0) | |
| Skinfold thickness measured by caliper (mm) | n = 42 # | n = 50 # | |
| Back of hand | |||
| mean (±SD) | 1.7 (±0.4) | 2.1 (±0.5) | <0.001 |
| Cubital fossa | |||
| mean (±SD) | 6.7 (±2.7) | 7.1 (±3.0) | 0.419 |
| Dorsal midfoot | |||
| mean (±SD) | 3.6 (±3.7) | 4.1 (±3.4) | 0.501 |
| Cutis thickness measured by HFUS (mm) | n = 53 | n = 53 | |
| Back of hand | |||
| mean (±SD) | 0.66 (±0.12) | 0.82 (±0.18) | <0.001 |
| Cubital fossa | |||
| mean (±SD) | 0.86 (±0.15) | 1.00 (±0.21) | <0.001 |
| Dorsal midfoot | |||
| mean (±SD) | 0.76 (±0.16) | 0.94 (±0.18) | <0.001 |
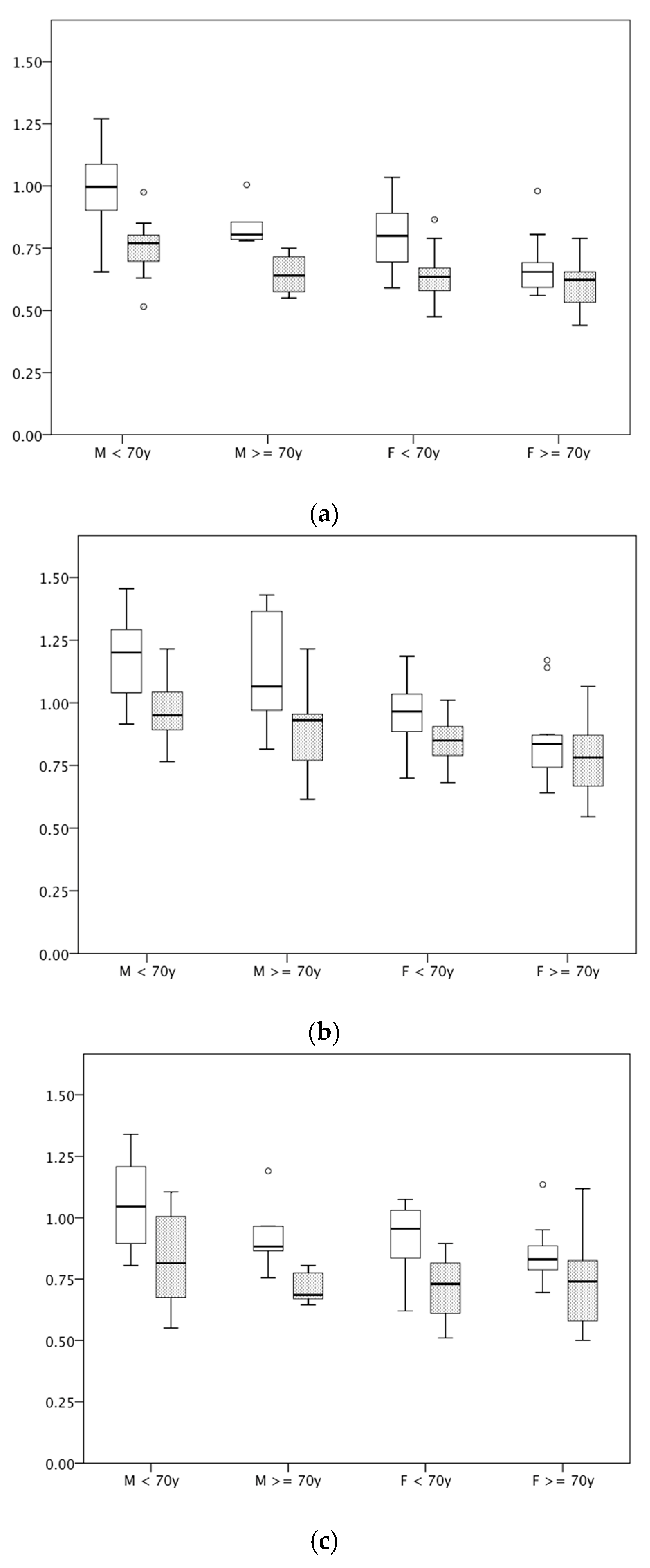
| Cutaneous Thickness by HFUS (in mm) | Patients with IRD and GC > 6 mo. | Subjects Without IRD and No Prior GC | p-Value * |
|---|---|---|---|
| Male, <70 years | n = 15 | n = 15 | |
| Back of hand | 0.75 (±0.11) | 0.99 (±0.17) | <0.001 |
| Cubital fossa | 0.97 (±0.11) | 1.18 (±0.16) | <0.001 |
| Dorsal midfoot | 0.84 (±0.19) | 1.05 (±0.18) | 0.004 |
| Male, ≥70 years | n = 5 | n = 6 | |
| Back of hand mean | 0.65 (±0.09) | 0.84 (±0.17) | 0.005 |
| Cubital fossa | 0.90 (±0.22) | 1.12 (±0.24) | 0.148 |
| Dorsal midfoot | 0.72 (±0.07) | 0.92 (±0.15) | 0.018 |
| Female, <70 years | n = 17 | n = 17 | |
| Back of hand | 0.64 (±0.10) | 0.80 (±0.13) | 0.001 |
| Cubital fossa | 0.84 (±0.09) | 0.94 (±0.13) | 0.016 |
| Dorsal midfoot | 0.72 (±0.12) | 0.93 (±0.13) | <0.001 |
| Female, ≥70 years | n = 16 | n = 15 | |
| Back of hand mean | 0.61 (±0.11) | 0.67 (±0.11) | 0.131 |
| Cubital fossa | 0.78 (±0.15) | 0.84 (±0.15) | 0.309 |
| Dorsal midfoot | 0.74 (±0.18) | 0.85 (±0.15) | 0.057 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schuster, A.; Horn, A.; Günther, F.; Fleck, M.; Hartung, W.; Ehrenstein, B. HF-Ultrasonography to Quantify Skin Atrophy in Patients with Inflammatory Rheumatic Diseases Treated with Courses of Glucocorticoids. Diagnostics 2025, 15, 619. https://doi.org/10.3390/diagnostics15050619
Schuster A, Horn A, Günther F, Fleck M, Hartung W, Ehrenstein B. HF-Ultrasonography to Quantify Skin Atrophy in Patients with Inflammatory Rheumatic Diseases Treated with Courses of Glucocorticoids. Diagnostics. 2025; 15(5):619. https://doi.org/10.3390/diagnostics15050619
Chicago/Turabian StyleSchuster, Antonia, Andreas Horn, Florian Günther, Martin Fleck, Wolfgang Hartung, and Boris Ehrenstein. 2025. "HF-Ultrasonography to Quantify Skin Atrophy in Patients with Inflammatory Rheumatic Diseases Treated with Courses of Glucocorticoids" Diagnostics 15, no. 5: 619. https://doi.org/10.3390/diagnostics15050619
APA StyleSchuster, A., Horn, A., Günther, F., Fleck, M., Hartung, W., & Ehrenstein, B. (2025). HF-Ultrasonography to Quantify Skin Atrophy in Patients with Inflammatory Rheumatic Diseases Treated with Courses of Glucocorticoids. Diagnostics, 15(5), 619. https://doi.org/10.3390/diagnostics15050619





