Prenatal Diagnosis and Management of Tuberous Sclerosis Complex with Cardiac Rhabdomyoma: A Case Report Highlighting the Role of Sirolimus and Postnatal Complications
Abstract
1. Introduction
2. Case
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sugalska, M.; Tomik, A.; Jóźwiak, S.; Werner, B. Treatment of Cardiac Rhabdomyomas with mTOR Inhibitors in Children with Tuberous Sclerosis Complex—A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 4907. [Google Scholar] [CrossRef] [PubMed]
- Yıldırım, S.; Aypar, E.; Aydın, B.; Akyüz, C.; Aykan, H.H.; Ertuğrul, İ.; Karagöz, T.; Alehan, D. Cardiac rhabdomyomas: Clinical progression, efficacy and safety of everolimus treatment. Turk. J. Pediatr. 2023, 65, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Cavalheiro, S.; da Costa, M.D.S.; Richtmann, R. Everolimus as a possible prenatal treatment of in utero diagnosed subependymal lesions in tuberous sclerosis complex: A case report. Child’s Nerv. Syst. 2021, 37, 3897–3899. [Google Scholar] [CrossRef] [PubMed]
- Montaguti, E.; Gesuete, V.; Perolo, A.; Balducci, A.; Fiorentini, M.; Donti, A.; Pilu, G. A case of massive fetal cardiac rhabdomyoma: Ultrasound features and management. J. Matern.-Fetal Neonatal Med. 2023, 36, 2197099. [Google Scholar] [CrossRef] [PubMed]
- Zungsontiporn, N.; Tantrachoti, P.; Puwanant, S. Foetal and maternal cardiac rhabdomyomas associated with tuberous sclerosis. Eur. Heart J. 2015, 37, 2036. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, H.J.; Javinani, A.; Morning, M.L.; D’Antonio, F.; Pagani, G.; Puranik, P.M.; Khalil, A.; Shamshirsaz, A.A. Characteristics and Outcomes of Fetal Cardiac Rhabdomyoma with or Without mTOR Inhibitors, a Systematic Review and Meta-Analysis. Prenat. Diagn. 2024, 44, 1251–1267. [Google Scholar] [CrossRef] [PubMed]
- Hinton, R.B.; Prakash, A.; Romp, R.L.; Krueger, D.A.; Knilans, T.K. Cardiovascular manifestations of tuberous sclerosis complex and summary of the revised diagnostic criteria and surveillance and management recommendations from the international tuberous sclerosis consensus group. J. Am. Heart Assoc. 2014, 3, e001493. [Google Scholar] [CrossRef] [PubMed]
- Schlaegel, F.; Takacs, Z.; Solomayer, E.F.; Abdul-Kaliq, H.; Meyberg-Solomayer, G. Prenatal diagnosis of giant cardiac rhabdomyoma with fetal hydrops in tuberous sclerosis. J. Prenat. Med. 2013, 7, 39. [Google Scholar] [PubMed]
- Chao, A.S.; Chao, A.; Wang, T.H.; Chang, Y.C.; Chang, Y.L.; Hsieh, C.C.; Lien, R.; Su, W.J. Outcome of antenatally diagnosed cardiac rhabdomyoma: Case series and a meta-analysis. Ultrasound Obstet. Gynecol. 2008, 31, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Racioppi, G.; Proietti Checchi, M.; Sforza, G.; Voci, A.; Mazzone, L.; Valeriani, M.; Moavero, R. Prenatal mTOR Inhibitors in Tuberous Sclerosis Complex: Current Insights and Future Directions. J. Clin. Med. 2024, 13, 6335. [Google Scholar] [CrossRef] [PubMed]
- Lucchesi, M.; Chiappa, E.; Giordano, F.; Mari, F.; Genitori, L.; Sardi, I. Sirolimus in infants with multiple cardiac rhabdomyomas associated with tuberous sclerosis complex. Case Rep. Oncol. 2018, 11, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Kohrman, M.H. Emerging treatments in the management of tuberous sclerosis complex. Pediatr. Neurol. 2012, 46, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Man, A.; Di Scipio, M.; Grewal, S.; Suk, Y.; Trinari, E.; Ejaz, R.; Whitney, R. The Genetics of Tuberous Sclerosis Complex and Related mTORopathies: Current Understanding and Future Directions. Genes 2024, 15, 332. [Google Scholar] [CrossRef] [PubMed]
- Hartinger, J.M.; Ryšánek, P.; Slanař, O.; Šíma, M. Pharmacokinetic principles of dose adjustment ofmTORinhibitors in solid organ transplanted patients. J. Clin. Pharm. Ther. 2022, 47, 1362–1367. [Google Scholar] [CrossRef] [PubMed]
- Maria, G.; Antonia, D.; Michael, A.; Kate, M.; Sian, E.; Sarah, F.E.; Mehul, D.; Pratik, S. Sirolimus: Efficacy and Complications in Children with Hyperinsulinemic Hypoglycemia: A 5-Year Follow-Up Study. J. Endocr. Soc. 2019, 3, 699–713. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Chang, C.S.; Choi, S.-J.; Oh, S.; Roh, C.-R. Sirolimus therapy for fetal cardiac rhabdomyoma in a pregnant woman with tuberous sclerosis. Obstet. Gynecol. Sci. 2019, 62, 280. [Google Scholar] [CrossRef] [PubMed]
- Dagge, A.; Silva, L.A.; Jorge, S.; Nogueira, E.; Rebelo, M.; Pinto, L. Fetal Tuberous Sclerosis: Sirolimus for the Treatment of Fetal rhabdomyoma. Fetal Pediatr. Pathol. 2021, 41, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi-Fakhari, D.; Stires, G.; Hahn, E.; Krueger, D.; Franz, D.N. Prenatal sirolimus treatment for rhabdomyomas in tuberous sclerosis. Pediatr. Neurol. 2021, 125, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Pluym, I.D.; Sklansky, M.; Wu, J.Y.; Afshar, Y.; Holliman, K.; Devore, G.R.; Walden, A.; Platt, L.D.; Krakow, D. Fetal cardiac rhabdomyomas treated with maternal sirolimus. Prenat. Diagn. 2020, 40, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Vachon-Marceau, C.; Guerra, V.; Jaeggi, E.; Chau, V.; Ryan, G.; Van Mieghem, T. In-utero treatment of large symptomatic rhabdomyoma with sirolimus. Ultrasound Obstet. Gynecol. 2019, 53, 420–421. [Google Scholar] [CrossRef] [PubMed]
- Will, J.; Siedentopf, N.; Schmid, O.; Gruber, T.; Henrich, W.; Hertzberg, C.; Weschke, B. Successful prenatal treatment of cardiac rhabdomyoma in a fetus with tuberous sclerosis. Pediatr. Rep. 2023, 15, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Mlczoch, E.; Hanslik, A.; Luckner, D.; Kitzmüller, E.; Prayer, D.; Michel-Behnke, I. Prenatal diagnosis of giant cardiac rhabdomyoma in tuberous sclerosis complex: A new therapeutic option with everolimus. Ultrasound Obstet. Gynecol. 2015, 45, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Peron, A.; Au, K.S.; Northrup, H. Genetics, genomics, and genotype–phenotype correlations of TSC: Insights for clinical practice. Am. J. Med. Genet. Part C Semin. Med. Genet. 2018, 178, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Hurtado-Sierra, D.; Ramos Garzón, J.X.; Rojas, L.Z.; Fernández-Gómez, O.; Manrique-Rincón, F. Case report: Accelerated regression of giant cardiac rhabdomyomas in neonates with low dose everolimus. Front. Pediatr. 2023, 11, 1109646. [Google Scholar] [CrossRef] [PubMed]
- Mao, B.; Zhang, Q.; Ma, L.; Zhao, D.-S.; Zhao, P.; Yan, P. Overview of Research into mTOR Inhibitors. Molecules 2022, 27, 5295. [Google Scholar] [CrossRef] [PubMed]
- Silva-Sánchez, M.P.; Alvarado-Socarras, J.L.; Castro-Monsalve, J.; Meneses, K.M.; Santiago, J.; Prada, C.E. Everolimus for severe arrhythmias in tuberous sclerosis complex related cardiac rhabdomyomas. Am. J. Med. Genet. Part A 2021, 185, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Bravo Oro, A.; Esmer Sánchez, M.d.C.; Rubio Hernández, M.E.; Morales Ibarra, J.J.; Reyes Vaca, J.G.; Villegas Valdez, D.M.M.; Gómez Elías, C.L. Respuesta a everolimus en un neonato con Rabdomioma cardiaco asociado con el Complejo Esclerosis Tuberosa. Acta Pediátrica México 2020, 41, 208–214. [Google Scholar] [CrossRef]
- Cai, X.; Golubkova, A.; Hunter, C.J. Advances in our understanding of the molecular pathogenesis of necrotizing enterocolitis. BMC Pediatrics 2022, 22, 210–225. [Google Scholar] [CrossRef] [PubMed]
- Kashif, H.; Abuelgasim, E.; Hussain, N.; Luyt, J.; Harky, A. Necrotizing enterocolitis and congenital heart disease. Ann. Pediatr. Cardiol. 2021, 14, 507–515. [Google Scholar] [CrossRef] [PubMed]
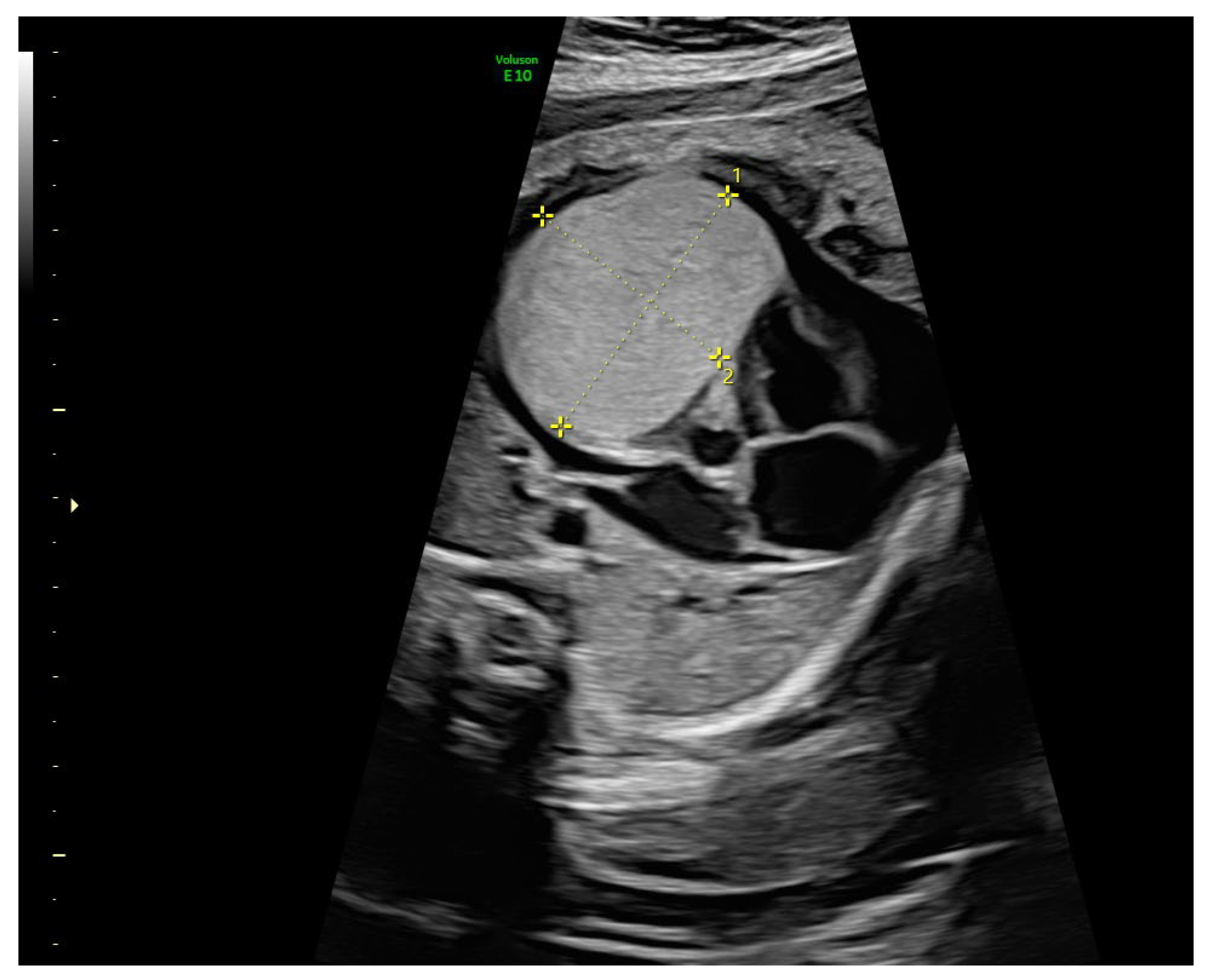
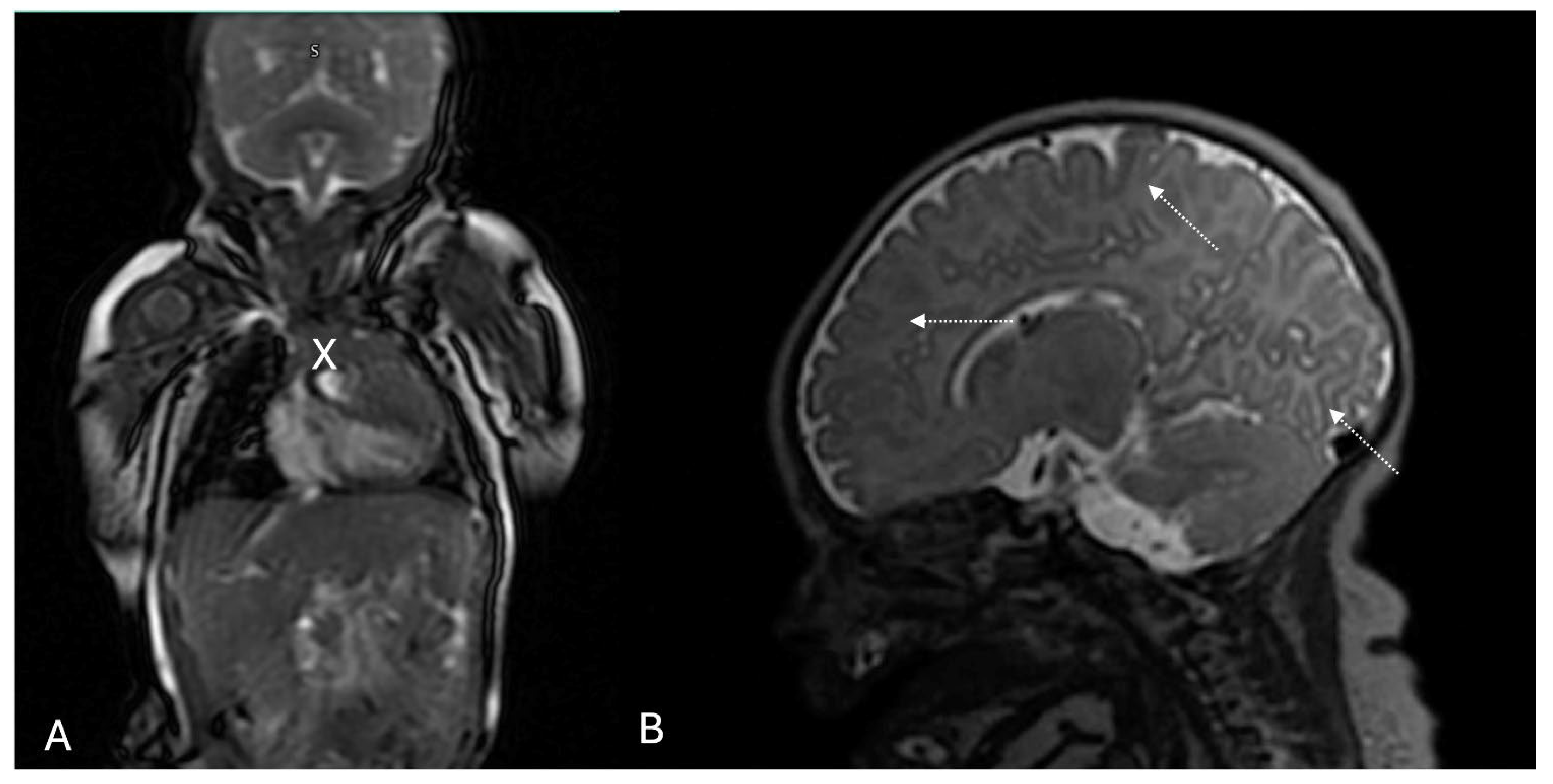
| Reference | Drug | Gestational Age at Initiation | Dose | Duration | Route | Fetal/Neonatal Outcomes | Adverse Events |
|---|---|---|---|---|---|---|---|
| Dagge et al., 2021 [17] | Sirolimus | 30 weeks | 4 mg/day (adjusted by levels) | 3 weeks | Maternal oral | Significant fetal tumor size reduction | Not reported |
| Ebrahimi-Fakhari et al., 2021 [18] | Sirolimus | 28–34 weeks (4 cases) | 1.5–4 mg/day (by levels) | 2–6 weeks | Maternal oral | Significant tumor reduction in all cases; no fetal arrhythmias | Mild maternal rash in one case; no serious complications |
| Pluym et al., 2020 [19] | Sirolimus | 30 weeks | 4 mg/day (adjusted) | 4 weeks | Maternal oral | Significant tumor regression; no neonatal complications | Maternal lipid reduction; no significant toxicity |
| Vachon-Marceau et al., 2019 [20] | Sirolimus | 30 weeks | 6 mg/day (by levels) | 4 weeks | Maternal oral | Tumor reduction; fetal clinical improvement | Maternal lipid reduction; no severe effects |
| Park et al., 2019 [16] | Sirolimus | 30 weeks | 2 mg/day | ~3 weeks | Maternal oral | Tumor shrinkage; improved signs of obstruction | None reported |
| Will et al., 2023 [21] | Sirolimus | 30 + 4 weeks | 3 mg/day (target: 10–15 ng/mL) | 3 weeks | Maternal oral | Rapid and complete tumor regression; uncomplicated delivery | Mild maternal nausea; no toxicity |
| Cavalheiro et al., 2021 [3] | Everolimus | 24 weeks | Not specified | 4 weeks | Maternal oral | Reduction of subependymal and cardiac lesions | Not reported |
| Mlczoch et al., 2015 [22] | Everolimus | 28 weeks | 10 mg/week (divided daily) | ~6 weeks | Maternal oral | Marked fetal tumor volume reduction | Mild maternal gastrointestinal discomfort |
| Present study | Sirolimus | 30 + 4 weeks | 3 mg/day (adjusted to 6 mg/day) plasma level 6 ng/mL | 4 weeks | Maternal oral | Not reduction in rhabdomyoma size; neonatal necrotizing enterocolitis | Not reported |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Torres, D.A.; Arenas-Estala, J.; Sánchez-Cortés, R.G.; Dávila-Escamilla, I.V.; Nieto-Sanjuanero, A.; López-Uriarte, G.A. Prenatal Diagnosis and Management of Tuberous Sclerosis Complex with Cardiac Rhabdomyoma: A Case Report Highlighting the Role of Sirolimus and Postnatal Complications. Diagnostics 2025, 15, 1811. https://doi.org/10.3390/diagnostics15141811
Rodríguez-Torres DA, Arenas-Estala J, Sánchez-Cortés RG, Dávila-Escamilla IV, Nieto-Sanjuanero A, López-Uriarte GA. Prenatal Diagnosis and Management of Tuberous Sclerosis Complex with Cardiac Rhabdomyoma: A Case Report Highlighting the Role of Sirolimus and Postnatal Complications. Diagnostics. 2025; 15(14):1811. https://doi.org/10.3390/diagnostics15141811
Chicago/Turabian StyleRodríguez-Torres, David Asael, Joel Arenas-Estala, Ramón Gerardo Sánchez-Cortés, Iván Vladimir Dávila-Escamilla, Adriana Nieto-Sanjuanero, and Graciela Arelí López-Uriarte. 2025. "Prenatal Diagnosis and Management of Tuberous Sclerosis Complex with Cardiac Rhabdomyoma: A Case Report Highlighting the Role of Sirolimus and Postnatal Complications" Diagnostics 15, no. 14: 1811. https://doi.org/10.3390/diagnostics15141811
APA StyleRodríguez-Torres, D. A., Arenas-Estala, J., Sánchez-Cortés, R. G., Dávila-Escamilla, I. V., Nieto-Sanjuanero, A., & López-Uriarte, G. A. (2025). Prenatal Diagnosis and Management of Tuberous Sclerosis Complex with Cardiac Rhabdomyoma: A Case Report Highlighting the Role of Sirolimus and Postnatal Complications. Diagnostics, 15(14), 1811. https://doi.org/10.3390/diagnostics15141811





