Validation of Axial Juvenile Spondyloarthropathy Criteria in Turkish Juvenile Spondyloarthropathy Patients
Abstract
1. Introduction
2. Materials and Methods
3. Statistical Analysis
4. Results
4.1. Demographic Features of the Patients
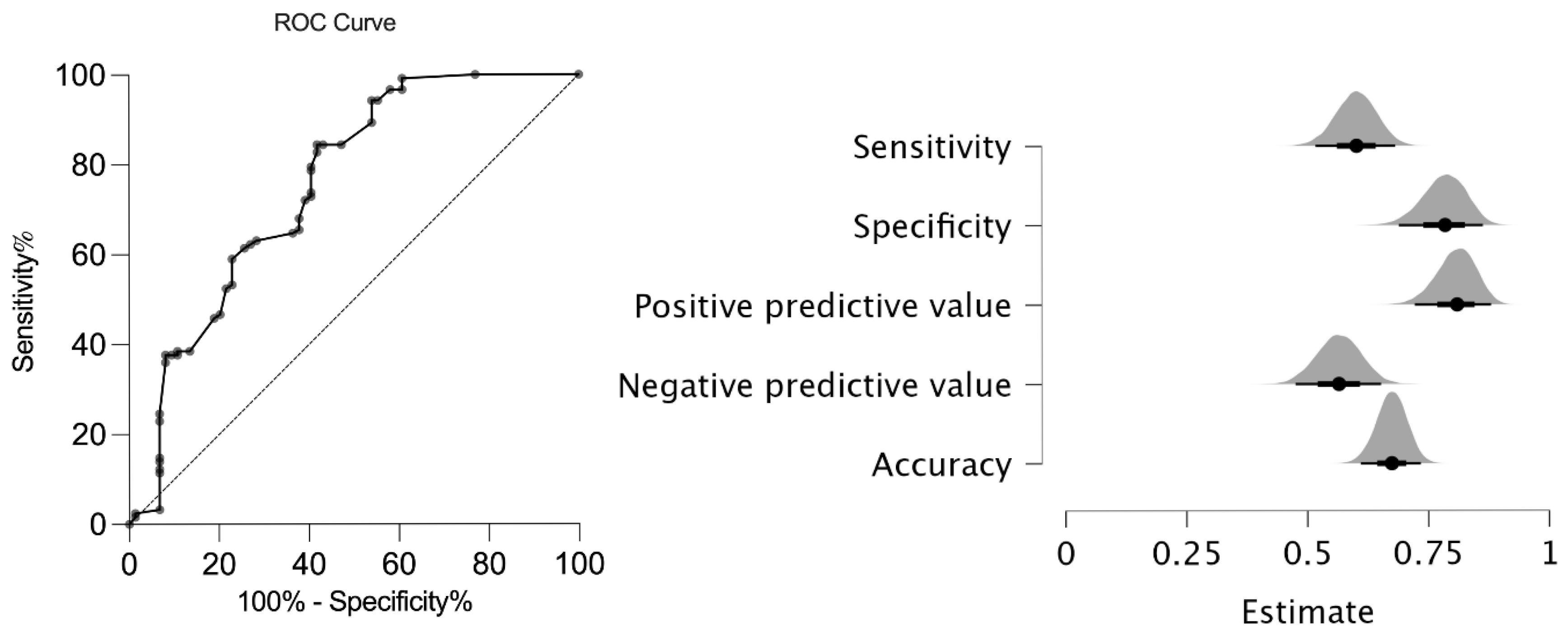
4.2. Sensitivity and Specificity of the New AxJSpA Classification Criteria in JSpA Patients
5. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nordal, E.; Zak, M.; Aalto, K.; Berntson, L.; Fasth, A.; Herlin, T.; Lahdenne, P.; Nielsen, S.; Straume, B.; Rygg, M. Ongoing disease activity and changing categories in a long-term nordic cohort study of juvenile idiopathic arthritis. Arthritis Rheum. 2011, 63, 2809–2818. [Google Scholar] [CrossRef] [PubMed]
- Modesto, C.; Antón, J.; Rodriguez, B.; Bou, R.; Arnal, C.; Ros, J.; Tena, X.; Rodrigo, C.; Rotés, I.; Hermosilla, E.; et al. Incidence and prevalence of juvenile idiopathic arthritis in Catalonia (Spain). Scand. J. Rheumatol. 2010, 39, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Oen, K.; Duffy, C.M.; Tse, S.M.; Ramsey, S.; Ellsworth, J.; Chédeville, G.; Chetaille, A.L.; Saint-Cyr, C.; Cabral, D.A.; Spiegel, L.R.; et al. Early outcomes and improvement of patients with juvenile idiopathic arthritis enrolled in a Canadian multicenter inception cohort. Arthritis Care Res. 2010, 62, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Kunjir, V.; Venugopalan, A.; Chopra, A. Profile of Indian patients with juvenile onset chronic inflammatory joint disease using the ILAR classification criteria for JIA: A community-based cohort study. J. Rheumatol. 2010, 37, 1756–1762. [Google Scholar] [CrossRef]
- Weiss, P.F.; Beukelman, T.; Schanberg, L.E.; Kimura, Y.; Colbert, R.A. Enthesitis-related arthritis is associated with higher pain intensity and poorer health status in comparison with other categories of juvenile idiopathic arthritis: The Childhood Arthritis and Rheumatology Research Alliance Registry. J. Rheumatol. 2012, 39, 2341–2351. [Google Scholar] [CrossRef]
- Weiß, A.; Minden, K.; Listing, J.; Foeldvari, I.; Sieper, J.; Rudwaleit, M. Course of patients with juvenile spondyloarthritis during 4 years of observation, juvenile part of GESPIC. RMD Open 2017, 3, e000366. [Google Scholar] [CrossRef]
- Gomez, K.S.; Raza, K.; Jones, S.D.; Kennedy, L.G.; Calin, A. Juvenile onset ankylosing spondylitis–more girls than we thought? J. Rheumatol. 1997, 24, 735–737. [Google Scholar]
- Hofer, M. Spondylarthropathies in children--are they different from those in adults? Best Pract. Res. Clin. Rheumatol. 2006, 20, 315–328. [Google Scholar] [CrossRef]
- Weiss, P.F.; Chauvin, N.A.; Roth, J. Imaging in Juvenile Spondyloarthritis. Curr. Rheumatol. Rep. 2016, 18, 75. [Google Scholar] [CrossRef]
- Weiss, P.F.; Xiao, R.; Biko, D.M.; Chauvin, N.A. Assessment of Sacroiliitis at Diagnosis of Juvenile Spondyloarthritis by Radiography, Magnetic Resonance Imaging, and Clinical Examination. Arthritis Care Res. 2016, 68, 187–194. [Google Scholar] [CrossRef]
- Petty, R.E.; Southwood, T.R.; Manners, P.; Baum, J.; Glass, D.N.; Goldenberg, J.; He, X.; Maldonado-Cocco, J.; Orozco-Alcala, J.; Prieur, A.M.; et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: Second revision, Edmonton, 2001. J. Rheumatol. 2004, 31, 390–392. [Google Scholar] [PubMed]
- Burgos-Vargas, R.; Rudwaleit, M.; Sieper, J. The place of juvenile onset spondyloarthropathies in the Durban 1997 ILAR classification criteria of juvenile idiopathic arthritis. International League of Associations for Rheumatology. J. Rheumatol. 2002, 29, 869–874. [Google Scholar] [PubMed]
- Rudwaleit, M.; Landewé, R.; van der Heijde, D.; Listing, J.; Brandt, J.; Braun, J.; Burgos-Vargas, R.; Collantes-Estevez, E.; Davis, J.; Dijkmans, B.; et al. The development of Assessment of SpondyloArthritis international Society classification criteria for axial spondyloarthritis (part I): Classification of paper patients by expert opinion including uncertainty appraisal. Ann. Rheum. Dis. 2009, 68, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Rudwaleit, M.; van der Heijde, D.; Landewé, R.; Listing, J.; Akkoc, N.; Brandt, J.; Braun, J.; Chou, C.T.; Collantes-Estevez, E.; Dougados, M.; et al. The development of Assessment of SpondyloArthritis international Society classification criteria for axial spondyloarthritis (part II): Validation and final selection. Ann. Rheum. Dis. 2009, 68, 777–783. [Google Scholar] [CrossRef]
- Rudwaleit, M.; van der Heijde, D.; Landewé, R.; Akkoc, N.; Brandt, J.; Chou, C.T.; Dougados, M.; Huang, F.; Gu, J.; Kirazli, Y.; et al. The Assessment of SpondyloArthritis International Society classification criteria for peripheral spondyloarthritis and for spondyloarthritis in general. Ann. Rheum. Dis. 2011, 70, 25–31. [Google Scholar] [CrossRef]
- Weiss, P.F.; Brandon, T.G.; Aggarwal, A.; Burgos-Vargas, R.; Colbert, R.A.; Horneff, G.; Laxer, R.M.; Minden, K.; Ravelli, A.; Ruperto, N.; et al. Classification Criteria for Axial Disease in Youth with Juvenile Spondyloarthritis. Arthritis Rheumatol. 2024, 76, 1797–1808. [Google Scholar] [CrossRef]
- Petty, R.E.; Southwood, T.R.; Baum, J.; Bhettay, E.; Glass, D.N.; Manners, P.; Maldonado-Cocco, J.; Suarez-Almazor, M.; Orozco-Alcala, J.; Prieur, A.M. Revision of the proposed classification criteria for juvenile idiopathic arthritis: Durban, 1997. J. Rheumatol. 1998, 25, 1991–1994. [Google Scholar]
- Dougados, M.; van der Linden, S.; Juhlin, R.; Huitfeldt, B.; Amor, B.; Calin, A.; Cats, A.; Dijkmans, B.; Olivieri, I.; Pasero, G.; et al. The European Spondylarthropathy Study Group preliminary criteria for the classification of spondylarthropathy. Arthritis Rheum. 1991, 34, 1218–1227. [Google Scholar] [CrossRef]
- Aydıngöz, Ü.; Yıldız, A.E.; Ayaz, E.; Batu, E.D.; Özen, S. Preferential involvement of the pelvis and hips along with active sacroiliitis in chronic nonbacterial osteomyelitis: MRI of 97 patients from a single tertiary referral center. Eur. Radiol. 2024, 34, 4979–4987. [Google Scholar] [CrossRef]
- Burgos-Vargas, R. The assessment of the spondyloarthritis international society concept and criteria for the classification of axial spondyloarthritis and peripheral spondyloarthritis: A critical appraisal for the pediatric rheumatologist. Pediatr. Rheumatol. Online J. 2012, 10, 14. [Google Scholar] [CrossRef]
- Althoff, C.E.; Feist, E.; Burova, E.; Eshed, I.; Bollow, M.; Hamm, B.; Hermann, K.G. Magnetic resonance imaging of active sacroiliitis: Do we really need gadolinium? Eur. J. Radiol. 2009, 71, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Demir, S.; Ergen, F.B.; Taydaş, O.; Sağ, E.; Bilginer, Y.; Aydıngöz, Ü.; Özen, S. Spinal involvement in juvenile idiopathic arthritis: What do we miss without imaging? Rheumatol. Int. 2022, 42, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Giménez-Roca, C.; Iglesias, E.; Torrente-Segarra, V.; Bou, R.; Sánchez-Manubens, J.; Calzada-Hernández, J.; Hernández, S.; Ricart, S.; Antón, J. Efficacy and safety of TNF-alpha antagonists in children with juvenile idiopathic arthritis who started treatment under 4 years of age. Rheumatol. Int. 2015, 35, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Donnithorne, K.J.; Cron, R.Q.; Beukelman, T. Attainment of inactive disease status following initiation of TNF-α inhibitor therapy for juvenile idiopathic arthritis: Enthesitis-related arthritis predicts persistent active disease. J. Rheumatol. 2011, 38, 2675–2681. [Google Scholar] [CrossRef]
- Bray, T.J.P.; Lopes, A.; Fisher, C.; Ciurtin, C.; Sen, D.; Hall-Craggs, M.A. Sacroiliac Joint Ankylosis in Young Spondyloarthritis Patients Receiving Biologic Therapy: Observation of Serial Magnetic Resonance Imaging Scans. Arthritis Rheumatol. 2019, 71, 594–598. [Google Scholar] [CrossRef]
- Goirand, M.; Breton, S.; Chevallier, F.; Duong, N.P.; Uettwiller, F.; Melki, I.; Mouy, R.; Wouters, C.; Bader-Meunier, B.; Job-Deslandre, C.; et al. Clinical features of children with enthesitis-related juvenile idiopathic arthritis/juvenile spondyloarthritis followed in a French tertiary care pediatric rheumatology centre. Pediatr. Rheumatol. Online J. 2018, 16, 21. [Google Scholar] [CrossRef]
- Zhao, L.; Liao, Z.; Yu, D.; Li, T.; Gu, J. Evaluation of the European Spondyloarthropathy Study Group (ESSG) classification criteria in a Chinese population. Clin. Exp. Rheumatol. 2005, 23, 397–399. [Google Scholar]
- Prieur, A.M.; Listrat, V.; Dougados, M.; Amor, B. Criteria for classification of spondylarthropathies in children. Arch. Fr. Pediatr. 1993, 50, 379–385. [Google Scholar]
| Criteria Domains and Levels | Maximum | |||
|---|---|---|---|---|
| Level | Weight | Domain Score | ||
| Imaging: active inflammation | ||||
| 0 | No unequivocal evidence of active lesions typical of sacroiliitis on MRI of the SIJs | 0 | ||
| 1 | Unequivocal evidence of active lesions typical of sacroiliitis on MRI of the SIJs | 23 | 23 | |
| Imaging: structural lesions | ||||
| 0 | No unequivocal evidence of structural lesions typical of sacroiliitis on MRI of the SIJs | 0 | ||
| 1 | Unequivocal evidence of sacroiliitis on radiograph ^ | 13 | ||
| 2 | Unequivocal evidence of structural lesions typical of sacroiliitis on MRI of the SIJs | 23 | 23 | |
| Pain chronicity | ||||
| 0 | No pain or pain < 4 days per week or for <6 weeks | 0 | ||
| 1 | Most days (≥4 days/week) for ≥6 but <12 weeks | 6 | ||
| 2 | Most days (≥4 days/week) ≥ 12 weeks | 9 | 9 | |
| Pain pattern | ||||
| 0 | No identifiable pain pattern | 0 | ||
| 1 | Awakens patient second half of the night OR insidious onset | 6 | ||
| 2 | Moderate to total relief with non-steroidal anti-inflammatory drugs | 10 | ||
| 3 | Improves with activity | 13 | 13 | |
| Pain location | ||||
| 0 | No back, groin, hip, or buttock pain | 0 | ||
| 1 | Lumbar spine pain (patient-reported) | 6 | ||
| 2 | Sacroiliac pain with deep palpation/FABER/Mennell/Gaenslen maneuvers # OR groin/hip pain (patient-reported) | 11 | ||
| 3 | Sacral/buttock pain (patient-reported) | 12 | 12 | |
| Stiffness (morning) | ||||
| 0 | No stiffness or stiffness < 15 min | 0 | ||
| 1 | ≥15 min | 9 | 9 | |
| Genetics | ||||
| 0 | No relevant family history in 1st degree relative and/or HLA-B27 unknown or negative | 0 | ||
| 1 | Presence of the following in a 1st degree relative: SpA or HLA-B27-associated acute (symptomatic) anterior uveitis | 8 | ||
| 2 | Presence of HLA-B27 | 11 | 11 | |
| ILAR Classification Criteria for Enthesitis-Related Arthritis [17] | ASAS Classification Criteria for Peripheral Spondyloarthritis [15] | ASAS Classification Criteria for Axial Spondyloarthritis [13] | ESSG Criteria [18] |
| Inclusion criteria Arthritis and enthesitis OR Arthritis or enthesitis plus two of the following:
Exclusion criteria
| Arthritis and/or enthesitis and/or dactylitis plus the following: ≥1 SpA feature
≥2 other SpA features
| In patients with ≥3 months of back pain and age < 45 years Sacroiliitis on imaging plus ≥ 1 SpA feature OR HLA-B27 plus ≥2 other SpA features SpA features
| Inflammatory back pain or synovitis (asymmetric or predominantly in the lower limbs) plus ≥1 of the following:
|
| ILAR sacroiliac joint arthritis (pediatric): Presence of tenderness upon direct compression over the sacroiliac joints [11]. | |||
| JSpA Patients (n = 123) | Control Group (n = 74) | p Value | |
|---|---|---|---|
| Sex, female n (%) | 32 (26%) | 35 (47.3%) | 0.002 |
| Age at the time of diagnosis (mean ± SD years) | 13 ± 2.8 | 10.7 ± 3.8 | <0.001 |
| Follow-up time (months) | 98 ± 43 | 37.6 ± 38.1 | <0.001 |
| HLA-B27 presence n (%) | 69 (56.1%) | 4 (5.4%) | <0.001 |
| Family history of SpA n (%) | 17 (13.8%) | 3 (4.1%) | 0.028 |
| Peripheral arthritis n (%) | 80 (65%) | 23 (31.1%) | <0.001 |
| Enthesitis n (%) | 59 (48%) | 10 (13.5%) | <0.001 |
| SIJ pain with direct palpation or FABER/Mennell/Gaenslen’s maneuvers * n (%) | 79 (64.2%) | 22 (29.7%) | <0.001 |
| Acute anterior uveitis n (%) | 4 (3.3%) | 0 | 0.149 |
| Inflammatory bowel disease n (%) | 5 (4.1%) | 3 (4.1%) | 0.652 |
| Psoriasis n (%) | 0 | 3 (4.1%) | 0.052 |
| Active sacroiliitis (MRI) n (%) | 78 (63.4%) | 33 (44.5%) | 0.010 |
| Chronic sacroiliitis (MRI) n (%) | 59 (47.9%) | 14 (18.9%) | <0.001 |
| Sensitivity (n = 123) | Specificity (n = 74) | Positive Predictive Value | Negative Predictive Value | |
|---|---|---|---|---|
| jAxSPA | 61% (74/123) | 77% (57/74) | 81.30% | 53.70% |
| ASAS axial | 74% (91/123) | 65% (48/74) | 77.80% | 60.80% |
| ASAS peripheral | 67% (83/123) | 84% (62/74) | 87.40% | 60.80% |
| ESSG | 92% (113/123) | 68% (50/74) | 82.50% | 83.30% |
| ILAR | 85% (105/123) | 81% (60/74) | 88.20% | 76.90% |
| ILAR + SI | 67% (82/123) | 74% (55/74) | 81.20% | 57.30% |
| Parameters of the AxJSpA Classification Criteria | Case Group (%) | Control Group (%) |
|---|---|---|
| Imaging: active inflammation | 63.4 | 45 |
| Imaging: structural lesions | 48 | 20 |
| Pain chronicity | ||
| No pain or pain < 4 days per week or for <6 weeks | 1.6 | 16.2 |
| Most days (≥4 days/week) for ≥6 but <12 weeks | 8.1 | 24.3 |
| Most days (≥4 days/week) ≥ 12 weeks | 90.2 | 59.5 |
| Pain pattern | ||
| No identifiable pain pattern | 54.5 | 47.3 |
| Awakens patient second half of the night OR insidious onset | 4.1 | 12.2 |
| Moderate to total relief with non-steroidal anti-inflammatory drugs | 25.2 | 23 |
| Improves with activity | 16.3 | 17.6 |
| Pain location | ||
| Lumbar spine pain (patient-reported) | 10.6 | 21.6 |
| Sacroiliac pain with deep palpation/FABER/Mennell/Gaenslen maneuvers # OR groin/hip pain | 1.6 | 6.8 |
| Sacral/buttock pain (patient-reported) | 78.9 | 41.9 |
| Stiffness (morning) | ||
| ≥15 min | 55.3 | 77 |
| Genetics | ||
| Presence of the following in a 1st degree relative: SpA or HLA-B27-associated acute anterior uveitis | 4.1 | 2.7 |
| Presence of HLA-B27 | 55.3 | 5.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Unal, D.; Tatar, C.A.; Sag, E.; Bayindir, Y.; Aliyev, E.; Cam, V.; Ercan Emreol, H.; Erkan, M.O.; Basaran, O.; Bilginer, Y.; et al. Validation of Axial Juvenile Spondyloarthropathy Criteria in Turkish Juvenile Spondyloarthropathy Patients. Diagnostics 2025, 15, 1498. https://doi.org/10.3390/diagnostics15121498
Unal D, Tatar CA, Sag E, Bayindir Y, Aliyev E, Cam V, Ercan Emreol H, Erkan MO, Basaran O, Bilginer Y, et al. Validation of Axial Juvenile Spondyloarthropathy Criteria in Turkish Juvenile Spondyloarthropathy Patients. Diagnostics. 2025; 15(12):1498. https://doi.org/10.3390/diagnostics15121498
Chicago/Turabian StyleUnal, Dilara, Cansu Ayten Tatar, Erdal Sag, Yagmur Bayindir, Emil Aliyev, Veysel Cam, Hulya Ercan Emreol, Mehmet Orhan Erkan, Ozge Basaran, Yelda Bilginer, and et al. 2025. "Validation of Axial Juvenile Spondyloarthropathy Criteria in Turkish Juvenile Spondyloarthropathy Patients" Diagnostics 15, no. 12: 1498. https://doi.org/10.3390/diagnostics15121498
APA StyleUnal, D., Tatar, C. A., Sag, E., Bayindir, Y., Aliyev, E., Cam, V., Ercan Emreol, H., Erkan, M. O., Basaran, O., Bilginer, Y., & Ozen, S. (2025). Validation of Axial Juvenile Spondyloarthropathy Criteria in Turkish Juvenile Spondyloarthropathy Patients. Diagnostics, 15(12), 1498. https://doi.org/10.3390/diagnostics15121498





