Predictive Value of Dual-Energy CT-Derived Metrics for the Use of Bone Substitutes in Distal Radius Fracture Surgery
Abstract
1. Introduction
2. Materials and Methods
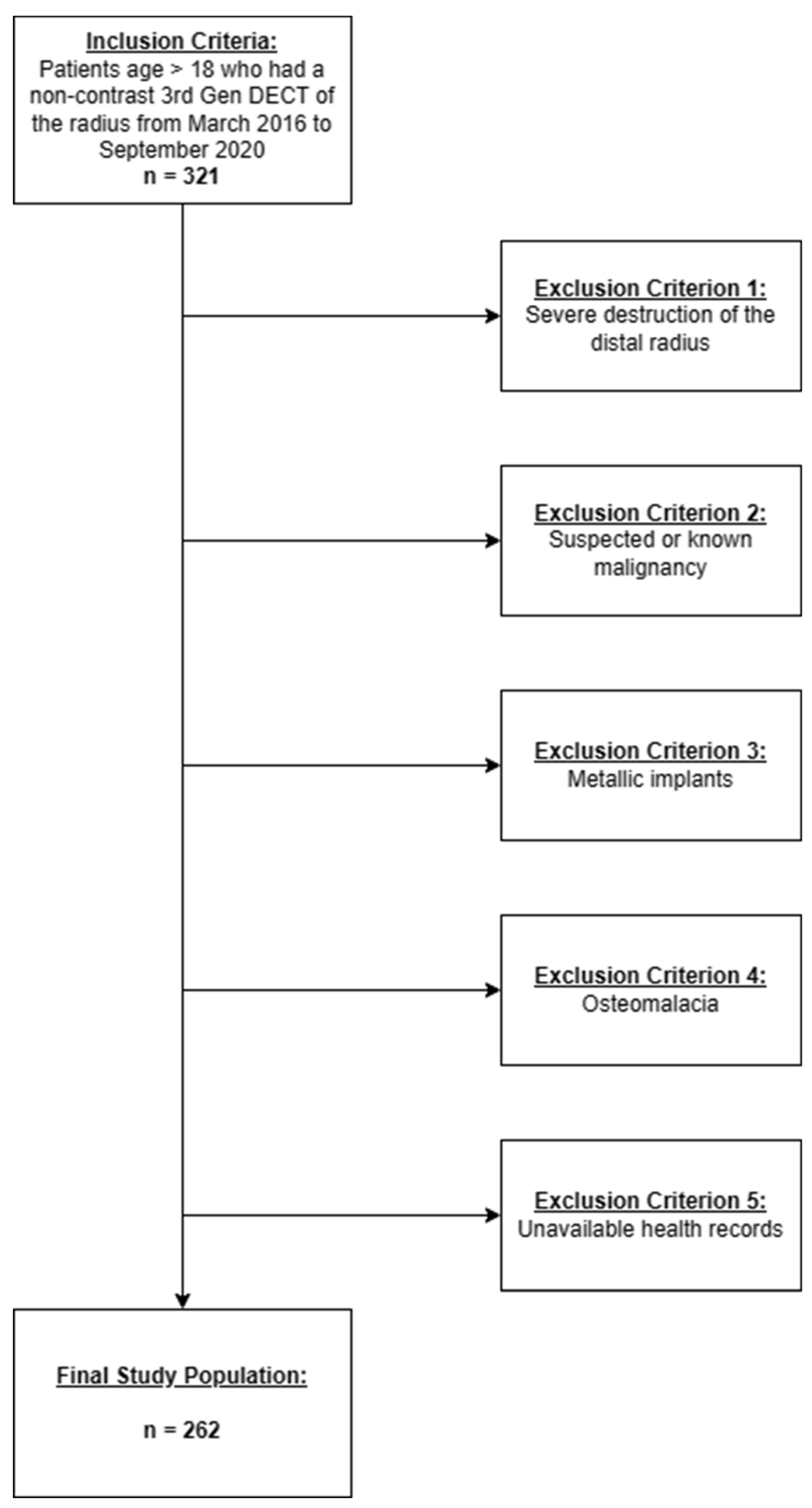
2.1. Patient Selection
2.2. Imaging Protocol
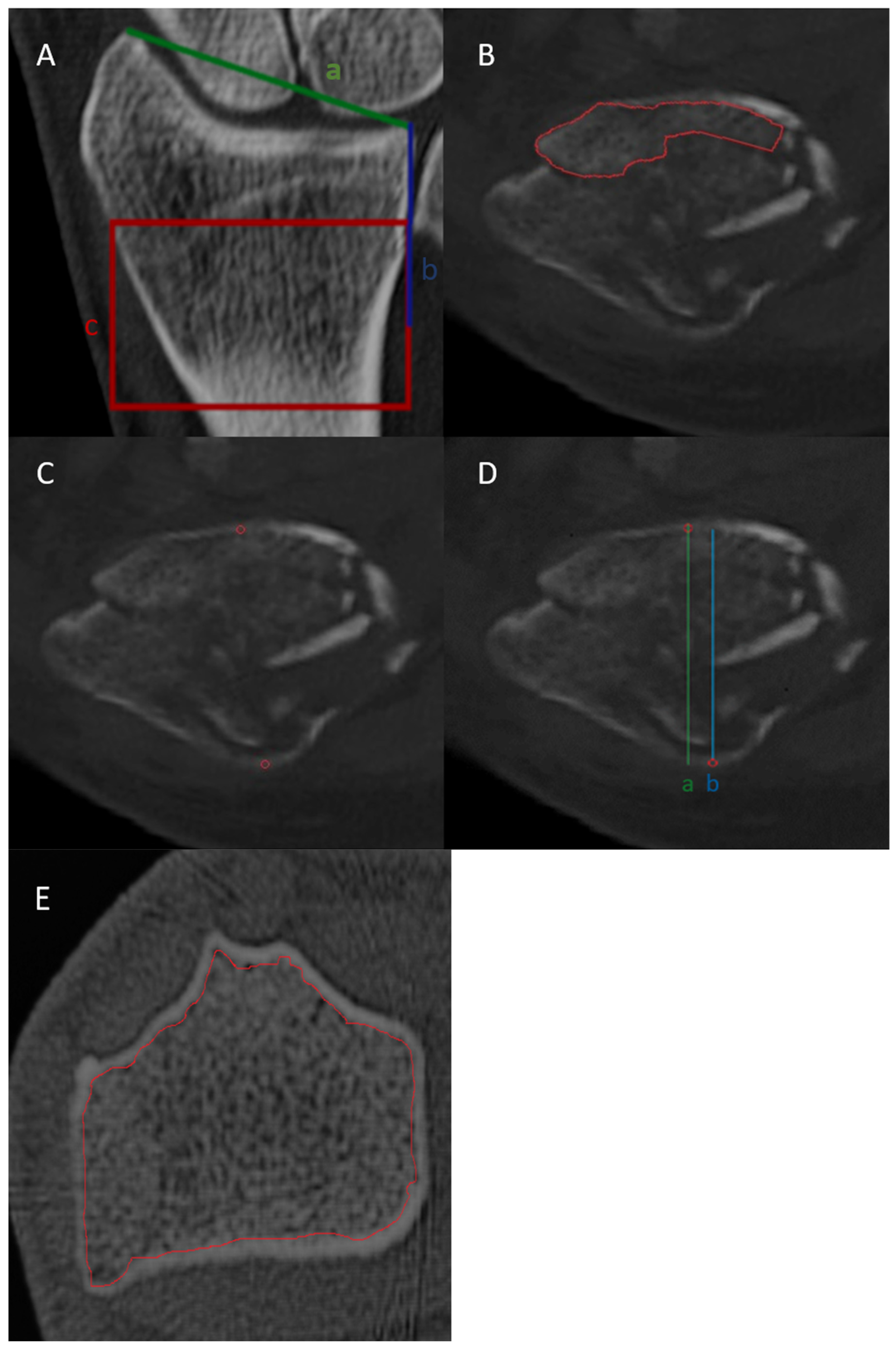
2.3. Image Interpretation
2.4. BMD Assessment
2.5. Statistical Analysis
3. Results
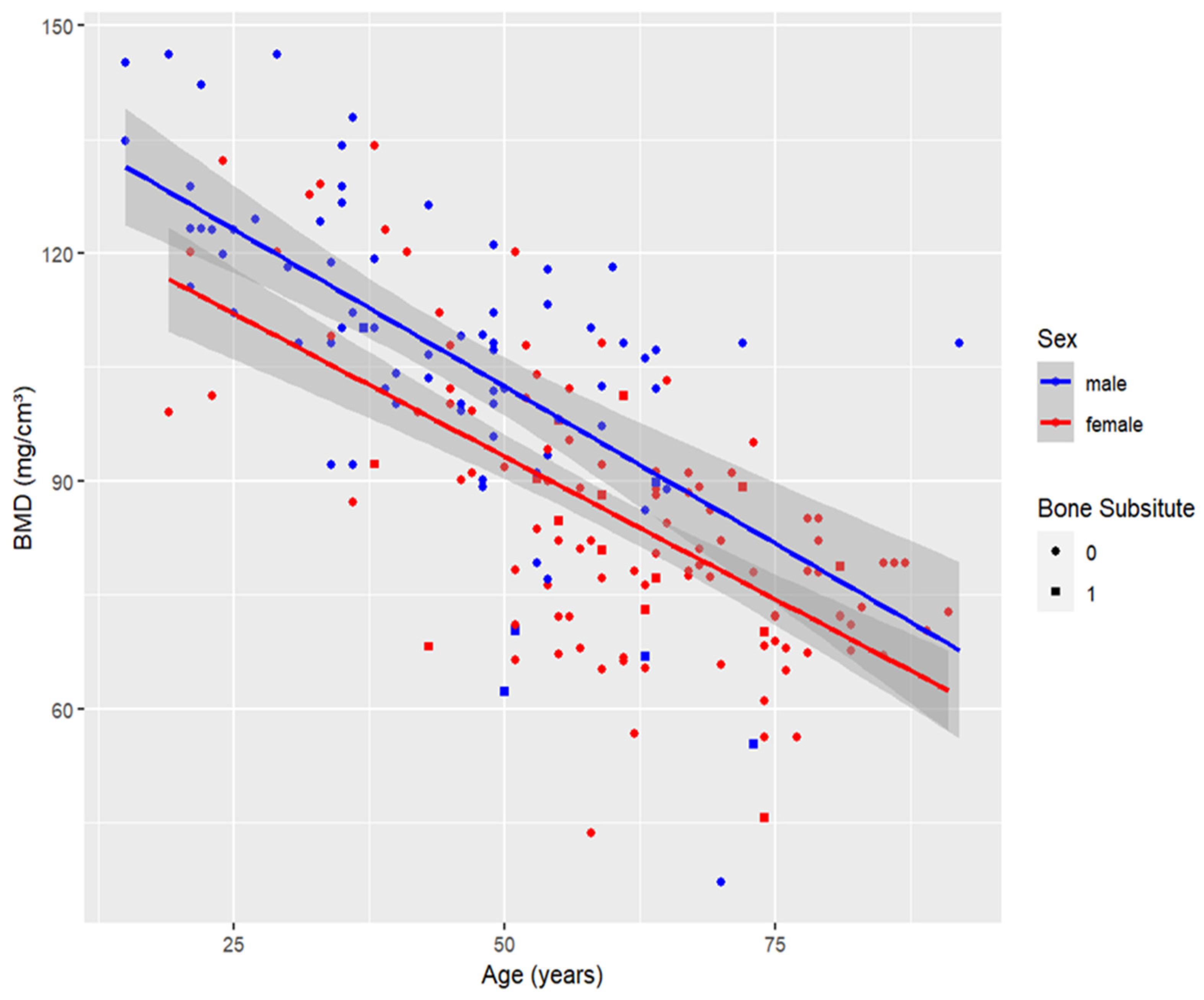
3.1. Patient Characteristics
3.2. Logistic Regression Analysis
3.3. Optimal Threshold
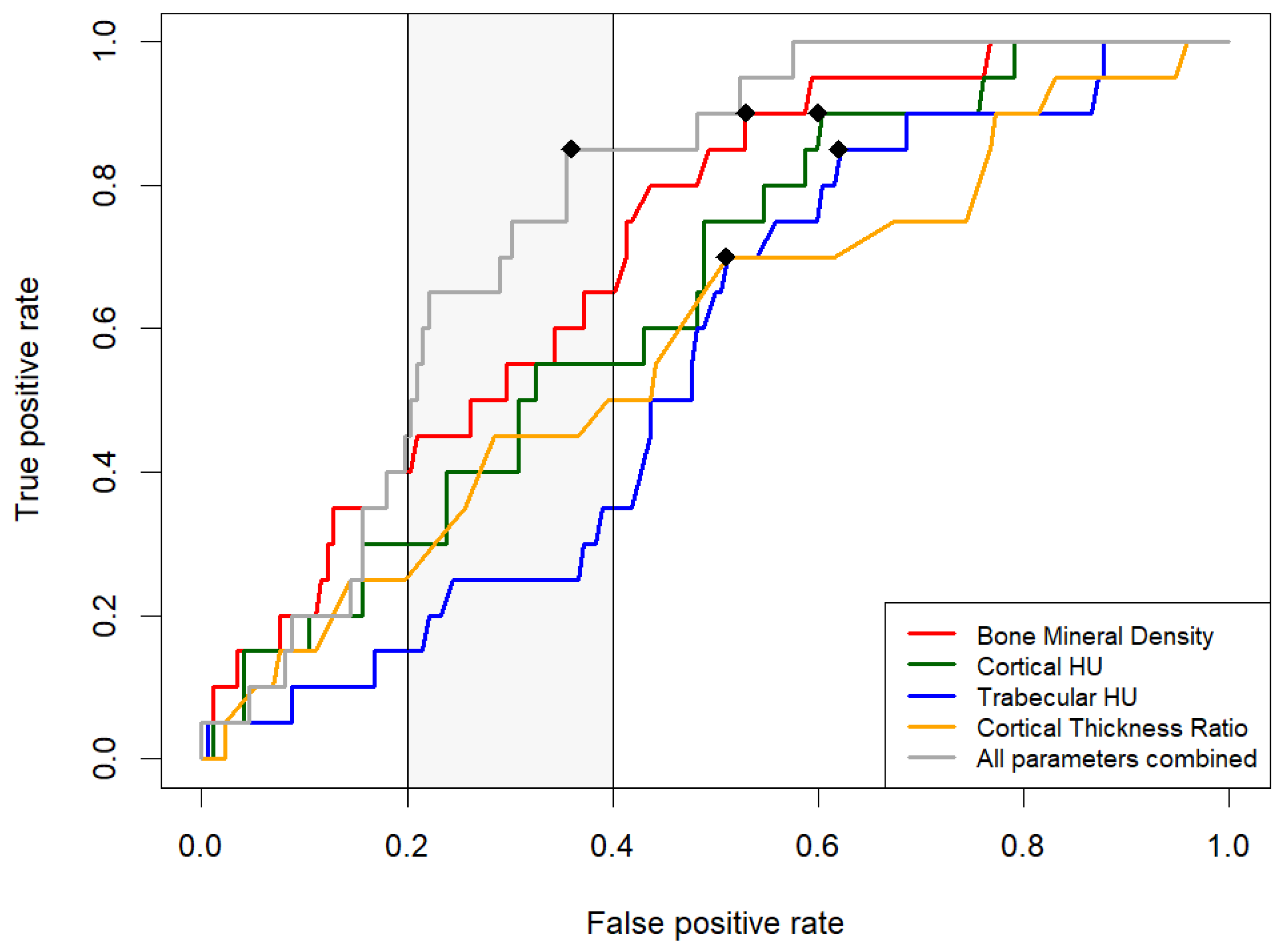
3.4. Comparative Analysis of Measurement Methods via ROC Curves
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Levin, L.S.; Rozell, J.C.; Pulos, N. Distal Radius Fractures in the Elderly. J. Am. Acad. Orthop. Surg. 2017, 25, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An Overview and Management of Osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Chang, F.-S.; Chen, C.-H.; Lee, C.-H.; Lee, K.-T.; Cho, Y.-C. Evaluating the Necessity of Bone Augmentation for Distal Radius Fracture Fixed with a Volar Locking Plate: A Retrospective Study. BMC Musculoskelet. Disord. 2020, 21, 180. [Google Scholar] [CrossRef] [PubMed]
- Kandeel, A.A.-M. Elderly Unstable Distal Radius Fractures a Prospective Cohort Study of Bone Substitutes-Augmented Percutaneous Pinning. BMC Musculoskelet. Disord. 2022, 23, 239. [Google Scholar] [CrossRef] [PubMed]
- Pawelke, J.; Vinayahalingam, V.; El Khassawna, T.; Heiss, C.; Eckl, L.; Knapp, G. Complication and Infection Risk Using Bone Substitute Materials to Treat Long Bone Defects in Geriatric Patients: An Observational Study. Medicina 2023, 59, 365. [Google Scholar] [CrossRef]
- Ciocca, L.; De Crescenzio, F.; Fantini, M.; Scotti, R. CAD/CAM and Rapid Prototyped Scaffold Construction for Bone Regenerative Medicine and Surgical Transfer of Virtual Planning: A Pilot Study. Comput. Med. Imaging Graph. 2009, 33, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-J.; Zhang, C.; Ma, C.; Qi, H.; Yang, Z.-H.; Wu, H.-Y.; Yang, K.-D.; Lin, J.-Y.; Wong, T.-M.; Li, Z.-Y.; et al. Automatic Phantom-Less QCT System with High Precision of BMD Measurement for Osteoporosis Screening: Technique Optimisation and Clinical Validation. J. Orthop. Transl. 2022, 33, 24–30. [Google Scholar] [CrossRef] [PubMed]
- O’Gorman, C.A.; Milne, S.; Lambe, G.; Sobota, A.; Beddy, P.; Gleeson, N. Accuracy of Opportunistic Bone Mineral Density Assessment on Staging Computed Tomography for Gynaecological Cancers. Medicina 2021, 57, 1386. [Google Scholar] [CrossRef] [PubMed]
- Pickhardt, P.J.; Pooler, B.D.; Lauder, T.; del Rio, A.M.; Bruce, R.J.; Binkley, N. Opportunistic Screening for Osteoporosis Using Abdominal Computed Tomography Scans Obtained for Other Indications. Ann. Intern. Med. 2013, 158, 588–595. [Google Scholar] [CrossRef]
- Merheb, J.; Van Assche, N.; Coucke, W.; Jacobs, R.; Naert, I.; Quirynen, M. Relationship between Cortical Bone Thickness or Computerized Tomography-Derived Bone Density Values and Implant Stability. Clin. Oral Implant. Res. 2010, 21, 612–617. [Google Scholar] [CrossRef]
- Xu, G.; Wang, D.; Zhang, H.; Xu, C.; Li, H.; Zhang, W.; Li, J.; Zhang, L.; Tang, P. Prediction of Osteoporosis from Proximal Femoral Cortical Bone Thickness and Hounsfield Unit Value with Clinical Significance. Front. Surg. 2022, 9, 1047603. [Google Scholar] [CrossRef] [PubMed]
- Mazess, R.B. Errors in Measuring Trabecular Bone by Computed Tomography Due to Marrow and Bone Composition. Calcif. Tissue Int. 1983, 35, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Wesarg, S.; Kirschner, M.; Becker, M.; Erdt, M.; Kafchitsas, K.; Khan, M.F. Dual-Energy CT-Based Assessment of the Trabecular Bone in Vertebrae. Methods Inf. Med. 2012, 51, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Nickoloff, E.L.; Feldman, F.; Atherton, J.V. Bone Mineral Assessment: New Dual-Energy CT Approach. Radiology 1988, 168, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Gruenewald, L.D.; Koch, V.; Martin, S.S.; Yel, I.; Eichler, K.; Gruber-Rouh, T.; Lenga, L.; Wichmann, J.L.; Alizadeh, L.S.; Albrecht, M.H.; et al. Diagnostic Accuracy of Quantitative Dual-Energy CT-Based Volumetric Bone Mineral Density Assessment for the Prediction of Osteoporosis-Associated Fractures. Eur. Radiol. 2022, 32, 3076–3084. [Google Scholar] [CrossRef] [PubMed]
- Reschke, P.; Gotta, J.; Stahl, A.; Koch, V.; Mader, C.; Martin, S.S.; Scholtz, J.-E.; Booz, C.; Yel, I.; Hescheler, D.A.; et al. Value of Dual-Energy CT-Derived Metrics for the Prediction of Bone Non-union in Distal Radius Fractures. Acad. Radiol. 2024. [Google Scholar] [CrossRef] [PubMed]
- Gruenewald, L.D.; Koch, V.; Martin, S.S.; Yel, I.; Mahmoudi, S.; Bernatz, S.; Eichler, K.; Gruber-Rouh, T.; Pinto Dos Santos, D.; D’Angelo, T.; et al. Dual-Energy CT-Based Opportunistic Volumetric Bone Mineral Density Assessment of the Distal Radius. Radiology 2023, 308, e223150. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, K.E.; Larkin, E.A.; Aznar, M.C.; Vasquez Osorio, E. Dual-Energy Computed Tomography: Survey Results on Current Uses and Barriers to Further Implementation. Br. J. Radiol. 2021, 94, 20210565. [Google Scholar] [CrossRef]
- KNellans, K.W.; Kowalski, E.; Chung, K.C. The Epidemiology of Distal Radius Fractures. Hand Clin. 2012, 28, 113–125. [Google Scholar] [CrossRef]
- Yu, E.W.; Thomas, B.J.; Brown, J.K.; Finkelstein, J.S. Simulated Increases in Body Fat and Errors in Bone Mineral Density Measurements by DXA and QCT. J. Bone Miner. Res. 2012, 27, 119–124. [Google Scholar] [CrossRef]
- Miclau, T.; Lu, C.; Thompson, Z.; Choi, P.; Puttlitz, C.; Marcucio, R.; Helms, J.A. The Effects of Delayed Stabilization on Fracture Healing. J. Orthop. Res. 2007, 25, 1552–1558. [Google Scholar] [CrossRef] [PubMed]

| Variables | Total DRF (n = 262) | No Bone Substitutes (n = 234) | Bone Substitutes (n = 28) | p-Value |
|---|---|---|---|---|
| Age (years) | 55 (43–67) | 54 (40–64) | 60 (52–66) | 0.15 |
| Male (n) | 103 | 94 | 9 | |
| Female (n) | 159 | 140 | 19 | 0.53 |
| BMD (mg/cm3) median (IQR) | 91.15 (77.98–108.2) | 93.75 (78.18–109.43) | 79.85 (69.63–89.9) | 0.002 |
| Trabecular HU median (IQR) | 31 (−19.5, –84.75) | 35 (−19.5–89) | 22.5 (−0.25–47) | 0.03 |
| Cortical HU median (IQR) | 1688 (1531.5–1837) | 1713 (1550.75–1845.25) | 1597.5 (1447–1712.5) | 0.43 |
| Cortical thickness ratio median (IQR) | 1.36 (1.29–1.45) | 1.37 (1.29–1.45) | 1.34 (1.27–1.42) | 0.23 |
| A Fracture (n) | 27 | 24 | 3 | 0.9 |
| B Fracture (n) | 39 | 36 | 3 | |
| C Fracture (n) | 196 | 174 | 22 |
| Bone Substitutes | Coefficient (β) | Odds Ratio | p-Value | n |
|---|---|---|---|---|
| BMD | −0.04 | 0.96 | 0.003 | 262 |
| Age 50–64 (n = 15) | −0.03 | 0.972 | 0.27 | 89 |
| Age 65–79 (n = 8) | −0.07 | 0.93 | 0.095 | 59 |
| Age ≥ 80 (n = 1) | −0.113 | 0.893 | 0.592 | 15 |
| Female Sex (n = 19) | −0.02 | 0.98 | 0.04 | 159 |
| Cortical HU | −0.001 | 0.99 | 0.06 | 262 |
| Age 50–64 (n = 15) | −0.003 | 0.99 | 0.02 | 81 |
| Age 65–79 (n = 8) | −0.003 | 0.99 | 0.13 | 50 |
| Age ≥ 80 (n = 1) | 0.001 | 1.001 | 0.863 | 15 |
| Female Sex (n = 19) | −0.001 | 0.99 | 0.17 | 159 |
| Trabecular HU | −0.003 | 0.99 | 0.33 | 262 |
| Age 50–64 (n = 15) | −0.002 | 0.99 | 0.02 | 81 |
| Age 65–79 (n = 8) | 0.008 | 1008 | 0.3 | 50 |
| Age ≥ 80 (n = 1) | −0.5 | 0.61 | 0.998 | 15 |
| Female Sex (n = 19) | −0.0003 | 1.0 | 0.94 | 159 |
| Cortical thickness ratio | −2.72 | 0.07 | 0.21 | 263 |
| Age 50–64 (n = 15) | −6.42 | 0.0016 | 0.07 | 81 |
| Age 65–79 (n = 8) | −1.03 | 0.036 | 0.84 | 50 |
| Age ≥ 80 (n = 1) | 3.439 | 3.02 | 0.999 | 15 |
| Female Sex (n = 19) | −3.28 | 0.04 | 0.22 | 159 |
| Variable | AUC [DeLong] | Partial AUC | Optimal Threshold | TPR | FPR | Specificity |
|---|---|---|---|---|---|---|
| BMD mg/cm3 | 0.71 (0.61–0.81) | 0.66 | 97.9 | 0.9 | 0.53 | 0.47 |
| Cortical HU | 0.65 (0.53–0.76) | 0.60 | 1778 | 0.9 | 0.6 | 0.4 |
| Trabecular HU | 0.55 (0.44–0.67) | NA | 55 | 0.85 | 0.62 | 0.38 |
| Cortical Thickness Ratio | 0.58 (0.45–0.72) | 0.58 | 1.37 | 0.7 | 0.51 | 0.49 |
| All variables combined | 0.76 (0.68–0.84) | 0.79 | - | 0.85 | 0.36 | 0.64 |
| Paired Variables | p-Value for AUC | p-Value for Partial AUC |
|---|---|---|
| BMD vs. Cortical HU | 0.43 | 0.44 |
| BMD vs. Trabecular HU | 0.04 | 0.05 |
| BMD vs. Cortical Thickness Ratio | 0.16 | 0.44 |
| Combined vs. Cortical HU | 0.06 | 0.04 |
| Combined vs. Trabecular HU | 0.003 | 0.001 |
| Combined vs. Cortical Thickness Ratio | 0.03 | 0.03 |
| Combined vs. BMD | 0.16 | 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reschke, P.; Koch, V.; Mahmoudi, S.; Booz, C.; Yel, I.; Gotta, J.; Stahl, A.; Reschke, R.; Scholtz, J.-E.; Martin, S.S.; et al. Predictive Value of Dual-Energy CT-Derived Metrics for the Use of Bone Substitutes in Distal Radius Fracture Surgery. Diagnostics 2024, 14, 697. https://doi.org/10.3390/diagnostics14070697
Reschke P, Koch V, Mahmoudi S, Booz C, Yel I, Gotta J, Stahl A, Reschke R, Scholtz J-E, Martin SS, et al. Predictive Value of Dual-Energy CT-Derived Metrics for the Use of Bone Substitutes in Distal Radius Fracture Surgery. Diagnostics. 2024; 14(7):697. https://doi.org/10.3390/diagnostics14070697
Chicago/Turabian StyleReschke, Philipp, Vitali Koch, Scherwin Mahmoudi, Christian Booz, Ibrahim Yel, Jennifer Gotta, Adrian Stahl, Robin Reschke, Jan-Erik Scholtz, Simon S. Martin, and et al. 2024. "Predictive Value of Dual-Energy CT-Derived Metrics for the Use of Bone Substitutes in Distal Radius Fracture Surgery" Diagnostics 14, no. 7: 697. https://doi.org/10.3390/diagnostics14070697
APA StyleReschke, P., Koch, V., Mahmoudi, S., Booz, C., Yel, I., Gotta, J., Stahl, A., Reschke, R., Scholtz, J.-E., Martin, S. S., Gruber-Rouh, T., Eichler, K., Vogl, T. J., & Gruenewald, L. D. (2024). Predictive Value of Dual-Energy CT-Derived Metrics for the Use of Bone Substitutes in Distal Radius Fracture Surgery. Diagnostics, 14(7), 697. https://doi.org/10.3390/diagnostics14070697








