Assessing Different PCR Master Mixes for Ultrarapid DNA Amplification: Important Analytical Parameters
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample
2.2. Primers
2.3. PCR Master Mixes
2.4. Plasmonic PCR Instrument
3. Results and Discussion
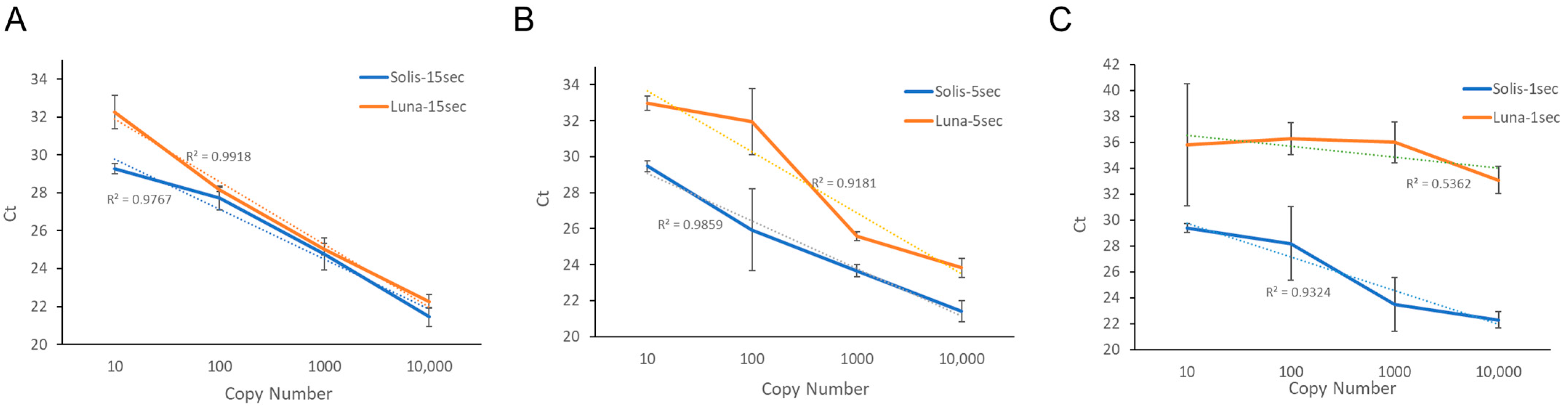
3.1. Thermal Cycling Profiles
3.2. Melting Curve Analysis—Amplification Accuracy of Solis PCR Assay
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahrberg, C.D.; Choi, J.W.; Lee, J.M.; Lee, K.G.; Lee, S.J.; Manz, A.; Chung, B.G. Plasmonic heating-based portable digital PCR system. Lab a Chip 2020, 20, 3560–3568. [Google Scholar] [CrossRef]
- An, Y.-Q.; Huang, S.-L.; Xi, B.-C.; Gong, X.-L.; Ji, J.-H.; Hu, Y.; Ding, Y.-J.; Zhang, D.-X.; Ge, S.-X.; Zhang, J.; et al. Ultrafast Microfluidic PCR Thermocycler for Nucleic Acid Amplification. Micromachines 2023, 14, 658. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Li, K.T. Analysis of PCR Kinetics inside a Microfluidic DNA Amplification System. Micromachines 2018, 9, 48. [Google Scholar] [CrossRef]
- Herold, K.E.; Sergeev, N.; Matviyenko, A.; Rasooly, A. Rapid DNA amplification using a battery-powered thin-film resistive thermocycler. Methods Mol. Biol. 2009, 504, 441–458. [Google Scholar]
- Hong, Y.; Kim, J.J.; Yu, Y.-C.; Kim, H.S.; Moon, G.; Park, E.M. Ultra-fast PCR method for the distinguishing between Miichthys miiuy and Sciaenops ocellatus. Food Sci. Biotechnol. 2021, 30, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Jalili, A.; Bagheri, M.; Shamloo, A.; Ashkezari, A.H.K. A plasmonic gold nanofilm-based microfluidic chip for rapid and inexpensive droplet-based photonic PCR. Sci. Rep. 2021, 11, 23338. [Google Scholar] [CrossRef]
- Liu, W.; Zhou, Z.; Zhang, L.; Li, L.; Wang, L.; Song, L.; Qiu, S.; Zhang, L.; Xu, D.; Tian, X.; et al. Establishment and evaluation of a 30-minute detection method for SARS-CoV-2 nucleic acid using a novel ultra-fast real-time PCR instrument. J. Thorac. Dis. 2021, 13, 6866–6875. [Google Scholar] [CrossRef]
- Wu, J.; Jiang, K.; Mi, H.; Qiu, Y.; Son, J.; Park, H.J.; Nam, J.-M.; Lee, J.-H. A rapid and sensitive fluorescence biosensor based on plasmonic PCR. Nanoscale 2021, 13, 7348–7354. [Google Scholar] [CrossRef] [PubMed]
- Mohammadyousef, P.; Paliouras, M.; Trifiro, M.A.; Kirk, A.G. Plasmonic and label-free real-time quantitative PCR for point-of-care diagnostics. Analyst 2021, 146, 5619–5630. [Google Scholar] [CrossRef] [PubMed]
- Roche, P.J.R.; Beitel, L.K.; Khan, R.; Lumbroso, R.; Najih, M.; Cheung, M.C.-K.; Thiemann, J.; Veerasubramanian, V.; Trifiro, M.; Chodavarapu, V.P.; et al. Demonstration of a plasmonic thermocycler for the amplification of human androgen receptor DNA. Analyst 2012, 137, 4475–4481. [Google Scholar] [CrossRef]
- Roche, P.J.; Najih, M.; Lee, S.S.; Beitel, L.K.; Carnevale, M.L.; Paliouras, M.; Kirk, A.G.; Trifiro, M.A. Real Time Plasmonic qPCR: How fast is Ultra-fast? 30 cycles in 54 seconds. Analyst 2017, 142, 1746–1755. [Google Scholar] [CrossRef]
- Uchehara, G.; Kirk, A.G.; Trifiro, M.; Paliouras, M.; Mohammadyousef, P. Real Time Label-Free Monitoring of Plasmonic Polymerase Chain Reaction Products; Nano-, Bio-, Info-Tech Sensors and 3d Systems Iii; Society of Photo-Optical Instrumentation Engineers (SPIE): Washington, DC, USA, 2019; p. 10969. [Google Scholar]
- Sarkar, S.L.; Alam, A.R.U.; Das, P.K.; Pramanik, M.H.A.; Al-Emran, H.M.; Jahid, I.K.; Hossain, M.A. Development and validation of cost-effective one-step multiplex RT-PCR assay for detecting the SARS-CoV-2 infection using SYBR Green melting curve analysis. Sci. Rep. 2022, 12, 6501. [Google Scholar] [CrossRef] [PubMed]
- Bulygin, A.A.; Kuznetsova, A.A.; Fedorova, O.S.; Kuznetsov, N.A. Comparative Analysis of Family A DNA-Polymerases as a Searching Tool for Enzymes with New Properties. Mol. Biol. 2023, 57, 185–196. [Google Scholar] [CrossRef]
- Millington, A.L.; Houskeeper, J.A.; Quackenbush, J.F.; Trauba, J.M.; Wittwer, C.T. The kinetic requirements of extreme qPCR. Biomol. Detect. Quantif. 2019, 17, 100081. [Google Scholar] [CrossRef] [PubMed]
- Farrar, J.S.; Wittwer, C.T. Extreme PCR: Efficient and Specific DNA Amplification in 15–60 Seconds. Clin. Chem. 2015, 61, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-H.; Liao, X.-J.; Chang, W.; Chiou, C.-C. Ultrafast DNA Amplification Using Microchannel Flow-Through PCR Device. Biosensors 2022, 12, 303. [Google Scholar] [CrossRef] [PubMed]
- Freire-Paspuel, B.; Morales-Jadan, D.; Zambrano-Mila, M.; Perez, F.; Garcia-Bereguiain, M.A. Analytical sensitivity and clinical performance of “COVID-19 RT-PCR Real TM FAST (CY5) (ATGen, Uruguay) and “ECUGEN SARS-CoV-2 RT-qPCR” (UDLA-STARNEWCORP, Ecuador)”: High quality-low cost local SARS-CoV-2 tests for South America. PLoS Neglected Trop. Dis. 2022, 16, e0010082. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, A.; Zhang, S.; Gao, S.; Rashid, M.; Li, Y.; Liu, J.; Ma, Q.; Li, Z.; Liu, Z.; et al. High resolution melting analysis of the 18S rRNA gene for the rapid diagnosis of bovine babesiosis. Parasites Vectors 2019, 12, 523. [Google Scholar] [CrossRef] [PubMed]
- Poritz, M.A.; Blaschke, A.J.; Byington, C.L.; Meyers, L.; Nilsson, K.; Jones, D.E.; Thatcher, S.A.; Robbins, T.; Lingenfelter, B.; Amiott, E.; et al. FilmArray, an automated nested multiplex PCR system for multi-pathogen detection: Development and application to respiratory tract infection. PLoS ONE 2011, 6, e26047. [Google Scholar] [CrossRef]
- Gong, L.; Yao, S.; He, Y.; Liu, C. Robust and Precise Quantitative Real-Time Polymerase Chain Reaction with an Amplification Efficiency-Aware Reaction Kinetics Model. Anal. Chem. 2023, 95, 5402–5410. [Google Scholar] [CrossRef]
- Jiang, K.; Wu, J.; Kim, J.-E.; An, S.; Nam, J.-M.; Peng, Y.-K.; Lee, J.-H. Plasmonic Cross-Linking Colorimetric PCR for Simple and Sensitive Nucleic Acid Detection. Nano Lett. 2023, 23, 3897–3903. [Google Scholar] [CrossRef] [PubMed]
- Katzman, B.M.; Wockenfus, A.M.; Kelley, B.R.; Karon, B.S.; Donato, L.J. Evaluation of the Visby medical COVID-19 point of care nucleic acid amplification test. Clin. Biochem. 2023, 117, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhao, S.; Jiang, L.; Wu, J.; Zhao, W.; Guo, X.; Peng, N.; Hu, F. A sample-to-answer DNA detection microfluidic system integrating sample pretreatment and smartphone-readable gradient plasmonic photothermal continuous-flow PCR. Analyst 2022, 147, 4876–4887. [Google Scholar] [CrossRef]
- Lim, H.; Jo, G.E.; Kim, K.S.; Back, S.M.; Choi, H. Miniaturized thermocycler based on thermoelectric heating for diagnosis of sexually transmitted disease by DNA amplification. Rev. Sci. Instrum. 2017, 88, 055001. [Google Scholar] [CrossRef] [PubMed]

| Luna Universal qPCR & RT-qPCR | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Copy Number | 15 s A/E | 5 s A/E | 1 s A/E | |||||||||
| Ct-s | Av. Ct | Tm-s | Tm CV | Ct-s | Av. Ct | Tm-s | Tm CV | Ct-s | Av. Ct | Tm-s | Tm CV | |
| 10 | 31.35 | 32.26 | 71.7/78.5 | 0.29 | 32.9 | 32.97 | 71/76 | n.d. | 37 | 35.77 | 71.3 | n.d. |
| 33.07 | 72.2/78.6 | 32.6 | 71.7 | 35.4 | 71.5 | |||||||
| 32.36 | 71.7/78.2 | 33.4 | 72 | 34.9 | 72.0 | |||||||
| 100 | 28.22 | 28.17 | 77.0 | 0.33 | 31.47 | 31.96 | 73.5/78.5 | 0.18 | 37.3 | 36.27 | 71.3 | n.d. |
| 28.05 | 76.8 | 33.7 | 70 * | 36.6 | 71.1 | |||||||
| 28.24 | 77.3 | 30.7 | 72.5/78.3 | 34.9 | 72.5 | |||||||
| 1000 | 24.7 | 25.04 | 77.9 | 0.46 | 25.35 | 25.59 | 77.83 | 0.51 | 37.53 | 36.04 | 73.8 | n.d. |
| 25.15 | 78.5 | 25.82 | 77.8 | 36.19 | 72.5 | |||||||
| 25.28 | 78.5 | 25.75 | 78.5 | 34.4 | 73.3 | |||||||
| 10,000 | 22.6 | 22.27 | 78.7 | 0.19 | 23.27 | 23.82 | 71.3/78 | 0.32 | 32.8 | 33.13 | 71.0 | n.d. |
| 22.3 | 78.8 | 24.3 | 71.9/78.3 | 32.3 | 72.4 | |||||||
| 21.9 | 78.5 | 23.9 | 71/78.5 | 34.3 | 72.2 | |||||||
| 0 | n.d. | n.d. | 72 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 72.3 | n.d. |
| n.d. | 71.6 | n.d. | n.d. | n.d. | 71.1 | |||||||
| n.d. | 72.1 | n.d. | n.d. | n.d. | 72.0 | |||||||
| Solis FAST qPCR Mix | ||||||||||||
| Copy Number | 15 s A/E | 5 s A/E | 1 s A/E | |||||||||
| Ct-s | Av. Ct | Tm-s | Tm CV | Ct-s | Av. Ct | Tm-s | Tm CV | Ct-s | Av. Ct | Tm-s | Tm CV | |
| 10 | 29.55 | 29.28 | 79.5 | 0.57 | 29.8 | 29.50 | 80 | 0.00 | 29 | 29.37 | 75/80 | 0.36 |
| 29.3 | 79.9 | 29.4 | 74/80 | 29.7 | 74/80 | |||||||
| 29 | 79 | 29.3 | 74/80 | 29.4 | 74/79.5 | |||||||
| 100 | 28.15 | 27.72 | 80 | 0.40 | 28.11 | 25.93 | 73/79 | 0.53 | 28.25 | 28.18 | 74/80 | 0.64 |
| 27 | 80 | 23.57 | 79.33 | 31 | 74/81 | |||||||
| 28 | 79.45 | 26.11 | 72.8/79.8 | 25.3 | 80.3 | |||||||
| 1000 | 24.18 | 24.78 | 79.1 | 0.94 | 23.41 | 23.66 | 80.8 | 0.67 | 21.96 | 23.52 | 80 | 0.32 |
| 25.37 | 80.7 | 23.9 | 79.9 | 25.86 | 79.3 | |||||||
| 24.8 | 80 | 27 * | 79.9 | 22.74 | 79.5 | |||||||
| 10,000 | 21.3 | 21.45 | 80.3 | 0.66 | 21.08 | 21.39 | 79.9 | 0.57 | 21.61 | 22.25 | 79.7 | 0.48 |
| 21.03 | 79.5 | 21.08 | 80.2 | 22.25 | 79.3 | |||||||
| 22.01 | 79.3 | 22 | 80.8 | 22.9 | 80.1 | |||||||
| 0 | n.d. | n.d. | 63 | n.d. | n.d. | n.d. | 73 | n.d. | n.d. | n.d. | 73 | n.d. |
| n.d. | 62.8 | n.d. | 63 | n.d. | 74 | |||||||
| n.d. | 63.2 | n.d. | 62.5 | n.d. | 62.5 | |||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brukner, I.; Paliouras, M.; Trifiro, M.; Bohbot, M.; Shamir, D.; Kirk, A.G. Assessing Different PCR Master Mixes for Ultrarapid DNA Amplification: Important Analytical Parameters. Diagnostics 2024, 14, 477. https://doi.org/10.3390/diagnostics14050477
Brukner I, Paliouras M, Trifiro M, Bohbot M, Shamir D, Kirk AG. Assessing Different PCR Master Mixes for Ultrarapid DNA Amplification: Important Analytical Parameters. Diagnostics. 2024; 14(5):477. https://doi.org/10.3390/diagnostics14050477
Chicago/Turabian StyleBrukner, Ivan, Miltiadis Paliouras, Mark Trifiro, Marc Bohbot, Daniel Shamir, and Andrew G. Kirk. 2024. "Assessing Different PCR Master Mixes for Ultrarapid DNA Amplification: Important Analytical Parameters" Diagnostics 14, no. 5: 477. https://doi.org/10.3390/diagnostics14050477
APA StyleBrukner, I., Paliouras, M., Trifiro, M., Bohbot, M., Shamir, D., & Kirk, A. G. (2024). Assessing Different PCR Master Mixes for Ultrarapid DNA Amplification: Important Analytical Parameters. Diagnostics, 14(5), 477. https://doi.org/10.3390/diagnostics14050477







