Pancreatic Adenocarcinoma: Imaging Modalities and the Role of Artificial Intelligence in Analyzing CT and MRI Images
Abstract
1. Introduction
2. Imaging Modalities
3. Methods
4. AI Applications in PDAC
4.1. Detection
| Author | Year | Modality | Approach | Sensitivity |
|---|---|---|---|---|
| Cao et al. [40] | 2023 | Non-contrast CT | Deep learning | 92.9% |
| Korifatis et al. [41] | 2023 | CECT | 3D-CNN | 75% |
| Alves et al. [46] | 2022 | CECT | CNN | N.A. |
| Wang et al. [48] | 2022 | CECT | Radiomics | 84% |
| Mukherjee et al. [47] | 2022 | CECT | Radiomics-ML | 95.5% |
| Viviers et al. [50] | 2022 | CECT | U-Net-Like Deep CNN | 99% |
| Ma et al. [45] | 2020 | Non-contrast CT | CNN | 91.58% |
| Chu et al. [42] | 2019 | CECT | Deep learning | 94.1% |
| Chu et al. [43] | 2019 | CECT | Radiomics | 100% |
4.2. Segmentation
| Author | Year | Modality | AI Method | DICE Score |
|---|---|---|---|---|
| Ni et al. [58] | 2023 | CECT | AX-Unet | 85.9% |
| Mahmoudi et al. [61] | 2022 | CECT | Hybrid 3D-CNN model | 60.6% |
| Turečková et al. [59] | 2020 | CECT | VNet-AG-DSV | 52.99% |
| Zhou et al. [53] | 2019 | CECT | 3D-ResDSN-multi-HPN | 63.94% |
| Isensee et al. [60] | 2018 | CECT | nnU-Net | 52.12% |
4.3. Classification
4.3.1. Differential Diagnosis
| Author | Year | Modality | Scope | Approach | AUC |
|---|---|---|---|---|---|
| Lu et al. [63] | 2023 | CECT | fAPI vs. PDAC | Radiomics | 0.83 |
| Shi et al. [68] | 2023 | MRI | PDAC vs. PNEN and SPN | CNN | 0.839 |
| Zhang et al. [70] | 2022 | CECT | PDAC vs. pNET | Radiomics | 0.930 |
| Anai et al. [65] | 2022 | CECT | fAPI vs. PDAC | Radiomics | 0.920 |
| Deng et al. [66] | 2021 | MRI | MFCP vs. PDAC | Radiomics | 0.962 |
| Ren et al. [67] | 2020 | CECT | PASC vs. PDAC | Radiomics | 0.82 |
| Park et al. [64] | 2020 | CECT | AIP vs. PDAC | Radiomics | 0.975 |
4.3.2. Histopathological Subtype and Genomic Features
| Author | Year | Modality | Scope | Approach | Performance |
|---|---|---|---|---|---|
| Cen et al. [79] | 2023 | CECT | Histopathological grade | Radiomics | AUC 0.76 |
| Deng et al. [86] | 2022 | MRI | MUC4 expression | Radiomics | AUC 0.861 |
| Hinzpeter et al. [81] | 2022 | CECT | Correlation with driver gene mutations | Radiomics | Youden Index 0.56 (KRAS), 0.67 (TP53), 0.5 (SMAD4 and CDKN2A) |
| Gao et al. [82] | 2021 | MRI | TP53 mutation | Radiomics | AUC 0.96 |
| Meng et al. [88] | 2021 | MRI | FAP expression | Radiomics | AUC 0.77 |
| Iwatate et al. [87] | 2020 | CECT | P53 and PD-L1 expression | Radiomics | AUC 0.795 and 0.683 |
| Qiu et al. [78] | 2019 | CECT | Histopathological grade | Radiomics | Accuracy 86% |
4.3.3. Prognosis
| Author | Year | Modality | Scope | Approach | Performance |
|---|---|---|---|---|---|
| Vezakis et al. [89] | 2023 | CECT | Survival prediction | Radiomics | C-index of 0.731 |
| Xu et al. [90] | 2023 | MRI | Survival prediction | Radiomics | C-index of 0.780 |
| Qiu et al. [91] | 2022 | MRI | Survival prediction | Radiomics | C-index of 0.814 |
| Li et al. [92] | 2022 | CECT | Risk of recurrence | Radiomics | AUC 0.764 for 1-year recurrence and AUC 0.773 for 2 year recurrence |
| Xie et al. [93] | 2020 | CECT | Survival prediction | Radiomics | C-index of 0.742 |
| Park et al. [94] | 2021 | CECT | Post-operative survival | Radiomics | C-index 0.7414 |
| Ni et al. [58] | 2023 | CECT | Recurrenceafter surgery | Radiomics | C-index 0.62 |
| Chen et al. [95] | 2020 | CECT | Surgical portal-superior mesenteric vein invasion | Radiomics | AUC 0.848 |
| Shi et al. [98] | 2022 | MRI | Lymph node metastasis | Radiomics | AUC 0.845 |
| Bian et al. [99] | 2022 | CECT | Lymph node metastasis | Radiomics | AUC 0.81 |
| Chang et al. [100] | 2022 | CECT | Lymph node metastasis and | 3D-CNN | Accuracy 90% for per-patient analysis and 75% for per-scan analysis |
5. Trends and Perspectives
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Author | Year | Type | Modality | Category |
|---|---|---|---|---|
| Cao et al. [40] | 2023 | Original Research | Non-contrast CT | Detection |
| Korifatis et al. [41] | 2023 | Original Research | CECT | Detection |
| Alves et al. [46] | 2022 | Original Research | CECT | Detection |
| Wang et al. [48] | 2022 | Original Research | CECT | Detection |
| Mukherjee et al. [47] | 2022 | Original Research | CECT | Detection |
| Viviers et al. [50] | 2022 | Original Research | CECT | Detection |
| Ma et al. [45] | 2020 | Original Research | Non-contrast CT | Detection |
| Chu et al. [42] | 2019 | Original Research | CECT | Detection |
| Chu et al. [43] | 2019 | Original Research | CECT | Detection |
| Ni et al. [58] | 2023 | Original Research | CECT | Segmentation |
| Mahmoudi et al. [61] | 2022 | Original Research | CECT | Segmentation |
| Turečková et al. [59] | 2020 | Original Research | CECT | Segmentation |
| Zhou et al. [53] | 2019 | Original Research | CECT | Segmentation |
| Isensee et al. [60] | 2018 | Original Research | CECT | Segmentation |
| Lu et al. [63] | 2023 | Original Research | CECT | Classification |
| Shi et al. [68] | 2023 | Original Research | MRI | Classification |
| Zhang et al. [70] | 2022 | Original Research | CECT | Classification |
| Anai et al. [65] | 2022 | Original Research | CECT | Classification |
| Deng et al. [66] | 2021 | Original Research | MRI | Classification |
| Ren et al. [67] | 2020 | Original Research | CECT | Classification |
| Park et al. [64] | 2020 | Original Research | CECT | Classification |
| Cen et al. [79] | 2023 | Original Research | CECT | Classification |
| Deng et al. [86] | 2022 | Original Research | MRI | Classification |
| Hinzpeter et al. [81] | 2022 | Original Research | CECT | Classification |
| Gao et al. [82] | 2021 | Original Research | MRI | Classification |
| Meng et al. [88] | 2021 | Original Research | MRI | Classification |
| Iwatate et al. [87] | 2020 | Original Research | CECT | Classification |
| Qiu et al. [78] | 2019 | Original Research | CECT | Classification |
| Vezakis et al. [89] | 2023 | Original Research | CECT | Classification |
| Xu et al. [90] | 2023 | Original Research | MRI | Classification |
| Qiu et al. [91] | 2022 | Original Research | MRI | Classification |
| Li et al. [92] | 2022 | Original Research | CECT | Classification |
| Xie et al. [93] | 2020 | Original Research | CECT | Classification |
| Park et al. [94] | 2021 | Original Research | CECT | Classification |
| Ni et al. [58] | 2023 | Original Research | CECT | Classification |
| Chen et al. [95] | 2020 | Original Research | CECT | Classification |
| Shi et al. [98] | 2022 | Original Research | MRI | Classification |
| Bian et al. [99] | 2022 | Original Research | CECT | Classification |
| Chang et al. [100] | 2022 | Original Research | CECT | Classification |
| Chen et al. [101] | 2022 | Review | N.A. | N.A. |
| Hayashi et al. [102] | 2021 | Review | N.A. | N.A. |
| Ladd et al. [103] | 2021 | Review | N.A. | N.A. |
| Kenner et al. [104] | 2021 | Review | N.A. | N.A. |
| Jiang et al. [105] | 2023 | Review | N.A. | N.A. |
| Pereira et al. [106] | 2020 | Review | N.A. | N.A. |
| Schuurmans et al. [74] | 2022 | Review | N.A. | N.A. |
| Sijithra et al. [107] | 2023 | Review | N.A. | N.A. |
| Anta et al. [108] | 2022 | Review | N.A. | N.A. |
| Marti-Bonmati et al. [10] | 2022 | Review | N.A. | N.A. |
| Barat et al. [55] | 2021 | Review | N.A. | N.A. |
| Faur et al. [109] | 2023 | Review | N.A. | N.A. |
| Enriquez et al. [110] | 2021 | Review | N.A. | N.A. |
| Qureshi et al. [111] | 2022 | Review | N.A. | N.A. |
| Barat et al. [112] | 2023 | Review | N.A. | N.A. |
| Pacella et al. [113] | 2023 | Review | N.A. | N.A. |
| Ramaekers et al. [38] | 2023 | Review | N.A. | N.A. |
| Bartoli et al. [114] | 2020 | Review | N.A. | N.A. |
| Cui et al. [115] | 2020 | Review | N.A. | N.A. |
| Mirza et al. [116] | 2023 | Review | N.A. | N.A. |
References
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef]
- Atlanta: American Cancer Society Facts & Figures 2024. Available online: https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/2024-cancer-facts-figures.html (accessed on 10 February 2024).
- Boursi, B.; Finkelman, B.; Giantonio, B.J.; Haynes, K.; Rustgi, A.K.; Rhim, A.D.; Mamtani, R.; Yang, Y.-X. A Clinical Prediction Model to Assess Risk for Pancreatic Cancer Among Patients with New-Onset Diabetes. Gastroenterology 2017, 152, 840–850.e3. [Google Scholar] [CrossRef]
- Klein, A.P.; Lindström, S.; Mendelsohn, J.B.; Steplowski, E.; Arslan, A.A.; Bueno-de-Mesquita, H.B.; Fuchs, C.S.; Gallinger, S.; Gross, M.; Helzlsouer, K.; et al. An Absolute Risk Model to Identify Individuals at Elevated Risk for Pancreatic Cancer in the General Population. PLoS ONE 2013, 8, e72311. [Google Scholar] [CrossRef]
- Pannala, R.; Basu, A.; Petersen, G.M.; Chari, S.T. New-Onset Diabetes: A Potential Clue to the Early Diagnosis of Pancreatic Cancer. Lancet Oncol. 2009, 10, 88–95. [Google Scholar] [CrossRef]
- Muhammad, W.; Hart, G.R.; Nartowt, B.; Farrell, J.J.; Johung, K.; Liang, Y.; Deng, J. Pancreatic Cancer Prediction Through an Artificial Neural Network. Front. Artif. Intell. 2019, 2, 2. [Google Scholar] [CrossRef]
- Adamska, A.; Domenichini, A.; Falasca, M. Pancreatic Ductal Adenocarcinoma: Current and Evolving Therapies. Int. J. Mol. Sci. 2017, 18, 1338. [Google Scholar] [CrossRef]
- Solomon, S.; Das, S.; Brand, R.; Whitcomb, D.C. Inherited Pancreatic Cancer Syndromes. Cancer J. 2012, 18, 485–491. [Google Scholar] [CrossRef]
- Rosato, V.; Polesel, J.; Bosetti, C.; Serraino, D.; Negri, E.; La Vecchia, C. Population Attributable Risk for Pancreatic Cancer in Northern Italy. Pancreas 2015, 44, 216–220. [Google Scholar] [CrossRef]
- Marti-Bonmati, L.; Cerdá-Alberich, L.; Pérez-Girbés, A.; Beveridge, R.D.; Orón, E.M.; Rojas, J.P.; Alberich-Bayarri, A. Pancreatic Cancer, Radiomics and Artificial Intelligence. Br. J. Radiol. 2022, 95, 20220072. [Google Scholar] [CrossRef]
- Kann, B.H.; Hosny, A.; Aerts, H.J.W.L. Artificial Intelligence for Clinical Oncology. Cancer Cell 2021, 39, 916–927. [Google Scholar] [CrossRef]
- Yip, S.S.F.; Aerts, H.J.W.L. Applications and Limitations of Radiomics. Phys. Med. Biol. 2016, 61, R150–R166. [Google Scholar] [CrossRef]
- Gillies, R.J.; Kinahan, P.E.; Hricak, H. Radiomics: Images Are More than Pictures, They Are Data. Radiology 2016, 278, 563–577. [Google Scholar] [CrossRef]
- Simpson, A.L.; Antonelli, M.; Bakas, S.; Bilello, M.; Farahani, K.; van Ginneken, B.; Kopp-Schneider, A.; Landman, B.A.; Litjens, G.; Menze, B.; et al. A Large Annotated Medical Image Dataset for the Development and Evaluation of Segmentation Algorithms. arXiv 2019, arXiv:1902.09063. [Google Scholar]
- Antonelli, M.; Reinke, A.; Bakas, S.; Farahani, K.; Kopp-Schneider, A.; Landman, B.A.; Litjens, G.; Menze, B.; Ronneberger, O.; Summers, R.M.; et al. The Medical Segmentation Decathlon. Nat. Commun. 2022, 13, 4128. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, Y.; Gu, S.; Zhu, C.; Ge, C.; Zhang, Y.; An, X.; Wang, C.; Wang, Q.; Liu, X.; et al. AbdomenCT-1K: Is Abdominal Organ Segmentation a Solved Problem? IEEE Trans. Pattern Anal. Mach. Intell 2022, 44, 6695–6714. [Google Scholar] [CrossRef]
- Roth, H.; Farag, A.; Turkbey, E.; Lu, L.; Liu, J.; Summers, R.M. Data From Pancreas-CT. (Version 2) [Data set]. Cancer Imaging Arch. 2016. [Google Scholar] [CrossRef]
- Szczypiński, P.M.; Klepaczko, A. MaZda. A Framework for Biomedical Image Texture Analysis and Data Exploration. In Biomedical Texture Analysis: Fundamentals, Tools and Challenges; Academic Press: Cambridge, MA, USA, 2017; pp. 315–347. [Google Scholar]
- Strzelecki, M.; Szczypinski, P.; Materka, A.; Klepaczko, A. A Software Tool for Automatic Classification and Segmentation of 2D/3D Medical Images. Nucl. Instrum. Methods Phys. Res. A 2013, 702, 137–140. [Google Scholar] [CrossRef]
- Szczypiński, P.M.; Strzelecki, M.; Materka, A.; Klepaczko, A. MaZda—The Software Package for Textural Analysis of Biomedical Images. In Computers in Medical Activity; Springer: Berlin, Germany, 2009; pp. 73–84. [Google Scholar]
- Szczypiński, P.M.; Strzelecki, M.; Materka, A.; Klepaczko, A. MaZda—A Software Package for Image Texture Analysis. Comput. Methods Programs Biomed. 2009, 94, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.slicer.org/ (accessed on 10 February 2024).
- Van Griethuysen, J.J.M.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.H.; Fillion-Robin, J.-C.; Pieper, S.; Aerts, H.J.W.L. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef]
- Available online: https://cerr.github.io/cerr/ (accessed on 10 February 2024).
- Nioche, C.; Orlhac, F.; Boughdad, S.; Reuzé, S.; Goya-Outi, J.; Robert, C.; Pellot-Barakat, C.; Soussan, M.; Frouin, F.; Buvat, I. LIFEx: A Freeware for Radiomic Feature Calculation in Multimodality Imaging to Accelerate Advances in the Characterization of Tumor Heterogeneity. Cancer Res. 2018, 78, 4786–4789. [Google Scholar] [CrossRef]
- Available online: https://monai.io/ (accessed on 10 February 2024).
- NCCN. Available online: https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf (accessed on 26 December 2023).
- Almeida, R.R.; Lo, G.C.; Patino, M.; Bizzo, B.; Canellas, R.; Sahani, D.V. Advances in Pancreatic CT Imaging. Am. J. Roentgenol. 2018, 211, 52–66. [Google Scholar] [CrossRef]
- Schoellnast, H.; Tillich, M.; Deutschmann, H.A.; Stessel, U.; Deutschmann, M.J.; Schaffler, G.J.; Schoellnast, R.; Uggowitzer, M.M. Improvement of Parenchymal and Vascular Enhancement Using Saline Flush and Power Injection for Multiple-Detector-Row Abdominal CT. Eur. Radiol. 2004, 14, 659–664. [Google Scholar] [CrossRef]
- Wessels, F.; van Delden, O.; Smithuis, R. Pancreatic Cancer—CT Staging 2.0. 2021. Available online: https://radiologyassistant.nl/abdomen/pancreas/pancreas-carcinoma-1 (accessed on 26 December 2023).
- Park, H.S.; Lee, J.M.; Choi, H.K.; Hong, S.H.; Han, J.K.; Choi, B.I. Preoperative Evaluation of Pancreatic Cancer: Comparison of Gadolinium-enhanced Dynamic MRI with MR Cholangiopancreatography versus MDCT. J. Magn. Reson. Imaging 2009, 30, 586–595. [Google Scholar] [CrossRef]
- Schima, W.; Függer, R. Evaluation of Focal Pancreatic Masses: Comparison of Mangafodipir-Enhanced MR Imaging and Contrast-Enhanced Helical CT. Eur. Radiol. 2002, 12, 2998–3008. [Google Scholar] [CrossRef]
- Horvat, N.; Ryan, D.E.; LaGratta, M.D.; Shah, P.M.; Do, R.K. Imaging for Pancreatic Ductal Adenocarcinoma. Chin. Clin. Oncol. 2017, 6, 62. [Google Scholar] [CrossRef]
- Chen, F.-M.; Ni, J.-M.; Zhang, Z.-Y.; Zhang, L.; Li, B.; Jiang, C.-J. Presurgical Evaluation of Pancreatic Cancer: A Comprehensive Imaging Comparison of CT Versus MRI. Am. J. Roentgenol. 2016, 206, 526–535. [Google Scholar] [CrossRef]
- Rickes, S.; Unkrodt, K.; Neye, H.; Ocran, K.W.; Wermke, W. Differentiation of Pancreatic Tumours by Conventional Ultrasound, Unenhanced and Echo-Enhanced Power Doppler Sonography. Scand. J. Gastroenterol. 2002, 37, 1313–1320. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S. Imaging Diagnosis of Pancreatic Cancer: A State-of-the-Art Review. World J. Gastroenterol. 2014, 20, 7864. [Google Scholar] [CrossRef] [PubMed]
- Dbouk, M.; Katona, B.W.; Brand, R.E.; Chak, A.; Syngal, S.; Farrell, J.J.; Kastrinos, F.; Stoffel, E.M.; Blackford, A.L.; Rustgi, A.K.; et al. The Multicenter Cancer of Pancreas Screening Study: Impact on Stage and Survival. J. Clin. Oncol. 2022, 40, 3257–3266. [Google Scholar] [CrossRef] [PubMed]
- Ramaekers, M.; Viviers, C.G.A.; Janssen, B.V.; Hellström, T.A.E.; Ewals, L.; van der Wulp, K.; Nederend, J.; Jacobs, I.; Pluyter, J.R.; Mavroeidis, D.; et al. Computer-Aided Detection for Pancreatic Cancer Diagnosis: Radiological Challenges and Future Directions. J. Clin. Med. 2023, 12, 4209. [Google Scholar] [CrossRef] [PubMed]
- Jang, K.M.; Kim, S.H.; Kim, Y.K.; Song, K.D.; Lee, S.J.; Choi, D. Missed Pancreatic Ductal Adenocarcinoma: Assessment of Early Imaging Findings on Prediagnostic Magnetic Resonance Imaging. Eur. J. Radiol. 2015, 84, 1473–1479. [Google Scholar] [CrossRef]
- Cao, K.; Xia, Y.; Yao, J.; Han, X.; Lambert, L.; Zhang, T.; Tang, W.; Jin, G.; Jiang, H.; Fang, X.; et al. Large-Scale Pancreatic Cancer Detection via Non-Contrast CT and Deep Learning. Nat. Med. 2023, 29, 3033–3043. [Google Scholar] [CrossRef]
- Korfiatis, P.; Suman, G.; Patnam, N.G.; Trivedi, K.H.; Karbhari, A.; Mukherjee, S.; Cook, C.; Klug, J.R.; Patra, A.; Khasawneh, H.; et al. Automated Artificial Intelligence Model Trained on a Large Data Set Can Detect Pancreas Cancer on Diagnostic Computed Tomography Scans as Well as Visually Occult Preinvasive Cancer on Prediagnostic Computed Tomography Scans. Gastroenterology 2023, 165, 1533–1546.e4. [Google Scholar] [CrossRef]
- Chu, L.C.; Park, S.; Kawamoto, S.; Wang, Y.; Zhou, Y.; Shen, W.; Zhu, Z.; Xia, Y.; Xie, L.; Liu, F.; et al. Application of Deep Learning to Pancreatic Cancer Detection: Lessons Learned From Our Initial Experience. J. Am. Coll. Radiol. 2019, 16, 1338–1342. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.C.; Park, S.; Kawamoto, S.; Fouladi, D.F.; Shayesteh, S.; Zinreich, E.S.; Graves, J.S.; Horton, K.M.; Hruban, R.H.; Yuille, A.L.; et al. Utility of CT Radiomics Features in Differentiation of Pancreatic Ductal Adenocarcinoma From Normal Pancreatic Tissue. Am. J. Roentgenol. 2019, 213, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.C.; Solmaz, B.; Park, S.; Kawamoto, S.; Yuille, A.L.; Hruban, R.H.; Fishman, E.K. Diagnostic Performance of Commercially Available vs. in-House Radiomics Software in Classification of CT Images from Patients with Pancreatic Ductal Adenocarcinoma vs. Healthy Controls. Abdom. Radiol. 2020, 45, 2469–2475. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Liu, Z.-X.; Zhang, J.-J.; Wu, F.-T.; Xu, C.-F.; Shen, Z.; Yu, C.-H.; Li, Y.-M. Construction of a Convolutional Neural Network Classifier Developed by Computed Tomography Images for Pancreatic Cancer Diagnosis. World J. Gastroenterol. 2020, 26, 5156–5168. [Google Scholar] [CrossRef] [PubMed]
- Alves, N.; Schuurmans, M.; Litjens, G.; Bosma, J.S.; Hermans, J.; Huisman, H. Fully Automatic Deep Learning Framework for Pancreatic Ductal Adenocarcinoma Detection on Computed Tomography. Cancers 2022, 14, 376. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Patra, A.; Khasawneh, H.; Korfiatis, P.; Rajamohan, N.; Suman, G.; Majumder, S.; Panda, A.; Johnson, M.P.; Larson, N.B.; et al. Radiomics-Based Machine-Learning Models Can Detect Pancreatic Cancer on Prediagnostic Computed Tomography Scans at a Substantial Lead Time before Clinical Diagnosis. Gastroenterology 2022, 163, 1435–1446.e3. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Lin, C.; Kolomaya, A.; Ostdiek-Wille, G.P.; Wong, J.; Cheng, X.; Lei, Y.; Liu, C. Compute Tomography Radiomics Analysis on Whole Pancreas Between Healthy Individual and Pancreatic Ductal Adenocarcinoma Patients: Uncertainty Analysis and Predictive Modeling. Technol. Cancer Res. Treat. 2022, 21, 15330338221126869. [Google Scholar] [CrossRef]
- Chen, P.-T.; Chang, D.; Yen, H.; Liu, K.-L.; Huang, S.-Y.; Roth, H.; Wu, M.-S.; Liao, W.-C.; Wang, W. Radiomic Features at CT Can Distinguish Pancreatic Cancer from Noncancerous Pancreas. Radiol. Imaging Cancer 2021, 3, e210010. [Google Scholar] [CrossRef]
- Viviers, C.G.A.; Ramaekers, M.; de With, P.H.N.; Mavroeidis, D.; Nederend, J.; Luyer, M.; van der Sommen, F. Improved Pancreatic Tumor Detection by Utilizing Clinically-Relevant Secondary Features. In MICCAI Workshop on Cancer Prevention through Early Detection; Springer: Cham, Switzerland, 2022; pp. 139–148. [Google Scholar]
- Weisberg, E.M.; Chu, L.C.; Park, S.; Yuille, A.L.; Kinzler, K.W.; Vogelstein, B.; Fishman, E.K. Deep Lessons Learned: Radiology, Oncology, Pathology, and Computer Science Experts Unite around Artificial Intelligence to Strive for Earlier Pancreatic Cancer Diagnosis. Diagn. Interv. Imaging 2020, 101, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Farag, A.; Lu, L.; Roth, H.R.; Liu, J.; Turkbey, E.; Summers, R.M. A Bottom-Up Approach for Pancreas Segmentation Using Cascaded Superpixels and (Deep) Image Patch Labeling. IEEE Trans. Image Process. 2017, 26, 386–399. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, Y.; Zhang, Z.; Wang, Y.; Wang, A.; Fishman, E.K.; Yuille, A.L.; Park, S. Hyper-Pairing Network for Multi-Phase Pancreatic Ductal Adenocarcinoma Segmentation. In Proceedings of the Medical Image Computing and Computer Assisted Intervention–MICCAI 2019: 22nd International Conference, Shenzhen, China, 13–17 October 2019; Shen, D., Ed.; Springer: Cham, Switzerland, 2019; pp. 155–163. [Google Scholar]
- Zhu, Z.; Xia, Y.; Xie, L.; Fishman, E.K.; Yuille, A.L. Multi-Scale Coarse-to-Fine Segmentation for Screening Pancreatic Ductal Adenocarcinoma. In Proceedings of the Medical Image Computing and Computer Assisted Intervention–MICCAI 2019: 22nd International Conference, Shenzhen, China, 13–17 October 2018; Shen, D., Ed.; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Barat, M.; Chassagnon, G.; Dohan, A.; Gaujoux, S.; Coriat, R.; Hoeffel, C.; Cassinotto, C.; Soyer, P. Artificial Intelligence: A Critical Review of Current Applications in Pancreatic Imaging. Jpn. J. Radiol. 2021, 39, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-C.; Papandreou, G.; Kokkinos, I.; Murphy, K.; Yuille, A.L. DeepLab: Semantic Image Segmentation with Deep Convolutional Nets, Atrous Convolution, and Fully Connected CRFs. IEEE Trans. Pattern Anal. Mach. Intell. 2018, 40, 834–848. [Google Scholar] [CrossRef]
- Chollet, F. Xception: Deep Learning with Depthwise Separable Convolutions. In Proceedings of the 2017 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Honolulu, HI, USA, 21–26 July 2017; IEEE: Piscataway, NJ, USA, 2017; pp. 1800–1807. [Google Scholar]
- Ni, H.; Zhou, G.; Chen, X.; Ren, J.; Yang, M.; Zhang, Y.; Zhang, Q.; Zhang, L.; Mao, C.; Li, X. Predicting Recurrence in Pancreatic Ductal Adenocarcinoma after Radical Surgery Using an AX-Unet Pancreas Segmentation Model and Dynamic Nomogram. Bioengineering 2023, 10, 828. [Google Scholar] [CrossRef]
- Turečková, A.; Tureček, T.; Komínková Oplatková, Z.; Rodríguez-Sánchez, A. Improving CT Image Tumor Segmentation Through Deep Supervision and Attentional Gates. Front. Robot. AI 2020, 7, 106. [Google Scholar] [CrossRef]
- Isensee, F.; Petersen, J.; Klein, A.; Zimmerer, D.; Jaeger, P.F.; Kohl, S.; Wasserthal, J.; Koehler, G.; Norajitra, T.; Wirkert, S.; et al. NnU-Net: Self-Adapting Framework for U-Net-Based Medical Image Segmentation. arXiv 2018, arXiv:1809.10486. [Google Scholar]
- Mahmoudi, T.; Kouzahkanan, Z.M.; Radmard, A.R.; Kafieh, R.; Salehnia, A.; Davarpanah, A.H.; Arabalibeik, H.; Ahmadian, A. Segmentation of Pancreatic Ductal Adenocarcinoma (PDAC) and Surrounding Vessels in CT Images Using Deep Convolutional Neural Networks and Texture Descriptors. Sci. Rep. 2022, 12, 3092. [Google Scholar] [CrossRef]
- Shen, C.; Roth, H.R.; Hayashi, Y.; Oda, M.; Miyamoto, T.; Sato, G.; Mori, K. A Cascaded Fully Convolutional Network Framework for Dilated Pancreatic Duct Segmentation. Int. J. Comput. Assist. Radiol. Surg. 2022, 17, 343–354. [Google Scholar] [CrossRef]
- Lu, J.; Jiang, N.; Zhang, Y.; Li, D. A CT Based Radiomics Nomogram for Differentiation between Focal-Type Autoimmune Pancreatitis and Pancreatic Ductal Adenocarcinoma. Front. Oncol. 2023, 13, 979437. [Google Scholar] [CrossRef]
- Park, S.; Chu, L.C.; Hruban, R.H.; Vogelstein, B.; Kinzler, K.W.; Yuille, A.L.; Fouladi, D.F.; Shayesteh, S.; Ghandili, S.; Wolfgang, C.L.; et al. Differentiating Autoimmune Pancreatitis from Pancreatic Ductal Adenocarcinoma with CT Radiomics Features. Diagn. Interv. Imaging 2020, 101, 555–564. [Google Scholar] [CrossRef]
- Anai, K.; Hayashida, Y.; Ueda, I.; Hozuki, E.; Yoshimatsu, Y.; Tsukamoto, J.; Hamamura, T.; Onari, N.; Aoki, T.; Korogi, Y. The Effect of CT Texture-Based Analysis Using Machine Learning Approaches on Radiologists’ Performance in Differentiating Focal-Type Autoimmune Pancreatitis and Pancreatic Duct Carcinoma. Jpn. J. Radiol. 2022, 40, 1156–1165. [Google Scholar] [CrossRef]
- Deng, Y.; Ming, B.; Zhou, T.; Wu, J.L.; Chen, Y.; Liu, P.; Zhang, J.; Zhang, S.Y.; Chen, T.W.; Zhang, X.M. Radiomics Model Based on MR Images to Discriminate Pancreatic Ductal Adenocarcinoma and Mass-Forming Chronic Pancreatitis Lesions. Front Oncol 2021, 11, 620981. [Google Scholar] [CrossRef]
- Ren, S.; Zhao, R.; Cui, W.; Qiu, W.; Guo, K.; Cao, Y.; Duan, S.; Wang, Z.; Chen, R. Computed Tomography-Based Radiomics Signature for the Preoperative Differentiation of Pancreatic Adenosquamous Carcinoma From Pancreatic Ductal Adenocarcinoma. Front. Oncol. 2020, 10, 1618. [Google Scholar] [CrossRef]
- Shi, Y.J.; Zhu, H.T.; Li, X.T.; Zhang, X.Y.; Liu, Y.L.; Wei, Y.Y.; Sun, Y.S. Histogram Array and Convolutional Neural Network of DWI for Differentiating Pancreatic Ductal Adenocarcinomas from Solid Pseudopapillary Neoplasms and Neuroendocrine Neoplasms. Clin. Imaging 2023, 96, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Younan, G. Pancreas Solid Tumors. Surg. Clin. N. Am. 2020, 100, 565–580. [Google Scholar] [CrossRef]
- Zhang, T.; Xiang, Y.; Wang, H.; Yun, H.; Liu, Y.; Wang, X.; Zhang, H. Radiomics Combined with Multiple Machine Learning Algorithms in Differentiating Pancreatic Ductal Adenocarcinoma from Pancreatic Neuroendocrine Tumor: More Hands Produce a Stronger Flame. J. Clin. Med. 2022, 11, 6789. [Google Scholar] [CrossRef] [PubMed]
- Macías, N.; Sayagués, J.M.; Esteban, C.; Iglesias, M.; González, L.M.; Quiñones-Sampedro, J.; Gutiérrez, M.L.; Corchete, L.A.; Abad, M.M.; Bengoechea, O.; et al. Histologic Tumor Grade and Preoperative Bilary Drainage Are the Unique Independent Prognostic Factors of Survival in Pancreatic Ductal Adenocarcinoma Patients After Pancreaticoduodenectomy. J. Clin. Gastroenterol. 2018, 52, e11–e17. [Google Scholar] [CrossRef]
- Kuhlmann, K.F.D.; de Castro, S.M.M.; Wesseling, J.G.; ten Kate, F.J.W.; Offerhaus, G.J.A.; Busch, O.R.C.; van Gulik, T.M.; Obertop, H.; Gouma, D.J. Surgical Treatment of Pancreatic Adenocarcinoma. Eur. J. Cancer 2004, 40, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Han, S.H.; Heo, J.S.; Choi, S.H.; Choi, D.W.; Han, I.W.; Han, S.; You, Y.H. Actual Long-Term Outcome of T1 and T2 Pancreatic Ductal Adenocarcinoma after Surgical Resection. Int. J. Surg. 2017, 40, 68–72. [Google Scholar] [CrossRef]
- Schuurmans, M.; Alves, N.; Vendittelli, P.; Huisman, H.; Hermans, J. Setting the Research Agenda for Clinical Artificial Intelligence in Pancreatic Adenocarcinoma Imaging. Cancers 2022, 14, 3498. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Mi, W.; Pan, B.; Guo, Y.; Li, J.; Xu, R.; Zheng, J.; Zou, C.; Zhang, T.; Liang, Z.; et al. Automatic Pancreatic Ductal Adenocarcinoma Detection in Whole Slide Images Using Deep Convolutional Neural Networks. Front. Oncol. 2021, 11, 665929. [Google Scholar] [CrossRef] [PubMed]
- Sántha, P.; Lenggenhager, D.; Finstadsveen, A.; Dorg, L.; Tøndel, K.; Amrutkar, M.; Gladhaug, I.P.; Verbeke, C. Morphological Heterogeneity in Pancreatic Cancer Reflects Structural and Functional Divergence. Cancers 2021, 13, 895. [Google Scholar] [CrossRef]
- Baxi, V.; Edwards, R.; Montalto, M.; Saha, S. Digital Pathology and Artificial Intelligence in Translational Medicine and Clinical Practice. Mod. Pathol. 2022, 35, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.; Duan, N.; Chen, X.; Ren, S.; Zhang, Y.; Wang, Z.; Chen, R. Pancreatic Ductal Adenocarcinoma: Machine Learning–Based Quantitative Computed Tomography Texture Analysis for Prediction of Histopathological Grade. Cancer Manag. Res. 2019, 11, 9253–9264. [Google Scholar] [CrossRef]
- Cen, C.; Wang, C.; Wang, S.; Wen, K.; Liu, L.; Li, X.; Wu, L.; Huang, M.; Ma, L.; Liu, H.; et al. Clinical-Radiomics Nomogram Using Contrast-Enhanced CT to Predict Histological Grade and Survival in Pancreatic Ductal Adenocarcinoma. Front. Oncol. 2023, 13, 1218128. [Google Scholar] [CrossRef]
- Makohon-Moore, A.; Iacobuzio-Donahue, C.A. Pancreatic Cancer Biology and Genetics from an Evolutionary Perspective. Nat. Rev. Cancer 2016, 16, 553–565. [Google Scholar] [CrossRef]
- Hinzpeter, R.; Kulanthaivelu, R.; Kohan, A.; Avery, L.; Pham, N.A.; Ortega, C.; Metser, U.; Haider, M.; Veit-Haibach, P. CT Radiomics and Whole Genome Sequencing in Patients with Pancreatic Ductal Adenocarcinoma: Predictive Radiogenomics Modeling. Cancers 2022, 14, 6224. [Google Scholar] [CrossRef]
- Gao, J.; Chen, X.; Li, X.; Miao, F.; Fang, W.; Li, B.; Qian, X.; Lin, X. Differentiating TP53 Mutation Status in Pancreatic Ductal Adenocarcinoma Using Multiparametric MRI-Derived Radiomics. Front. Oncol. 2021, 11, 632130. [Google Scholar] [CrossRef]
- Chaturvedi, P.; Singh, A.P.; Moniaux, N.; Senapati, S.; Chakraborty, S.; Meza, J.L.; Batra, S.K. MUC4 Mucin Potentiates Pancreatic Tumor Cell Proliferation, Survival, and Invasive Properties and Interferes with Its Interaction to Extracellular Matrix Proteins. Mol. Cancer Res. 2007, 5, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Mimeault, M.; Johansson, S.L.; Senapati, S.; Momi, N.; Chakraborty, S.; Batra, S.K. MUC4 Down-Regulation Reverses Chemoresistance of Pancreatic Cancer Stem/Progenitor Cells and Their Progenies. Cancer Lett. 2010, 295, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Saitou, M. MUC4 Expression Is a Novel Prognostic Factor in Patients with Invasive Ductal Carcinoma of the Pancreas. J. Clin. Pathol. 2005, 58, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Li, Y.; Wu, J.L.; Zhou, T.; Tang, M.Y.; Chen, Y.; Zuo, H.D.; Tang, W.; Chen, T.W.; Zhang, X.M. Radiomics Models Based on Multi-Sequence MRI for Preoperative Evaluation of MUC4 Status in Pancreatic Ductal Adenocarcinoma: A Preliminary Study. Quant. Imaging Med. Surg. 2022, 12, 5129–5139. [Google Scholar] [CrossRef] [PubMed]
- Iwatate, Y.; Hoshino, I.; Yokota, H.; Ishige, F.; Itami, M.; Mori, Y.; Chiba, S.; Arimitsu, H.; Yanagibashi, H.; Nagase, H.; et al. Radiogenomics for Predicting P53 Status, PD-L1 Expression, and Prognosis with Machine Learning in Pancreatic Cancer. Br. J. Cancer 2020, 123, 1253–1261. [Google Scholar] [CrossRef]
- Meng, Y.; Zhang, H.; Li, Q.; Xing, P.; Liu, F.; Cao, K.; Fang, X.; Li, J.; Yu, J.; Feng, X.; et al. Noncontrast Magnetic Resonance Radiomics and Multilayer Perceptron Network Classifier: An Approach for Predicting Fibroblast Activation Protein Expression in Patients with Pancreatic Ductal Adenocarcinoma. J. Magn. Reson. Imaging 2021, 54, 1432–1443. [Google Scholar] [CrossRef]
- Vezakis, I.; Vezakis, A.; Gourtsoyianni, S.; Koutoulidis, V.; Polydorou, A.A.; Matsopoulos, G.K.; Koutsouris, D.D. An Automated Prognostic Model for Pancreatic Ductal Adenocarcinoma. Genes 2023, 14, 1742. [Google Scholar] [CrossRef]
- Xu, X.; Qu, J.; Zhang, Y.; Qian, X.; Chen, T.; Liu, Y. Development and Validation of an MRI-Radiomics Nomogram for the Prognosis of Pancreatic Ductal Adenocarcinoma. Front. Oncol. 2023, 13, 1074445. [Google Scholar] [CrossRef]
- Qiu, H.; Xu, M.; Wang, Y.; Wen, X.; Chen, X.; Liu, W.; Zhang, N.; Ding, X.; Zhang, L. Original Article A Novel Preoperative MRI-Based Radiomics Nomogram Outperforms Traditional Models for Prognostic Prediction in Pancreatic Ductal Adenocarcinoma. Am. J. Cancer Res. 2022, 12, 2032. [Google Scholar]
- Li, X.; Wan, Y.; Lou, J.; Xu, L.; Shi, A.; Yang, L.; Fan, Y.; Yang, J.; Huang, J.; Wu, Y.; et al. Preoperative Recurrence Prediction in Pancreatic Ductal Adenocarcinoma after Radical Resection Using Radiomics of Diagnostic Computed Tomography. EClinicalMedicine 2022, 43, 101215. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Wang, X.; Li, M.; Tong, T.; Yu, X.; Zhou, Z. Pancreatic Ductal Adenocarcinoma: A Radiomics Nomogram Outperforms Clinical Model and TNM Staging for Survival Estimation after Curative Resection. Eur. Radiol. 2020, 30, 2513–2524. [Google Scholar] [CrossRef]
- Park, S.; Sham, J.G.; Kawamoto, S.; Blair, A.B.; Rozich, N.; Fouladi, D.F.; Shayesteh, S.; Hruban, R.H.; He, J.; Wolfgang, C.L.; et al. CT Radiomics–Based Preoperative Survival Prediction in Patients With Pancreatic Ductal Adenocarcinoma. Am. J. Roentgenol. 2021, 217, 1104–1112. [Google Scholar] [CrossRef]
- Chen, F.; Zhou, Y.; Qi, X.; Zhang, R.; Gao, X.; Xia, W.; Zhang, L. Radiomics-Assisted Presurgical Prediction for Surgical Portal Vein-Superior Mesenteric Vein Invasion in Pancreatic Ductal Adenocarcinoma. Front. Oncol. 2020, 10, 523543. [Google Scholar] [CrossRef]
- Jang, J.K.; Byun, J.H.; Kang, J.H.; Son, J.H.; Kim, J.H.; Lee, S.S.; Kim, H.J.; Yoo, C.; Kim, K.; Hong, S.-M.; et al. CT-Determined Resectability of Borderline Resectable and Unresectable Pancreatic Adenocarcinoma Following FOLFIRINOX Therapy. Eur. Radiol. 2021, 31, 813–823. [Google Scholar] [CrossRef]
- Zins, M.; Matos, C.; Cassinotto, C. Pancreatic Adenocarcinoma Staging in the Era of Preoperative Chemotherapy and Radiation Therapy. Radiology 2018, 287, 374–390. [Google Scholar] [CrossRef]
- Shi, L.; Wang, L.; Wu, C.; Wei, Y.; Zhang, Y.; Chen, J. Preoperative Prediction of Lymph Node Metastasis of Pancreatic Ductal Adenocarcinoma Based on a Radiomics Nomogram of Dual-Parametric MRI Imaging. Front. Oncol. 2022, 12, 927077. [Google Scholar] [CrossRef] [PubMed]
- Bian, Y.; Guo, S.; Jiang, H.; Gao, S.; Shao, C.; Cao, K.; Fang, X.; Li, J.; Wang, L.; Ma, C.; et al. Radiomics Nomogram for the Preoperative Prediction of Lymph Node Metastasis in Pancreatic Ductal Adenocarcinoma. Cancer Imaging 2022, 22, 4. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Liu, Y.; Saey, S.A.; Chang, K.C.; Shrader, H.R.; Steckly, K.L.; Rajput, M.; Sonka, M.; Chan, C.H.F. Machine-Learning Based Investigation of Prognostic Indicators for Oncological Outcome of Pancreatic Ductal Adenocarcinoma. Front. Oncol. 2022, 12, 6660. [Google Scholar] [CrossRef]
- Chen, X.; Fu, R.; Shao, Q.; Chen, Y.; Ye, Q.; Li, S.; He, X.; Zhu, J. Application of Artificial Intelligence to Pancreatic Adenocarcinoma. Front. Oncol. 2022, 12, 960056. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, H.; Uemura, N.; Matsumura, K.; Zhao, L.; Sato, H.; Shiraishi, Y.; Yamashita, Y.; Baba, H. Recent Advances in Artificial Intelligence for Pancreatic Ductal Adenocarcinoma. World J. Gastroenterol. 2021, 27, 7480–7496. [Google Scholar] [CrossRef] [PubMed]
- Ladd, A.M.; Diehl, D.L. Artificial Intelligence for Early Detection of Pancreatic Adenocarcinoma: The Future Is Promising. World J. Gastroenterol. 2021, 27, 1283–1295. [Google Scholar] [CrossRef]
- Kenner, B.; Chari, S.T.; Kelsen, D.; Klimstra, D.S.; Pandol, S.J.; Rosenthal, M.; Rustgi, A.K.; Taylor, J.A.; Yala, A.; Abul-Husn, N.; et al. Artificial Intelligence and Early Detection of Pancreatic Cancer: 2020 Summative Review. Pancreas 2021, 50, 251–279. [Google Scholar] [CrossRef]
- Jiang, J.; Chao, W.-L.; Culp, S.; Krishna, S.G. Artificial Intelligence in the Diagnosis and Treatment of Pancreatic Cystic Lesions and Adenocarcinoma. Cancers 2023, 15, 2410. [Google Scholar] [CrossRef]
- Pereira, S.P.; Oldfield, L.; Ney, A.; Hart, P.A.; Keane, M.G.; Pandol, S.J.; Li, D.; Greenhalf, W.; Jeon, C.Y.; Koay, E.J.; et al. Early Detection of Pancreatic Cancer. Lancet Gastroenterol. Hepatol. 2020, 5, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Sijithra, P.; Santhi, N.; Ramasamy, N. A Review Study on Early Detection of Pancreatic Ductal Adenocarcinoma Using Artificial Intelligence Assisted Diagnostic Methods. Eur. J. Radiol. 2023, 166, 110972. [Google Scholar] [CrossRef]
- Anta, J.A.; Martínez-Ballestero, I.; Eiroa, D.; García, J.; Rodríguez-Comas, J. Artificial Intelligence for the Detection of Pancreatic Lesions. Int. J. Comput. Assist. Radiol. Surg. 2022, 17, 1855–1865. [Google Scholar] [CrossRef] [PubMed]
- Faur, A.C.; Lazar, D.C.; Ghenciu, L.A. Artificial Intelligence as a Noninvasive Tool for Pancreatic Cancer Prediction and Diagnosis. World J. Gastroenterol. 2023, 29, 1811–1823. [Google Scholar] [CrossRef]
- Enriquez, J.S.; Chu, Y.; Pudakalakatti, S.; Hsieh, K.L.; Salmon, D.; Dutta, P.; Millward, N.Z.; Lurie, E.; Millward, S.; McAllister, F.; et al. Hyperpolarized Magnetic Resonance and Artificial Intelligence: Frontiers of Imaging in Pancreatic Cancer. JMIR Med. Inform. 2021, 9, e26601. [Google Scholar] [CrossRef]
- Qureshi, T.A.; Javed, S.; Sarmadi, T.; Pandol, S.J.; Li, D. Artificial Intelligence and Imaging for Risk Prediction of Pancreatic Cancer: A Narrative Review. Chin. Clin. Oncol. 2022, 11, 1. [Google Scholar] [CrossRef]
- Barat, M.; Marchese, U.; Pellat, A.; Dohan, A.; Coriat, R.; Hoeffel, C.; Fishman, E.K.; Cassinotto, C.; Chu, L.; Soyer, P. Imaging of Pancreatic Ductal Adenocarcinoma: An Update on Recent Advances. Can. Assoc. Radiol. J. 2023, 74, 351–361. [Google Scholar] [CrossRef]
- Pacella, G.; Brunese, M.C.; D’Imperio, E.; Rotondo, M.; Scacchi, A.; Carbone, M.; Guerra, G. Pancreatic Ductal Adenocarcinoma: Update of CT-Based Radiomics Applications in the Pre-Surgical Prediction of the Risk of Post-Operative Fistula, Resectability Status and Prognosis. J. Clin. Med. 2023, 12, 7380. [Google Scholar] [CrossRef] [PubMed]
- Bartoli, M.; Barat, M.; Dohan, A.; Gaujoux, S.; Coriat, R.; Hoeffel, C.; Cassinotto, C.; Chassagnon, G.; Soyer, P. CT and MRI of Pancreatic Tumors: An Update in the Era of Radiomics. Jpn. J. Radiol. 2020, 38, 1111–1124. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.-J.; Tang, T.-Y.; Zou, X.-W.; Su, Q.-M.; Feng, L.; Gong, X.-Y. Role of Imaging Biomarkers for Prognostic Prediction in Patients with Pancreatic Ductal Adenocarcinoma. Clin. Radiol. 2020, 75, 478.e1–478.e11. [Google Scholar] [CrossRef] [PubMed]
- Mirza-Aghazadeh-Attari, M.; Madani, S.P.; Shahbazian, H.; Ansari, G.; Mohseni, A.; Borhani, A.; Afyouni, S.; Kamel, I.R. Predictive Role of Radiomics Features Extracted from Preoperative Cross-Sectional Imaging of Pancreatic Ductal Adenocarcinoma in Detecting Lymph Node Metastasis: A Systemic Review and Meta-Analysis. Abdom. Radiol. 2023, 48, 2570–2584. [Google Scholar] [CrossRef]
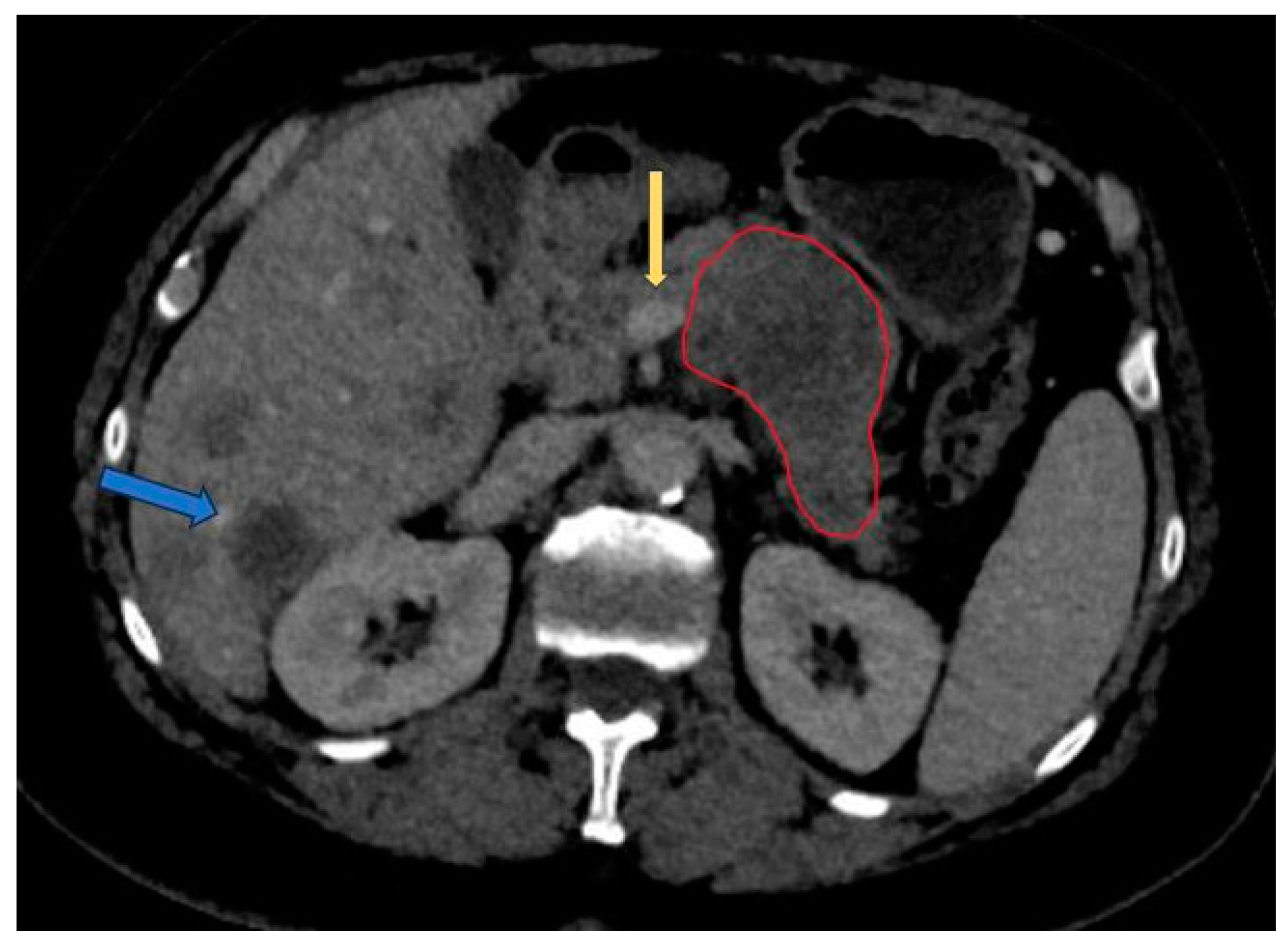
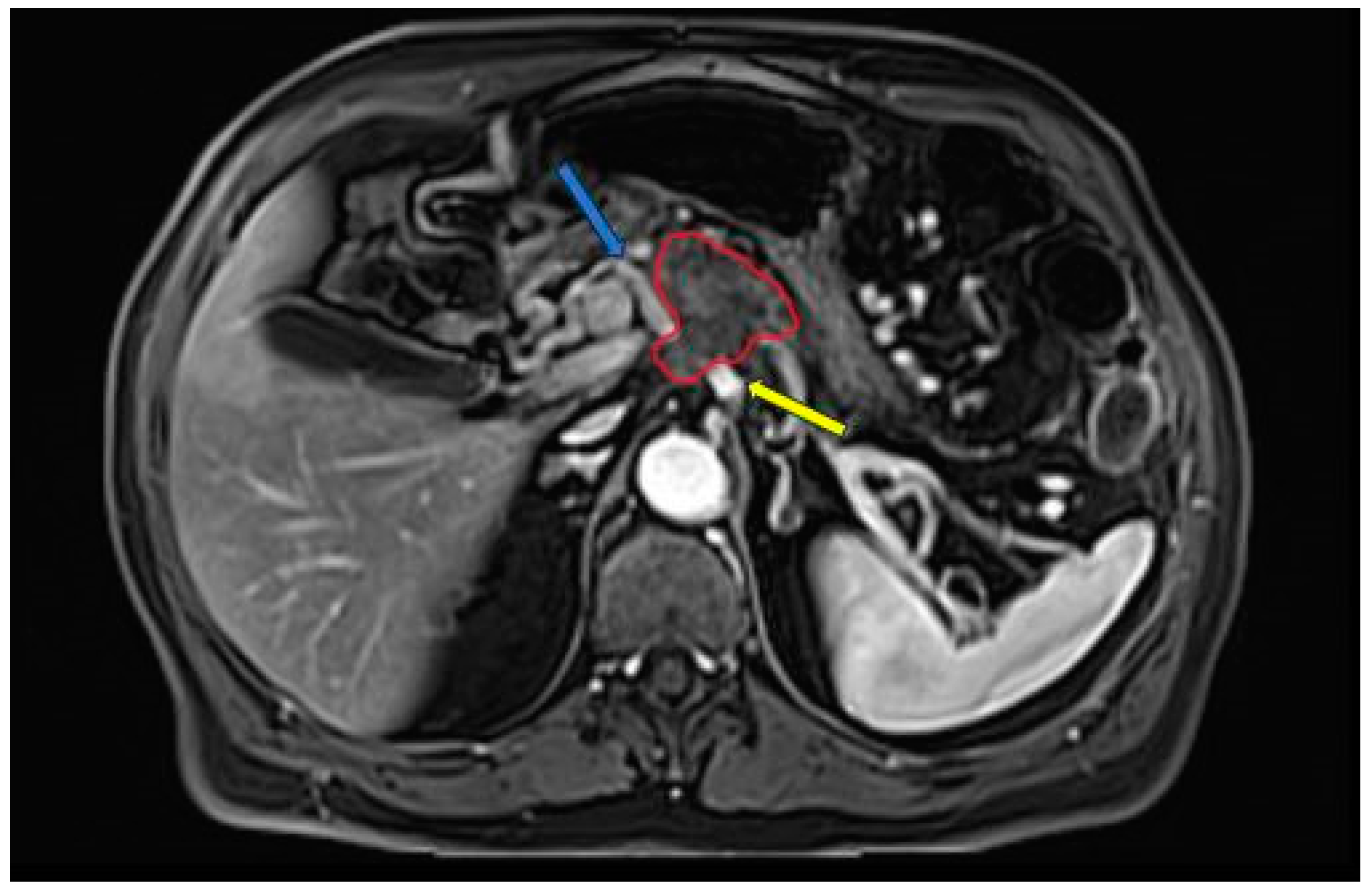
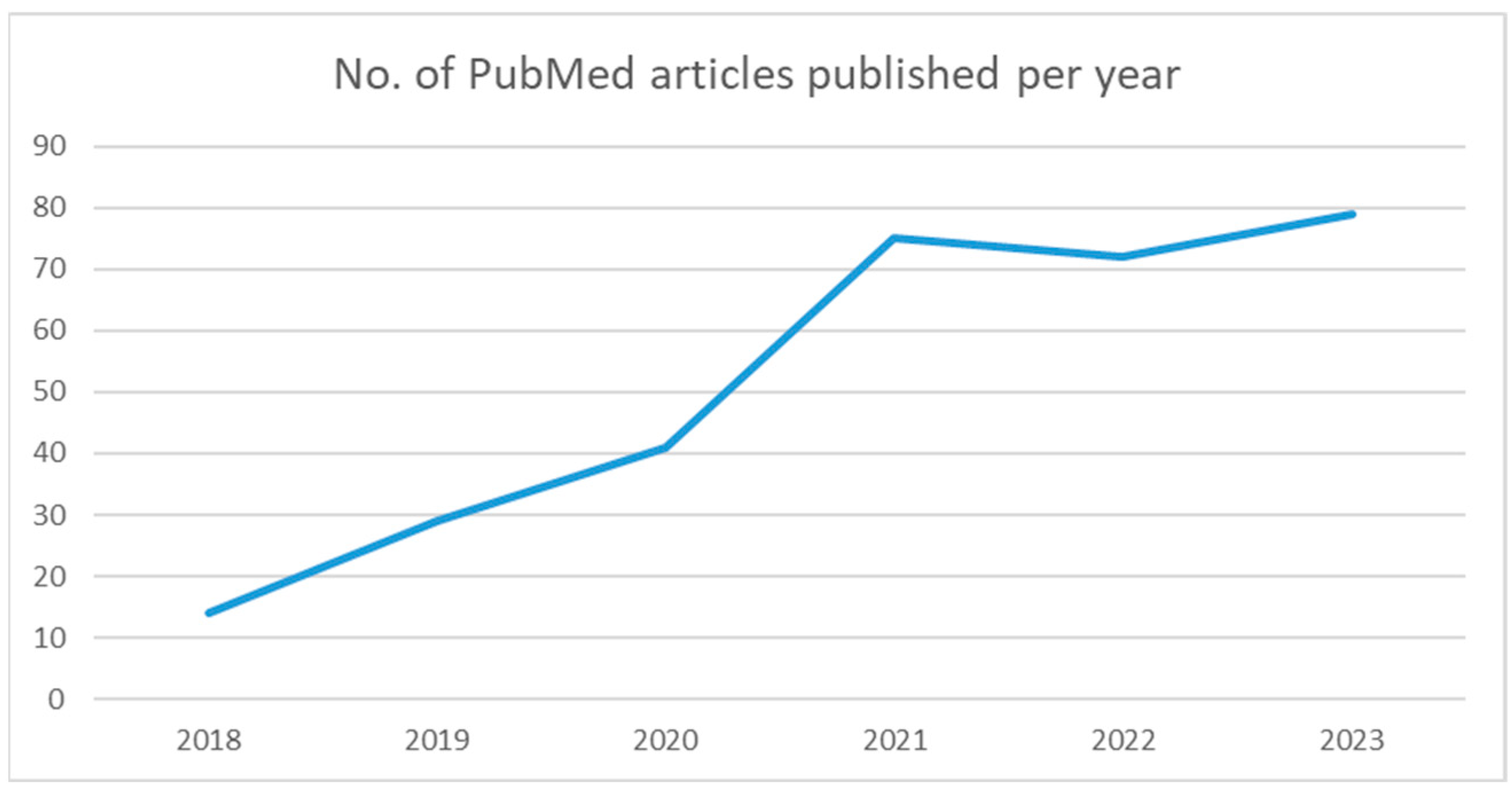
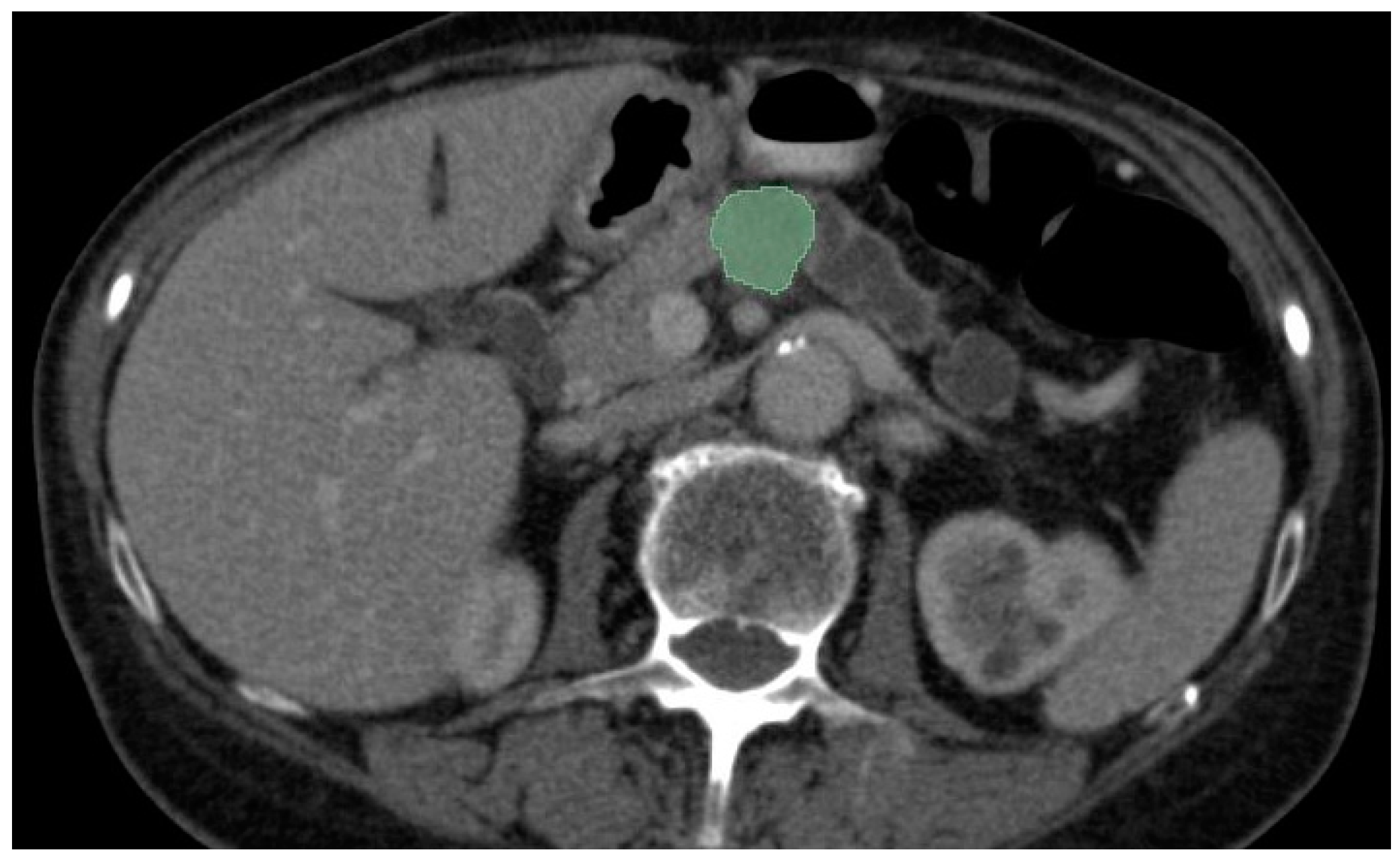
| Name | Website |
|---|---|
| MaZda ver. 4.6 [18,19,20,21] | https://www.eletel.p.lodz.pl/programy/mazda/ (accessed on 10 February 2024) |
| 3D Slicer ver. 5.6.1 [22] | https://www.slicer.org/ (accessed on 10 February 2024) |
| PyRadiomics ver. 3.1.0 [23] | https://pyradiomics.readthedocs.io/en/latest/ (accessed on 10 February 2024) |
| A Computational Environment for Radiological Research (updated on 25 February 2020) [24] | https://cerr.github.io/CERR/ (accessed on 10 February 2024) |
| LIFEx ver. 7.4.0 [25] | https://www.lifexsoft.org/ (accessed on 10 February 2024) |
| MONAI ver. 0.8.0 [26] | https://monai.io/ (accessed on 10 February 2024) |
| Resectable | Borderline | Locally Advanced |
|---|---|---|
| Arterial No contact | Head/uncinate process: | Head/uncinate process: |
| Tumor contact with common hepatic artery without extension to celiac artery (CA) or hepatic artery bifurcation. Tumor contact with SMA ≤ 180°. Tumor contact with variant arterial anatomy. | >180° SMA or CA | |
| Body/tail: Tumor contact with the CA ≤ 180° | Body/tail: >180° SMA or CA or ≤180° CA and aortic involvement | |
| Venous ≤180° without contour irregularity | >180° or with contour irregularity/thrombosis resection & reconstruction possible. Tumor contact with IVC | Unreconstructible SMV/PV due to tumor involvement or occlusion (can be due to tumor or bland thrombus) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anghel, C.; Grasu, M.C.; Anghel, D.A.; Rusu-Munteanu, G.-I.; Dumitru, R.L.; Lupescu, I.G. Pancreatic Adenocarcinoma: Imaging Modalities and the Role of Artificial Intelligence in Analyzing CT and MRI Images. Diagnostics 2024, 14, 438. https://doi.org/10.3390/diagnostics14040438
Anghel C, Grasu MC, Anghel DA, Rusu-Munteanu G-I, Dumitru RL, Lupescu IG. Pancreatic Adenocarcinoma: Imaging Modalities and the Role of Artificial Intelligence in Analyzing CT and MRI Images. Diagnostics. 2024; 14(4):438. https://doi.org/10.3390/diagnostics14040438
Chicago/Turabian StyleAnghel, Cristian, Mugur Cristian Grasu, Denisa Andreea Anghel, Gina-Ionela Rusu-Munteanu, Radu Lucian Dumitru, and Ioana Gabriela Lupescu. 2024. "Pancreatic Adenocarcinoma: Imaging Modalities and the Role of Artificial Intelligence in Analyzing CT and MRI Images" Diagnostics 14, no. 4: 438. https://doi.org/10.3390/diagnostics14040438
APA StyleAnghel, C., Grasu, M. C., Anghel, D. A., Rusu-Munteanu, G.-I., Dumitru, R. L., & Lupescu, I. G. (2024). Pancreatic Adenocarcinoma: Imaging Modalities and the Role of Artificial Intelligence in Analyzing CT and MRI Images. Diagnostics, 14(4), 438. https://doi.org/10.3390/diagnostics14040438












